Foundayo Reviews: Honest 2026 Verdict on Cost, Side Effects, and Who It's Best For
Our verdict
Foundayo (orforglipron) is the real deal — a legitimate, FDA-approved GLP-1 weight loss pill from Eli Lilly.
Take it anytime, with food, without water restrictions. People on the highest dose in clinical trials lost an average of 25 lbs (11.1%) over 72 weeks. Starts at $149/mo.
But before you start: read this first.
Foundayo does not produce the most weight loss in the oral GLP-1 category. The Wegovy pill shows ~13.6% vs Foundayo's 11.1%. Plus, it has 3 hidden label frictions that most review pages skip entirely — including a birth control interaction that affects 5+ months of your medication routine.
Because Foundayo eliminates the empty-stomach routine entirely — no fasting, no 30-minute wait, no 4-oz water rule — it may be the oral GLP-1 that fits more people's lives well enough to keep taking it long-term. A GLP-1 you stay on at 11% beats one you quit after 3 months at 0%. That convenience-vs-efficacy tradeoff is the entire decision, and this page will help you make it.
Jump to: vs Wegovy pill · Pricing · Weight loss data · Side effects · Hidden frictions · Who qualifies · How to get it · Dosage · Switching · FAQ

Foundayo vs Wegovy Pill: Head-to-Head Comparison
We pulled every data point below from FDA prescribing information, Eli Lilly's official pricing, Novo Nordisk's published data, and CMS Medicare guidance — then assembled them into one decision matrix.

| Foundayo | Wegovy Pill | |
|---|---|---|
| Generic name | Orforglipron | Semaglutide |
| Manufacturer | Eli Lilly | Novo Nordisk |
| FDA approved | April 1, 2026 | December 22, 2025 |
| How you take it | Once daily, any time, with or without food | Once daily, empty stomach in morning, ≤4 oz water, wait 30 min before food/drinks/other meds |
| Dose steps | 6 steps: 0.8 → 2.5 → 5.5 → 9 → 14.5 → 17.2 mg | 4 steps: 1.5 → 4 → 9 → 25 mg |
| Months to max dose | ~5–6 months | ~4 months |
| Avg weight loss (max dose) | 11.1% / ~25 lbs (treatment-policy, 72 wks) | 13.6% / ~32 lbs (OASIS 4, 64 wks) |
| % losing ≥15% body weight | 35.9% | 47% |
| Self-pay starting dose | $149/mo | $149/mo |
| Self-pay max dose | $349/mo (or $299 with 45-day refill offer) | $299/mo |
| With savings card | As low as $25/mo | As low as $25/mo |
| Medicare (from July 1, 2026) | $50/mo via GLP-1 Bridge | $50/mo via GLP-1 Bridge |
| Cardiovascular label | No current CV indication | Approved for MACE reduction in eligible adults |
| Birth control warning | Yes — add barrier or switch to non-oral for 30 days after start AND each dose increase | No specific oral contraceptive warning |
| Simvastatin limit | Max 20 mg/day simvastatin | No simvastatin-specific restriction |
| CYP3A4 interaction | Max dose capped at 9 mg with strong CYP3A4 inhibitors | No CYP3A4 interaction (peptide drug) |
| Discontinued due to adverse reactions | 6–10% depending on dose | 6.9% at 25 mg |
Sources: Foundayo US Prescribing Information (DailyMed, April 2026); Eli Lilly investor press release (April 1, 2026); LillyDirect pricing; Wegovy tablet US Prescribing Information (FDA, 2025); Novo Nordisk pricing guide; CMS Medicare GLP-1 Bridge guidance; ATTAIN-1 and OASIS 4 trial data.
Note on discontinuation rates: Both drugs show adverse-reaction discontinuation in the 6–10% range. The two numbers come from separate trials with different protocols — close enough that discontinuation alone shouldn't be the deciding factor between them.
Free 60-second GLP-1 matching quiz · No email required
How Much Does Foundayo Actually Cost? (Every Pricing Path, April 2026)
Foundayo pricing is more complicated than the $149 headline number most pages quote. The starting dose is $149, yes. But you don't stay on the starting dose — here's what happens as your doctor escalates you over the first 6 months, and the hidden refill rule that determines whether you pay $299 or $349 at the highest doses.
