Head-to-Head Comparison · Pricing verified April 13, 2026
Foundayo vs Ozempic: Which Is Better in 2026?
Last reviewed:
For most adults searching Foundayo vs Ozempic for weight loss, Foundayo is usually the better fit — and the reason goes deeper than the pill-vs-shot debate. Foundayo (orforglipron) is FDA-approved specifically for chronic weight management, starts at $149/month self-pay, and you take it as a daily pill with no food or water restrictions. Ozempic injection (semaglutide) is FDA-approved for type 2 diabetes — not weight loss — and its list price is $1,027.51/month before savings programs.
But here’s what most comparison pages won’t tell you: if you specifically want semaglutide for weight loss, the closer comparison isn’t Ozempic at all — it’s Wegovy, which is the semaglutide brand actually labeled for weight management. And Ozempic is genuinely the better option if you have type 2 diabetes and need its proven cardiovascular and kidney-protective benefits.
That distinction changes your insurance options, your out-of-pocket cost, and whether you’re even comparing the right drugs. We’ll walk through every verified difference so you can make the right call for your situation.
Disclosure: Some links on this page are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you.

A note on Ozempic formulations
In February 2026, the FDA approved Ozempic tablets (oral semaglutide 1.5 mg, 4 mg, and 9 mg) for type 2 diabetes, replacing the Rybelsus brand name. Ozempic tablets require empty-stomach morning dosing with water restrictions — different rules from the injection. This comparison focuses on Ozempic injection (the once-weekly pen), since that is what most people mean when they search “Foundayo vs Ozempic.”
3 paths — find yours fast:
- →Weight loss + pill preference: Foundayo is the FDA-approved option. See current Foundayo pricing and availability →
- →Type 2 diabetes + semaglutide coverage: Ozempic injection is the on-label choice. Check your Ozempic savings on NovoCare →
- →Want semaglutide for weight loss? You may be comparing the wrong brand. Compare Foundayo vs Wegovy instead →
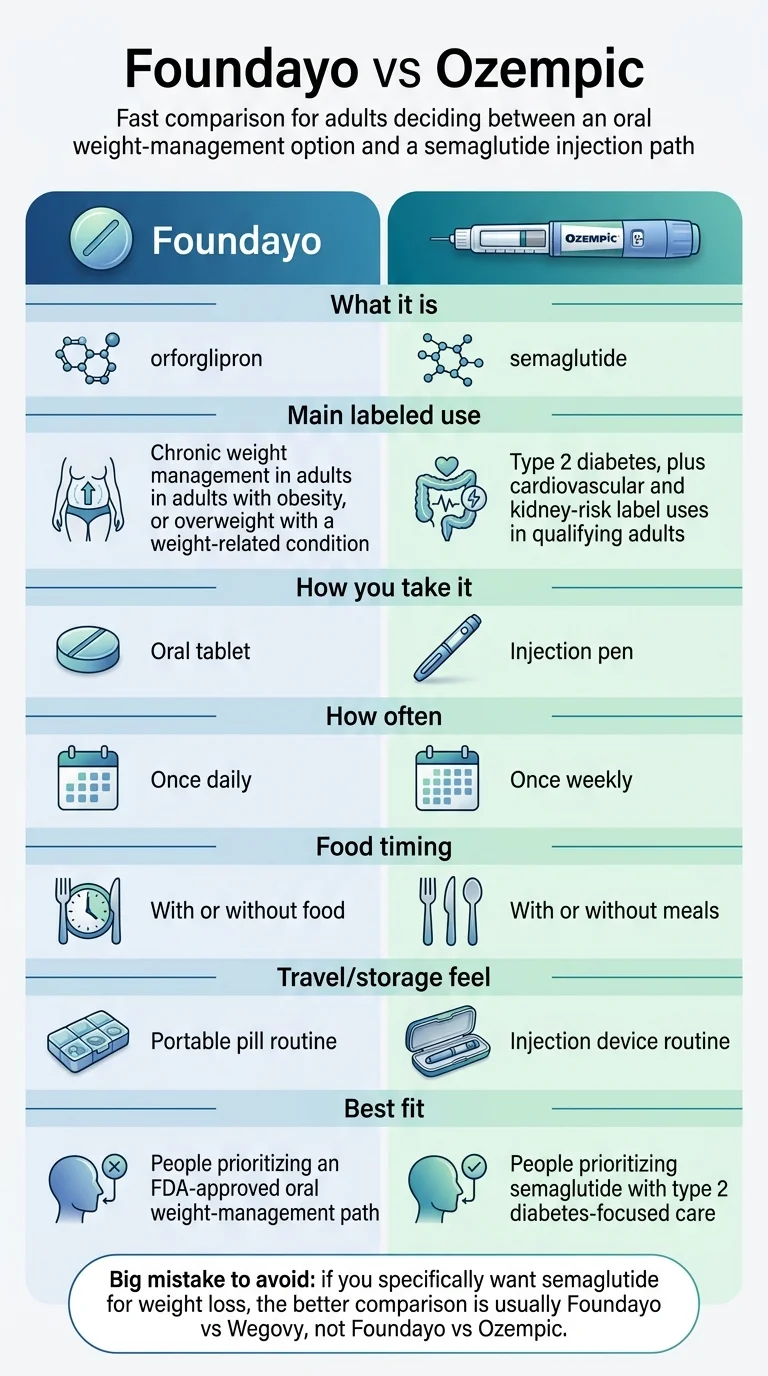
Foundayo vs Ozempic at a Glance: The Full Decision Matrix
Foundayo and Ozempic injection are both GLP-1 receptor agonist medications, but they are not interchangeable — and they are not different versions of the same drug. Foundayo contains orforglipron (Eli Lilly). Ozempic injection contains semaglutide (Novo Nordisk). Different molecules, different companies, different FDA labels, different access logic.
We built this comparison by checking the current prescribing information for both medications and official manufacturer pricing pages.
