GLP-1 Comparison · April 29, 2026 · Prices Verified
Mounjaro vs Ozempic: Which Is Better in 2026?
Published:
Written by The RX Index Editorial Team
Last verified: April 29, 2026 · FDA labels checked via DailyMed · Pricing verified · See methodology · Affiliate disclosure
Bottom line, verified April 29, 2026
Mounjaro vs Ozempic comes down to one question: are you after the largest possible drop in weight and A1C, or do you have a heart or kidney problem that needs the specific FDA-labeled indications only Ozempic carries?
Mounjaro (tirzepatide) wins for raw weight and A1C reduction. Ozempic (semaglutide) wins when its labeled cardiovascular and kidney indications matter. Neither is FDA-approved for weight loss alone — for that, you're actually shopping Zepbound vs Wegovy.
List prices: $1,112.16 per 28-day fill (Mounjaro) and $1,027.51 per fill (Ozempic) before any insurance, savings cards, or cash-pay programs.
There's also a third group reading this page who shouldn't be comparing these two at all. We'll be honest about who that is in a minute, even if it costs us the click. Stay with us — the answer is faster and more useful than the page-1 results that brought you here.
Affiliate disclosure. The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We may earn a commission if you use some of the links on this page. Commissions never change which medication is medically right for you, and our editorial conclusions are based on FDA labels, published trial evidence, and current public pricing — not on what pays best.
Medical disclaimer. This is not medical advice. Talk to a licensed clinician before starting, switching, or stopping any prescription medication.
The 30-second decision
Before you scroll, this is the answer for most people:
| If your main question is… | Better first discussion | Why |
|---|---|---|
| “Which lowers A1C and weight more?” | Mounjaro | Beat Ozempic 1 mg head-to-head on both in SURPASS-2 |
| “Which has heart-attack and stroke protection?” | Ozempic | FDA-labeled to reduce major cardiovascular events in T2D + heart disease |
| “Which protects my kidneys?” | Ozempic | FDA-labeled to slow kidney disease worsening in T2D + CKD |
| “Which is FDA-approved for weight loss?” | Neither — compare Zepbound vs Wegovy | Mounjaro and Ozempic are diabetes-labeled brands |
| “Which is cheaper without insurance?” | Ozempic ($349–$499/mo via NovoCare); Mounjaro has no DTC program | Different manufacturers, different self-pay strategies |
| “I have insurance — which will it cover?” | Whichever has the diagnosis to match (almost always T2D) | Coverage is decided by your diagnosis and plan, not the drug name |
| “Should I switch from Ozempic to Mounjaro?” | Maybe — depends on why you're switching | Clinician-guided restart at lower dose; expect new GI adjustment |
Not sure which one your insurance actually covers? Use Ro's free GLP-1 Insurance Coverage Checker. You enter your name, plan info, and a few eligibility details. Ro runs the check and emails you a personalized report covering Ozempic, Wegovy, and Zepbound — including whether prior authorization is required and what your real copay would be. (For Mounjaro coverage specifically, see the FAQ below — Ro doesn't currently offer Mounjaro, so we'll show you the right path for that one.)
Check my GLP-1 coverage — free →What we verified for this page
We don't expect you to take our word for any of this. Here's the receipt.
Verified on April 29, 2026:
- FDA labels for Mounjaro and Ozempic via DailyMed (the NIH's official drug-label database).
- Mounjaro list price of $1,112.16 per 28-day fill via Eli Lilly's official Mounjaro pricing page and FAQ. One fill is defined as a month's supply of four pens.
- Ozempic list price of $1,027.51 and self-pay tiers ($349/mo for 0.25–1 mg, $499/mo for 2 mg, $199 intro for the first two months on starting doses through June 30, 2026) via NovoCare's official Ozempic savings and pricing pages.
- SURPASS-2 head-to-head trial results via the original New England Journal of Medicine publication and Eli Lilly's clinical-trial press release.
- SURPASS-CVOT cardiovascular outcomes (tirzepatide vs dulaglutide) via the American College of Cardiology summary and the published primary endpoint.
- November 2025 White House GLP-1 pricing agreement via the Novo Nordisk and Eli Lilly press releases announcing implementation.
- Medicare Part D rules and 2026 $2,100 out-of-pocket cap via CMS.gov; Medicare GLP-1 Bridge announcement (effective July 1, 2026) via CMS.
- Ro's GLP-1 Insurance Coverage Checker, Body program pricing, and Mounjaro availability status via ro.co.
Needs final verification before going live:
- Live Bing autocomplete and PAA exact wording on publish day.
- Whether Eli Lilly has launched a consumer Mounjaro program through LillyDirect (the November 2025 commitment was to bring Mounjaro to LillyDirect at 50–60% off list; as of late April 2026 only Zepbound has the consumer vial program at $299–$449/month).
- Current Sesame Care branded GLP-1 pricing and Costco-member pricing on Wegovy and Ozempic.
We re-verify every commercial fact on this page on a 30-day cadence. The "Last verified" date at the top of the page updates when we do.