Foundayo Price by Dose (Self-Pay Through LillyDirect)
| Dose | Month | Regular Price | With Purchase Offer* |
|---|---|---|---|
| 0.8 mg | Month 1 | $149 | $149 |
| 2.5 mg | Month 2 | $199 | $199 |
| 5.5 mg | Month 3 | $299 | $299 |
| 9 mg | Month 4 | $299 | $299 |
| 14.5 mg | Month 5 | $349 ⚠️ | $299 |
| 17.2 mg | Month 6+ | $349 ⚠️ | $299 |
Source: Eli Lilly LillyDirect pricing, verified April 8, 2026. *Purchase offer: $299 for first order of 14.5/17.2 mg; keep by refilling within 45 days of delivery.
The 45-day refill rule most pages miss
Lilly automatically applies a $299 purchase offer to your first order of 14.5 mg or 17.2 mg Foundayo — saving you $50/month at the doses that matter most. But to keep that $299 price on subsequent refills, you must reorder within 45 days of your previous delivery date.
Miss the window and you revert to $349. This creates price surprises for travelers, people with pharmacy delays, or anyone who doesn't set a refill reminder. Not a dealbreaker — but it's the kind of detail that creates frustration when you discover it on month 5 instead of month 1.
First 6 Months: Foundayo vs Wegovy Pill — Actual Cost Math
| Month | Foundayo (45-day offer) | Foundayo (regular price) | Wegovy Pill (current promos) |
|---|---|---|---|
| Month 1 | $149 | $149 | $149 (1.5 mg) |
| Month 2 | $199 | $199 | $149 (4 mg promo) |
| Month 3 | $299 | $299 | $299 (9 mg) |
| Month 4 | $299 | $299 | $299 (25 mg) |
| Month 5 | $299 | $349 | $299 |
| Month 6 | $299 | $349 | $299 |
| 6-Month Total | $1,544 | $1,644 | $1,494 |
Wegovy 4 mg promo pricing active through August 31, 2026. After promo expiration, Wegovy 6-month total rises to ~$1,544 — essentially identical to Foundayo with the purchase offer. Verify both pricing structures before your first fill — offer terms can change without notice.
The cost bottom line: Over 6 months, Foundayo runs roughly $50–$150 more than the Wegovy pill under current promotions. Whether that delta matters depends on whether the no-fasting convenience is worth it to you. For someone who quit an oral GLP-1 because the empty-stomach routine didn't fit their life, paying $50 more over 6 months for a pill they'll actually take is money well spent.
Insurance and Medicare Paths
Commercial insurance with Foundayo Savings Card
As low as $25/mo. Eligibility required — the card has monthly and annual limits, and terms can change. This is a manufacturer discount program, not insurance.
Medicare Part D — GLP-1 Bridge Program (July 1 – December 31, 2026)
Eligible Medicare beneficiaries can access Foundayo and Wegovy through the CMS Medicare GLP-1 Bridge for $50/month. This drops the monthly cost by $100–$299 compared to self-pay. If you're on Medicare and can wait a few months, this dramatically changes the math.
Free · No email · 60-second matching quiz
How Much Weight Can You Actually Lose on Foundayo?
In the ATTAIN-1 trial — the primary obesity study behind Foundayo's FDA approval — 3,127 adults without diabetes took Foundayo or placebo for 72 weeks alongside a reduced-calorie diet and increased physical activity.
Weight Loss by Dose (ATTAIN-1, Adults Without Diabetes, 72 Weeks)
| Dose | Avg Start Wt | Avg Loss (lbs) | Avg Loss (%) | ≥5% wt loss | ≥10% wt loss | ≥15% wt loss |
|---|---|---|---|---|---|---|
| 5.5 mg | 227 lbs | 17.2 lbs | 7.4% | 59.6% | 32.5% | 14.3% |
| 9 mg | 225 lbs | 18.9 lbs | 8.3% | 63.1% | 39.8% | 20.1% |
| 17.2 mg | 227 lbs | 24.9 lbs | 11.1% | 71.5% | 54.5% | 35.9% |
| Placebo | 229 lbs | 5.3 lbs | 2.1% | 26.8% | 13.0% | 6.0% |
Source: Foundayo US Prescribing Information, ATTAIN-1 trial (treatment-policy estimand — includes all randomized participants whether or not they completed the full trial).
Treatment-policy vs on-treatment: why both numbers exist
11.1% / ~25 lbs (treatment-policy)
Includes everyone randomized to the highest dose — whether they stayed on it or not. This is the more conservative, real-world-representative number. We use this throughout this page.
