INFLAMMATION & GLP-1 MEDICATIONS — 2026 GUIDE
Best GLP-1 for Inflammation: Which Drug Fits Your Type of Inflammation?
No GLP-1 is FDA-approved specifically for treating inflammation. But the anti-inflammatory evidence behind these medications is now strong enough that the question has shifted from "do GLP-1s reduce inflammation?" to "which GLP-1 is best for inflammation — and for my situation specifically?"
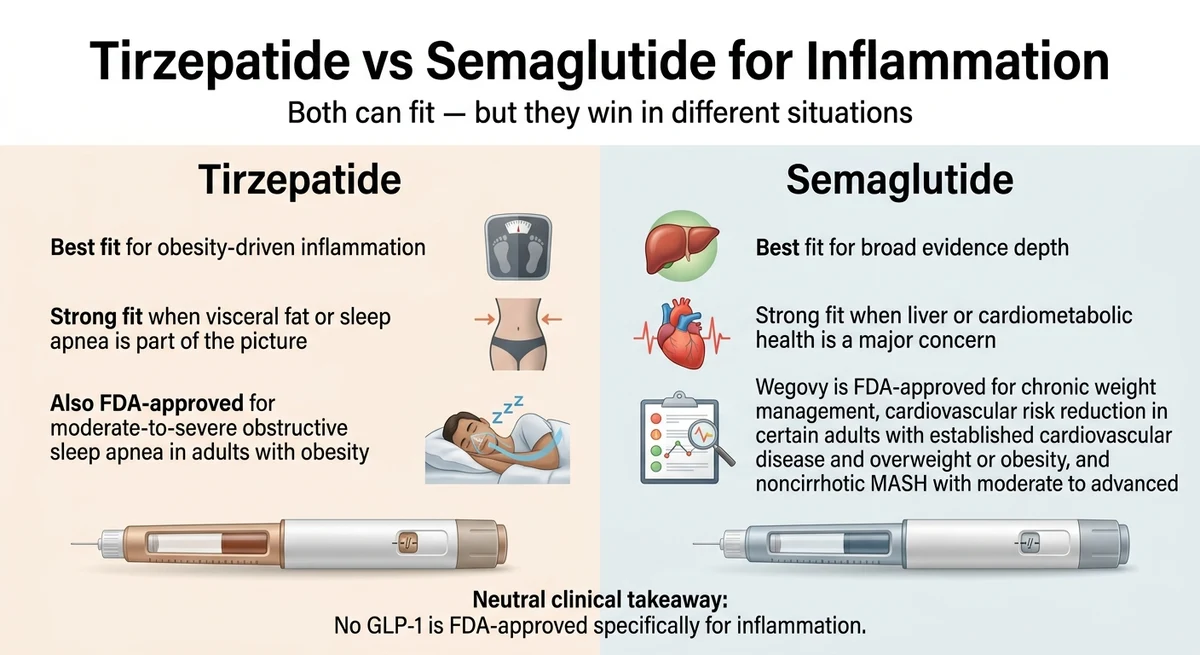
If obesity, visceral fat, or sleep apnea is fueling your inflammation: tirzepatide (Zepbound, Mounjaro) is the strongest practical pick. It delivers the most aggressive weight and waist reduction, removing the metabolic fuel that keeps inflammation elevated. Zepbound is also the first FDA-approved medication for moderate-to-severe obstructive sleep apnea in adults with obesity.
If you want the broadest published anti-inflammatory evidence — especially for liver and cardiovascular inflammation: semaglutide (Wegovy, Ozempic) is the better evidence-first choice. The SELECT trial showed up to 38% CRP reduction, often within weeks. Wegovy is FDA-approved for treating noncirrhotic MASH with moderate to advanced liver fibrosis (F2–F3) — the first GLP-1 approved for liver disease.
If budget matters most: liraglutide (Saxenda) is the established fallback. Generic liraglutide for weight management became available in 2025, improving the cost picture.
If your main problem is active autoimmune disease without significant metabolic drivers: a GLP-1 is an add-on at best, not the main treatment. Specialist care comes first.
By The RX Index Research Team · Last updated: March 28, 2026 · Pricing verified: March 28, 2026
Sources: JCI 2025 anti-inflammatory review; SELECT trial (NEJM); SURMOUNT-OSA (NEJM); ACR Open Rheumatology 2025; FDA prescribing information; Frontiers in Cardiovascular Medicine 2024 meta-analysis

Disclosure: Some links on this page are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you.
Bottom line
Tirzepatide (Zepbound/Mounjaro) wins for obesity-driven, visceral fat, and sleep apnea inflammation — strongest weight/waist reduction, dual GLP-1+GIP pathway, FDA-approved for OSA. Semaglutide (Wegovy/Ozempic) wins for evidence depth — SELECT trial 38% CRP reduction, FDA-approved for MASH and CV risk. No GLP-1 is FDA-approved specifically for inflammation.
Which GLP-1 Is Best for Inflammation?
