If you searched “GLP-3 peptide retatrutide,” here's the short version: Retatrutide is an investigational once-weekly triple agonist from Eli Lilly that activates the body's receptors for GIP, GLP-1, and glucagon. In the Phase 3 TRIUMPH-4 trial — which enrolled adults with obesity or overweight and knee osteoarthritis — participants on the 12 mg dose lost an average of 28.7% of their body weight at 68 weeks (Eli Lilly press release, Dec. 11, 2025; detailed results not yet peer-reviewed). “GLP-3” is a scientifically inaccurate nickname, not a formal classification. Retatrutide is not FDA-approved, it is not available at any pharmacy, and anything sold online as “retatrutide peptide” is not legitimate medical access. Eli Lilly says retatrutide is legally available only through Lilly-sponsored clinical trials, and public availability depends on remaining Phase 3 results and regulatory review.
That's the bottom line. But there's a lot more to this story — and if you're weighing your options, the details matter. We built this guide to answer every question you'd normally have to piece together from ten different tabs. What the drug actually does. What the latest Phase 3 data shows. What the new safety signal is that most sites aren't covering. Whether you should wait for it or start treatment now. And most importantly — what your safest, smartest next step actually looks like.
What people search vs. what's actually true
| What you typed | What you probably mean | Current reality | Honest next step |
|---|---|---|---|
| GLP-3 peptide retatrutide | “Tell me about retatrutide” | “GLP-3” is informal; retatrutide is investigational — not approved | Read the data below, then evaluate approved options |
| Retatrutide for sale | “Can I buy this?” | Not through any legitimate pharmacy or prescriber | Do not buy online; consider clinical trials or approved GLP-1s |
| Retatrutide results | “Does it work?” | Phase 3 data: 28.7% weight loss at 68 weeks in adults with obesity and knee OA | Strong results, but long-term data is still being collected |
| Retatrutide side effects | “What could go wrong?” | GI issues similar to other GLP-1 drugs, plus a new skin-sensation signal | See our full side-effects breakdown below |
| Retatrutide vs tirzepatide | “Is it better than what's available?” | Possibly more effective, but not approved and not directly compared yet | Tirzepatide is FDA-approved and available today |
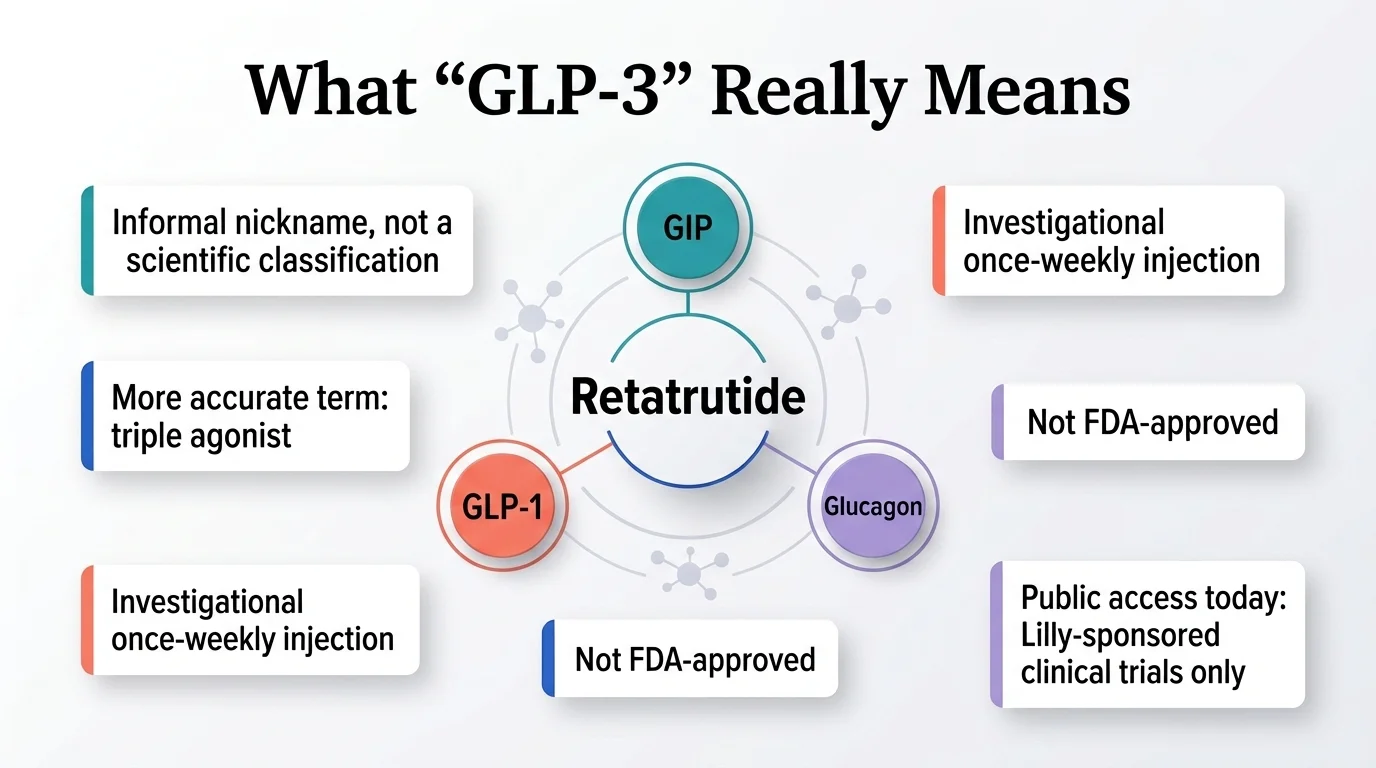
What Does “GLP-3 Peptide Retatrutide” Actually Mean?
Let's clear up the biggest source of confusion first.
“GLP-3” is not a medical term. You won't find it in a textbook or on an FDA label. It's a scientifically inaccurate media nickname. Eli Lilly has stated that the nickname emerged because retatrutide targets three receptors — GLP-1, GIP, and glucagon — not because “GLP-3” is a recognized scientific class. So when you see “GLP-3 peptide retatrutide,” what people actually mean is: retatrutide, the triple hormone receptor agonist.