| Factor | Foundayo (orforglipron) | Ozempic injection (semaglutide) |
|---|---|---|
| FDA-approved use | Chronic weight management in adults with obesity (BMI ≥30) or overweight (BMI ≥27) with ≥1 weight-related condition | Type 2 diabetes glycemic control; CV event reduction in adults with T2D + established CVD; kidney-risk reduction in adults with T2D + CKD |
| Molecule type | Small-molecule, non-peptide oral GLP-1 receptor agonist | Peptide-based injectable GLP-1 receptor agonist |
| How you take it | Once-daily pill, any time, with or without food | Once-weekly injection (pre-filled pen), any time, with or without meals |
| Dose levels | 0.8 → 2.5 → 5.5 → 9 → 14.5 → 17.2 mg (≥30-day steps) | 0.25 → 0.5 → 1 → 2 mg (4-week steps) |
| Storage | Room temperature. No refrigeration. Protect from light. | Refrigerate before first use. After first use: room temp or fridge up to 56 days. |
| Self-pay starting price | $149/month (0.8 mg starter) | List: $1,027.51/month. Self-pay program: $199/mo intro (first 2 fills); then $349/mo (0.25–1 mg) or $499/mo (2 mg). |
| With commercial insurance + savings card | As low as $25/month | As low as $25/month (for T2D indication) |
| Avg weight loss (clinical trials) | 11.1% body weight (all doses, 72 wks); 12.4% on-treatment at highest dose (ATTAIN-1, obesity population) | Not the primary endpoint. T2D trials (SUSTAIN program): ~3.8–6.4 kg depending on dose and study |
| Cardiovascular benefit (FDA-labeled) | Not established | ✅ Yes — reduces major CV events in adults with T2D + established CVD (SUSTAIN-6 trial) |
| Kidney benefit (FDA-labeled) | Not established | ✅ Yes — reduces kidney-risk worsening + CV death in adults with T2D + CKD (FLOW trial) |
| Safety data maturity | Approved April 1, 2026. Phase 3 data from 4,500+ patients across two global trials. | On market since December 2017. Extensive real-world data. Multiple completed outcome trials. |
| Missed-dose rule | If you miss 7+ consecutive days, contact prescriber — may need to restart at lower dose | Missed dose can be taken within 5 days of scheduled day |
| Key drug interactions | CYP3A4-metabolized: strong inhibitors cap dose at 9 mg. Simvastatin capped at 20 mg/day. May reduce effectiveness of oral contraceptives. | Slows gastric emptying; may affect absorption of other oral meds. Retinopathy monitoring in T2D patients. |
| Manufacturer | Eli Lilly | Novo Nordisk |
Sources: Foundayo prescribing information (Eli Lilly, April 2026); Ozempic injection prescribing information (Novo Nordisk); LillyDirect official pricing; NovoCare official pricing and savings; FDA press releases. All pricing verified April 13, 2026.