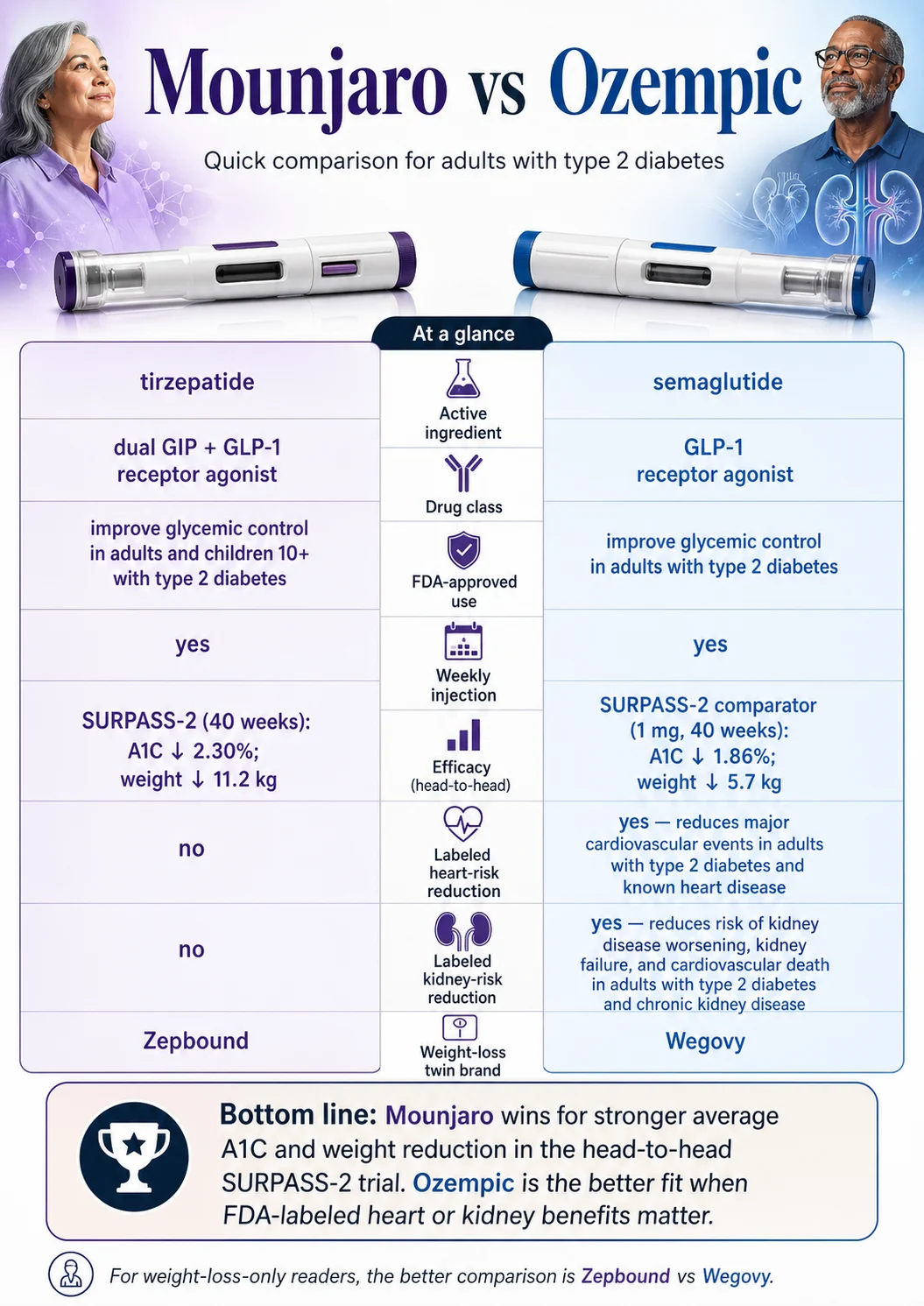
The 2026 Mounjaro vs Ozempic master comparison
Every dimension that should drive your decision, in one place.
| Dimension | Mounjaro | Ozempic |
|---|---|---|
| Active ingredient | Tirzepatide | Semaglutide |
| Manufacturer | Eli Lilly | Novo Nordisk |
| Mechanism | Dual GIP + GLP-1 receptor agonist | GLP-1 receptor agonist |
| FDA-approved use(s) | Adults and children 10+ with type 2 diabetes (glycemic control) | Adults with T2D; reducing major cardiovascular events in T2D + heart disease; slowing kidney disease worsening in T2D + CKD (injection form) |
| Weight-management-labeled twin | Zepbound (also tirzepatide) | Wegovy (also semaglutide) |
| Average A1C drop, top dose, head-to-head (SURPASS-2) | −2.30% (15 mg) | −1.86% (1 mg) |
| Average weight loss, top dose, head-to-head (SURPASS-2, 40 weeks) | −11.2 kg | −5.7 kg |
| % reaching A1C ≤6.5% + ≥10% weight loss (SURPASS-2) | 60% (15 mg) | 22% (1 mg) |
| Cardiovascular event reduction (FDA label) | Not approved | Approved (T2D + CVD) |
| Kidney disease progression (FDA label) | Not approved | Approved (T2D + CKD, injection) |
| Dose range | 2.5 → 5 → 7.5 → 10 → 12.5 → 15 mg weekly | 0.25 → 0.5 → 1 → 2 mg weekly |
| Pen room-temp stability | 21 days | 56 days |
| Oral form | No — injection only | Yes — both injection (weekly) and oral tablet forms exist; Rybelsus is a related oral semaglutide brand. Oral and injectable forms are not mg-to-mg interchangeable. |
| List price | $1,112.16/fill (28 days) | $1,027.51/fill |
| Manufacturer cash-pay program (no insurance) | None currently | NovoCare: $349/mo (0.25–1 mg), $499/mo (2 mg); $199/mo intro for 2 months at 0.25–0.5 mg through June 30, 2026 |
| Same-molecule cash-pay alternative for weight loss | LillyDirect Zepbound vials: $299–$449/mo (post-Nov 2025 pricing) | NovoCare Wegovy: $349/mo standard; $199 intro |
| Commercial insurance + manufacturer savings (T2D) | As low as $25/mo (commercial only; government insurance excluded) | As low as $25/mo |
| Medicare Part D, T2D (2026) | About 8 in 10 Part D patients pay $0–$50 per 28-day supply; remaining patients average ~$262 (Lilly data); 2026 Part D out-of-pocket cap: $2,100 | Plan-specific; covered for T2D under most Part D formularies; same $2,100 annual cap applies |
| Medicare for weight loss alone | Not covered under standard Part D | Not covered under standard Part D (the new Medicare GLP-1 Bridge launches July 1, 2026 with a $50 copay for eligible weight-management uses outside Part D) |
| Common GI side effects (rates from prescribing info) | Nausea 12–18%, diarrhea 12–17%, vomiting 5–9%, constipation 6–7% | Nausea 16–20%, diarrhea 8.5–8.8%, vomiting 5–9.2%, constipation 3.1–5% |
| Boxed warning | Thyroid C-cell tumors (rodent data; human relevance unknown) | Thyroid C-cell tumors (rodent data; human relevance unknown) |
The next sections turn this table into a decision for your situation.
The single most important fact about Mounjaro vs Ozempic
Here's the honest version, which is the one most ranking pages bury six headings deep:
Neither Mounjaro nor Ozempic is FDA-approved for weight loss. Both are approved for type 2 diabetes. Their weight-management-approved twins are Zepbound (same molecule as Mounjaro — tirzepatide) and Wegovy (same molecule as Ozempic — semaglutide). That distinction is the single biggest predictor of three things you're going to care about: whether your insurance covers the prescription, whether your doctor will write it, and what you'll actually pay.