12.4% / 27.3 lbs (on-treatment)
Only counts people who stayed on the medication as directed. Technically accurate but paints a rosier picture than most people will experience. If a competing review page quotes 12.4% without this context, they're presenting the best-case subset.
How Foundayo Compares to Other GLP-1 Options
| GLP-1 Option | Form | Avg Weight Loss (%) |
|---|---|---|
| Foundayo (17.2 mg) | Daily pill, no fasting | 11.1% |
| Wegovy pill (25 mg) | Daily pill, empty stomach | 13.6% |
| Wegovy injection (2.4–7.2 mg) | Weekly shot | ~15–18% |
| Zepbound / tirzepatide (15 mg) | Weekly shot | ~20–22% |
These numbers come from separate clinical trials with different populations and durations. Cross-trial comparisons are inherently imperfect — no head-to-head obesity trial exists between Foundayo and the Wegovy pill.
The pattern is clear: injectables produce more weight loss, and the Wegovy pill currently shows a stronger efficacy headline than Foundayo. But over 7 in 10 people (71.5%) on the highest Foundayo dose lost at least 5% of their body weight, and more than 1 in 3 (35.9%) lost 15% or more. Those are clinically significant numbers — the kind that move blood pressure, cholesterol, and how your clothes fit.
Ro offers GLP-1 concierge with insurance navigation and prior authorization support
What Are the Real Side Effects of Foundayo?
GI side effects are the main tradeoff with every GLP-1 medication, and Foundayo is no exception. The good news: they're worst during dose escalation and tend to improve as your body adjusts. The slow 30-day step-up schedule is specifically designed to minimize this.
Most Common Side Effects (Pooled Weight-Management Trials, by Dose)
| Side Effect | 5.5 mg | 9 mg | 17.2 mg | Placebo |
|---|---|---|---|---|
| Nausea | 26% | 34% | 35% | 8% |
| Constipation | 20% | 27% | 24% | 6% |
| Diarrhea | 21% | 23% | 25% | 10% |
| Vomiting | 13% | 21% | 24% | 3% |
| Abdominal pain | 13% | 14% | 14% | 5% |
| Headache | 8% | 9% | 9% | 7% |
| Abdominal distension | 7% | 9% | 8% | 3% |
| Fatigue | 6% | 7% | 9% | 4% |
| Hair loss | 4% | 4% | 5% | 2% |
Source: Foundayo US Prescribing Information, pooled weight-management trial data.
How Many People Quit Because of Side Effects?
| Dose | Discontinued (Any Adverse Reaction) | GI-Related Discontinuation |
|---|---|---|
| 5.5 mg | 6% | 3% |
| 9 mg | 9% | 6% |
| 17.2 mg | 10% | 6% |
| Placebo | 3% | 1% |
GI adverse reactions were reported in 60–69% of Foundayo patients across doses vs 37% on placebo — that sounds high, but most were mild to moderate and concentrated in the first weeks after each dose increase. At the highest dose, only 1 in 10 stopped due to any adverse reaction, and only 6% stopped specifically because of GI issues. That means 94 out of 100 people found the stomach side effects manageable enough to continue.
About hair loss
Hair loss showed up in trials at 4–5% depending on dose. This isn't unique to Foundayo — it's a known phenomenon with significant weight loss from any cause, including surgery and calorie restriction. The medical term is telogen effluvium (a temporary disruption of the hair growth cycle triggered by rapid weight change), and it's typically reversible as your body stabilizes at its new weight.
Serious Warnings
⚠️ Boxed warning — thyroid C-cell tumors
GLP-1 receptor agonist activity caused thyroid tumors in rodent studies. Do NOT take Foundayo if you or a family member has had medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2). Watch for a lump in the neck, hoarseness, or trouble swallowing.
Pancreatitis
Stop Foundayo and contact your doctor immediately if you experience severe, persistent abdominal pain that radiates to your back.
Gallbladder problems
Gallstones and gallbladder inflammation have been reported with GLP-1 medications including Foundayo.
Acute kidney injury
Usually related to dehydration from vomiting or diarrhea. Stay hydrated — this matters more than it sounds during the first weeks at each new dose.
Managing GI side effects in practice: Eat smaller meals. Avoid greasy, fried, and spicy foods during dose escalation. Stay hydrated — especially if you experience diarrhea or vomiting. The slow titration schedule exists for a reason; don't rush through it. Most people find the nausea peaks in the first 1–2 weeks after each dose increase and then fades significantly.
