For the GLP-1 and GIP/GLP-1 medications most people are searching about — Wegovy, Zepbound, Ozempic, Mounjaro, Rybelsus, and Saxenda — the formal FDA hard stops come down to two categories:
Personal or family history of medullary thyroid carcinoma (MTC), or a diagnosis of MEN2
Section 4, FDA prescribing information via DailyMed
Serious hypersensitivity reaction to the specific drug or any of its ingredients
Exact label wording varies by brand. Section 4, FDA prescribing information via DailyMed

For those six mainstream brands, those are the two Section 4 categories. Older diabetes-focused GLP-1 drugs have some different or additional contraindications, which we cover in the brand-by-brand table below.
Everything else you've seen called a “contraindication” online — pregnancy, pancreatitis history, gastroparesis, kidney disease, upcoming surgery — lives in a different part of the label. Those are warnings, precautions, or situations described as “not recommended.” They matter. They're serious. But they're not Section 4 hard stops, and confusing them is exactly why you're still searching.
Am I Disqualified? The 30-Second Hard-Stop Check
Before you read another word, answer these five questions. Questions 1–3 screen for Section 4 hard contraindications on the six mainstream brands most people compare for GLP-1 weight loss and diabetes. Question 4 screens for a label-based caution that can change next steps. Question 5 handles the most common ambiguity that prevents a clean yes/no answer.
Hard-Stop Screening Tool
Answer all 5 questions to see your result. This screens for Section 4 contraindications only.
Have you or anyone in your family ever been diagnosed with medullary thyroid carcinoma (MTC)?
Wegovy, Ozempic, Rybelsus, Mounjaro, Zepbound, Saxenda prescribing information — Section 4
Have you been diagnosed with Multiple Endocrine Neoplasia syndrome type 2 (MEN2)?
Same sources — Section 4
Have you ever had a serious allergic reaction — anaphylaxis, angioedema, severe swelling, trouble breathing — to the specific GLP-1 drug you are considering or any of its ingredients?
Same sources — Section 4
Have you ever had a serious allergic reaction to a different GLP-1 drug than the one you're considering?
See yellow-lane note below
Are you unsure what type of thyroid cancer you or a family member had?
This is an educational screening tool, not a medical diagnosis. Always confirm with your healthcare provider.
What Does “Contraindication” Actually Mean on an FDA Drug Label?
This distinction is where almost every competing resource drops the ball — and it's the reason so many search results conflict with each other.
Every FDA-approved drug label is organized into numbered sections. Here are the four that matter for this page:
| Label Section | What It Means | Severity | Example |
|---|---|---|---|
| Section 4 — Contraindications | The drug must not be used in this situation. | Hard stop | Personal or family history of MTC |
| Section 5 — Warnings & Precautions | Serious concern. Requires monitoring, specialist input, or avoidance in certain cases. | Discuss with clinician | History of pancreatitis |
| Section 6 — Adverse Reactions | Something that may happen during normal use. | Side effect | Nausea, constipation |
| Section 8 — Use in Specific Populations | Guidance for pregnancy, lactation, pediatric use, etc. Important, but not automatically a Section 4 hard stop. | Review with clinician | Pregnancy |

This page covers Section 4 only.
When other sites list pregnancy, pancreatitis, gastroparesis, or kidney disease as “absolute contraindications,” they're pulling from Sections 5 or 8 — not Section 4. Those are genuinely important. But labeling them as hard stops either scares people away from medications they may still be eligible for if their healthcare provider determines they are appropriate, or dilutes the severity of the actual hard stops. You deserve to know the difference.
Why MTC, MEN2, and Serious Hypersensitivity Are the Hard Stops
Now that you know what the hard stops are, here's why the FDA singled these out.
What Is Medullary Thyroid Carcinoma (MTC)?
