Semaglutide vs Tirzepatide Dosage Chart: the 2026 Dose Comparison That Actually Helps
The honest answer first
There is no validated semaglutide vs tirzepatide dosage chart that converts milligrams between these two drugs. 2.4 mg of semaglutide and 2.5 mg of tirzepatide are not “about the same.” They are different molecules with different receptor targets, different binding strengths, and different potency scales.
For weight loss, the best direct obesity evidence compares tirzepatide 10–15 mg with semaglutide 1.7–2.4 mg in the SURMOUNT-5 trial: tirzepatide produced 20.2% vs 13.7% average weight loss over 72 weeks.¹
If your top priority is the highest average weight-loss result at standard doses, tirzepatide has the stronger direct evidence. If you want a semaglutide path, a needle-free pill option, or an FDA-labeled cardiovascular risk reduction indication, semaglutide now has real advantages it didn't have a year ago. This page is for weight-loss decision-making, not a DIY prescribing chart. Every dose change should be guided by a licensed provider.
Jump to: Overview table · No real conversion · Dose ladders · Trial evidence · 5 mg vs 2.4 mg · Which path fits · Switching · Pricing · Units vs mg · FAQ

Semaglutide vs tirzepatide: 2026 overview
| Semaglutide injection (Wegovy) | Semaglutide pill (Wegovy pill) | Tirzepatide injection (Zepbound) | |
|---|---|---|---|
| Best for | FDA-labeled MACE reduction in obesity/overweight with CVD + weight loss; new HD option for plateaus | Needle-free; low FDA-approved entry price ($149/mo) | Highest average weight loss at standard doses |
| Obesity dose ladder | 0.25 → 0.5 → 1.0 → 1.7 → 2.4 mg weekly; 7.2 mg HD after 4+ wks on 2.4 | 1.5 → 4 → 9 → 25 mg daily | 2.5 → 5 → 7.5 → 10 → 12.5 → 15 mg weekly |
| Direct head-to-head obesity trial vs the other? | Yes — at 1.7/2.4 mg (SURMOUNT-5)¹. Not yet for 7.2 mg HD | No direct trial vs tirzepatide | Yes — at 10/15 mg (SURMOUNT-5)¹ |
| Oral option? | Yes (Wegovy pill) | This IS the oral option | No. Foundayo (orforglipron) is a different oral GLP-1, approved April 1, 2026 — not oral tirzepatide |
| Cash-pay entry (April 2026) | ~$199/mo starter (NovoCare intro); HD at $399/mo | $149/mo for 1.5 mg and 4 mg via NovoCare | $299/mo at 2.5 mg; $399 at 5 mg; $449 at 7.5–15 mg (LillyDirect Self Pay Journey) |
| Biggest tradeoff | Lower average weight loss than tirzepatide at standard doses; HD has dysesthesia risk (~22%) | ~14% average weight loss — lower ceiling than injections of either drug | No FDA-labeled MACE indication; no oral option; no 7.2 mg equivalent |
Sources: ¹SURMOUNT-5, NEJM 2025. NovoCare pricing verified April 2026. Zepbound.lilly.com/savings verified April 2026. Wegovy prescribing information 2026 label. FDA approval of Foundayo, April 1, 2026.
Is there a real semaglutide-to-tirzepatide conversion chart?
Annoying truth
This page is not going to give you one magic number that converts semaglutide milligrams into tirzepatide milligrams. That chart does not exist in validated form. No regulatory body has published one, no head-to-head trial was designed to produce one, and the clinical switching guidance that does exist says to use individualized titration rather than a formula.
The reason the internet is full of “approximate equivalency” charts is because people desperately want one. The charts you find on other pages are extrapolations from clinical experience and trial dose arms — not validated conversion ratios. Some of those extrapolations are reasonable. But presenting them as settled facts, which most pages do, is overselling the certainty.
The RX Index Confidence Matrix: direct evidence vs extrapolation

Is 5 mg tirzepatide equal to 1 mg semaglutide?
SURPASS-2 directly compared those doses in type 2 diabetes patients on metformin. Tirzepatide 5 mg performed better on average for A1C and weight. But SURPASS-2 was a diabetes trial, not an obesity trial.
Is 10 or 15 mg tirzepatide stronger than 1.7/2.4 mg semaglutide for obesity?
SURMOUNT-5 directly compared max tolerated tirzepatide (10 or 15 mg) vs max tolerated semaglutide (1.7 or 2.4 mg) in adults with obesity without diabetes. Tirzepatide produced 20.2% vs 13.7% average weight loss at 72 weeks.
Is 2.5 mg tirzepatide equal to 0.25 mg semaglutide?