What Most Pages Miss: Foundayo's Hidden Label Frictions
Most Foundayo review pages mention big-picture side effects and move on. But the prescribing information contains three specific friction points that affect real people making real decisions — and almost nobody is surfacing them clearly.

1. The Birth Control Warning
If you take oral hormonal contraceptives (the pill), Foundayo's label says you should switch to a non-oral method (patch, ring, IUD, implant) or add a barrier method (condoms) for 30 days after starting Foundayo AND for 30 days after every dose increase.
With 6 dose steps, that means potential contraception disruption across your first 5–6 months on the medication. Foundayo slows gastric emptying (the speed at which food and medications move through your stomach), which can reduce how well oral medications are absorbed. The effect on oral contraceptive absorption hasn't been directly studied, so the label takes a cautious approach.
2. The Simvastatin Cap
If you take simvastatin for cholesterol, your dose cannot exceed 20 mg/day while on Foundayo. Foundayo increases the active metabolite of simvastatin — even when dosing is staggered by 2 hours. This interaction is specific to simvastatin; atorvastatin and rosuvastatin do not have clinically relevant interactions with Foundayo.
3. The CYP3A4 Drug Interaction Ceiling
Foundayo is metabolized by the CYP3A4 enzyme system in your liver that processes many common medications. This creates two important rules:
Strong CYP3A4 inhibitors (e.g., clarithromycin, certain antifungals like ketoconazole, some HIV medications) cap your Foundayo dose at 9 mg — meaning you can never reach the highest doses where the strongest weight loss data exists.
Strong CYP3A4 inhibitors that also inhibit OATP1B should be avoided entirely with Foundayo.
Strong CYP3A4 inducers (e.g., rifampin, certain seizure medications) reduce Foundayo's effectiveness and should be avoided.
A clinical consultation covers drug interactions — this quiz helps you find the right provider
Who Qualifies for Foundayo — and Who Should Choose Something Else?
Foundayo is FDA-approved for adults with obesity (BMI ≥30) or overweight (BMI ≥27) with at least one weight-related medical condition such as high blood pressure, type 2 diabetes, high cholesterol, or obstructive sleep apnea. But qualification is just step one. The real question is whether Foundayo specifically is the right GLP-1 for you.
Foundayo Is Ideal If You...
Hate needles or refuse to self-inject
Foundayo eliminates injections entirely — it's a daily pill, full stop.
Tried an oral GLP-1 and quit because of the empty-stomach routine
As one Reddit user described: "the empty stomach rule finally broke me." Foundayo removes that friction completely. Take it with dinner, with your morning coffee, at bedtime. It works the same.
Travel frequently or have an unpredictable schedule
No refrigeration, no injection supplies, no fasting timer. One trial participant described the pill as easy to fit into a travel-heavy routine.
Want an FDA-approved brand-name medication at a cash-pay price starting under $200/month
$149/mo at the starting dose — the same entry price as the Wegovy pill.
Accept the convenience–efficacy tradeoff
~11% average weight loss is meaningful and clinically significant — even if injectables and the Wegovy pill produce more. A GLP-1 you stick with beats a more effective one you quit.
Foundayo Probably Isn't Your Best Path If...
Maximum weight loss is your only priority
Injectable tirzepatide (Zepbound) produces ~20–22% weight loss. Injectable semaglutide (Wegovy) produces ~15–18%. Even the Wegovy pill outperforms Foundayo's headline number. If the number on the scale matters most, injectables are the strongest path.
See our injectable GLP-1 provider comparison →You take oral birth control and don't want to change methods
The 30-day backup requirement after starting AND after each dose escalation creates months of contraception planning. The Wegovy pill doesn't have this specific issue.
You take simvastatin above 20 mg/day
You'd need a statin switch first. Atorvastatin and rosuvastatin are fine with Foundayo; simvastatin specifically is the issue.
You take a strong CYP3A4 inhibitor
Your max Foundayo dose would be capped at 9 mg, limiting your results to the 8.3% average weight loss tier — not the 11.1% maximum.
You specifically need a cardiovascular risk-reduction label
The Wegovy pill (or injection) is approved to reduce major cardiovascular events in eligible adults. Foundayo does not have this indication.
Budget is your absolute priority and you don't have insurance
The Wegovy pill runs approximately $50–$150 less over 6 months under current promotions. Under the Medicare GLP-1 Bridge (July 1, 2026), both are $50/month — so the gap closes entirely.