The best answer depends on what is driving your inflammation. Find your situation in the table below.
| Your Inflammation Driver | Best GLP-1 Pick | Why | Evidence |
|---|---|---|---|
| Obesity / visceral fat / sleep apnea | Tirzepatide (Zepbound, Mounjaro) | Strongest weight/waist reduction removes the metabolic engine; dual GLP-1/GIP pathway; FDA-approved for OSA | Strong |
| Liver inflammation (MASH) / cardiovascular / broad cardiometabolic | Semaglutide (Wegovy, Ozempic) | Deepest human evidence; SELECT trial CRP data; FDA-approved for MASH and CV risk reduction | Strong |
| Budget-first / need affordable FDA-approved option | Liraglutide (Saxenda, Victoza) | Established, FDA-approved, generic available since 2025; moderate anti-inflammatory signal | Moderate |
| Rheumatoid / psoriatic arthritis / joint pain with obesity | Semaglutide or tirzepatide (depends on metabolic profile) | ACR 2025 data: fewer flares, improved disease activity; weight-independent mechanism suggested | Promising |
| Active autoimmune disease without metabolic drivers | Not GLP-1 first | Specialist treatment is primary; GLP-1 may help as an add-on if metabolic factors coexist | Limited |
This is not a ranking pulled from thin air. Below, we walk through the evidence for every row — study by study, condition by condition.
Not sure which row fits your situation?
Take the Free 60-Second GLP-1 Matching Quiz →How GLP-1 Medications Reduce Inflammation
Before you choose a drug, it helps to understand why weight-loss medications affect inflammation at all. There are two distinct pathways.
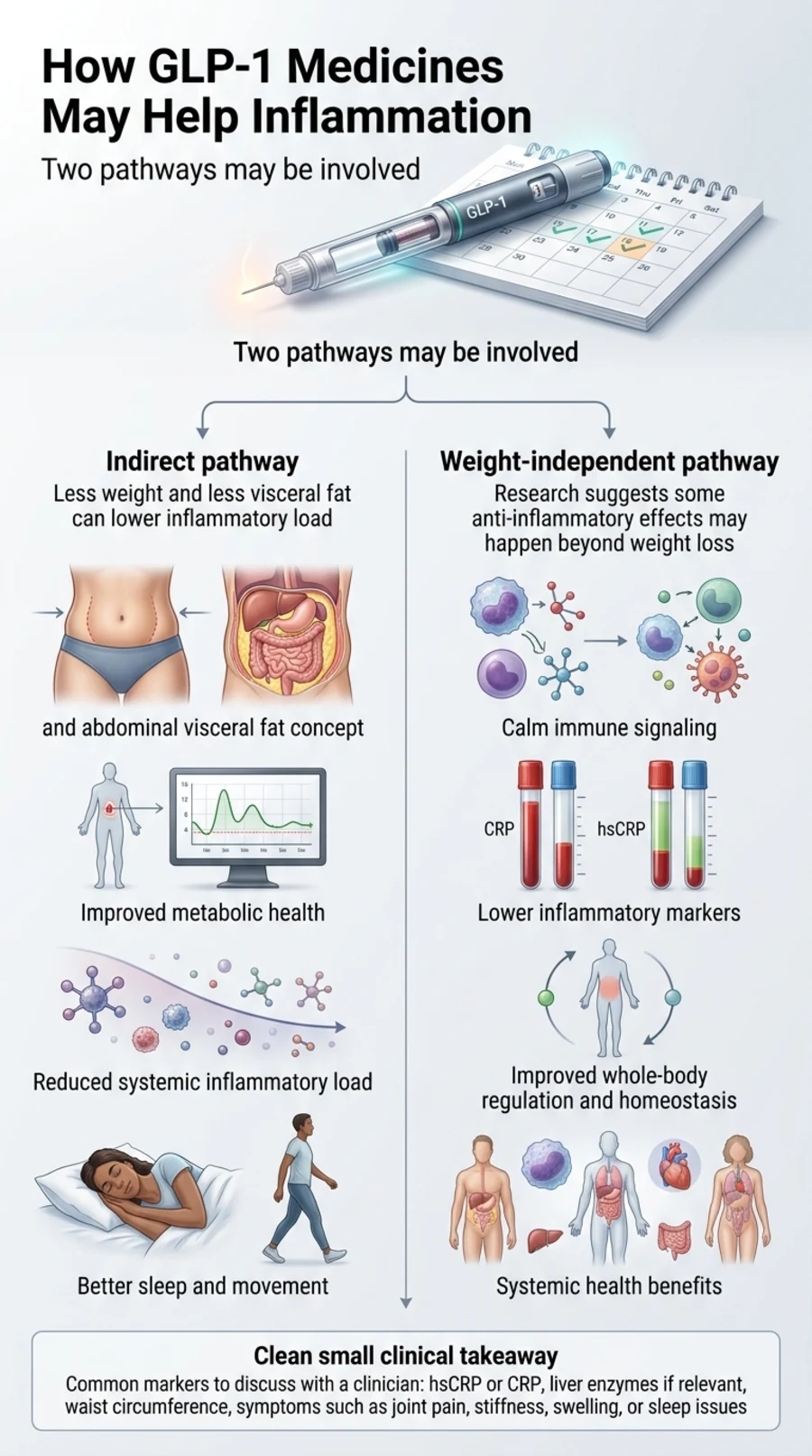
The indirect path: less fat means less inflammation
Visceral fat — the deep abdominal fat surrounding your organs — is not just storage. It functions like an inflammatory organ, releasing cytokines such as IL-6, TNF-α, and IL-17 around the clock. The more visceral fat you carry, the higher your baseline inflammatory load.
GLP-1 medications reduce visceral fat aggressively. Tirzepatide achieves roughly 20–22% average weight loss in clinical trials; semaglutide averages 15–17%. As visceral fat drops, so does the body's chronic inflammatory output.