Both are labeled initiation doses meant for GI tolerance building. Neither is a therapeutic maintenance dose. There is no direct equivalence trial. Calling them 'equivalent' because they're both starter doses is like saying a car in first gear and a truck in first gear are going the same speed.
What tirzepatide dose matches Wegovy pill 25 mg?
No direct obesity head-to-head trial exists between the Wegovy pill (any dose) and any tirzepatide dose.
What tirzepatide dose matches Wegovy HD 7.2 mg?
No direct head-to-head trial exists. Cross-trial reference: Wegovy HD produced an 18.8% LS mean weight reduction from baseline in Study 8 per Wegovy PI, while tirzepatide max tolerated dose produced 20.2% in SURMOUNT-5. Different trial populations and designs make this an inference, not equivalence.
Sources: SURMOUNT-5 (NEJM, 2025); SURPASS-2 (NEJM, Frías et al. 2021); Wegovy prescribing information 2026 label (Study 8); STEP UP (Lancet Diabetes & Endocrinology, Nov 2025); Whitley HP, Clinical Diabetes 2023;41(3):467-473; Pharmaceutical Journal switching guidance.
What does the 2026 semaglutide vs tirzepatide dosage chart actually look like?
Here are the three current FDA-approved obesity dose ladders as of April 2026, including options that most competing pages haven't added yet.
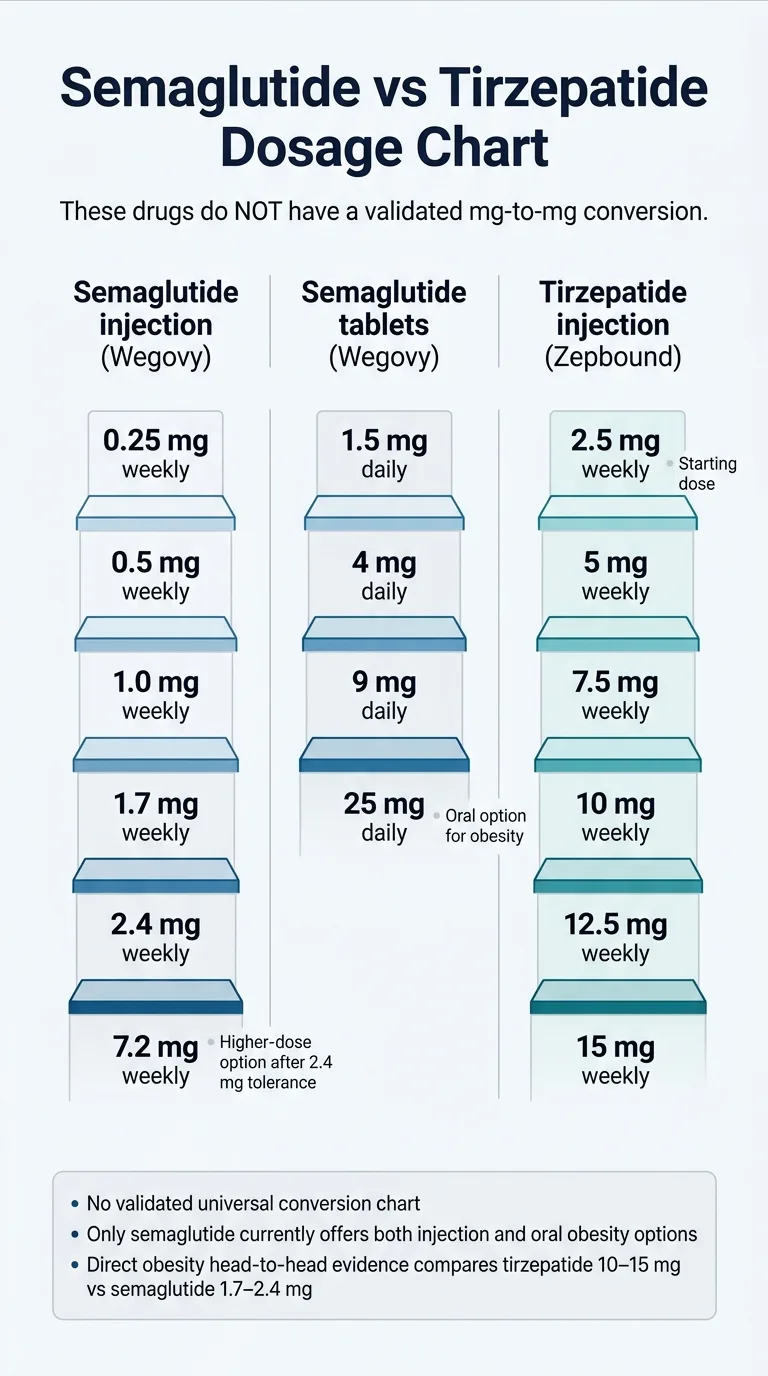
Semaglutide injection (Wegovy / Wegovy HD)
| Week | Dose | Purpose |
|---|---|---|
| Weeks 1–4 | 0.25 mg weekly | Initiation — GI tolerance building |
| Weeks 5–8 | 0.5 mg weekly | Titration |
| Weeks 9–12 | 1.0 mg weekly | Titration |
| Weeks 13–16 | 1.7 mg weekly | First maintenance option |
| Week 17+ | 2.4 mg weekly | Standard maintenance |
| Week 21+ (after ≥4 wks at 2.4 mg) | 7.2 mg weekly (Wegovy HD) | Higher maintenance — only if tolerated 2.4 mg and additional weight loss is clinically indicated |
Source: FDA-approved Wegovy prescribing information (2026 label revision). Wegovy HD approved March 19, 2026.