No email required · Personalized GLP-1 path recommendation
How Do You Actually Get Foundayo Right Now?
Foundayo is available today. Eli Lilly began accepting prescriptions through LillyDirect immediately upon FDA approval and started shipping on April 6, 2026, with broader retail pharmacy and telehealth availability expanding.
LillyDirect (Eli Lilly's Direct Platform)
LillyDirect works with licensed third-party pharmacies including Amazon Pharmacy and Prescryptive. Your doctor sends a prescription to LillyDirect Pharmacy (NPI: 1912889320, NCPDP: 1574056). A pharmacy partner confirms details, you choose home delivery (free shipping, 1–4 days) or retail pickup. Works for both insured and self-pay patients.
Your Doctor or Specialist
Any licensed prescriber — your primary care doctor, an endocrinologist, an obesity medicine specialist — can prescribe Foundayo and route it to LillyDirect or your retail pharmacy.
Telehealth Providers
Telehealth platforms prescribing FDA-approved GLP-1 medications are adding Foundayo as availability expands. Most require BMI ≥30, or BMI ≥27 with at least one weight-related condition — the same FDA-approved criteria as in-person visits.
Retail Pharmacies
CVS, Walgreens, Amazon Pharmacy, and other retail chains — broad availability expanding in the weeks following the April 6 launch.
Medicare (Starting July 1, 2026)
Through the CMS Medicare GLP-1 Bridge program, eligible Medicare beneficiaries can access Foundayo for $50/month starting July 1, 2026 through December 31, 2026. If you're on Medicare and can wait a few months, this cuts your monthly cost dramatically.
What to gather before your appointment
Your current height and weight (for BMI calculation)
A full list of current medications — especially statins, CYP3A4-interacting drugs, and oral contraceptives
Your current contraception method (if applicable)
Any history of medullary thyroid cancer in yourself or family
A recent blood pressure reading
Your insurance information (or decision to self-pay)
Walking in prepared means you leave with a prescription — not a follow-up appointment.
Foundayo, the Wegovy pill, injectable, or something else — personalized in under a minute
Foundayo Dosage: How You Take It and How Fast It Escalates
Foundayo (orforglipron) is a once-daily pill taken orally. Swallow the tablet whole — do not crush, break, or chew it. Store tablets in the original bottle and protect from light.
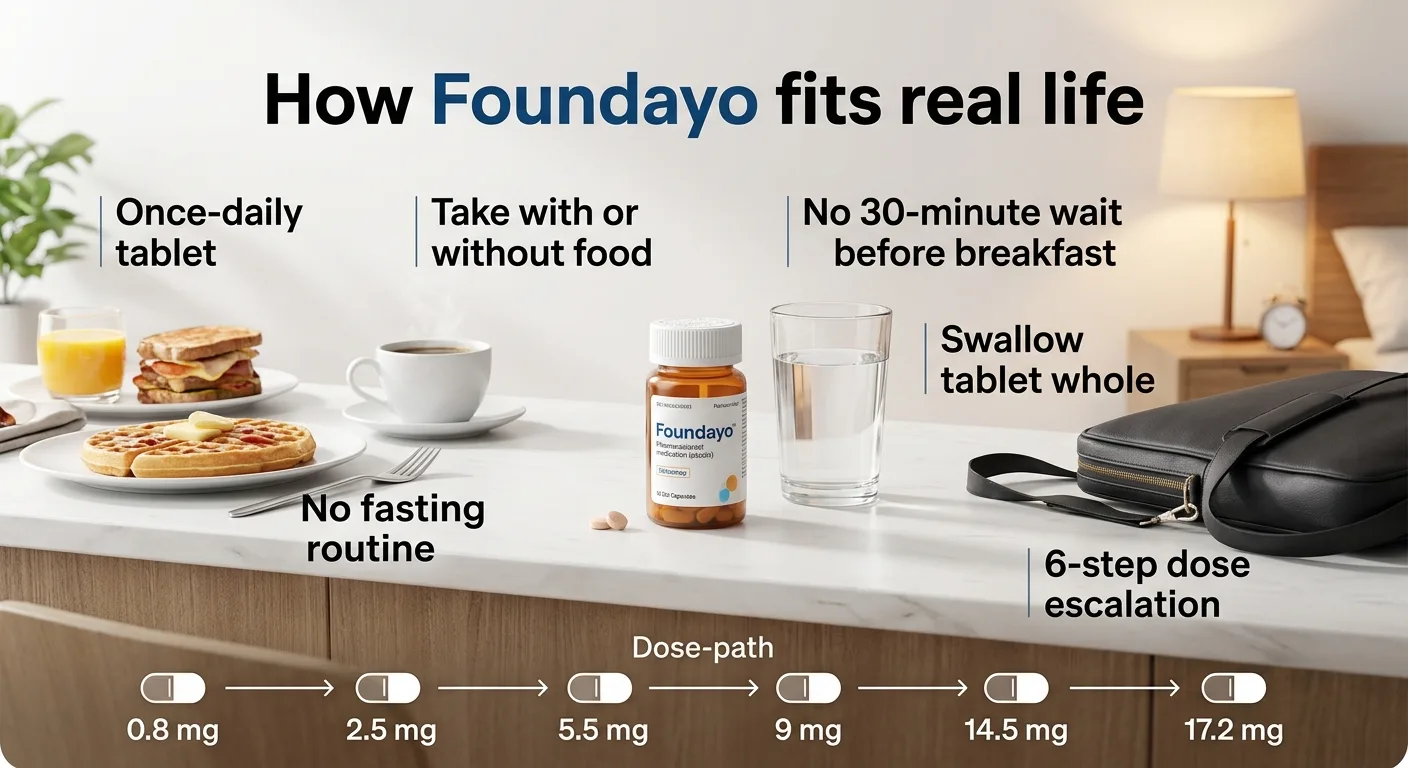
The Foundayo Titration Schedule
| Month | Dose | Notes |
|---|---|---|
| Month 1 | 0.8 mg | Starting dose — expect some GI adjustment |
| Month 2 | 2.5 mg | First escalation — GI side effects may briefly return |
| Month 3 | 5.5 mg | First maintenance-eligible dose (7.4% avg weight loss) |
| Month 4+ | 9 mg (optional) | Based on response and tolerability |