Not sure which GLP-1 path fits your goals and insurance? Our free 60-second quiz matches you to the right program — no sign-up required.
Take our free matching quiz →The Biggest Mistake in This Comparison
Most people searching “Foundayo vs Ozempic” are really asking: Is the new weight-loss pill better than the famous GLP-1 shot? Reasonable question. But it has a problem built into it.
Important: Ozempic injection is not FDA-approved for weight loss.
Its official website states exactly that. Ozempic is labeled for type 2 diabetes, cardiovascular risk reduction in adults with T2D and established heart disease, and kidney-risk reduction in adults with T2D and chronic kidney disease.
The semaglutide brand that is FDA-approved for chronic weight management is Wegovy — same active ingredient, higher doses, specifically studied and labeled for weight loss. Wegovy is available as both a weekly injection and a daily pill (approved December 22, 2025).
Why this matters for your wallet and your coverage
Insurance coverage.
Most commercial plans cover Ozempic injection for type 2 diabetes. Many plans deny coverage for off-label weight loss use. If you don’t have T2D and your doctor writes an Ozempic prescription for weight loss, there’s a meaningful chance your insurer says no.
Clinical data context.
Foundayo’s weight-loss numbers come from obesity trials in people seeking weight management. Ozempic’s body-weight data come from diabetes trials where weight loss was a secondary outcome. Comparing those numbers directly is misleading — and we won’t do it.
The comparison you probably actually want.
If your main goal is weight loss and you’re interested in semaglutide, your real comparison is Foundayo vs Wegovy — not Foundayo vs Ozempic.
Who should keep reading this page
Stay here if you have type 2 diabetes and you’re weighing Ozempic against Foundayo, you’re exploring all GLP-1 options and want to understand where Ozempic fits, your doctor mentioned both names, or you’re on Ozempic now and wondering whether to switch.
If your only goal is weight loss and you don’t have diabetes, you already have your answer: Foundayo is the FDA-approved weight-loss option here, and Ozempic is not. But keep reading — the cost, routine, and side-effect details matter too.
When Foundayo Is the Better Choice
Foundayo is usually the better choice when your main goal is chronic weight management, you want an FDA-approved pill, and you care about avoiding injections, simplifying your daily routine, or keeping costs predictable on a cash-pay budget.
You want an FDA-approved weight-loss medication
This is the cleanest distinction. Foundayo is labeled for chronic weight management. Ozempic injection is not. If weight loss is your primary treatment goal, Foundayo puts you on an FDA-approved pathway — and that matters for insurance arguments, medical records, and the simple fact that the drug was studied in people like you.
In Eli Lilly’s ATTAIN-1 trial, adults with obesity taking Foundayo’s highest marketed dose lost an average of 27.3 pounds (12.4% of body weight) over 72 weeks on-treatment. Across all participants regardless of whether they completed treatment, the average was 25 pounds (11.1%). At the highest marketed dose, 71.5% of participants achieved at least 5% body weight loss and 54.5% achieved at least 10%.
You want a pill, not a needle
Needle aversion is one of the most common reasons people delay GLP-1 treatment. Online communities and patient surveys reflect the same pattern: needle phobia, injection anxiety, and the appeal of a pill option are constant themes. Foundayo eliminates that barrier entirely. One tablet a day.
You travel, have a busy routine, or just want simplicity
Foundayo stores at room temperature. No refrigeration. Toss it in your bag. Take it whenever — morning, lunch, bedtime. No fasting. No water restrictions. No injection device to manage.
You’re paying cash and want the lowest branded starting price
Important context
Foundayo does NOT produce as much weight loss as injectable Wegovy or Zepbound. Published data show roughly 11% vs 15–20% for the injectables. If maximum weight loss is your only priority and you’re comfortable with injections, those options will likely produce better results.
| Dose | Monthly cost (self-pay program) |
|---|---|
| 0.8 mg (starter) | $149 |
| 2.5 mg | $199 |
| 5.5 mg – 9 mg | $299 |
| 14.5 mg – 17.2 mg | $299 (refilled within 45 days) or $349 |
Foundayo self-pay pricing verified April 13, 2026 from LillyDirect. With commercial insurance that covers Foundayo, savings card can reduce cost to as low as $25/month. Available through LillyDirect (free home delivery) and telehealth providers including Ro.
Does a daily pill starting at $149/month sound like it fits your situation?