Semaglutide reaches standard 2.4 mg maintenance at approximately week 17. Reaching the new 7.2 mg HD dose takes at least 21 weeks if titrated as fast as the label allows.
Semaglutide pill (Wegovy oral tablet)
| Day | Dose | Purpose |
|---|---|---|
| Days 1–30 | 1.5 mg daily | Initiation |
| Days 31–60 | 4 mg daily | Titration |
| Days 61–90 | 9 mg daily | Titration |
| Day 91+ | 25 mg daily | Maintenance |
Source: FDA-approved Wegovy oral tablet prescribing information. Launched January 2026.
Important: The Wegovy pill must be taken on an empty stomach with no more than 4 oz of water, and you must wait 30 minutes before eating, drinking, or taking other oral medications. The pill reaches maintenance at approximately day 91 — about 13 weeks.
Tirzepatide injection (Zepbound)
| Week | Dose | Purpose |
|---|---|---|
| Weeks 1–4 | 2.5 mg weekly | Initiation — not a maintenance dose |
| Weeks 5–8 | 5 mg weekly | First maintenance option |
| Weeks 9–12 | 7.5 mg weekly | Titration |
| Weeks 13–16 | 10 mg weekly | Common maintenance |
| Weeks 17–20 | 12.5 mg weekly | Titration |
| Week 21+ | 15 mg weekly | Maximum maintenance |
Source: FDA-approved Zepbound prescribing information (2025 label revision).
Key insight from these ladders: Semaglutide reaches its standard maintenance dose (2.4 mg) slightly sooner than tirzepatide reaches common maintenance (10 mg). But both drugs can take roughly 20–21 weeks to reach their highest approved obesity dose if titrated as fast as allowed.
Only semaglutide currently gives you both an injection and a pill path for obesity. Foundayo (orforglipron) is an oral GLP-1 approved April 1, 2026 — but it is a different molecule than tirzepatide.
Free GLP-1 coverage check — Wegovy, Zepbound, Ozempic, and Mounjaro
Which doses were actually compared in clinical trials?
This is where most “conversion charts” fall apart. Only a few specific dose pairings have been directly tested against each other — and the context of those trials matters.
Obesity: the one head-to-head that matters most
SURMOUNT-5 — the only completed head-to-head obesity trial
Study population
751 adults with obesity, no type 2 diabetes
Duration
72 weeks
Tirzepatide result
20.2% avg weight lost
Semaglutide result
13.7% avg weight lost
Waist: tirzepatide
−18.4 cm
Waist: semaglutide
−13.0 cm
Source: SURMOUNT-5, NEJM 2025; ClinicalTrials.gov NCT05822830. Tirzepatide: max tolerated 10 or 15 mg. Semaglutide: max tolerated 1.7 or 2.4 mg.
That 6.5 percentage-point gap is not small. For someone starting at 250 lbs, it's roughly the difference between losing 34 lbs (semaglutide) and losing 50 lbs (tirzepatide) on average. More tirzepatide patients also achieved ≥10%, ≥15%, ≥20%, and ≥25% weight loss thresholds.
Type 2 diabetes: useful context, different question
SURPASS-2 (NEJM, Frías et al. 2021) compared tirzepatide 5, 10, and 15 mg against semaglutide 1 mg — not 2.4 mg — in patients with type 2 diabetes on metformin.
| Drug & dose | Weight loss at 40 weeks | A1C reduction |
|---|---|---|
| Semaglutide 1 mg | −5.7 kg | ~1.9% |
| Tirzepatide 5 mg | −7.6 kg | ~2.0% |
| Tirzepatide 10 mg | −9.3 kg | ~2.2% |
| Tirzepatide 15 mg | −11.2 kg | ~2.5% |
This gives you a “dose anchor” — tirzepatide 5 mg beat semaglutide 1 mg in a diabetes population — but it does not tell you what happens in obesity at different dose combinations.