| Month 5+ | 14.5 mg (optional) | Based on response and tolerability |
| Month 6+ | 17.2 mg (optional) | Maximum dose — 11.1% avg weight loss |
Each escalation requires at least 30 days on the current dose. Not everyone needs or reaches the maximum dose — the 5.5 mg dose alone showed 7.4% average weight loss.
“Take anytime, with or without food” — what that actually means in practice
This is Foundayo's single biggest practical advantage over the Wegovy pill. The Wegovy pill must be taken on an empty stomach in the morning with no more than 4 oz of plain water, followed by a 30-minute fast from all food, drink, and other oral medications.
Foundayo has zero timing rules. Take it with breakfast. Take it at 2 PM with a sandwich. Take it at midnight after a snack. For people with unpredictable schedules, shift work, early-morning medication regimens, or a simple dislike of fasting — this is the feature that makes Foundayo worth considering even with a slightly lower weight loss headline.
If you miss a dose
Take it as soon as you remember. Do not double up (no more than one tablet per day). If you miss 7 or more consecutive days, contact your clinician — you may need to go back to a lower dose and retitrate.
Can You Switch From Injections or the Wegovy Pill to Foundayo?
This is already one of the most common questions in GLP-1 communities on Reddit. Here's the honest answer.
Switching from the Wegovy pill to Foundayo
This makes the most sense if the empty-stomach routine is causing you to miss doses or feel frustrated. If you're adherent and losing weight on the Wegovy pill, switching to Foundayo means potentially less weight loss in exchange for more dosing flexibility. Talk to your clinician about whether the convenience gain justifies the potential difference for your situation.
Switching from injectable GLP-1s (Wegovy or Zepbound) to Foundayo
If you're on injectable Wegovy or Zepbound and it's working well, switching to Foundayo would likely mean less weight loss. The data gap is significant: injectables produce 15–22% vs Foundayo's 11%. The main reasons to consider: needle fatigue, travel convenience, or cost (if your injectable is more expensive than Foundayo).
Switching from compounded GLP-1 injections to Foundayo
Foundayo is a legitimate path if you want to move from compounded semaglutide or tirzepatide to an FDA-approved oral option. Tradeoff: you lose the injectable efficacy advantage but gain FDA-approved status, manufacturing consistency, and the convenience of a pill.
Important: do not take Foundayo with another GLP-1
The prescribing information explicitly states Foundayo should not be used with other GLP-1 receptor agonists simultaneously. Discuss any switching plan with your clinician before making changes — they'll advise on timing and dose restart protocol.