The direct path: GLP-1 appears to calm the immune system independently of weight loss
A November 2025 review published in the Journal of Clinical Investigation examined how GLP-1 medicines reduce inflammation through both weight-dependent and weight-independent mechanisms.[1] Key findings:
- In the SELECT trial, CRP dropped significantly even among participants who lost less than 2% body weight — strong evidence the drug itself has anti-inflammatory properties beyond weight loss.[7]
- The JCI review estimates that only 20–60% of CRP improvement can be explained by weight or glucose changes. The rest appears to involve direct immune modulation.[1]
- Some preclinical studies have shown anti-inflammatory effects within hours of exposure, before meaningful weight loss is possible.[1]
- GLP-1 medications appear to suppress the NF-κB signaling pathway — a central regulator of inflammatory gene expression.[1]
Why this matters for you
If you start a GLP-1 for weight management or another approved indication and you also have chronic inflammation, you are likely getting a dual benefit. This is consistent with reports from patients who notice improvements in joint pain, energy, and puffiness before they have lost significant weight.
What inflammatory markers do GLP-1 medications affect?
hsCRP (high-sensitivity C-reactive protein)
The most commonly tracked marker of systemic inflammation. Semaglutide reduced hsCRP by up to 38% in the SELECT trial.[7] A meta-analysis of 13 randomized trials (26,000+ patients) confirmed this effect across diabetic and non-diabetic populations.[4]
TNF-α (tumor necrosis factor alpha)
A pro-inflammatory cytokine elevated in obesity, diabetes, and autoimmune conditions. GLP-1 medications reduce TNF-α, with some preclinical models demonstrating effects within hours of exposure.[1]
IL-6 (interleukin-6)
A key inflammatory cytokine. Semaglutide reduced circulating IL-6 levels in a study of 40 men with type 2 diabetes.[10] IL-6 drives inflammation in joints, the cardiovascular system, and the liver.
IL-1β (interleukin-1 beta)
Implicated in joint destruction in RA and cardiovascular disease. GLP-1 receptor agonists suppress IL-1β in preclinical models through inhibition of the NLRP3 inflammasome.[10]
Practical takeaway
If you want to objectively track whether your GLP-1 is reducing inflammation, ask your doctor to check hsCRP at baseline and again at 8–12 weeks. It is the simplest and most reliable way to measure improvement.
Best GLP-1 by Inflammation Driver: The Full Breakdown
The "best" answer depends on what is fueling your inflammation. Here is the evidence-backed breakdown for each driver.
If Obesity, Visceral Fat, or Sleep Apnea Is the Engine
Our pick: Tirzepatide (Zepbound, Mounjaro)
When inflammation is driven primarily by excess body weight and the metabolic dysfunction that comes with it, the most effective anti-inflammatory move is removing the fuel. Tirzepatide does this more aggressively than any other GLP-1 on the market.
In the SURMOUNT trials, tirzepatide achieved roughly 20–22% average weight loss — approximately 5 percentage points more than semaglutide in comparable populations.[8] It also demonstrated significant waist circumference reduction, a direct proxy for visceral fat loss.
For sleep apnea specifically, the SURMOUNT-OSA trial showed tirzepatide reduced sleep apnea severity by up to 63% as measured by the apnea-hypopnea index. Nearly half of participants no longer had OSA symptoms after one year of treatment.[8] Zepbound is now FDA-approved for moderate-to-severe obstructive sleep apnea in adults with obesity — the first medication ever approved for this indication. Sleep apnea is both a consequence and a driver of systemic inflammation — treating it can break the inflammatory cycle.
Tirzepatide's dual mechanism (it activates both GLP-1 and GIP receptors) also appears to contribute to anti-inflammatory effects. GIP receptor signaling may directly modulate immune cells, according to the JCI 2025 review, though this research is still early.[1]
What tirzepatide is NOT best for
If your primary concern is liver-specific inflammation (MASH) and you want the drug with the deepest published evidence and an actual FDA-approved label for that condition, semaglutide currently has a stronger position. Tirzepatide's liver data is promising but still catching up.
What to expect
Appetite changes within 1–2 weeks. Puffiness and energy improvements often reported within 4–8 weeks. Meaningful weight and waist reduction over 3–6 months.
Check if you qualify for FDA-approved tirzepatide (Zepbound) or semaglutide (Wegovy)
Check Eligibility & Pricing on Ro →If Liver Inflammation (MASH) or Broad Cardiometabolic Inflammation Is the Problem
Our pick: Semaglutide (Wegovy, Ozempic)
Semaglutide has the largest body of published anti-inflammatory evidence of any GLP-1 medication.
The SELECT trial — over 17,600 patients — showed a 20% reduction in major cardiovascular events (heart attack, stroke, cardiovascular death).[7] It also demonstrated up to 38% CRP reduction, with effects appearing within 4–8 weeks. Even participants who lost less than 2% body weight still showed significant CRP reductions.[7]
For liver inflammation, the evidence is no longer just "promising." In August 2025, Wegovy received FDA accelerated approval for treating noncirrhotic MASH with moderate to advanced liver fibrosis (stages F2–F3) — making it the first GLP-1 ever approved for liver disease. In the ESSENCE trial, 63% of participants on semaglutide achieved resolution of liver inflammation without worsening fibrosis, versus 34% on placebo.
A 2024 meta-analysis in Frontiers in Cardiovascular Medicine pooled 13 randomized controlled trials (over 26,000 patients) and confirmed that semaglutide consistently reduces CRP regardless of the population studied or dosing regimen used.[4]
What semaglutide is NOT best for
If maximum weight-loss speed is your priority, tirzepatide typically outperforms semaglutide by several percentage points. Semaglutide wins on evidence breadth; tirzepatide wins on metabolic potency.