MTC is a specific and uncommon type of thyroid cancer. It starts in the C-cells of the thyroid gland — cells that produce a hormone called calcitonin. MTC accounts for roughly 3–4% of all thyroid cancers. [National Cancer Institute]
In rodent studies, GLP-1 receptor agonists caused dose-dependent thyroid C-cell tumors, including MTC. That finding was significant enough for the FDA to place a Boxed Warning (sometimes called a “black-box warning”) — the strongest safety alert the FDA issues — on many mainstream GLP-1 products. [Wegovy prescribing information — Boxed Warning]
Whether this risk translates to humans is still being studied. But because the potential consequence is cancer, the FDA took the most protective position: if you have a personal or family history of MTC, the drug is contraindicated.
Note: Not every GLP-1 product carries this specific Boxed Warning. Older exceptions exist, including Byetta and Adlyxin, which do not list MTC/MEN2 in their Section 4. See the brand table for details.
What Is MEN2?
Multiple Endocrine Neoplasia syndrome type 2 is a rare inherited genetic condition associated with MTC and other endocrine tumors (like pheochromocytoma and parathyroid tumors). It is often identified through genetic testing and/or characteristic family or tumor patterns. [MedlinePlus Genetics]
Because MEN2 is tightly linked to MTC risk, the FDA extended the same Section 4 contraindication.
Why MTC/MEN2 Specifically — Not All Thyroid Problems?
The FDA label language is specific. It says medullary thyroid carcinoma. It says MEN2. It does not say “thyroid problems,” “thyroid disease,” or even “thyroid cancer” broadly.
MTC arises from thyroid C-cells. The far more common papillary and follicular thyroid cancers arise from follicular cells — a different cell type entirely. [National Cancer Institute; American Thyroid Association] That biological distinction is why the label names MTC specifically. We break this down condition-by-condition in the thyroid decoder below.
What Counts as a Serious Hypersensitivity Reaction?
The second hard-stop category is a serious hypersensitivity reaction to the specific GLP-1 drug you are considering or any of its ingredients. Exact label wording varies by brand. [Section 4, all six mainstream brands] In practice, the reactions that matter here include:
- Anaphylaxis — severe whole-body response involving throat swelling, difficulty breathing, rapid blood-pressure drop
- Angioedema — deep swelling beneath the skin, especially face, lips, tongue, or throat
- Severe systemic reaction requiring emergency intervention
What this does not mean: injection-site redness, mild itching, or the nausea that's common when starting a GLP-1. Those are side effects or local reactions — not hypersensitivity in the Section 4 sense. [Wegovy prescribing information — Section 5.7]
One thing worth knowing: the Section 4 contraindication is product-specific. Semaglutide (Ozempic, Wegovy, Rybelsus) is a different molecule from tirzepatide (Mounjaro, Zepbound), which is different from liraglutide (Saxenda, Victoza). The inactive ingredients also vary between formulations. If you had a serious reaction to one, a different GLP-1 may still be possible — but current labels advise caution because it's unknown whether cross-predisposition exists. That decision belongs to your prescribing clinician, not a website. [Wegovy prescribing information — Section 5.7]
Which GLP-1 Brands Have Which Hard Contraindications?
Not every GLP-1 carries exactly the same Section 4 language. The six mainstream brands most people compare for GLP-1 weight loss and diabetes are nearly identical, but some legacy diabetes-focused drugs differ.