A note on head-to-head evidence: The strongest direct orforglipron head-to-head evidence Lilly has publicized was against oral semaglutide 14 mg in type 2 diabetes (the ACHIEVE-3 trial), not against the 25 mg Wegovy obesity tablet in the same population. Until there's a same-population head-to-head obesity trial, the comparisons between Foundayo and the Wegovy pill are educated estimates, not certainty.
What Are People Saying About Foundayo So Far?
Foundayo only started shipping on April 6, 2026, so broad real-world review data is still early. We'll update this section monthly as verified user experiences accumulate.
Clinical trial participant (Business Insider)
“One trial participant described how easily the pill fit into a travel-heavy work routine — no fasting, no injection supplies, no refrigeration. Just a pill with whatever else you're doing.”
Reddit GLP-1 communities — early adopter sentiment
Reddit discussions show active interest in switching to Foundayo. The recurring theme: routine frustration. People who tried oral semaglutide and found the empty-stomach fasting rule unsustainable are the most interested early audience.
Physician perspective
Dr. Deborah Horn, director of the Center for Obesity Medicine at UTHealth Houston, noted that Foundayo addresses both clinical realities of obesity and practical daily challenges. Dr. Tara Narula (Good Morning America) emphasized that having a pill option helps normalize obesity treatment the same way blood pressure and cholesterol pills are normalized.
Why we don't use star ratings on this page
Foundayo has been shipping for days — not months. Using star ratings without meaningful real-world review volume is misleading structured data. When verified patient feedback reaches the point where a rating is meaningful, we'll add one. Until then, the comparison data and verified facts above are more useful than a made-up score.
This section last updated: April 8, 2026
How We Evaluated Foundayo
Every claim on this page traces to a specific, verifiable source. Here's our methodology.
| Source | What We Checked | Verified |
|---|---|---|
| Foundayo US Prescribing Information (DailyMed) | Dosing, side effects, warnings, drug interactions, clinical trial data | April 8, 2026 |
| Eli Lilly investor press release | Approval details, trial headline numbers | April 8, 2026 |
| LillyDirect pricing pages | Self-pay cost by dose, savings card terms, 45-day offer rule | April 8, 2026 |
| CMS Medicare GLP-1 Bridge guidance | Medicare eligibility, $50/mo pricing, July 1 start date, coverage of both drugs | April 8, 2026 |
| Wegovy tablet US Prescribing Information (FDA) | Adverse-reaction discontinuation rate, dosing, warnings | April 8, 2026 |
| Novo Nordisk pricing guide | Wegovy pill self-pay pricing, 4 mg promo end date (Aug 31, 2026), savings card terms | April 8, 2026 |
| ATTAIN-1 trial data | Foundayo efficacy by dose, responder rates, treatment-policy vs on-treatment estimands | April 8, 2026 |
| OASIS 4 trial data | Oral Wegovy (semaglutide 25 mg) obesity efficacy | April 8, 2026 |
Our Decision-Weight Framework
Efficacy (clinical trial data)
The primary reason anyone considers a GLP-1
Convenience / Adherence (dosing flexibility)
The most effective drug is the one you actually take
Cash Economics (real cost over 6 months)
Actual cost, not headline price
Access (availability, channel options)
Matters most in the first months after launch
Safety Friction (drug interactions, contraception)
The hidden dealbreakers
Evidence Quality (trial design, data transparency)
How much we trust the numbers
Label Breadth (obesity + CV indication)
Additional clinical utility
Switching Flexibility (from other GLP-1s)
Relevant for the large existing GLP-1 user base
Foundayo FAQ
Is Foundayo FDA-approved?
Yes. The FDA approved Foundayo (orforglipron) on April 1, 2026 for adults with obesity (BMI ≥30) or overweight (BMI ≥27) with at least one weight-related medical condition. It was the fastest new drug approval since 2002 — reviewed in just 50 days under the FDA Commissioner's National Priority Voucher program.
Can you take Foundayo with food?
Yes. Foundayo can be taken at any time of day, with or without food, and without water restrictions. This is its primary advantage over the Wegovy pill, which must be taken on an empty stomach with no more than 4 oz of water, followed by a 30-minute fast from all food, drink, and other oral medications.
How much does Foundayo cost without insurance?
Self-pay through LillyDirect starts at $149/mo for the 0.8 mg starting dose, escalates to $199 at 2.5 mg, $299 at 5.5–9 mg, and up to $349 at 14.5–17.2 mg (or $299 with Lilly's purchase offer if you refill within 45 days). Over the first 6 months with the purchase offer, expect approximately $1,544 total.