Here's the brand map, because the four-way confusion ends up costing real people real money:
| Active ingredient | Diabetes brand | Weight-management brand |
|---|---|---|
| Tirzepatide (Eli Lilly) | Mounjaro | Zepbound |
| Semaglutide (Novo Nordisk) | Ozempic | Wegovy |
If you're reading this page because you want to lose weight and you don't have type 2 diabetes, here's our damaging admission: you're researching the wrong two drugs. You should be on our Wegovy vs Zepbound comparison instead. Wegovy and Zepbound are FDA-approved for chronic weight management. Insurance is more likely to cover them for that purpose. Zepbound also has an FDA-approved indication for moderate-to-severe obstructive sleep apnea in adults with obesity. Both have manufacturer cash-pay programs that genuinely make them affordable — Wegovy via NovoCare ($199 intro, $349 standard for most doses) and Zepbound vials via LillyDirect ($299–$449 after the November 2025 pricing reset). Mounjaro doesn't have a cash-pay program at all right now. Ozempic does, but its self-pay tiers are higher than Wegovy's.
We say this knowing some of you will leave. That's fine. The right reader for the rest of this page is going to trust everything we say next more for it, because we said this out loud instead of pretending the search term you typed was always the right question.
For everyone still here — readers with type 2 diabetes, readers already prescribed off-label, readers comparing both drugs for a specific medical reason — the Mounjaro vs Ozempic comparison is the right comparison, and we're going to do it properly.
Mounjaro vs Ozempic for weight loss
Answer up front
In the only major head-to-head clinical trial (SURPASS-2, 1,879 adults with type 2 diabetes), Mounjaro at its top dose produced about twice the weight loss of Ozempic at the top dose available at the time — roughly 11.2 kg (24.7 lb) vs 5.7 kg (12.6 lb) over 40 weeks. Real-world data has gone in the same direction. Mounjaro wins on raw weight loss. Ozempic isn't far behind, especially when titrated to its current top dose.
Now the nuance, because anyone who tells you "Mounjaro is twice as effective" is overselling.
SURPASS-2 compared Mounjaro to Ozempic 1 mg, not Ozempic 2 mg. The 2 mg dose of Ozempic was approved in March 2022, around the same time that trial was wrapping up. The closest data we have on Ozempic 2 mg comes from SUSTAIN FORTE, which compared semaglutide 2 mg vs 1 mg in adults with type 2 diabetes and found an average weight loss of 6.9 kg vs 6.0 kg at week 40 (trial-product estimand). The 2 mg dose adds modestly to Ozempic's weight-loss effect — narrowing the gap with Mounjaro, but not closing it.
Two clean numbers worth remembering:
SURPASS-2 composite goal
60% vs 22%
60% of patients on Mounjaro 15 mg hit the composite goal of A1C ≤6.5% plus ≥10% body-weight loss without serious low blood sugar. 22% of patients on Ozempic 1 mg hit the same goal.
2024 JAMA Internal Medicine real-world study
Greater weight loss with tirzepatide that widened over time
In routine clinical practice, the gap between tirzepatide and semaglutide widened the longer patients stayed on therapy.
Plain-English bottom line on weight loss:
- If your top priority is the largest average weight loss and your insurance situation is comparable for both drugs, Mounjaro is the medication to ask your clinician about first.
- If you've plateaued on Ozempic and your weight isn't moving anymore, that's a legitimate reason to ask about switching to Mounjaro.
- If you don't have type 2 diabetes and your only goal is weight loss, the right comparison is Zepbound vs Wegovy.
Mounjaro vs Ozempic for diabetes (A1C and blood sugar)
Answer up front
Mounjaro reduces A1C more on average than Ozempic, with the same head-to-head caveat that the SURPASS-2 comparator was Ozempic 1 mg. At top doses studied, A1C dropped 2.30% on Mounjaro 15 mg vs 1.86% on Ozempic 1 mg. For most adults with type 2 diabetes whose main goal is glycemic control, Mounjaro is the stronger first discussion. Ozempic remains a strong choice — especially when its cardiovascular or kidney indications also matter.
Both drugs are FDA-approved as add-ons to diet and exercise to improve glycemic control. Both work weekly. Both reduce A1C significantly in trials and in clinical practice. The differences that matter:
Mounjaro's A1C edge. Across the three doses studied in SURPASS-2 (5, 10, 15 mg), Mounjaro reduced A1C by 2.01%, 2.24%, and 2.30% respectively. Ozempic 1 mg reduced A1C by 1.86%. That's a real, statistically significant gap — and it's why endocrinologists have leaned toward tirzepatide in patients who need aggressive A1C lowering.
Ozempic's dose ceiling. Ozempic now goes up to 2 mg weekly, which produces additional A1C reduction beyond 1 mg. That narrows but doesn't eliminate the gap.
The dose-equivalence question. There is no FDA-approved or universally accepted conversion chart between Mounjaro doses and Ozempic doses. Some clinicians use a rough rule of thumb (Mounjaro 5 mg ≈ Ozempic 1 mg in A1C effect), but this isn't validated. If you're switching, your clinician will almost certainly start you at the new drug's lowest dose and titrate up.
If Mounjaro is your first ask, your next step is a clinician visit. Lilly's Mounjaro Savings Card brings eligible commercially insured patients to as low as $25/month for type 2 diabetes — verify Mounjaro coverage directly with your plan or your prescriber's office before the appointment. If your plan won't cover Mounjaro, Ozempic is the strong fallback for diabetes — and Ro's free coverage checker tells you what your plan covers for Ozempic, Wegovy, and Zepbound in one report.
Check Ozempic, Wegovy & Zepbound coverage — free →Mounjaro vs Ozempic side effects
Answer up front
Both drugs cause GI side effects in about 1 in 3 people — nausea, diarrhea, vomiting, constipation, abdominal pain — concentrated during dose increases. Most fade within weeks. Mounjaro at the highest doses (12.5–15 mg) tends to be the harder of the two for many people to tolerate. Both share the same FDA boxed warning for thyroid C-cell tumors based on rodent studies (human relevance is unknown). Neither is "safer" universally; tolerability is individual.
Here are the actual side-effect rates from each drug's FDA prescribing information. They're not perfectly comparable across drugs (different trials, different populations) but they're the cleanest data publicly available.
| Side effect | Mounjaro (vs placebo) | Ozempic (vs placebo) |
|---|---|---|
| Nausea | 12–18% (vs 4%) | 16–20% (vs 6%) |
| Diarrhea | 12–17% (vs 9%) | 8.5–8.8% (vs 1.9%) |
| Decreased appetite | 5–11% (vs 1%) | — |
| Vomiting | 5–9% (vs 2%) | 5–9.2% (vs 2.3%) |
| Constipation | 6–7% (vs 1%) | 3.1–5% (vs 1.5%) |
| Heartburn (dyspepsia) | 5–8% (vs 3%) | 2.7–3.5% (vs 1.9%) |
| Abdominal pain | 5–6% (vs 4%) | 5.7–7.3% (vs 4.6%) |
The discontinuation rate — patients who stopped because of GI side effects — is in the low single digits for both drugs at typical doses, slightly higher for Mounjaro at the 15 mg dose.