Mainstream Brands
| Brand | Active Ingredient | Typical Use | Section 4 Contraindications | Source |
|---|---|---|---|---|
| Wegovy | Semaglutide | Chronic weight management; CV risk reduction; MASH (injection only) | Personal or family history of MTC, or in patients with MEN 2 · Prior serious hypersensitivity reaction to semaglutide or to any of the ingredients in WEGOVY injection or WEGOVY tablet | DailyMed, rev. 2/2026 |
| Ozempic | Semaglutide | Type 2 diabetes | Personal or family history of MTC, or in patients with MEN 2 · A serious hypersensitivity reaction to semaglutide or to any of the ingredients in OZEMPIC | DailyMed |
| Rybelsus | Semaglutide (oral) | Type 2 diabetes | Personal or family history of MTC, or in patients with MEN 2 · A prior serious hypersensitivity reaction to semaglutide or to any of the ingredients in RYBELSUS or OZEMPIC tablets | DailyMed |
| Zepbound | Tirzepatide | Weight loss | Personal or family history of MTC, or in patients with MEN 2 · Known serious hypersensitivity to tirzepatide or any of the ingredients in ZEPBOUND | DailyMed |
| Mounjaro | Tirzepatide | Type 2 diabetes | Personal or family history of MTC, or in patients with MEN 2 · Known serious hypersensitivity to tirzepatide or any of the ingredients in MOUNJARO | DailyMed |
| Saxenda | Liraglutide | Weight loss | Personal or family history of MTC, or in patients with MEN 2 · Serious hypersensitivity reaction to liraglutide or to any of the ingredients in SAXENDA | DailyMed |
Legacy & Less Common GLP-1 Brands
These are primarily prescribed for Type 2 diabetes. Some share the mainstream core contraindications, while others differ:
| Brand | Active Ingredient | Section 4 Contraindications | How This Brand Differs |
|---|---|---|---|
| Victoza | Liraglutide | Personal or family history of MTC, or in patients with MEN 2 · Serious hypersensitivity reaction to liraglutide or to any of the ingredients | Same as Saxenda (same active ingredient, lower dose) |
| Trulicity | Dulaglutide | Personal or family history of MTC, or in patients with MEN 2 · Serious hypersensitivity reaction to dulaglutide or any of the product components | Standard class contraindications |
| Bydureon BCise | Exenatide ER | Personal or family history of MTC, or in patients with MEN 2 · Prior serious hypersensitivity reaction to exenatide or to any of the ingredients of BYDUREON BCISE · History of drug-induced immune-mediated thrombocytopenia from exenatide products | Adds a blood-disorder contraindication not found in other GLP-1s; discontinued in 2024 |
| Byetta | Exenatide | A prior severe hypersensitivity reaction to exenatide or to any of the ingredients in BYETTA · A history of drug-induced immune-mediated thrombocytopenia from exenatide products | Does not list MTC/MEN2 in Section 4; discontinued in 2024 |
| Adlyxin | Lixisenatide | Known severe hypersensitivity to lixisenatide or to any component of ADLYXIN | Does not list MTC/MEN2 in Section 4; discontinued in 2023 |
All entries verified against current official prescribing information (DailyMed and FDA labeling resources, as applicable) as of March 2026.
Does My Thyroid History Actually Count as a Disqualifier?