Ro offers Foundayo with a $39 first-month trial, then as low as $74/month with an annual membership plan. Online assessment, provider visit, and medication shipped to your door.
Check Foundayo Eligibility on Ro →$39 first month · then as low as $74/mo with annual plan
When Ozempic Is the Better Choice
Ozempic injection is the right choice when you have type 2 diabetes and need glycemic control, or when your specific cardiovascular or kidney condition requires what Ozempic is FDA-labeled for.
You have type 2 diabetes
Ozempic is FDA-approved for type 2 diabetes management. If T2D is your primary condition, Ozempic was studied for your exact situation — glycemic control in adults with T2D. Foundayo is not approved for type 2 diabetes.
You need the cardiovascular or kidney benefit
Cardiovascular risk reduction: Ozempic injection is FDA-approved to reduce the risk of major adverse cardiovascular events (MACE) in adults with type 2 diabetes and established cardiovascular disease. This is based on the SUSTAIN-6 trial.
Kidney-risk reduction: In April 2024, the FDA approved Ozempic injection to reduce the risk of worsening kidney disease and cardiovascular death in adults with type 2 diabetes and CKD, based on the FLOW trial (N=3,534).
Foundayo has no FDA-labeled cardiovascular or kidney benefit.
You have commercial insurance covering Ozempic for T2D
If you have type 2 diabetes and commercial insurance that covers Ozempic, you may qualify to pay as little as $25/month with the Novo Nordisk savings card (conditions apply). That’s a substantial value argument.
Foundayo’s $25/month savings-card price is also available for commercially insured patients whose plan covers Foundayo — but Foundayo was only approved April 1, 2026, and formulary decisions take time. Ozempic has had years of formulary placement.
You want the longest track record
Ozempic injection has been on the market since December 2017. There are completed large-scale outcome trials (SUSTAIN-6, FLOW), years of real-world prescribing data, and extensive post-market safety monitoring. Foundayo was approved April 1, 2026 — its Phase 3 data covered 4,500+ patients, which is meaningful, but it’s a new drug.
Side by Side: What Daily Life Looks Like
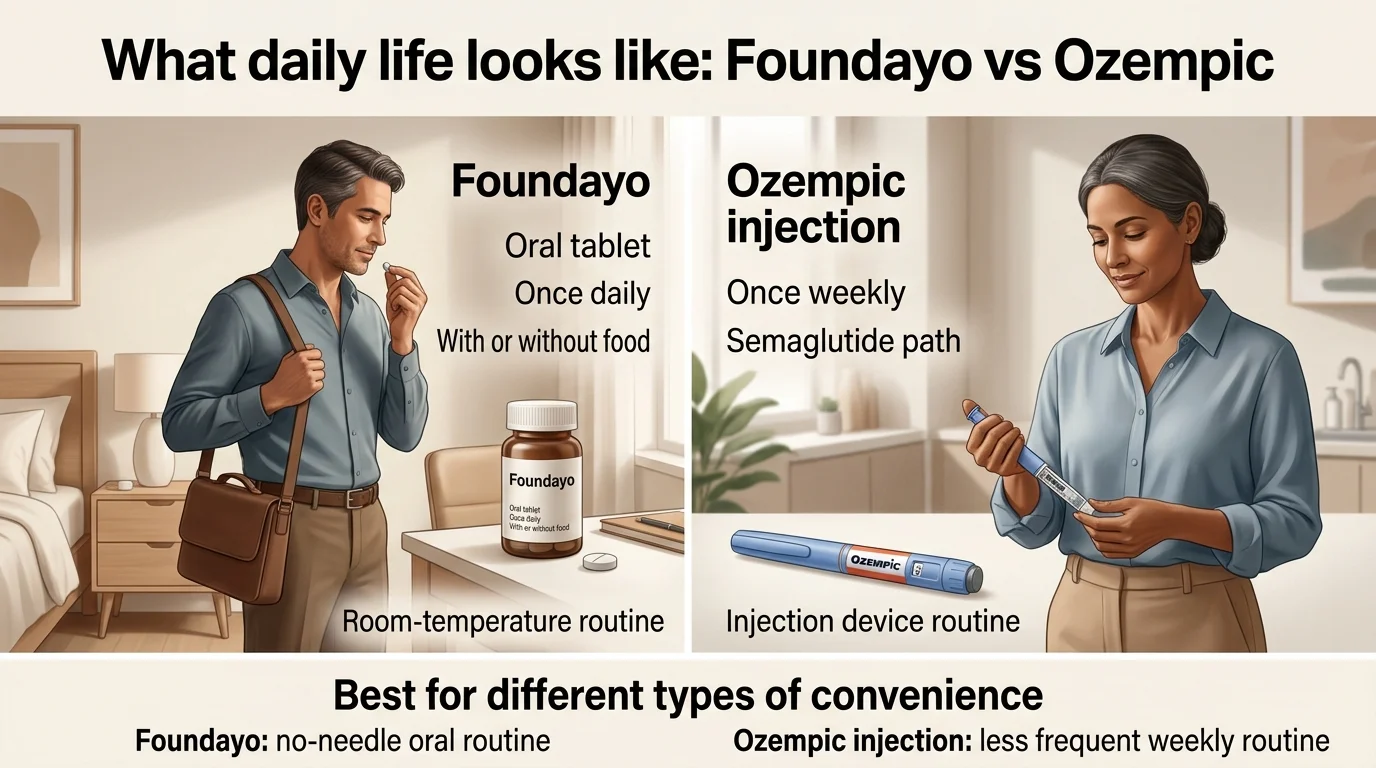
Foundayo daily routine
- ✓Swallow one tablet daily — any time, no food or water restrictions
- ✓Store in your bag, nightstand, or desk drawer at room temperature
- ✓No device, no needles, no refrigeration
- ✓One missed dose: take it when you remember, skip if almost time for next
- ✓Seven+ consecutive days missed: contact your prescriber
Ozempic injection weekly routine
- •Choose your injection day and stick to it (up to 5 days of flexibility)
- •Keep the pen refrigerated before first use; up to 56 days after opening