Both drugs share serious warnings that you and your prescriber should review:
- Thyroid C-cell tumors: Boxed warning. Don't take either if you or a family member has had medullary thyroid carcinoma or have Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- Pancreatitis: Stop the medication and call your clinician immediately for severe abdominal pain.
- Gallbladder disease: Both can increase risk.
- Diabetic retinopathy worsening: A specific warning on the Ozempic label, especially in patients with established retinopathy.
- Kidney injury: Severe vomiting or diarrhea can cause dehydration that worsens kidney function.
- Low blood sugar (hypoglycemia): Especially when combined with insulin or sulfonylureas. Your clinician may reduce other diabetes meds when starting either drug.
- Allergic reactions: Including anaphylaxis and angioedema.
Mounjaro-specific warning to know
Mounjaro can reduce the effectiveness of oral hormonal contraceptives (birth control pills) because it slows down stomach emptying. The FDA label advises switching to a non-oral method of contraception, or adding a barrier method, for 4 weeks after starting Mounjaro and for 4 weeks after each dose increase. This is important for anyone of reproductive age starting or titrating Mounjaro.
What real users are asking on Reddit and patient forums: "Which drug has had less side effects? I am so fatigued on Ozempic." and "Which was easier to take, or which had better efficacy?" — these are the exact questions readers bring to this comparison. We include them not as evidence (they're not), but to acknowledge that tolerability varies enormously person to person. Some people sail through both with minor nausea on day 2 of each dose increase. Others can't tolerate Mounjaro 15 mg or Ozempic 2 mg at all and have to step back. Your clinician will work with you to find a tolerable dose.
Practical advice that helps almost everyone:
- Titrate slowly — don't push to the next dose if you're still queasy at the current one.
- Eat smaller, lower-fat meals, especially in the first few days after a shot.
- Hydrate aggressively. GI losses can sneak up on you.
- Take an antiemetic (like ondansetron) if your clinician prescribes one for the first dose increase.
- Call your clinician for severe or persistent symptoms.
Mounjaro vs Ozempic if you have heart disease or kidney disease
Answer up front
Ozempic is the clear winner if you have type 2 diabetes plus established cardiovascular disease or chronic kidney disease. The FDA approved Ozempic to reduce major cardiovascular events (heart attack, stroke, cardiovascular death) in T2D + CVD, and to slow kidney disease worsening, kidney failure, and cardiovascular death in T2D + CKD (injection form). Mounjaro carries neither indication on its label as of April 2026.
This is the dimension where the comparison flips most decisively. Mounjaro has the bigger A1C and weight numbers. Ozempic has the bigger label.
Ozempic's cardiovascular indication comes from the SUSTAIN-6 trial, which showed semaglutide reduced major adverse cardiovascular events in adults with T2D and existing cardiovascular disease. The FDA approved this indication in 2020.
Ozempic's kidney indication comes from the FLOW trial, published in 2024. Semaglutide reduced the risk of major kidney disease events, a sustained drop in kidney function, and death from cardiovascular causes in adults with T2D and chronic kidney disease.
What about Mounjaro's cardiovascular trial? SURPASS-CVOT results are now published. The trial tested tirzepatide against dulaglutide (a different GLP-1, not a placebo) in adults with type 2 diabetes and established atherosclerotic cardiovascular disease. The primary endpoint of major adverse cardiovascular events occurred in 12% of tirzepatide patients vs 13% of dulaglutide patients (hazard ratio 0.92). Tirzepatide met the bar for noninferiority but did not show superiority to dulaglutide for cardiovascular event reduction. That doesn't mean Mounjaro doesn't help the heart — it means the trial showed equivalence to another GLP-1, not superiority over standard care, and the FDA has not added a cardiovascular risk-reduction indication to the Mounjaro label.
What this means for you:
- If you have T2D plus a history of heart attack, stroke, coronary artery disease, peripheral artery disease, or established cardiovascular disease — ask about Ozempic first. The label backs it up.
- If you have T2D plus chronic kidney disease (eGFR <60 or albuminuria) — ask about Ozempic first.
- If your primary issue is A1C and weight without cardiovascular or kidney complications — Mounjaro is a reasonable first ask.
If your situation calls for Ozempic specifically and your insurance won't cover it on the first try, this is the exact friction Ro's insurance concierge is built for. They submit prior authorization paperwork, work appeals on denials, and source the medication through pharmacies that have it in stock. Ro's process for handling coverage and paperwork takes about 2–3 weeks.
Have Ro fight for my Ozempic coverage →How much do Mounjaro and Ozempic cost in 2026?
Answer up front
List price is $1,112.16 per 28-day fill for Mounjaro and $1,027.51 per fill for Ozempic. Most commercially insured patients with type 2 diabetes pay $25/month via the manufacturer savings card. Without insurance, Ozempic has a real direct-from-manufacturer cash-pay program ($349–$499/mo through NovoCare, with a $199 intro for the first two months on starting doses through June 30, 2026). Mounjaro currently does not have an equivalent direct cash-pay program. That's the single biggest financial difference between the two drugs in 2026.
This is the section most pages on page 1 of search results get wrong, because they were written before the November 2025 White House pricing reset. Here's the reality, with timestamps.
The 2026 cost reality matrix
| Access path | Mounjaro | Ozempic | Notes |
|---|---|---|---|
| List price (cash, no programs) | $1,112.16/fill | $1,027.51/fill | All doses priced the same |
| Commercial insurance + savings card (T2D, covered) | As low as $25/mo | As low as $25/mo | Government insurance excluded |
| Commercial insurance + savings card (T2D, plan doesn't cover) | Up to $499 saved per fill | $25/mo with limits | Eligibility varies |
| Manufacturer cash-pay (uninsured, T2D) | No DTC program currently | $349/mo (0.25–1 mg), $499/mo (2 mg) | NovoCare rolled this out Nov 2025 |
| Manufacturer cash-pay intro offer | None | $199/mo for first 2 months at 0.25–0.5 mg, through June 30, 2026 | Limited-time |
| Same-molecule cash-pay alternative for weight loss | LillyDirect Zepbound vials: $299–$449/mo | NovoCare Wegovy: $349/mo standard, $199 intro | Different FDA indication |
| Medicare Part D for T2D (2026) | About 8 in 10 Part D patients pay $0–$50; remaining patients average ~$262 | Plan-specific; covered for T2D | 2026 annual out-of-pocket cap: $2,100 |
| Medicare for weight loss alone | Not covered under standard Part D | Not covered under standard Part D | New Medicare GLP-1 Bridge launches July 1, 2026 ($50 copay for eligible weight uses) |
| Pharmacy discount cards (GoodRx, SingleCare) | $900–$1,100/mo | $850–$995/mo | Varies by pharmacy |
Every commercial number above was verified against the manufacturer's official pricing pages within the last 30 days.