Thyroid confusion is the #1 reason people either panic unnecessarily or assume they're disqualified when they're not. Here's a condition-by-condition breakdown.
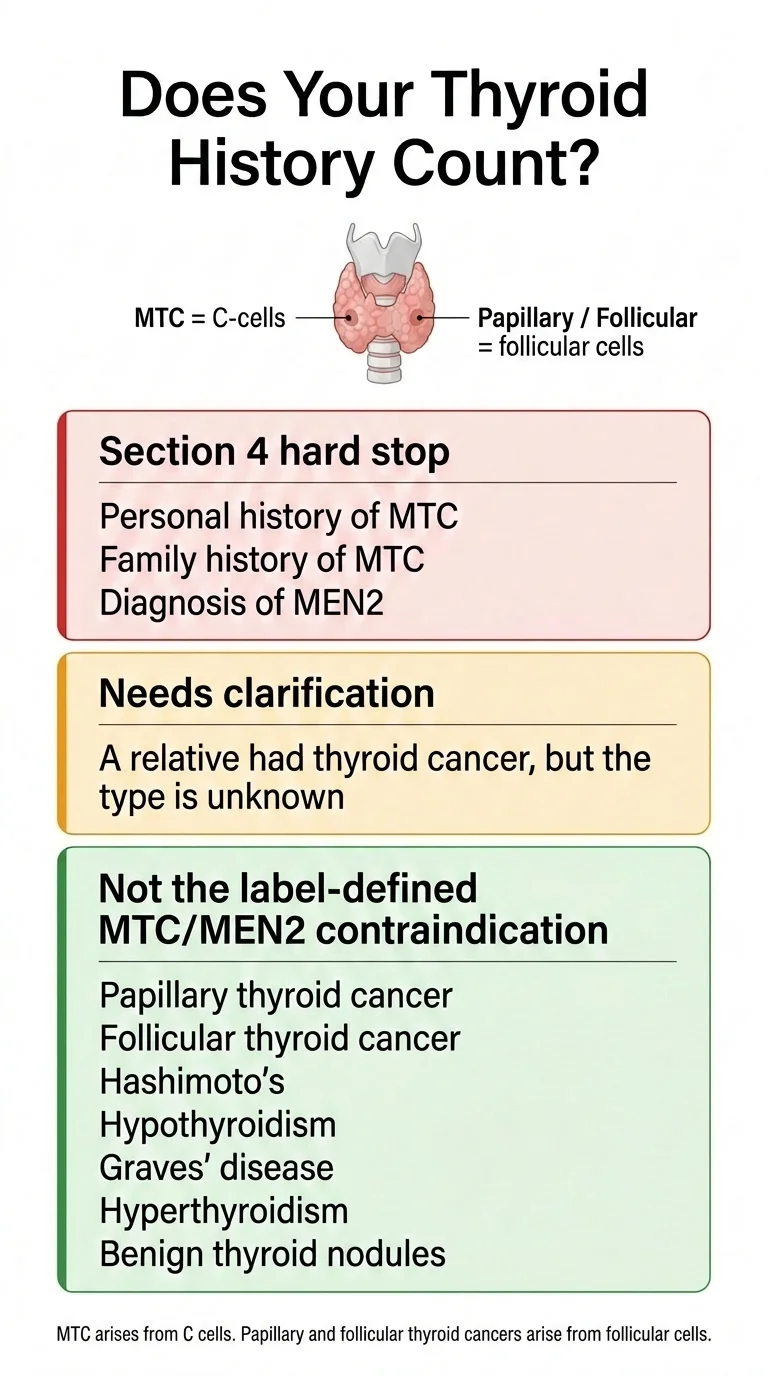
| Your Thyroid History | Section 4 Contraindication? | Why / Why Not | What to Do |
|---|---|---|---|
| Personal history of MTC | Yes | Directly named in Boxed Warning and Section 4 | Treat as a label-based hard stop. Do not start the drug; speak with the prescribing clinician about alternatives or specialist review. |
| Family history of MTC | Yes | “Personal or family history” is the exact label language | Same as above |
| Diagnosed with MEN2 | Yes | “Patients with MEN 2” is named in Section 4 | Same as above |
| Papillary thyroid cancer | Not the Section 4 contraindication | Arises from follicular cells, not C-cells. Most common type (~80% of thyroid cancers). | Not the label-defined MTC/MEN2 contraindication. Still review with your clinician. |
| Follicular thyroid cancer | Not the Section 4 contraindication | Same reasoning — different cell type than MTC. | Not the label-defined MTC/MEN2 contraindication. Still review with your clinician. |
| Hashimoto's / Hypothyroidism | Not the Section 4 contraindication | Hashimoto's is an autoimmune thyroid disease; hypothyroidism is an underactive-thyroid state with several possible causes. Neither is the label-defined MTC/MEN2 contraindication. | Not the label-defined MTC/MEN2 contraindication. Still review with your clinician. |
| Graves' disease / Hyperthyroidism | Not the Section 4 contraindication | Graves' disease is an autoimmune thyroid disease; hyperthyroidism is an overactive-thyroid state with several possible causes. Neither is the label-defined MTC/MEN2 contraindication. | Not the label-defined MTC/MEN2 contraindication. Still review with your clinician. |
| Thyroid nodules (benign) | Not the Section 4 contraindication | Common finding. Not the same as MTC. | Not the label-defined MTC/MEN2 contraindication. Mention to your provider for awareness. |
| “My relative had thyroid cancer but I don't know the type” | Needs clarification | The Section 4 contraindication specifically names MTC. If your relative had papillary or follicular (far more common), it likely does not trigger Section 4. But you need to verify. | Get the pathology report or diagnosis details before spending money on treatment or assuming you're disqualified. |
What Definitely Counts
Medullary thyroid carcinoma (personal or family history) and MEN2. These are the conditions named in the FDA Boxed Warning and Section 4 contraindication. [DailyMed — Wegovy, Ozempic, Mounjaro, Zepbound, Rybelsus, Saxenda prescribing information]
What the Label Does Not Name
These conditions are not the label-defined MTC/MEN2 contraindication. Papillary and follicular thyroid cancers arise from follicular cells rather than C-cells, while autoimmune thyroid disease, hypothyroidism, hyperthyroidism, and benign nodules are different thyroid conditions altogether. [NCI; ATA; NIDDK]
Any thyroid history is still worth mentioning to your prescriber. Transparency with your provider is always the right call — even when the label doesn't formally exclude you.