What this still does NOT tell you
- ✗No direct obesity head-to-head for Wegovy pill (oral semaglutide 25 mg) vs any tirzepatide dose
- ✗No direct head-to-head for Wegovy HD 7.2 mg vs any tirzepatide dose
- ✗No trial that validates a universal dose-conversion ratio for switching between drugs
Is 5 mg tirzepatide equal to 1 mg or 2.4 mg semaglutide?
This is the most-searched specific dose question. Here's the evidence-based answer in three parts.
5 mg tirzepatide vs 1 mg semaglutide
Medium confidence — diabetes evidence only
Directly compared in SURPASS-2 — but only in type 2 diabetes patients on metformin, not in an obesity-focused trial. Tirzepatide 5 mg produced more weight loss (7.6 kg vs 5.7 kg) and better A1C reduction. That makes tirzepatide 5 mg the 'winner' in that specific comparison, but it doesn't make them 'equivalent.' They were compared; one performed better.
5 mg tirzepatide vs 2.4 mg semaglutide
Low confidence — no direct trial
Not directly compared head-to-head in any published trial. Any chart that presents this as a settled equivalence is overselling certainty. The closest support comes from indirect treatment comparisons, which are statistical estimates, not direct evidence. SURMOUNT-5 compared higher tirzepatide doses (10/15 mg) against semaglutide 1.7/2.4 mg — not tirzepatide 5 mg.
2.5 mg tirzepatide vs 0.25 mg semaglutide
Low confidence — same role, not equivalent potency
Both are labeled initiation doses designed for GI tolerance building. Neither is meant to produce meaningful weight loss on its own. Calling them 'equivalent' because they're both starter doses is like saying a car in first gear and a truck in first gear are going the same speed. Same purpose, completely different machines.
Bottom line: If someone tells you “5 mg tirzepatide = X mg semaglutide,” ask them which trial proved it and for which condition. If they can't answer, the number is an estimate — possibly a useful one, but not a fact.
Semaglutide or tirzepatide: which path fits your situation?
The right answer depends less on pharmacology and more on your specific constraints.
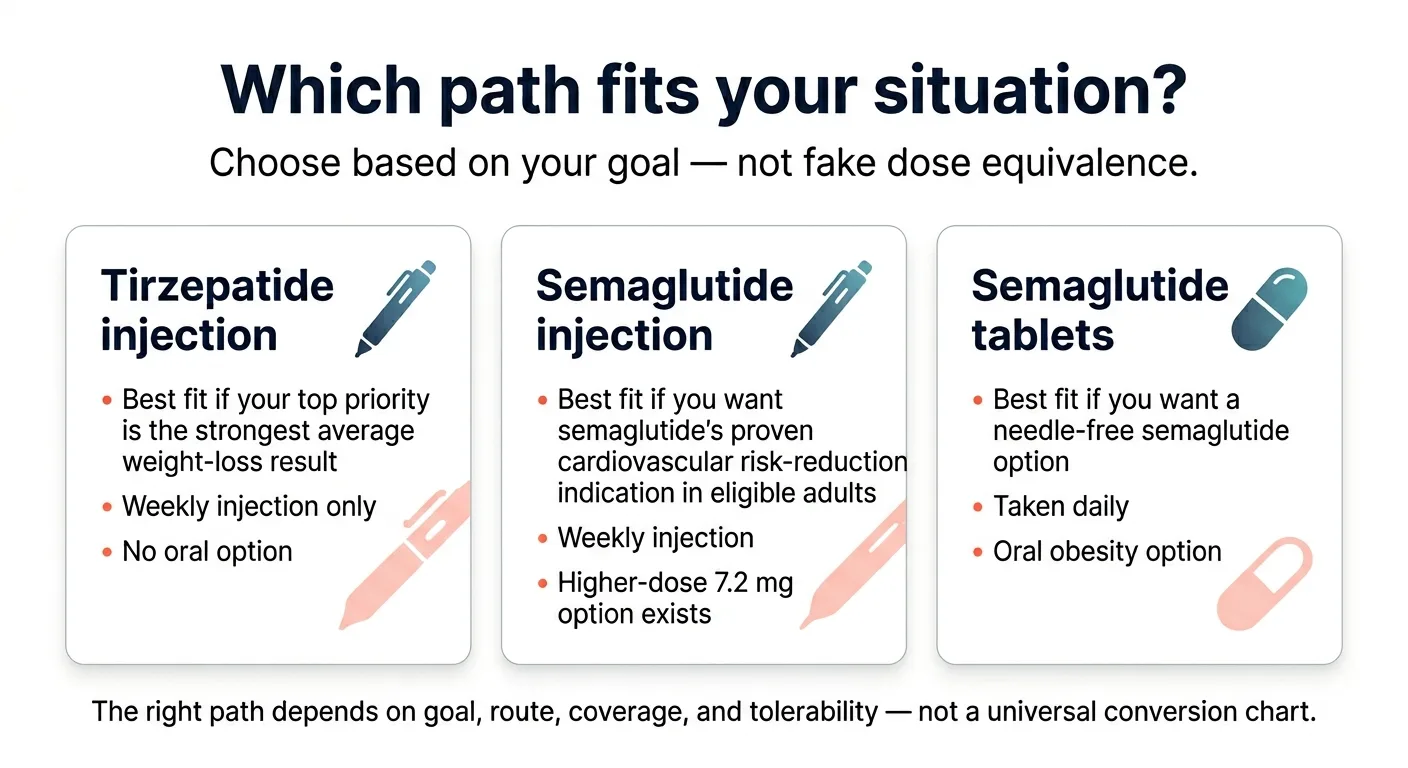
Pick tirzepatide if your #1 priority is the strongest average weight-loss ceiling
Tirzepatide's dual GIP/GLP-1 receptor mechanism produces meaningfully higher average weight loss than semaglutide at standard max doses. SURMOUNT-5 showed 20.2% vs 13.7%.¹ SURMOUNT-1 showed up to 22.5% average weight loss at the 15 mg dose over 72 weeks. If maximizing total weight lost is your driving goal and you can handle weekly injections, tirzepatide has the strongest published evidence.
Includes insurance navigation and GLP-1 concierge for prior authorizations
Pick semaglutide if a pill option, cardiovascular protection, or delivery flexibility matters more
Semaglutide now offers something no other GLP-1 weight-loss drug does: three distinct delivery paths. You can start with the Wegovy pill at $149/month, move to injectable Wegovy at 2.4 mg, and — if you plateau — escalate to Wegovy HD 7.2 mg. That full semaglutide pathway didn't exist 6 months ago.