The November 2025 White House pricing reset
In November 2025, the federal government negotiated pricing agreements with both Eli Lilly and Novo Nordisk on GLP-1s. Novo Nordisk implemented immediately — that's why the new $349/$499 NovoCare tiers and the $199 intro exist. The standard self-pay price for Ozempic dropped from $499 to $349 in the same announcement. Eli Lilly committed to bringing diabetes-side discounts on Trulicity and Mounjaro to LillyDirect at 50–60% off list, but as of late April 2026 the consumer-facing Mounjaro program has not launched. The closest legitimate cash-pay path for tirzepatide remains LillyDirect's Zepbound vial program at $299–$449/month — but that's a different FDA-approved drug for a different FDA-approved indication (chronic weight management and obstructive sleep apnea).
If you're a self-pay Mounjaro patient watching this space, this is the single biggest update to track between now and the end of 2026.
Why "list price" is almost never what you'll actually pay
Three numbers to keep straight:
- List price (also called WAC, wholesale acquisition cost): What Eli Lilly and Novo Nordisk charge wholesalers. Almost no patient pays this.
- Cash-pay price (manufacturer programs): What you pay if you skip insurance and buy directly from the manufacturer's program. This is the NovoCare $349/$499 number for Ozempic.
- Insurance copay: What you actually pay at the pharmacy after insurance, prior authorization, and any savings card.
Most insured T2D patients fall into bucket 3 and pay $25/mo with the manufacturer savings card. Most uninsured Ozempic patients fall into bucket 2 and pay $349/mo. Almost nobody pays bucket 1.
The single biggest mistake people make on this page: assuming list price is what they'll pay. Before you commit to either drug, find out what your specific insurance plan will actually cover and what your real out-of-pocket would be. Ro's GLP-1 Insurance Coverage Checker is free, takes about 3 minutes, and shows your coverage and prior-authorization picture for Ozempic, Wegovy, and Zepbound before you commit.
See what my plan actually covers — free →Will insurance cover Mounjaro or Ozempic?
Answer up front
Yes, almost always — if you have type 2 diabetes and your plan includes either drug on its formulary. Coverage for off-label weight loss without a diabetes diagnosis is nearly universal in its denial. Medicare Part D covers both for T2D (the 2026 out-of-pocket cap is $2,100) but legally cannot cover either for weight loss alone — though the new Medicare GLP-1 Bridge launching July 1, 2026 will offer a $50 copay for eligible weight-management uses outside the standard Part D benefit. State Medicaid programs vary widely. The fastest way to know is to run a coverage check before picking a drug.
What commercial insurance usually covers
Most commercial health plans cover both Ozempic and Mounjaro for adults with type 2 diabetes, often with prior authorization and step therapy. Common requirements:
- Diagnosis on file: ICD-10 code for T2D (E11.x).
- Prior authorization: Your prescriber submits documentation proving medical necessity. Approval typically takes 1–10 business days.
- Step therapy (often called fail-first): Some plans require you to try metformin first, and sometimes a second-line drug, before they'll cover a GLP-1. Documentation that earlier therapies were ineffective or not tolerated unlocks coverage.
- Quantity limits: Usually one box of pens per 28 days.
What commercial insurance usually doesn't cover
Off-label weight loss without T2D. If your prescription is written for weight management and you don't have a diabetes diagnosis, expect a denial. The standard advice: ask your clinician whether Wegovy or Zepbound (the FDA-approved weight-loss versions) would be more appropriate. Plans that exclude weight-loss drugs entirely will deny both, but plans that include obesity benefits are far more likely to cover Wegovy or Zepbound for documented obesity than Ozempic or Mounjaro off-label.
Medicare in 2026
- Part D for T2D: Both drugs are covered. For Mounjaro specifically, Eli Lilly reports that about 8 in 10 Medicare Part D patients pay $0–$50 per 28-day supply; remaining patients average about $262. The 2026 Part D benefit redesign capped annual out-of-pocket spending on covered drugs at $2,100.
- Part D for weight loss alone: Not covered. Federal law excludes weight-loss drugs from Part D. CMS proposed changing this in late 2024; the final rule in April 2025 declined to do so.
- Medicare GLP-1 Bridge: Beginning July 1, 2026, CMS is launching a separate program that will offer eligible Medicare beneficiaries a $50/month copay for certain GLP-1 medications used for weight management — outside the standard Part D benefit. Eligibility and covered drugs are still being finalized.
- Manufacturer savings cards: Cannot be used by Medicare, Medicaid, VA, TRICARE, or other government beneficiaries. This is a federal anti-kickback rule.
Medicaid
State by state. As of late 2025, only about 13 states cover obesity medications under Medicaid, and several (including California and Pennsylvania) eliminated coverage effective January 1, 2026. T2D coverage is more consistent but typically requires prior authorization. Check your state's Medicaid formulary.
How Ro fits into the insurance picture
Ro doesn't replace your insurance — it works alongside it. The Ro Body program is cash-pay only ($39 first month, then $149/mo or as low as $74/mo with annual prepay; medication priced separately). Ro's insurance concierge handles prior authorization paperwork on FDA-approved GLP-1s and works with your insurance company to get coverage approved; the process takes about 2–3 weeks.
Important to know: Ro does not currently offer Mounjaro. The drugs Ro can prescribe and provide insurance support for are Ozempic, Wegovy, and Zepbound (plus Foundayo, the new oral GLP-1). For Mounjaro specifically, your access path is your primary care or specialist clinician plus the Lilly Mounjaro Savings Card if you're commercially insured. For everything else — Ozempic, Wegovy, Zepbound — Ro is the cleanest single-stop telehealth option.
For self-pay Ozempic specifically, NovoCare is the most direct path ($349/mo or $199 intro). Sesame Care has been a strong secondary option for self-pay Wegovy and Ozempic, especially with Costco-member pricing — and Sesame's branded formulary is broader than Ro's. Verify current offers at the source before committing.
Which one should you ask about? Decision tree by situation
Answer up front
Average results are useful; your actual choice depends on your situation. Below are six common reader profiles and the medication that fits each — based on FDA labels, head-to-head efficacy, real-world cost, and tolerability.

Pick Mounjaro first if this sounds like you
You have type 2 diabetes. Your A1C is above target. You also want to lose weight. You don't have established cardiovascular disease or chronic kidney disease that would push you toward Ozempic specifically. Your insurance covers Mounjaro, or your out-of-pocket cost is acceptable. You're prepared to titrate slowly and tolerate possible GI side effects.