“My Mom Had Thyroid Cancer — Am I Out?”
Maybe not. The vast majority of thyroid cancers (~80%+) are papillary, which is not the type named in Section 4. MTC accounts for only about 3–4%.
If you don't know the specific type:
- Ask your family member (or their doctor) for the pathology report or diagnosis name.
- Look for the word “medullary.” Papillary, follicular, or anaplastic are different.
- If MEN2 runs in the family, genetic testing (RET proto-oncogene) can provide definitive answers.
Don't assume you're disqualified until you've confirmed the type. And don't assume you're clear until you have, either.
Calcitonin Testing or Thyroid Ultrasound Before Starting?
Current labels state that routine serum calcitonin monitoring or thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with these drugs. [DailyMed — Wegovy prescribing information, Section 5.1] Some providers may order baseline testing if your clinical picture warrants it, but it is not a label-required pre-treatment step.
Why Do Other Sites List Pregnancy, Pancreatitis, Gastroparesis, or Surgery Here?
Because they can carry significant risks. They just live in a different part of the label.
We're not minimizing these — we're categorizing them correctly so you can make better decisions.
| Condition | Where It Lives on the Label | What the Label Actually Says | Section 4? | Learn More |
|---|---|---|---|---|
| Pregnancy | Section 8 (Use in Specific Populations) | Product instructions vary. Wegovy (for CV risk/weight): discontinue when pregnancy is recognized. Semaglutide labels: discontinue at least 2 months before a planned pregnancy due to long half-life. Mounjaro: use during pregnancy only if potential benefit justifies potential risk. | No | GLP-1 & Pregnancy |
| Breastfeeding | Section 8 | Product instructions vary. Wegovy tablets: breastfeeding not recommended during treatment. Mounjaro: weigh maternal need and infant risk. Saxenda: patient and clinician decide whether to use the drug or breastfeed. | No | GLP-1 & Pregnancy |
| History of pancreatitis | Section 5 (Warnings & Precautions) | Discontinue if pancreatitis is suspected. It is not known if Ozempic can be used in people who have had pancreatitis. | No | Talk to Your Doctor |
| Severe gastroparesis | Section 5 | GLP-1s slow gastric emptying. Wegovy prescribing information: “not recommended in patients with severe gastroparesis.” | No | Talk to Your Doctor |
| Kidney disease | Section 5 | GLP-1 medications have also been shown to provide some kidney protection. In the label context, the concern is usually dehydration-related acute kidney injury from GI side effects. Some exenatide products are not recommended in severe renal impairment or low eGFR, but this is a warning/precaution, not Section 4. | No | — |
| Upcoming surgery / anesthesia | Section 5 | GLP-1 drugs can delay gastric emptying, which may increase aspiration risk during general anesthesia or deep sedation. Current labels instruct patients to inform their surgical/anesthesia team before planned procedures. | No — but requires coordination with your surgical team | Talk to Your Doctor |
The point isn't that these don't matter. The point is that someone with a history of pancreatitis and someone with a family history of medullary thyroid cancer are in fundamentally different positions — and they deserve to know that.
Will This Actually Stop Me From Getting Prescribed?
Here's how these categories typically play out — whether you're seeing a provider in person or through a telehealth platform.
When a Provider Will Usually Say No
If you have a confirmed personal or family history of MTC, a diagnosis of MEN2, or a documented serious allergic reaction to the specific drug you're requesting. That's the label doing its job.
When They'll Say “We Need More Information First”
This is more common than an outright no. You might land here if:
- A family member had “thyroid cancer” but you don't know the type
- You had an allergic reaction to a GLP-1 but you're not sure which ingredient or how severe it was
- Your medical history is complex and the provider wants specialist input
Don't interpret “we need more info” as rejection. It's the responsible path forward.