Wegovy also has an FDA-labeled indication for reducing the risk of major adverse cardiovascular events — heart attack, stroke, and cardiovascular death — in adults with established cardiovascular disease and obesity or overweight. That labeled indication is unique to Wegovy among GLP-1 weight-loss drugs as of April 2026.
Includes insurance navigation and NovoCare self-pay pricing pathway
Pick based on insurance if coverage is likely to decide the answer anyway
For most people, the drug their insurance covers — or covers at a lower copay — is the drug they should take. Both medications are highly effective. The difference between 15% and 20% average weight loss matters in clinical trials, but in the real world, the medication you can actually afford to stay on for 12+ months produces better results than the theoretically superior one you can't maintain.
Run a free GLP-1 coverage check through Ro →Personalized coverage report for Wegovy, Zepbound, Ozempic, and Mounjaro on your specific plan
Leave this page if your real question is diabetes management
This page is scoped to weight-loss dose comparison. If your primary goal is blood sugar control for type 2 diabetes, the dose ladders, brand names (Ozempic/Mounjaro vs Wegovy/Zepbound), and insurance dynamics are different. Read our Best GLP-1 for Diabetes guide →
If I'm switching, what dose do people usually start on?
Switching between semaglutide and tirzepatide is common — often triggered by a plateau, a cost change, an insurance formulary shift, or a shortage. Here's what the published guidance and real-world experience suggest.
What published switching guidance says
There is no validated semaglutide-to-tirzepatide equivalence for switching purposes. The most-cited clinical guidance (Whitley HP, Clinical Diabetes 2023) offers suggested comparative doses based on A1C effect, but these are estimates based on trial-arm dose anchors, not a validated conversion.
Conservative: start tirzepatide at 2.5 mg
Many clinicians default to the labeled initiation dose to minimize GI side effects, especially for patients with a history of GI sensitivity on their prior GLP-1.
Direct switch to 5 mg
There is published prospective evidence (Endocrine Practice 2024) for switching patients on stable GLP-1 RA therapy directly to tirzepatide 5 mg. Some switching guidance explicitly allows 5 mg as a starting dose for established GLP-1 users.
Universal rule
Do not jump to a high tirzepatide dose (10+ mg) to “match” a high semaglutide dose. Titrate up based on tolerability and response, not based on a conversion formula.
Additional switching considerations
- ·Wait at least one week after your last semaglutide dose (semaglutide's half-life is ~7 days)
- ·Do not switch from a submaximal dose of one drug to the maximum dose of another
Sources: Whitley HP, Clinical Diabetes 2023;41(3):467-473; Endocrine Practice 2024; Pharmaceutical Journal switching guidance.
Why people feel hungrier at first after switching
If you search Reddit or patient forums, you'll find a pattern: people switching from max-dose semaglutide to starting-dose tirzepatide describe returning “food noise,” increased hunger, and anxiety about regain during the re-titration period.