Why Mounjaro: Stronger head-to-head A1C drop and meaningfully greater average weight loss. The dual GIP + GLP-1 mechanism is the differentiator.
Next step: Ask your clinician about Mounjaro by name. Bring the SURPASS-2 numbers if you want a data-backed conversation. If you have commercial insurance, the Lilly Mounjaro Savings Card brings eligible patients to as low as $25/month — verify Mounjaro coverage with your plan before the visit.
Pick Ozempic first if this sounds like you
You have type 2 diabetes plus established heart disease (history of heart attack, stroke, coronary disease, or peripheral arterial disease) OR chronic kidney disease (eGFR below 60, or albuminuria). You want a medication with FDA-approved cardiovascular and kidney benefits — not just glycemic control.
Why Ozempic: It's the only one of the two with FDA-approved indications to reduce major cardiovascular events in T2D + CVD and to slow kidney disease progression in T2D + CKD. The label backs the medical reasoning.
Next step: Ask your clinician about Ozempic specifically — and ask whether the cardiovascular or kidney indication applies to you. Ro's coverage checker will tell you what your plan covers, including step therapy requirements.
Check Ozempic coverage on Ro →Pick neither — switch to comparing Zepbound vs Wegovy if this sounds like you
You don't have type 2 diabetes. Your goal is weight loss. You're researching Mounjaro and Ozempic because they're the names you've seen in headlines and on social media.
Why neither: Mounjaro and Ozempic are diabetes-labeled drugs. Insurance is far more likely to deny them off-label for weight loss than to cover their FDA-approved twins. Zepbound (tirzepatide for obesity, now also approved for moderate-to-severe obstructive sleep apnea) and Wegovy (semaglutide for obesity, now also approved for cardiovascular risk reduction in adults with overweight or obesity plus heart disease) are the on-label, more-likely-to-be-covered options.
Next step: Read our Wegovy vs Zepbound comparison. For cash-pay, Wegovy via NovoCare ($199 intro / $349 standard) and Zepbound via LillyDirect ($299–$449 vials) are the most affordable legitimate paths.
Find which one your insurance covers — free →Pick a path, not just a drug, if this sounds like you
You're paying cash, you're going to use a GLP-1 anyway, and your top constraint is monthly price.
Why path matters more than drug name:
Your cheapest legitimate FDA-approved options in April 2026:
- Wegovy via NovoCare: $199/mo intro for first 2 months at 0.25–0.5 mg (through June 30, 2026), then $349/mo standard. Approved for chronic weight management.
- Zepbound vials via LillyDirect: $299/mo (2.5 mg), $399/mo (5 mg), $449/mo (7.5–15 mg with the Self Pay Journey Program). Approved for chronic weight management and OSA.
- Ozempic via NovoCare if you specifically need semaglutide injection labeled for diabetes: $349/mo (0.25–1 mg), $499/mo (2 mg).
- Mounjaro: Currently no manufacturer cash-pay program. List price $1,112.16/fill.
Next step: Ro Body offers Ozempic, Wegovy, Zepbound, and Foundayo at the same NovoCare and LillyDirect pricing, plus insurance navigation.
See Ro Body pricing →Ask about switching if this sounds like you
You're already on Ozempic and you've plateaued. The weight stopped moving 4–6 months ago. Your A1C drift is creeping up. You're not getting the satiety effect you used to.
Why switching is reasonable: Real-world data and SURPASS-2 both suggest tirzepatide produces additional weight loss in patients who plateau on semaglutide. Switching is medically sound when there's a clinical reason.
Next step: Read the switching section below before you ask. Don't try to self-convert doses.
Read the switching guide →Take the matching quiz if you're not sure where you fit
You read everything above and you still don't know if you're a Mounjaro person, an Ozempic person, a Zepbound person, or a Wegovy person. That's normal — the drug landscape has changed three times in 18 months.
Take our free 60-second GLP-1 matching quiz. It asks about your goals, insurance, medical history, and budget, then routes you to the medication and provider path that fits your specific situation. No email required to see your result.
Find my GLP-1 path →Can you switch from Ozempic to Mounjaro (or Mounjaro to Ozempic)?
Answer up front
Yes — under your clinician's guidance. There's no FDA-approved or universally accepted dose-conversion chart, so most clinicians start the new drug at a lower dose and titrate up over weeks to months. Expect 1–4 weeks of adjustment and possibly a return of early-treatment GI side effects. The four most common reasons people switch are plateau, side-effect intolerance, insurance changes, or a new comorbidity (CVD or CKD developing) that pushes the choice toward Ozempic.
Reasons people switch (in order of frequency)
- Plateau. Weight loss stalled on the current drug. Switching to the other class can reset progress.
- GI tolerability. Severe nausea, vomiting, or diarrhea on the current drug. Some people genuinely tolerate one molecule and not the other.
- Insurance coverage change. Your formulary changed at open enrollment, or your employer switched plans, and the drug you were on is no longer covered.
- New diagnosis. Heart disease or kidney disease appeared, and Ozempic's labeled indications now matter more.
How the switch usually works
Mounjaro stays in your system for about 25 days; Ozempic stays for about 5 weeks. Most clinicians:
- Wait one weekly dose cycle past your last shot of the old drug.
- Start you at a lower dose of the new drug than the equivalent on the old drug — often the lowest starting dose (Mounjaro 2.5 mg or Ozempic 0.25 mg).
- Re-titrate over 4-week intervals, checking tolerance.
If you're switching because of GI side effects, your clinician may want symptoms to fully resolve before starting the new drug — that can mean a 2–4 week pause.
Things to know before you ask
- No validated dose-equivalence chart. Don't trust internet graphics that say "Ozempic 1 mg = Mounjaro 5 mg." There isn't one.
- Switching may not restart weight loss. Most people see additional loss; some see equivalent results; a few see worse. There's no guarantee.
- Plan for the insurance back-and-forth. A switch often triggers a new prior authorization. Build in 2–4 weeks before your current supply runs out.
- GI side effects can return on the new drug. Restarting at a lower dose helps, but the first 4–8 weeks of any new GLP-1 typically include nausea on dose increases.
Switching to Ozempic from Mounjaro? Ro can handle the prior authorization and step therapy paperwork for Ozempic, and the insurance concierge process takes about 2–3 weeks. (For switching the other way — to Mounjaro — you'll work directly with your prescriber, since Ro doesn't currently offer Mounjaro.)