What to expect
CRP reductions measurable within 4–8 weeks. Weight loss of 15–17% average over 12–18 months. Liver enzyme improvement often seen within 3–6 months. Cardiovascular risk reduction documented over longer follow-up.
Check access to FDA-approved semaglutide (Wegovy) — including insurance navigation and current pricing
Check Coverage & Pricing Through Ro →If Budget and FDA-Approved Access Matter Most
Our pick: Liraglutide (Saxenda, Victoza)
Liraglutide is the most established GLP-1 on the market, with the longest safety track record. It is FDA-approved for both diabetes (Victoza) and weight management (Saxenda), and generic liraglutide for weight management became available in 2025, meaningfully improving the cost story.
On the anti-inflammatory front, liraglutide has demonstrated reductions in TNF-α, IL-1β, and IL-6 in human studies — and notably, some of these reductions occurred independently of weight loss or glycemic control.[12] The tradeoff: liraglutide is a daily injection (not weekly), achieves more moderate weight loss (approximately 8–10% average), and has a lower ceiling for anti-inflammatory benefit. But it is more affordable with the generic option and may be the right fit if you cannot access or afford newer agents. For broader cost comparisons, see our GLP-1 cost without insurance guide.
If Rheumatoid Arthritis, Psoriatic Arthritis, or Joint Pain Are the Focus
Our pick: Semaglutide or tirzepatide (depends on your metabolic profile)
This is the category where the evidence has evolved fastest — and where the 2025 research has been genuinely exciting. But we want to be straightforward: the current human evidence is still early, consisting mainly of one retrospective study and conference-level presentations. GLP-1s are not standard rheumatoid arthritis or psoriatic arthritis therapy.
At ACR Convergence 2025, multiple studies presented data on GLP-1s in rheumatic diseases. RA patients on DMARDs who also received GLP-1 agonists had fewer disease flares. A large multi-center study found GLP-1 use was linked to lower risk of developing immune-mediated inflammatory diseases.[6]
A 2025 study in ACR Open Rheumatology examined 173 patients with RA and BMI ≥27 who were prescribed semaglutide or tirzepatide. GLP-1 users experienced significant improvements in disease activity scores, pain scores, and cardiovascular risk factors compared to controls. The improvements in inflammation markers and pain were not significantly correlated with weight loss — suggesting a weight-independent mechanism.[5]
For osteoarthritis specifically, the STEP-9 trial showed semaglutide produced significant pain reduction and improved physical function in patients with obesity and knee OA.[9]
For psoriatic arthritis and psoriasis, patients report skin improvement before significant weight change — suggesting a direct anti-inflammatory effect. GLP-1 therapy helps both skin clearance and joint inflammation in PsA, and weight loss from GLP-1 amplifies the benefits of existing PsA medications.[6]
Important context
GLP-1s are not standard rheumatoid arthritis, psoriatic arthritis, or osteoarthritis therapy. The current human evidence is mainly a retrospective study plus conference presentations — not definitive randomized trials designed with arthritis as the primary endpoint. But if you have inflammatory arthritis AND you qualify for a GLP-1 based on weight or metabolic criteria, the anti-inflammatory benefit may be substantial.
If Active Autoimmune Disease Is the Primary Driver
Our pick: Not a GLP-1-first answer
If your inflammation is primarily driven by active autoimmune disease — RA with minimal metabolic contribution, lupus, Crohn's — a GLP-1 is not the best first-line anti-inflammatory tool. Specialist treatment with DMARDs, biologics, or other immunomodulatory therapies remains the standard of care.
But here is where it gets nuanced: many people with autoimmune conditions also carry metabolic risk factors — excess weight, insulin resistance, visceral fat — that amplify their inflammatory load. In those cases, a GLP-1 may work powerfully as an add-on. It removes the metabolic fuel that keeps the inflammatory fire burning hotter than it needs to. Patients with RA and obesity report fewer flares and reduced need for steroids after starting a GLP-1 — but the GLP-1 did not replace their RA treatment, it supported it.[5][6]
If Lipedema, Swelling, or "Whole-Body Inflammation" Is What You Feel
People describe feeling puffy, swollen, heavy, achey — like their entire body is inflamed. Often, this maps to excess visceral fat driving systemic inflammatory cytokine production, insulin resistance creating a chronic low-grade inflammatory state, undiagnosed fatty liver, or undiagnosed sleep apnea causing overnight inflammation spikes.
For this population, tirzepatide or semaglutide can produce some of the most noticeable subjective improvements. The puffiness reduction, energy improvement, and "whole-body inflammation" relief people describe is consistent with what happens when you lower CRP, reduce visceral fat, improve insulin sensitivity, and calm the NF-κB inflammatory cascade simultaneously.
The key: get a baseline workup — hsCRP, fasting insulin, liver panel, and a sleep study if snoring or daytime fatigue is present. That workup will tell you and your prescriber which inflammation driver to target.
Does this sound like your situation?
Find the GLP-1 Path That Matches Your Inflammation Driver →What to Bring to Your Doctor's Appointment
Many prescribers are still catching up to the anti-inflammatory evidence for GLP-1 medications. If your doctor is not yet familiar with this data, here are the key references to bring.