When You Can Move to the Next Question
If no formal hard contraindication applies, the conversation shifts to eligibility, side effect management, cost, insurance, and choosing the right medication. That's a different — and much more productive — set of questions.
Heading into a provider visit? Here's what to gather beforehand:
- The specific type of thyroid cancer in your family (if applicable) — not just “thyroid cancer”
- A detailed medication allergy history: which drug caused the reaction and what symptoms you experienced
- If thyroid cancer was involved: pathology report or diagnosis letter if available
- Your current medication list (especially insulin, sulfonylureas, or other diabetes drugs)
- Pregnancy status or plans, if relevant
Having these details ready can be the difference between leaving with a prescription and leaving with a follow-up appointment. Read our full doctor conversation guide
What Should I Do Now?
If You Hit a Red-Lane Hard Stop
A formal contraindication doesn't mean your weight-loss journey is over — it means GLP-1 medications aren't the right tool for you specifically. Other FDA-approved options exist, including naltrexone-bupropion (Contrave), phentermine-topiramate (Qsymia), orlistat (Alli/Xenical), medically supervised weight management programs, and bariatric surgery evaluation.
Talk to your provider about which path makes sense.
If You're in the Yellow Lane
Get the missing detail before paying for treatment or assuming you're disqualified. Verify the type of thyroid cancer in your family. Clarify your allergy history with the prescribing clinician; allergy specialist input may be helpful if the prior reaction was serious or unclear. Review gray-area conditions with the appropriate specialist.
If You're in the Green Lane
No formal Section 4 hard contraindication was found on this page. The next step is a provider consultation to confirm eligibility, discuss side effects, compare medications, and find the most affordable path.
“Green lane” means no Section 4 hard contraindication was identified here. It does not mean “approved” or “safe for you specifically.” A healthcare provider still needs to evaluate your complete medical history, including any Section 5 warnings and precautions that may apply.
Frequently Asked Questions
Who should not take GLP-1 medications?
For the six mainstream brands (Wegovy, Ozempic, Rybelsus, Mounjaro, Zepbound, Saxenda), the Section 4 hard stops are a personal or family history of medullary thyroid carcinoma (MTC), a diagnosis of MEN2, or a serious hypersensitivity reaction to the specific drug or its ingredients. Exact hypersensitivity wording varies by product label.
What are the absolute contraindications for semaglutide (Ozempic, Wegovy, Rybelsus)?
Two categories: a personal or family history of MTC, or a diagnosis of MEN2; and a serious hypersensitivity reaction to semaglutide or its ingredients. Ozempic injection says "a serious hypersensitivity reaction"; Wegovy and semaglutide tablets say "a prior serious hypersensitivity reaction." Pregnancy, pancreatitis, and gastroparesis are in other label sections — serious, but categorized differently.
What are the contraindications for tirzepatide (Mounjaro, Zepbound)?
Two categories: a personal or family history of MTC, or a diagnosis of MEN2; and known serious hypersensitivity to tirzepatide or its ingredients.
Is pregnancy an absolute contraindication to GLP-1 drugs?
FDA labels classify pregnancy under Section 8 (Use in Specific Populations), not Section 4 (Contraindications). Product instructions vary: Wegovy (for weight/CV) says discontinue when pregnancy is recognized; semaglutide labels recommend stopping at least 2 months before a planned pregnancy; Mounjaro says use only if benefit justifies risk.
Is pancreatitis a contraindication to semaglutide?
Not in the Section 4 sense. Pancreatitis is in Warnings and Precautions (Section 5). Labels say to discontinue if pancreatitis is suspected. The Ozempic Medication Guide notes it is not known if Ozempic can be used in people who have had pancreatitis.
Does hypothyroidism or Hashimoto's disqualify me from Ozempic or Wegovy?
Hashimoto's is an autoimmune thyroid disease. Hypothyroidism is an underactive-thyroid state with several possible causes, including Hashimoto's. Neither is the label-defined MTC/MEN2 contraindication. Still review with your clinician.
Does papillary thyroid cancer count as a GLP-1 contraindication?