Is Foundayo covered by Medicare?
Starting July 1, 2026, eligible Medicare beneficiaries can access Foundayo for $50/month through the CMS Medicare GLP-1 Bridge program. This program runs through December 31, 2026 and provides access outside the normal Part D benefit payment flow.
Is Foundayo better than the Wegovy pill?
It depends on what matters most to you. Foundayo is more convenient — no fasting, take it anytime. The Wegovy pill currently shows stronger average weight loss (13.6% vs 11.1%) and has a cardiovascular risk-reduction label that Foundayo doesn't. There is no head-to-head obesity trial between the two.
Can you switch from Wegovy or injections to Foundayo?
Potentially, but discuss with your clinician first. The prescribing information says Foundayo should not be used with another GLP-1 simultaneously. Switching from the Wegovy pill makes most sense if the fasting routine is causing missed doses. Switching from injectables means accepting less weight loss in exchange for more convenience.
What are the most common side effects of Foundayo?
Nausea (35% at highest dose), diarrhea (25%), constipation (24%), vomiting (24%), and abdominal pain (14%). These are most pronounced during dose escalation and typically improve as your body adjusts. Permanent discontinuation due to adverse reactions was 10% at the highest dose; GI-related discontinuation specifically was 6%.
Can you take Foundayo with birth control pills?
The prescribing information advises women on oral hormonal contraceptives to switch to a non-oral method (patch, ring, IUD, implant) or add a barrier method (condoms) for 30 days after starting Foundayo and after each dose increase. With 6 dose steps, this means potential contraception disruption across your first 5–6 months. Non-oral hormonal contraceptives are not affected.
Can you take Foundayo with simvastatin?
Yes, but simvastatin must not exceed 20 mg/day while taking Foundayo. Foundayo increases the active metabolite of simvastatin even when dosing is staggered by 2 hours. Atorvastatin and rosuvastatin do not have clinically relevant interactions with Foundayo.
Can you take Foundayo with another GLP-1?
No. The prescribing information states Foundayo should not be used with other GLP-1 receptor agonist medicines.
How long does Foundayo take to work?
Most people begin noticing appetite changes within the first few weeks. Clinically meaningful weight loss typically becomes apparent over several months as the dose escalates. In clinical trials, the full 72-week treatment period produced the headline results — this is a long-term medication, not a quick fix.
Is Foundayo the same as Ozempic or Wegovy?
No. Foundayo contains orforglipron, a small-molecule, non-peptide GLP-1 receptor agonist made by Eli Lilly. Ozempic and Wegovy contain semaglutide, a peptide-based GLP-1 made by Novo Nordisk. They target the same receptor but are completely different drugs with different molecular structures, different manufacturers, and different dosing schedules.
Who qualifies for Foundayo?
Adults with obesity (BMI ≥30) or adults who are overweight (BMI ≥27) with at least one weight-related medical condition, such as high blood pressure, type 2 diabetes, high cholesterol, or obstructive sleep apnea. It is used alongside a reduced-calorie diet and increased physical activity.
Is Foundayo available through LillyDirect right now?
Yes. Lilly began accepting prescriptions through LillyDirect immediately upon FDA approval and started shipping on April 6, 2026. Broader retail pharmacy and telehealth availability is expanding. LillyDirect works with Amazon Pharmacy and Prescryptive.
Still Not Sure Which GLP-1 Program Is Right for You?
You've read the data. You've seen the tradeoffs. If Foundayo sounds right, you know what to do. If you're still weighing options — that's smart, and we built something for exactly that.
Take our free 60-second GLP-1 matching quiz
Answer a few questions about your goals, budget, medications, and preferences. We'll show you the GLP-1 path that fits your specific situation — whether that's Foundayo, the Wegovy pill, an injectable option, or a compounded alternative.
Take the Free GLP-1 Matching Quiz →No email required · No signup · Personalized recommendation in under a minute
Medical disclaimer: This page is for informational purposes only and does not replace professional medical advice, diagnosis, or treatment. All pricing, access, and clinical data were last verified on April 8, 2026. Always make medical decisions with a qualified healthcare provider.
Related guides
The RX Index is an independent editorial publisher covering GLP-1 medications and telehealth providers. We are not affiliated with Eli Lilly, Novo Nordisk, or any drug manufacturer. We may earn a commission if you visit a provider through our links, but this does not influence our editorial content.