- The JCI 2025 review — the most comprehensive peer-reviewed analysis of GLP-1 anti-inflammatory mechanisms (both weight-dependent and weight-independent)[1]
- The SELECT trial CRP data — up to 38% CRP reduction, effects seen even with minimal weight loss[7]
- ACR 2025 research — for rheumatic conditions: RA disease activity improvement, fewer flares, improved joint outcomes[5][6]
- The ESSENCE trial and Wegovy MASH approval — for fatty liver concerns[11]
- Your own baseline labs — hsCRP, liver panel, fasting insulin. Having your own numbers makes the conversation concrete.
You are not asking your doctor to prescribe off-label for inflammation. You are asking them to prescribe for a qualifying indication — weight management, diabetes, cardiovascular risk, OSA, or MASH — with the understanding that anti-inflammatory benefit is a documented and measurable secondary effect.
Pro tip
Ask specifically for a baseline hsCRP before starting, and a follow-up at 8–12 weeks. That single lab gives you and your doctor an objective way to see whether the medication is reducing your inflammation — not just your weight.
Who Should NOT Use a GLP-1 for Inflammation?
Not everyone is a candidate. Being clear about that is part of what makes this page trustworthy.
Hard-stop contraindications
- Personal or family history of medullary thyroid carcinoma (MTC) or multiple endocrine neoplasia type 2 (MEN2) — GLP-1 medications are contraindicated
- Known hypersensitivity to the specific drug or its components
When extra caution is needed
- History of pancreatitis — discuss with your prescriber; GLP-1s may not be appropriate
- Gallbladder disease — weight loss itself increases gallstone risk; GLP-1s may compound this
- Severe gastroparesis — GLP-1s slow gastric emptying and could worsen symptoms
- Pregnancy or trying to conceive — GLP-1 medications are generally not recommended during pregnancy; if pregnancy occurs, contact your clinician
- Type 1 diabetes — GLP-1s are not a substitute for insulin therapy
If you have unexplained elevated inflammatory markers without obesity or metabolic risk factors, the right first step is a thorough medical workup — not a GLP-1 prescription. Inflammatory markers can be elevated by infections, undiagnosed autoimmune conditions, certain cancers, and other causes that require specific treatment. See our full GLP-1 contraindications guide for the complete list.
GLP-1 Anti-Inflammatory Evidence Matrix
Every claim is traceable to published research. Focus on the "Best-Fit Inflammation Driver" column first.
| Medication | Best-Fit Driver | Key Evidence | CRP Data | FDA-Approved For |
|---|---|---|---|---|
| Semaglutide Wegovy, Ozempic, Rybelsus | Liver (MASH), cardiovascular, broad cardiometabolic | SELECT (17,600 pts)[7]; 26,000+ pt meta-analysis[4]; STEP-9 (knee OA)[9]; ESSENCE (MASH) | Up to 38%; significant even with <2% weight loss | T2D, weight management, CV risk reduction, MASH (F2–F3, accelerated) |
| Tirzepatide Zepbound, Mounjaro | Obesity-driven, visceral fat, sleep apnea | SURMOUNT trials[8]; SURMOUNT-OSA[8]; ACR 2025[6] | Significant in SURMOUNT; dual pathway may amplify | T2D, weight management, moderate-to-severe OSA with obesity |
| Liraglutide Saxenda, Victoza (generic available) | Budget-first; established option | LEADER trial (CV outcomes); human cytokine studies[12] | TNF-α, IL-1β, IL-6 reductions; some independent of glycemic control | T2D, weight management |
| Dulaglutide Trulicity | General cardiometabolic (less studied) | REWIND trial (CV outcomes in T2D) | Limited CRP-specific data | T2D |
Tirzepatide vs. Semaglutide for Inflammation: The Head-to-Head
This is the comparison most readers actually want.
| Factor | Semaglutide (Wegovy/Ozempic) | Tirzepatide (Zepbound/Mounjaro) |
|---|---|---|
| Mechanism | GLP-1 receptor only | Dual GLP-1 + GIP receptors |
| Anti-inflammatory evidence | Largest (SELECT, STEP-9, meta-analyses, ESSENCE) | Growing (SURMOUNT, SURMOUNT-OSA, ACR 2025) |
| CRP reduction | Up to 38% in SELECT; significant even with <2% weight loss | Significant in SURMOUNT; dual pathway may enhance |
| Average weight loss | ~15–17% | ~20–22% |
| Best inflammation fit | Liver/MASH (FDA-approved), cardiovascular (FDA-approved) | Obesity-driven, visceral fat, sleep apnea (FDA-approved for OSA) |
| OA pain data | STEP-9 trial (direct evidence for knee OA) | No completed OA-specific trial yet (one underway) |
| Dosing | Weekly injection (or daily pill — oral semaglutide) | Weekly injection |
The simplest honest rule
- If your inflammation is mostly a metabolic load problem (excess weight, visceral fat, sleep apnea, insulin resistance), lean tirzepatide.
- If your inflammation question is tied to liver health, cardiovascular risk, or evidence depth, lean semaglutide.
- If the problem is autoimmune-first with minimal metabolic involvement, specialist care comes first.
You genuinely cannot go wrong with either for inflammation. The differences are real but not dramatic. What matters most is starting — because the anti-inflammatory benefits begin appearing within weeks.
Check your eligibility for FDA-approved tirzepatide or semaglutide
Check Eligibility on Ro →How Quickly Does GLP-1 Reduce Inflammation?
The timeline varies by marker, but the evidence is clear that inflammation changes begin well before significant weight loss.