- •Inject into abdomen, thigh, or upper arm (rotate sites)
- •Needles are part of the routine; pens come with needles
- •No food or timing restrictions around the injection
The real-world impact: people with active travel schedules, phobias of needles, or busy non-routine lives often find Foundayo easier to maintain. People who are already comfortable with injection devices — or who are already using other injectable medications — find the once-weekly Ozempic routine straightforward.
Which Has Fewer Side Effects?
Both Foundayo and Ozempic injection are GLP-1 receptor agonists. The most common side effects across GLP-1 class drugs are nausea, vomiting, diarrhea, constipation, and decreased appetite.
| Side effect | Foundayo (ATTAIN-1 trial data) | Ozempic injection (prescribing information) |
|---|---|---|
| Nausea | ~45% (any severity, all doses) | ~20% (1 mg); ~15% (0.25/0.5 mg) |
| Vomiting | ~25% | ~9% (1 mg) |
| Diarrhea | ~22% | ~9% (1 mg) |
| Constipation | ~10% | ~5% (1 mg) |
| Decreased appetite | ~52% | Not separately listed |
Source: Foundayo prescribing information (Eli Lilly, April 2026); Ozempic prescribing information (Novo Nordisk). Higher rates in ATTAIN-1 may partly reflect study design, dose titration schedule, and patient population — not necessarily a direct comparison to Ozempic at equivalent doses.
Both carry a boxed warning about thyroid C-cell tumors (seen in animal studies; human relevance is not established). Both are contraindicated in patients with personal or family history of MTC or MEN2.
Which Is Safer Long-Term?
Ozempic injection
Approved December 2017. Large completed outcome trials (SUSTAIN-6, FLOW). Years of real-world post-market surveillance. The ongoing safety profile is well-understood.
Foundayo
Approved April 1, 2026. Phase 3 evidence from 4,500+ patients across two global studies. Long-term real-world data will accumulate over time.
Neither drug should be used during pregnancy. Both require ongoing monitoring for specific risks (retinopathy risk monitoring is noted in Ozempic’s labeling for patients with diabetic retinopathy). This is not a reason to avoid Foundayo — FDA approval requires a meaningful benefit-risk assessment. But if having years of real-world safety data matters more to you than convenience, Ozempic’s longer track record is a legitimate differentiator.
The Verdict: Which Should You Choose?

If your goal is weight loss and you’re not managing type 2 diabetes:
Foundayo is the FDA-approved option. It’s a once-daily pill starting at $149/month self-pay, studied in adults with obesity, and now available through LillyDirect and telehealth providers. Ozempic injection is not approved for weight loss. Compare Foundayo to Wegovy if you want semaglutide.