This is real. It is not a sign that tirzepatide is “weaker” — it's a sign that you're starting at an initiation dose while your body was adapted to a therapeutic dose of a different drug. The appetite suppression typically returns as the tirzepatide dose increases. Talk to your prescriber about whether starting at 5 mg (rather than 2.5 mg) is appropriate given your history.
Can you take semaglutide and tirzepatide together?
No. Zepbound's prescribing information explicitly states that coadministration with any other GLP-1 receptor agonist is not recommended. Semaglutide's label carries the same warning. Taking both simultaneously increases GI side effect risk without evidence of additive benefit.
No email required · Personalized recommendation
Does the dose change the cost enough to change the decision?
Yes — especially in April 2026, when manufacturer pricing has shifted dramatically.
| Drug path | Starter dose (self-pay) | Mid-range dose (self-pay) | Highest dose (self-pay) |
|---|---|---|---|
| Wegovy pill | $149/mo (1.5 mg and 4 mg via NovoCare) | $299/mo (25 mg maintenance) | $299/mo (25 mg is the max oral dose) |
| Wegovy injection | ~$199/mo for first fills (NovoCare intro) | ~$349/mo (2.4 mg via NovoCare) | $399/mo (7.2 mg Wegovy HD) |
| Zepbound injection | $299/mo (2.5 mg via LillyDirect) | $399/mo (5 mg) | $449/mo (7.5, 10, 12.5, or 15 mg) |
| Foundayo (orforglipron) | $149/mo (lowest dose via LillyDirect) | Dose-tiered, up to $399/mo | Up to $399/mo at highest doses |
| With commercial insurance + savings card | As low as $25/mo | As low as $25/mo | As low as $25/mo |
Sources: NovoCare pricing page verified April 2026. Zepbound.lilly.com/savings (Self Pay Journey terms) verified April 2026. Foundayo pricing per LillyDirect announcement, April 2026. Bloomberg April 7, 2026. Savings card terms per Wegovy.com and Zepbound.lilly.com/savings.
Why Wegovy HD pricing matters
At $399/month cash-pay, Novo Nordisk deliberately undercut Zepbound's highest-dose pricing ($449/month at 7.5–15 mg). For uninsured and self-pay patients, semaglutide's highest-dose injection path is now cheaper than tirzepatide's.
Medicare GLP-1 Bridge (July 1 – December 31, 2026)
CMS has confirmed the Medicare GLP-1 Bridge program covers all Wegovy formulations, all Foundayo formulations, and Zepbound KwikPen only (not single-dose vials) at a $50 copay for eligible Medicare Part D beneficiaries. This is the first time Medicare has covered GLP-1 medications for weight loss.
GLP-1 concierge handles prior authorizations and shows your exact out-of-pocket cost
What if your vial uses units instead of mg?
If you're using a compounded semaglutide or tirzepatide vial from a compounding pharmacy, you may see your dose expressed in “units” on an insulin syringe rather than milligrams. This is where dosing errors happen — and the FDA has issued specific warnings about it.