Weeks 1–2: Early signals
Appetite changes and reduced cravings are usually the first noticeable effect. Some patients report reduced puffiness and better energy, though GI adjustment (nausea, changes in digestion) is also common.
Weeks 4–8: Measurable inflammation changes
In the SELECT trial, CRP reductions were detectable within this window — before participants had achieved significant weight loss.[7] If your doctor orders hsCRP labs at baseline and again at 6–8 weeks, you may see measurable improvement. Patients with joint pain often report improvement in this timeframe.
Months 2–6: Compounding benefits
As weight loss accelerates, the indirect anti-inflammatory pathway compounds with the direct immune effects. Lab improvements become more pronounced — CRP, liver enzymes, lipids, A1c. Joint pain and physical function continue to improve. Sleep apnea severity may decrease.
Months 6–12+: Durable change
Deeper metabolic improvements. Liver inflammation markers show continued improvement. Cardiovascular risk reduction becomes measurable. The compounding effect of sustained weight loss plus ongoing anti-inflammatory activity creates durable change — as long as treatment continues.

What to track so you know it is working
Ask your doctor to monitor at baseline and at regular intervals:
- hsCRP or CRP — the most direct marker of systemic inflammation
- Liver panel (ALT, AST) — especially if fatty liver is a concern
- Waist circumference — a proxy for visceral fat (more informative than body weight alone)
- Fasting insulin / HOMA-IR — insulin resistance drives inflammation
- Joint symptoms — pain scale, morning stiffness duration, flare frequency
- Weight — important, but not the only metric that matters
The Honest Tradeoffs (What GLP-1s Will NOT Do for Inflammation)
GLP-1s are not approved for treating inflammation as a standalone condition
You will not receive a GLP-1 prescription with "inflammation" as the diagnosis. Current FDA-approved indications include type 2 diabetes, chronic weight management, cardiovascular risk reduction (Wegovy), moderate-to-severe OSA with obesity (Zepbound), and noncirrhotic MASH with F2–F3 fibrosis (Wegovy under accelerated approval). The majority of people searching for "best GLP-1 for inflammation" also have conditions that qualify them through these standard on-label pathways.
Side effects are real — but manageable for most
Every GLP-1 medication shares a similar side effect profile: nausea, diarrhea, constipation, and decreased appetite are most common. These are typically dose-dependent and improve within 4–6 weeks. In the ACR Open Rheumatology study, about one-third of RA patients discontinued their GLP-1 due to GI side effects or insurance difficulties.[5] That is a real number. If you are in that group, your doctor can adjust the approach.
GLP-1s do not regenerate damaged tissue
For osteoarthritis patients: GLP-1 medications reduce inflammation and pain, but they do not reverse existing cartilage damage. Preclinical evidence suggests chondroprotective effects — less inflammation plus less weight on the joint equals less further damage and meaningfully better quality of life now.
Stopping means inflammation may return
Like blood pressure medication, the benefits persist while you take the medication. Stopping typically leads to weight regain and return of inflammatory markers over time — unless you have independently established lifestyle changes that sustain some of the gains.
None of these limitations change the fundamental picture. The anti-inflammatory evidence for GLP-1 medications is now among the most exciting developments in metabolic medicine. The tradeoffs are manageable. And the potential benefit — measurably lower inflammation, less pain, better energy, reduced cardiovascular risk, improved liver health — is substantial enough that researchers at Harvard, the American College of Rheumatology, and the Journal of Clinical Investigation are actively investigating it.
If you qualify for a GLP-1 through any on-label pathway and chronic inflammation is part of your health picture, this is worth a serious conversation with your clinician.
See If You Qualify for FDA-Approved GLP-1 Access on Ro →How to Actually Get Started
Step 1. Determine if you qualify
The most common on-label pathways:
- Obesity (BMI ≥30)
- Overweight + comorbidity (BMI ≥27 with a weight-related condition)
- Type 2 diabetes (Ozempic/Mounjaro)
- Established cardiovascular disease with overweight/obesity (Wegovy)
- Moderate-to-severe OSA with obesity (Zepbound)
- Noncirrhotic MASH with F2–F3 fibrosis (Wegovy, accelerated approval)
Step 2. Choose your access path
For FDA-approved medications with clinical support: Ro offers FDA-approved GLP-1 treatment online, including clinician evaluation, insurance-support tools, and transparent pricing. They offer both semaglutide (Wegovy) and tirzepatide (Zepbound) through FDA-approved channels.
If cost is the primary barrier: generic liraglutide (Saxenda) became available in 2025 and offers a lower-cost FDA-approved entry point. For broader cost comparisons, see our GLP-1 cost without insurance guide.
Step 3. Talk to your provider about inflammation monitoring
Do not just track your weight. Ask your doctor to establish baseline inflammatory markers:
- hsCRP at baseline and at 8–12 weeks — the single most useful inflammation marker
- Liver panel (ALT, AST) — especially for MASH/fatty liver concerns
- Fasting insulin or HOMA-IR — insulin resistance tracking
- Waist circumference — measure monthly; more direct than body weight for visceral fat
Step 4. Set realistic expectations
What Dose Is Used for Inflammation?
There is no FDA-approved "inflammation dose" for any GLP-1 medication. All current dosing follows the approved indications — diabetes management or weight management.
Both semaglutide and tirzepatide use gradual dose escalation. Semaglutide starts at 0.25 mg weekly, titrated up to 2.4 mg for weight management. Tirzepatide starts at 2.5 mg weekly, titrated up to 15 mg. The titration exists to minimize GI side effects.