If you have type 2 diabetes:
Ozempic injection is the on-label treatment. It has proven cardiovascular and kidney benefits, deep formulary placement, and years of clinical experience. If you prefer an oral option for T2D, ask your provider about Ozempic tablets (approved February 2026).
If you specifically want semaglutide for weight loss:
Your comparison is Foundayo vs Wegovy — not Foundayo vs Ozempic. Both Wegovy injection and Wegovy pill are FDA-approved for weight management.
If you’re paying cash and want the lowest self-pay price:
Foundayo at $149/month (starter dose) vs Ozempic at $199/month (intro period, through June 30, 2026) then $349–$499/month ongoing. Foundayo wins on ongoing self-pay price at every dose tier.
Ready to see if Foundayo is the right fit for you?
Ro offers Foundayo starting at $39 for the first month, then as low as $74/month with an annual membership. Free home delivery, licensed providers, no in-person visit required.
Check Foundayo Eligibility on Ro →$39 first month · then as low as $74/mo with annual plan
Frequently Asked Questions: Foundayo vs Ozempic
Is Foundayo approved for weight loss?
Yes. Foundayo (orforglipron) received FDA approval on April 1, 2026 for chronic weight management in adults with obesity (BMI ≥30) or overweight (BMI ≥27) with at least one weight-related condition such as high blood pressure, type 2 diabetes, or high cholesterol. It is not approved as a type 2 diabetes treatment.
Is Ozempic approved for weight loss?
No. Ozempic injection is FDA-approved for type 2 diabetes management, cardiovascular risk reduction in adults with T2D and established heart disease, and kidney-risk reduction in adults with T2D and CKD. Ozempic's official website states it is not indicated for weight loss. The semaglutide brand labeled for weight loss is Wegovy — available as injection or daily pill.
Is Foundayo the same as semaglutide?
No. Foundayo contains orforglipron, a small-molecule non-peptide drug made by Eli Lilly. Semaglutide is the active ingredient in Ozempic and Wegovy, made by Novo Nordisk. Different molecules, different companies, same receptor target (GLP-1).
Which is cheaper without insurance?
Foundayo's ongoing self-pay pricing ($149–$349/month by dose) is lower than Ozempic injection's ongoing self-pay pricing ($349–$499/month after introductory fills). During the introductory period (first 2 fills through June 30, 2026), Ozempic's starter dose is $199/month vs Foundayo's $149/month. Foundayo is cheaper on ongoing self-pay pricing at every dose tier.
Which is easier to travel with?
Foundayo. It stores at room temperature with no refrigeration required and travels like a standard pill bottle. Ozempic injection requires refrigeration before first use and temperature awareness during transport.
Can you take Foundayo and Ozempic together?
No. Foundayo's prescribing information states it should not be used with other GLP-1 receptor agonist medicines.
Can you switch from Ozempic to Foundayo?
Possibly, with your prescriber's guidance. It's a molecule switch (semaglutide → orforglipron), not a simple format change. The labeled starting dose for Foundayo is 0.8 mg, and there is no official conversion chart. Discuss your goals, insurance, and transition plan with a licensed provider.
What if I want semaglutide for weight loss?
Compare Foundayo to Wegovy, not Ozempic. Wegovy is the semaglutide brand FDA-approved for chronic weight management. It's available as both a weekly injection (approved 2021) and a daily pill (approved December 22, 2025).
Do Foundayo and Ozempic have the same boxed warning?
Both carry a boxed warning about thyroid C-cell tumors in rodents. Both are contraindicated in patients with personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2). The class-wide warning language is similar, though the specific studies cited differ.
Still not sure which GLP-1 program is right for you?
Our free 60-second quiz considers your diagnosis, insurance, budget, and medication preferences to match you to the right path. No sign-up required.
Take Our Free Matching Quiz →Related comparisons
Affiliate disclosure: The RX Index may earn a commission when you use provider links on this page. This does not affect the accuracy of the information presented or our editorial independence. If we removed every link on this page, this comparison would still hold up — and we believe it would still be the most complete version available.
Medical disclaimer: This article is for informational purposes only and does not constitute medical advice, diagnosis, or treatment. Foundayo is a trademark of Eli Lilly and Company. Ozempic and Wegovy are trademarks of Novo Nordisk. Consult a licensed healthcare provider about your specific health situation. All pricing is subject to change.
The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We re-verify pricing and availability monthly.