Have Ro check my Ozempic coverage →For a deeper switching framework, see our How to Switch from Semaglutide to Tirzepatide guide.
Can you take Mounjaro and Ozempic together?
Answer up front
No. Don't combine GLP-1 receptor agonists unless a prescribing clinician specifically directs you to. They're both incretin-pathway drugs that work on overlapping mechanisms; stacking them increases the risk of severe GI side effects, hypoglycemia, and dehydration without adding meaningful efficacy. The right move is choosing one — not running both.
People ask this question for predictable reasons: they think more must be better, they're plateauing and looking for any edge, they're switching and confused about overlap, or they have leftover pens. None of these are reasons to combine the drugs. If you're plateauing, ask about switching. If you're switching, your clinician will tell you when to stop the first drug and start the second. If you have leftover pens from a previous prescription, don't use them to stack therapy — store them per the label until your clinician advises otherwise, or use an approved medication take-back option for safe disposal.
How we made this comparison (methodology)
We're showing our work because health pages should.
FDA labels for both drugs were pulled from DailyMed (the NIH's official drug-label database) on April 29, 2026. Anything we say about FDA-approved indications, dosing, or safety warnings is sourced to the current label.
Head-to-head clinical evidence is from the SURPASS-2 trial, published June 25, 2021 in The New England Journal of Medicine. Real-world reinforcement comes from a 2024 JAMA Internal Medicine study comparing tirzepatide to semaglutide in routine clinical practice. Cardiovascular outcomes data on tirzepatide comes from SURPASS-CVOT.
Side-effect rates are from each drug's current FDA prescribing information. We disclosed that rates aren't directly comparable across separate trials.
Pricing was verified on the official manufacturer pages on April 29, 2026:
- Mounjaro: pricinginfo.lilly.com/mounjaro and the Mounjaro FAQ page.
- Ozempic: novocare.com (Ozempic Savings Offer page and explaining-list-price page).
Insurance and Medicare coverage rules were verified via CMS.gov, current Medicare Part D plan documents, and the FDA's published rules on manufacturer savings cards.
The November 2025 White House pricing agreement implementation status was verified via the Novo Nordisk press release ("Novo Nordisk launches introductory self-pay offer for Wegovy and Ozempic for $199 per month," November 17, 2025) and Eli Lilly's announcement of the same.
Provider details for Ro were verified directly on ro.co, including the GLP-1 Insurance Coverage Checker, Ro Body pricing, the Ro Mounjaro page (which states Ro does not currently offer Mounjaro), and the insurance navigation timeline.
What we didn't do: We didn't take payment from any drug manufacturer in exchange for placement. We didn't fabricate testimonials. We didn't cite forum posts as evidence for medical claims — we used Reddit and patient-forum quotes only to surface the real questions readers are asking, with explicit framing that they aren't medical evidence.
Our editorial judgment standard: Editorial conclusions on this page are based on verified label facts, published evidence, and current public pricing/access information. They are not medical advice and do not replace a clinician's judgment.
Our re-verification cadence: Every commercial number on this page is re-checked on a 30-day cycle. The "Last verified" date at the top of the page updates when we re-check. If you're reading this 6+ months after that date, treat the pricing numbers as approximate and click through to the manufacturer page to confirm current pricing.
Mounjaro vs Ozempic FAQ
Is Mounjaro better than Ozempic?
For most adults with type 2 diabetes whose main goal is the largest A1C drop and the most weight loss, Mounjaro is the stronger first ask, based on the SURPASS-2 head-to-head trial. For adults with T2D plus established heart disease or chronic kidney disease, Ozempic is the stronger first ask because of its FDA-approved cardiovascular and kidney indications. Neither is universally better.
Is Mounjaro better than Ozempic for weight loss?
In adults with type 2 diabetes, yes — Mounjaro produced about twice the weight loss of Ozempic 1 mg in SURPASS-2 (11.2 kg vs 5.7 kg over 40 weeks). The gap narrows somewhat at Ozempic's higher 2 mg dose. If your only goal is weight loss and you don't have diabetes, the more accurate comparison is Zepbound vs Wegovy, not Mounjaro vs Ozempic.
Is Ozempic safer than Mounjaro?
Neither is universally safer. Both share the same FDA boxed warning for thyroid C-cell tumors and similar warnings about pancreatitis, gallbladder disease, kidney injury, and hypoglycemia when combined with insulin. Side-effect rates overlap, with Mounjaro at 15 mg producing slightly higher rates of nausea, vomiting, and diarrhea than Ozempic at typical doses. Tolerability is highly individual.
Which has fewer side effects?
There's no universal answer. Most people experience GI side effects on either drug during dose increases that fade within weeks. Some tolerate one drug and not the other. Your clinician will help you titrate slowly and adjust if side effects don't resolve.
Does Ozempic protect the heart?
Yes, when used in adults with type 2 diabetes plus established cardiovascular disease. Ozempic's FDA label includes reducing the risk of major adverse cardiovascular events (heart attack, stroke, cardiovascular death) in this population. Mounjaro has not received this indication.
Does Ozempic help protect the kidneys?
Yes, when used in adults with type 2 diabetes plus chronic kidney disease. The injection form of Ozempic is FDA-approved to reduce the risk of kidney disease worsening, kidney failure, and cardiovascular death in this population. Mounjaro has not received this indication.
Is Mounjaro approved for weight loss?
No. Mounjaro is FDA-approved for type 2 diabetes only. Zepbound — the same molecule (tirzepatide) under a different brand — is the FDA-approved version for chronic weight management and obstructive sleep apnea.
Is Ozempic approved for weight loss?
No. Ozempic is FDA-approved for type 2 diabetes (with cardiovascular and kidney indications in specific T2D populations). Wegovy — the same molecule (semaglutide) under a different brand — is the FDA-approved version for chronic weight management and cardiovascular risk reduction in adults with overweight or obesity plus heart disease.
How much does Mounjaro cost without insurance in 2026?
List price is $1,112.16 per 28-day fill (one fill = four pens). As of April 2026, Eli Lilly does not offer a direct-to-consumer cash-pay program for Mounjaro. Pharmacy discount cards (GoodRx, SingleCare) bring the price to roughly $900–$1,100/month. Patients seeking cash-pay tirzepatide for weight loss often use LillyDirect's Zepbound vial program ($299–$449/month) — a different FDA-approved drug for a different FDA-approved use.
How much does Ozempic cost without insurance in 2026?
Through NovoCare, uninsured or self-paying patients pay $349/month for the 0.25, 0.5, or 1 mg pen, or $499/month for the 2 mg pen. New patients can pay $199/month for the first two months at 0.25 mg or 0.5 mg through June 30, 2026.