Most of the published anti-inflammatory data comes from standard therapeutic doses used in large trials like SELECT and SURMOUNT. There is no established "low-dose for inflammation" protocol. Start at the recommended starting dose, titrate per your prescriber's guidance, and monitor your inflammatory markers.
Will Insurance Cover a GLP-1 for Inflammation?
The short answer: not under the label "inflammation." But there are real pathways. Insurance coverage is based on the FDA-approved indication, not your personal reason for wanting it.
The Zepbound OSA pathway deserves special attention
Since December 2024, Zepbound has been FDA-approved for moderate-to-severe obstructive sleep apnea in adults with obesity. If you have OSA (even undiagnosed — consider a sleep study if you snore, have daytime fatigue, or wake unrefreshed), this indication may create a more accessible insurance coverage pathway than weight management alone for some patients.
The Wegovy MASH pathway is new
As of August 2025, Wegovy is approved under accelerated approval for noncirrhotic MASH with F2–F3 fibrosis. If you have been diagnosed with fatty liver disease that has progressed to MASH with moderate-to-advanced fibrosis, this provides another on-label coverage pathway.
The FDA-approved path vs. the compounded path
Both semaglutide and tirzepatide have been removed from FDA's drug shortage list. The enforcement discretion periods that previously allowed pharmacies to compound copies of these medications have expired. FDA's posture toward mass-marketed compounded GLP-1s has tightened significantly — including warning letters to several firms.
Red flags to watch for
- Marketing language like "same active ingredient" applied to compounded products
- Vague pharmacy sourcing
- No clear disclosure of whether the product is FDA-approved or compounded
- Aggressive discounting with no explanation of what you are receiving
Check FDA-approved options and current availability
Check FDA-Approved Options on Ro →What People Report
The clinical trial data is compelling, but so are the stories from real patients. Individual experiences are not clinical proof, and your results may differ — but these reports are consistent with what the research shows.
“I am considering it myself for both inflammation and weight loss.”
— Reddit user, r/lipedema
“My inflammation is way down and most labs were normal.”
— Reddit user, r/lipedema
“I noticed when I first started… my inflammation & pain subsided almost immediately.”
— Reddit user, r/lipedema
These are anecdotal — they reflect individual experience, not clinical proof. But the pattern is consistent with what large-scale trials show: anti-inflammatory effects that often appear early, sometimes before significant weight loss.
How We Evaluated the Evidence
Our evidence hierarchy
- Current FDA labels and approved indications — the regulatory floor
- Large randomized controlled trials (SELECT, SURMOUNT, STEP-9, LEADER, ESSENCE) — the gold standard
- Peer-reviewed meta-analyses and systematic reviews (Frontiers 2024, JCI 2025)
- Observational human studies (ACR 2025 presentations, ACR Open Rheumatology 2025)
- Patient experience — context only, clearly labeled as anecdotal
What we verified
- All study citations link to PubMed, NEJM, JCI, or official proceedings
- FDA label information cross-referenced with current FDA Access Data
- Provider access claims verified against provider websites (March 2026)
- Compounding landscape verified against current FDA shortage list and enforcement guidance
- This page is not sponsored by any pharmaceutical company or telehealth provider
Frequently Asked Questions
Which GLP-1 works best for inflammation?
It depends on the driver. For obesity-driven inflammation, tirzepatide has the strongest practical impact. For the broadest published anti-inflammatory evidence across liver, heart, and metabolic conditions, semaglutide leads. Liraglutide is the budget-friendly established option with a generic now available. No single GLP-1 is universally best.
Do GLP-1 drugs really reduce inflammation?
Yes, and the evidence is substantial. A 2024 meta-analysis of 13 randomized trials confirmed semaglutide consistently reduces CRP. The JCI 2025 review documented both weight-dependent and weight-independent anti-inflammatory mechanisms. The SELECT trial showed up to 38% CRP reduction. Multiple trials and real-world studies now support anti-inflammatory effects across the GLP-1 class.
Is tirzepatide better than semaglutide for inflammation?
Not proven across all inflammation types. Tirzepatide achieves more weight loss (which itself reduces inflammation) and has dual-receptor activity that may enhance anti-inflammatory effects. But semaglutide has significantly more published anti-inflammatory data and FDA-approved indications for both MASH and cardiovascular risk reduction. For obesity-driven inflammation, tirzepatide may have an edge. For evidence depth and liver or cardiovascular inflammation, semaglutide is stronger.
Can GLP-1 help rheumatoid arthritis?
Early evidence is promising. A 2025 study in ACR Open Rheumatology showed GLP-1 medications improved RA disease activity, pain, and cardiovascular risk in patients with BMI 27 or higher. ACR 2025 data showed fewer flares in RA patients on DMARDs who added a GLP-1. However, these are not definitive randomized trials, and GLP-1s are not standard RA therapy. They may be a valuable add-on when metabolic factors coexist.
Can GLP-1 help autoimmune disease more broadly?
The evidence is early but accumulating. A large multi-center study found GLP-1 use was linked to lower risk of developing new immune-mediated inflammatory diseases. Cleveland Clinic researchers found about 42% lower uveitis risk in GLP-1 users versus metformin or insulin users. For active autoimmune conditions, GLP-1 medications are add-ons, not replacements for specialist treatment. But when obesity or metabolic dysfunction is amplifying autoimmune inflammation, a GLP-1 can reduce that metabolic fuel.
How quickly does GLP-1 reduce inflammation?