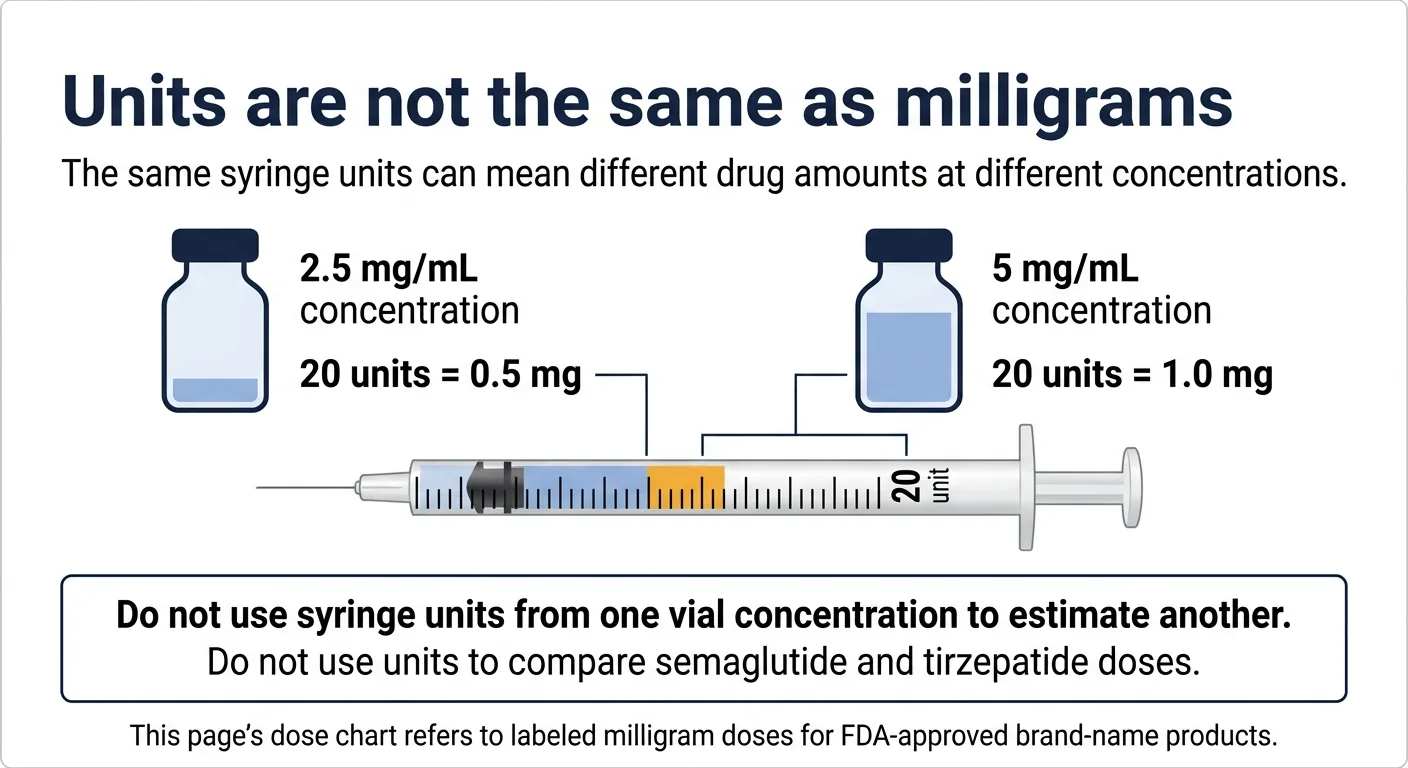
⚠️ FDA warning on compounded GLP-1 dosing errors
The FDA has warned about dosing errors, including hospitalizations, associated with compounded semaglutide. The confusion: “units” on an insulin syringe measure volume, not drug amount. The same number of units delivers a completely different milligram dose depending on the concentration of your vial.
Do not assume that a “units” number from one vial concentration applies to a different concentration. And definitely do not use a units-based dose from semaglutide to estimate a tirzepatide dose or vice versa. The drugs have different concentrations, different potency scales, and different vial formulations.
This dosage chart covers FDA-approved brand-name formulations expressed in milligrams, as labeled. If you're on a compounded formulation, the milligram dose should be confirmed by your prescribing provider and the dispensing pharmacy before you compare it to anything on this page.
How we built and verified this chart
Direct evidence
FDA-approved prescribing information (Wegovy 2026 label, Wegovy oral tablet, Zepbound 2025 label); SURMOUNT-5 (NEJM 2025); SURPASS-2 (NEJM 2021); STEP UP (Lancet Diabetes & Endocrinology, Nov 2025); SURMOUNT-1 (NEJM 2022); STEP-1 (NEJM 2021)
Switching guidance
Whitley HP, Clinical Diabetes 2023;41(3):467-473; Endocrine Practice 2024 (direct switch to tirzepatide 5 mg from prior GLP-1 RA); Pharmaceutical Journal switching guidance
Pricing sources
NovoCare Pharmacy official pricing (verified April 2026); LillyDirect / Zepbound.lilly.com official pricing (verified April 2026); Bloomberg report on Wegovy HD pricing (April 7, 2026)
Regulatory sources
FDA alert on compounded semaglutide dosing errors; FDA approval of Foundayo (orforglipron), April 1, 2026; CMS.gov Medicare GLP-1 Bridge program page (verified April 2026)
Statistical models
Adjusted indirect comparison, PMC 2022 (tirzepatide vs semaglutide 2 mg in T2D) — labeled as extrapolation throughout
What counts as direct evidence on this page: a dose pairing tested in a randomized trial with published results.
What counts as extrapolation: dose comparisons inferred from separate trials, adjusted indirect comparisons, or clinical experience. These are clearly labeled throughout.
Last verified: April 8, 2026. Updated for Wegovy HD 7.2 mg (approved March 19, 2026), Foundayo/orforglipron (approved April 1, 2026), Wegovy pill (launched January 2026), Medicare GLP-1 Bridge confirmation, and current 2026 cash-pay pricing. Next audit: May 8, 2026.
Frequently asked questions
Is there an official semaglutide-to-tirzepatide conversion chart?