Can you switch from Ozempic to Mounjaro?
Yes, with your clinician's guidance. You'll typically restart at a lower Mounjaro dose and titrate up over weeks. Expect possible new GI side effects during the adjustment period.
Can you take Mounjaro and Ozempic together?
No. Don't combine GLP-1 receptor agonists unless a clinician specifically directs you to. The risks (severe GI side effects, hypoglycemia, dehydration) outweigh any potential benefit.
Does Ro offer Mounjaro?
No. According to Ro's own Mounjaro page, Ro does not currently offer Mounjaro. Ro can prescribe and provide insurance support for Ozempic, Wegovy, Zepbound, and Foundayo. For Mounjaro specifically, work with your primary care or specialist clinician and use the Lilly Mounjaro Savings Card if you're commercially insured.
Is there a generic version of Mounjaro or Ozempic in the U.S.?
In the U.S., there is no FDA-approved generic version of tirzepatide or semaglutide as of April 2026. Both are patent-protected. Generic entry is years away in the U.S.
What about compounded semaglutide or tirzepatide?
The FDA enforcement-discretion period that allowed widespread compounded GLP-1s during the shortage has ended. The 503A enforcement-discretion period for tirzepatide ended after the March 2025 court update, and 503B ended on March 19, 2025. For semaglutide, 503A ended after the April 2025 court update, and 503B on May 22, 2025. Mass-market compounded copies of Mounjaro or Ozempic are no longer permitted.
Will Medicare cover Mounjaro or Ozempic?
For type 2 diabetes, yes — both are typically covered under Part D plans. For Mounjaro specifically, Eli Lilly reports about 8 in 10 Medicare Part D patients pay $0–$50 per 28-day supply. The 2026 Part D out-of-pocket cap on covered drugs is $2,100. For weight loss alone, neither is covered under standard Part D. The new Medicare GLP-1 Bridge launching July 1, 2026 will offer a $50/month copay for eligible weight-management uses outside Part D.
Are Mounjaro and Wegovy / Ozempic and Zepbound the same drug?
Same active ingredients, different FDA-approved uses. Mounjaro and Zepbound are both tirzepatide; Ozempic and Wegovy are both semaglutide. The drugs are produced by the same manufacturers in the same molecule, but they're approved for different indications, dosed differently, and priced differently.
Still not sure which GLP-1 is right for you?
You've made it this far. You know more about Mounjaro vs Ozempic than 99% of the people who searched for it today. If the right answer for you is still fuzzy, that's because the right answer depends on your situation — and we don't know yours.
Take our free 60-second GLP-1 matching quiz. It asks about your goals (weight loss, blood sugar, both), your insurance, your medical history, your budget, and your tolerance for monthly injections. Then it routes you to the FDA-approved medication and provider path that actually fits — Mounjaro, Ozempic, Zepbound, Wegovy, Foundayo, or a different option entirely. No email required to see your result.
Take the free 60-second quiz →If you already know what you need, here's where to go next:
- Insurance coverage check for Ozempic, Wegovy, and Zepbound (free, all in one report): Run my coverage check →
- Cash-pay branded GLP-1 access (Wegovy, Zepbound, Foundayo, Ozempic at NovoCare/LillyDirect pricing, with insurance concierge): Start a Ro Body intake →
- Switching from one to the other: How to Switch from Semaglutide to Tirzepatide
- Weight-loss-only readers: Wegovy vs Zepbound: 2026 Comparison
- Live pricing tracker: GLP-1 Pricing Change Log (coming soon — updated each time NovoCare or LillyDirect posts a new price).
This page was produced by The RX Index Editorial Team. The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We re-verify every commercial fact on this page on a 30-day cadence; the "Last verified" date at the top of the page updates when we re-check.
Last verified: .
Sources
- Eli Lilly. "Mounjaro Cost Information | With or Without Insurance." pricinginfo.lilly.com/mounjaro and mounjaro.lilly.com/faq. Mounjaro list price: $1,112.16 per 28-day fill (one fill = four pens).
- Novo Nordisk. "Ozempic Pricing." novopricing.com/ozempic.html. Ozempic WAC: $1,027.51 per fill.
- Frías JP, Davies MJ, Rosenstock J, et al. "Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes." N Engl J Med 2021;385:503-515. SURPASS-2.
- DailyMed. "MOUNJARO (tirzepatide) injection prescribing information." Updated 2026. dailymed.nlm.nih.gov.
- NovoCare. "Get savings and support for Ozempic." novocare.com/diabetes/products/ozempic/savings-offer.html. Self-pay tiers and intro offer dates.
- Ro. "GLP-1 Insurance Coverage Checker." ro.co/weight-loss/glp1-insurance-checker.
- DailyMed. "OZEMPIC (semaglutide) injection prescribing information." Updated 2026. dailymed.nlm.nih.gov.
- American College of Cardiology. "SURPASS-CVOT: Is Tirzepatide Superior to Dulaglutide in Patients With T2D and ASCVD?" Primary endpoint: 12% (tirzepatide) vs 13% (dulaglutide); HR 0.92; noninferiority met, superiority not met.
- Novo Nordisk press release, November 17, 2025: "Novo Nordisk launches introductory self-pay offer for Wegovy and Ozempic for $199 per month." Eli Lilly Zepbound LillyDirect pricing announcement, November 2025.
- Centers for Medicare & Medicaid Services. 2026 Part D benefit redesign and out-of-pocket cap. Medicare GLP-1 Bridge effective July 1, 2026. cms.gov.
- Ro. "Weight Loss Program Pricing." ro.co/weight-loss/pricing. Ro Body membership: $39 first month, then $149/month or as low as $74/month with annual prepay.
- Ro. "Mounjaro for Weight Loss" page. ro.co/weight-loss/mounjaro. Ro states Mounjaro is not currently offered.
- Frías JP, Auerbach P, Bajaj HS, et al. "Efficacy and safety of once-weekly semaglutide 2.0 mg versus 1.0 mg in patients with type 2 diabetes (SUSTAIN FORTE)." Lancet Diabetes Endocrinol 2021. Weight loss: 6.9 kg vs 6.0 kg at week 40 (trial-product estimand).
- Rodriguez PJ, Goodwin Cartwright BM, Gratzl S, et al. "Semaglutide vs Tirzepatide for Weight Loss in Adults With Overweight or Obesity." JAMA Internal Medicine, 2024.
- Ro. Insurance navigation page. ro.co/weight-loss/insurance. Ro states the concierge process takes about 2–3 weeks.
- U.S. Food and Drug Administration. "FDA clarifies policies for compounders as national GLP-1 supply begins to stabilize." fda.gov.