Papillary thyroid cancer is not the type named in the FDA Boxed Warning or Section 4 contraindication. It originates in follicular cells, not the C-cells involved in MTC, and accounts for about 80% of thyroid cancers. Not the label-defined Section 4 contraindication. Still review with your oncologist or endocrinologist.
What if my mother had thyroid cancer but I don't know the type?
You're in the yellow lane. The Section 4 contraindication specifically names MTC, which is rare (~3–4% of thyroid cancers). Most thyroid cancers are papillary. But you need to verify the type before proceeding — get the pathology report or specific diagnosis name.
Can I take a GLP-1 if I had an allergic reaction to a different GLP-1?
The Section 4 contraindication is product-specific — it applies to the drug you reacted to and its ingredients. Semaglutide, tirzepatide, and liraglutide are different molecules. However, current labels advise caution with other GLP-1 receptor agonists if you've had anaphylaxis or angioedema, because cross-predisposition is unknown. This requires clinician evaluation; allergy specialist input may be helpful.
Should I stop a GLP-1 before surgery?
Current product labels note that GLP-1 drugs can delay gastric emptying and that pulmonary aspiration has been reported in patients undergoing procedures with general anesthesia or deep sedation. Labels instruct patients to inform their surgical and anesthesia teams before planned procedures. This is not a Section 4 contraindication — it requires coordination with your surgical team.
Do the hard contraindications apply to oral GLP-1s too?
Yes. Oral semaglutide products — Rybelsus, Ozempic tablets, and Wegovy tablets — carry the same semaglutide Boxed Warning and Section 4 contraindications as injectable semaglutide. The route of administration does not change the medical disqualifiers.
Do I need calcitonin testing before starting a GLP-1?
Current labels state that routine serum calcitonin monitoring or thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with these drugs. It is not a label-required pre-treatment step.
How We Built This Page
We reviewed the current FDA-approved prescribing information for each GLP-1 and GIP/GLP-1 receptor agonist listed on this page, accessed through DailyMed (the National Library of Medicine's drug label database). We treated Section 4 (Contraindications) as the sole source of truth for what qualifies as a formal hard contraindication.
We cross-referenced the National Cancer Institute and American Thyroid Association for the biological distinction between MTC and other thyroid cancers, MedlinePlus Genetics for MEN2, the NIDDK for thyroid-condition definitions, and the FDA's labeling FAQ for the Section 4/5/6/8 framework.
We deliberately excluded common side effects and Section 5 precautions from the “hard contraindication” designation because the FDA prescribing information categorizes them differently — and we believe you deserve to know that distinction.
Sources Referenced
- Official Prescribing Information (DailyMed and FDA labeling resources, as applicable): Wegovy (semaglutide injection and tablets, rev. 2/2026), Ozempic (semaglutide injection and tablets), Rybelsus (semaglutide tablets), Mounjaro (tirzepatide injection), Zepbound (tirzepatide injection), Saxenda (liraglutide injection), Victoza (liraglutide injection), Trulicity (dulaglutide injection), Byetta (exenatide injection), Bydureon BCise (exenatide ER injection), Adlyxin (lixisenatide injection)
- FDA: Frequently Asked Questions About Labeling Prescription Medicines
- National Cancer Institute (NCI): Medullary Thyroid Cancer
- American Thyroid Association (ATA): Thyroid Cancer Overview
- MedlinePlus Genetics: Multiple Endocrine Neoplasia
- NIDDK: Hashimoto's Disease; Graves' Disease
Editorial Standards
Author: The RX Index Editorial Team
Responsible Editor: Kaden Coziar, Founder & Managing Editor
Medical Reviewer: Jillian Foglesong Stabile, MD, FAAFP, DABOM · Medical Review Policy
Last Reviewed: March 2026
Change Log: Initial publication March 18, 2026. Will be updated when FDA label language changes or new GLP-1 drugs are approved.
Disclosure: Some links on this page are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you.
This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before starting, stopping, or changing any medication. If you believe you are experiencing a medical emergency, call 911 or your local emergency number immediately.
Have a question we didn't cover? Contact us — we update this page based on real reader questions.