CRP reductions have been documented within 4 to 8 weeks, sometimes before significant weight loss. Joint pain improvement is commonly reported within 2 to 3 months. Full compounding benefits build over 6 to 12 months.
What dose is used for inflammation?
There is no specific inflammation dose. Anti-inflammatory benefits have been documented at standard therapeutic doses used in trials like SELECT and SURMOUNT. There is no established evidence base for low-dose or microdosing protocols for inflammation specifically. Work with your prescriber to find the right dose for your overall health profile.
Will insurance cover GLP-1 for inflammation?
Not under the diagnosis of inflammation. But most people with chronic inflammation also qualify under weight management, type 2 diabetes, cardiovascular risk reduction, OSA, or MASH criteria. The anti-inflammatory benefit comes as a secondary benefit of an on-label prescription. The Zepbound OSA pathway and the Wegovy MASH pathway are both relatively new and may provide coverage options that did not exist a year ago.
Can GLP-1 medications help with gut inflammation or IBD?
Preliminary evidence suggests potential benefit. Some observational studies show reduced steroid use, fewer hospitalizations, and fewer IBD-related surgeries in patients taking GLP-1s for diabetes. GLP-1 improves mucosal integrity and calms the gut immune response. However, no GLP-1 is approved for IBD, more research is needed, and some researchers have raised questions about microbiome changes. If you have IBD, discuss this with your gastroenterologist before starting a GLP-1.
Are compounded GLP-1s safe?
Compounded GLP-1 medications have not undergone FDA premarket review for safety, effectiveness, or quality. The drug shortages that previously allowed wide-scale compounding have been resolved, and FDA has tightened enforcement including warning letters to several firms. Compounding remains legal for documented individual patient needs, but mass-market compounded GLP-1s are no longer in the enforcement discretion window.
What if I only want FDA-approved options?
That is the cleanest path. Wegovy (semaglutide) and Zepbound (tirzepatide) are FDA-approved for weight management with additional approved indications for MASH and OSA respectively. Ro offers access to FDA-approved GLP-1 medications with clinical support, insurance navigation, and transparent pricing.
What is the most affordable GLP-1 for inflammation?
Generic liraglutide (Saxenda) became available in 2025 and is generally the most affordable FDA-approved option. For semaglutide and tirzepatide, manufacturer savings programs can significantly reduce cost. Checking eligibility through a provider like Ro is the fastest way to get your actual out-of-pocket price.
The Bottom Line
If you have chronic inflammation and you also carry excess weight, have insulin resistance, fatty liver, sleep apnea, or metabolic syndrome — a GLP-1 medication may be one of the most impactful decisions you can make for your health.
The evidence is no longer theoretical. Large-scale human trials, peer-reviewed meta-analyses, and real-world clinical data all point in the same direction: GLP-1 medications reduce inflammation through both weight loss and direct immune modulation.
With both injectable semaglutide and tirzepatide shortages resolved, FDA-approved access is clearer than it has been in three years. If you and your clinician decide a GLP-1 fits your situation, earlier treatment may help address both metabolic risk and inflammation sooner.
Still not sure which GLP-1 program is right for you?
We'll ask about your inflammation drivers, health goals, insurance situation, and budget — and give you a personalized recommendation you can take to your doctor or use to get started today.
Sources
- Wong CK, et al. “Antiinflammatory actions of glucagon-like peptide-1–based therapies beyond metabolic benefits.” Journal of Clinical Investigation. November 2025. jci.org/articles/view/194751
- Komaroff AL. “Do GLP-1 drugs reduce inflammation?” Harvard Health. October 2025. health.harvard.edu
- FDA. “FDA Approves First Medication for Obstructive Sleep Apnea.” December 20, 2024. fda.gov
- Irfan H, et al. “Anti-inflammatory effect of semaglutide: updated systematic review and meta-analysis.” Frontiers in Cardiovascular Medicine. 2024. frontiersin.org
- Kellner DA, et al. “Effect of glucagon-like peptide 1 receptor agonists on patients with rheumatoid arthritis.” ACR Open Rheumatology. September 2025. acrjournals.onlinelibrary.wiley.com
- ACR. “New Research Highlights Potential of GLP-1 Therapies in Rheumatic Disease Management.” ACR Convergence 2025. rheumatology.org
- SELECT trial: Lincoff AM, et al. “Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes.” NEJM. 2023. 17,604 patients. Up to 38% CRP reduction; 20% reduction in MACE.
- SURMOUNT-OSA: Malhotra A, et al. “Tirzepatide for the Treatment of Obstructive Sleep Apnea and Obesity.” NEJM. 2024. Up to 63% AHI reduction.
- STEP-9 trial: Semaglutide demonstrated significant pain reduction and improved physical function in patients with obesity and knee osteoarthritis. Published 2024.
- Yaribeygi H, et al. “Anti-inflammatory benefits of semaglutide: State of the art.” Pharmacological Research. 2024. PMC10992717
- FDA. “FDA Approves Treatment for Serious Liver Disease Known as MASH.” August 2025. fda.gov
- Drucker DJ, et al. “Glucagon-like peptide-1: a multi-faceted anti-inflammatory agent.” Frontiers in Endocrinology. 2023. PMC10230051
- FDA Drug Shortage and Compounding Guidance. fda.gov
Affiliate disclosure: The RX Index maintains affiliate relationships with some providers mentioned on this page. These relationships do not affect our evidence analysis, recommendations, or editorial content. Our analysis is based on published peer-reviewed research and verified FDA information.