No. No regulatory body or medical society has published a validated mg-to-mg conversion ratio. Published 'equivalent dose' charts are clinical estimates based on trial dose arms and clinical experience, not a validated formula. Use them as rough reference points with your prescriber, not as DIY switching instructions.
Is 5 mg tirzepatide the same as 1 mg semaglutide?
They were directly compared in SURPASS-2 (type 2 diabetes), and tirzepatide 5 mg outperformed semaglutide 1 mg on both A1C and weight. They are not 'the same' — they were compared, and one performed better. This comparison was in diabetes, not obesity.
Is 5 mg tirzepatide the same as 2.4 mg semaglutide?
This dose pairing has not been directly compared in any published trial. Any chart presenting them as equivalent is extrapolating from indirect data.
Can I switch from semaglutide to tirzepatide the next week?
Generally yes, with provider guidance. Wait at least one week after your last semaglutide dose (approximately one half-life). Your provider may start you at 2.5 mg (the labeled initiation dose) or at 5 mg if you were stable on a GLP-1 RA — there is published evidence supporting both approaches. Do not jump to a high tirzepatide dose to 'match' your semaglutide dose.
Can I take semaglutide and tirzepatide together?
No. Both prescribing labels advise against coadministration with other GLP-1 receptor agonists.
Which causes more weight loss on average — semaglutide or tirzepatide?
Tirzepatide, at standard max doses. SURMOUNT-5 showed 20.2% vs 13.7% for semaglutide at max tolerated doses. Wegovy HD 7.2 mg narrows that gap — the Wegovy prescribing information reports an 18.8% LS mean weight reduction from baseline — but a direct head-to-head of 7.2 mg semaglutide vs 15 mg tirzepatide has not been conducted.
Does the Wegovy pill change the semaglutide vs tirzepatide comparison?
It expands semaglutide's advantages — $149/mo entry price (tied with Foundayo as the lowest FDA-approved GLP-1 weight-loss entry point), no needles, oral daily dosing — but does not change the weight-loss comparison. The Wegovy pill produces approximately 14% average weight loss, which is lower than either injectable drug at max doses.
Does Wegovy HD 7.2 mg change the comparison?
Yes. Before Wegovy HD, semaglutide's maximum injectable weight loss was about 15–17% at 2.4 mg. The 7.2 mg dose produces an 18.8% LS mean weight reduction from baseline per the Wegovy prescribing information, narrowing the gap with tirzepatide's 20.2% in SURMOUNT-5. This is a cross-trial inference, not a head-to-head result. New tradeoff: Wegovy HD has a dysesthesia (skin sensitivity) rate of about 22% at 7.2 mg vs 6% at 2.4 mg.
What about the new oral GLP-1, Foundayo (orforglipron)?
Foundayo was FDA-approved on April 1, 2026. It is Eli Lilly's oral GLP-1 receptor agonist — a different molecule from tirzepatide. It starts at $149/month via LillyDirect. Clinical trials showed approximately 12.4% average weight loss at the highest dose. It gives you another oral option alongside the Wegovy pill, but it is not oral Zepbound.
What if my compounded vial uses units instead of mg?
Units on a syringe measure volume, not drug amount. The same number of units delivers different milligram doses at different vial concentrations. Never assume unit equivalence across concentrations or across drugs. Confirm your milligram dose with your pharmacy and prescriber before comparing it to any chart.
Which path is cheapest without insurance in April 2026?
Wegovy pill and Foundayo both start at $149/month, tied as the lowest FDA-approved GLP-1 weight-loss entry points. Wegovy HD at $399/month is cheaper than Zepbound at 7.5–15 mg ($449/month). For a full cost breakdown by insurance status, see our GLP-1 cost without insurance guide.
Will Medicare cover these drugs?
Starting July 1, 2026, the Medicare GLP-1 Bridge program covers Wegovy (all formulations), Foundayo (all formulations), and Zepbound (KwikPen only) at a $50 copay for eligible Part D beneficiaries. The program runs through December 31, 2026.
Still deciding?
If you've read this far, you have a more accurate picture of semaglutide vs tirzepatide dosing than most people who search for a conversion chart. The real decision isn't about finding the right mg-to-mg equivalent — it's about matching a path to your goals, your insurance, and your preferences.
No email required · Personalized recommendation
GLP-1 concierge can handle prior authorizations and help you get to $25/mo through insurance
Medical disclaimer: This content is for informational purposes only and does not replace professional medical advice, diagnosis, or treatment. Always consult your healthcare provider before starting, stopping, or changing any medication. Dosing information reflects FDA-approved prescribing information and published clinical trial data as of April 8, 2026.
Related guides
The RX Index is an independent editorial publisher covering GLP-1 medications and telehealth providers. We are not medical professionals. We may earn a commission if you visit a provider through our links, but this does not influence our editorial content.