To understand where retatrutide fits, it helps to see the progression of GLP-based weight loss drugs. The table below is an informal teaching aid — not a formal medical classification — but it captures how the science has evolved:
| Informal label | Drug | Receptors targeted | How it's known |
|---|---|---|---|
| Single agonist | Semaglutide (Wegovy, Ozempic) | GLP-1 only | The drug that started the weight-loss medication wave |
| Dual agonist | Tirzepatide (Mounjaro, Zepbound) | GLP-1 + GIP | Eli Lilly's first blockbuster; outperforms semaglutide in trials |
| Triple agonist | Retatrutide (LY3437943) | GLP-1 + GIP + Glucagon | Investigational; strongest weight loss data in trials so far |
Each step adds a receptor, and each receptor adds a distinct mechanism for weight loss. We'll unpack exactly what that means in the section on how it works.
So is “GLP-3” wrong? Technically, yes. Scientifically, “GLP-3” doesn't refer to any recognized receptor or peptide class. But practically, if you searched that term and landed here, you're in the right place. Retatrutide is what you're looking for. We'll use the proper name from here on.
Key fact: Retatrutide (also known by its research code LY3437943) is made by Eli Lilly and Company — the same company behind Mounjaro and Zepbound. It is currently in Phase 3 clinical trials. It has not been submitted to the FDA for approval. There is no brand name yet.
Why Is Retatrutide Everywhere Right Now?
If it feels like retatrutide appeared out of nowhere, here's why.
The Phase 3 data has been striking. In December 2025, Eli Lilly released topline results from the TRIUMPH-4 trial showing that participants on the highest dose of retatrutide lost an average of 28.7% of their body weight at 68 weeks, in a population of adults with obesity or overweight and knee osteoarthritis. That's roughly 71 pounds from a starting weight of about 249 pounds. For cross-trial context: semaglutide (Wegovy) has achieved about 15% weight loss, and tirzepatide (Zepbound) about 21%, though these were tested in different populations and trial designs.
Then on March 19, 2026, Eli Lilly released topline results from the TRANSCEND-T2D-1 trial, the first Phase 3 study of retatrutide in people with Type 2 diabetes. The results: up to 2.0% A1C reduction and up to 16.8% weight loss (efficacy estimand) at 40 weeks, with no sign of a weight-loss plateau (Eli Lilly press release; detailed results not yet peer-reviewed). For people with Type 2 diabetes — a population that historically struggles to lose weight on medication — these numbers are significant.
These results have driven enormous search interest. People are hearing about retatrutide from the news, social media, friends, even their doctors. But the gap between the early data and the reality of access is where confusion — and risk — live.
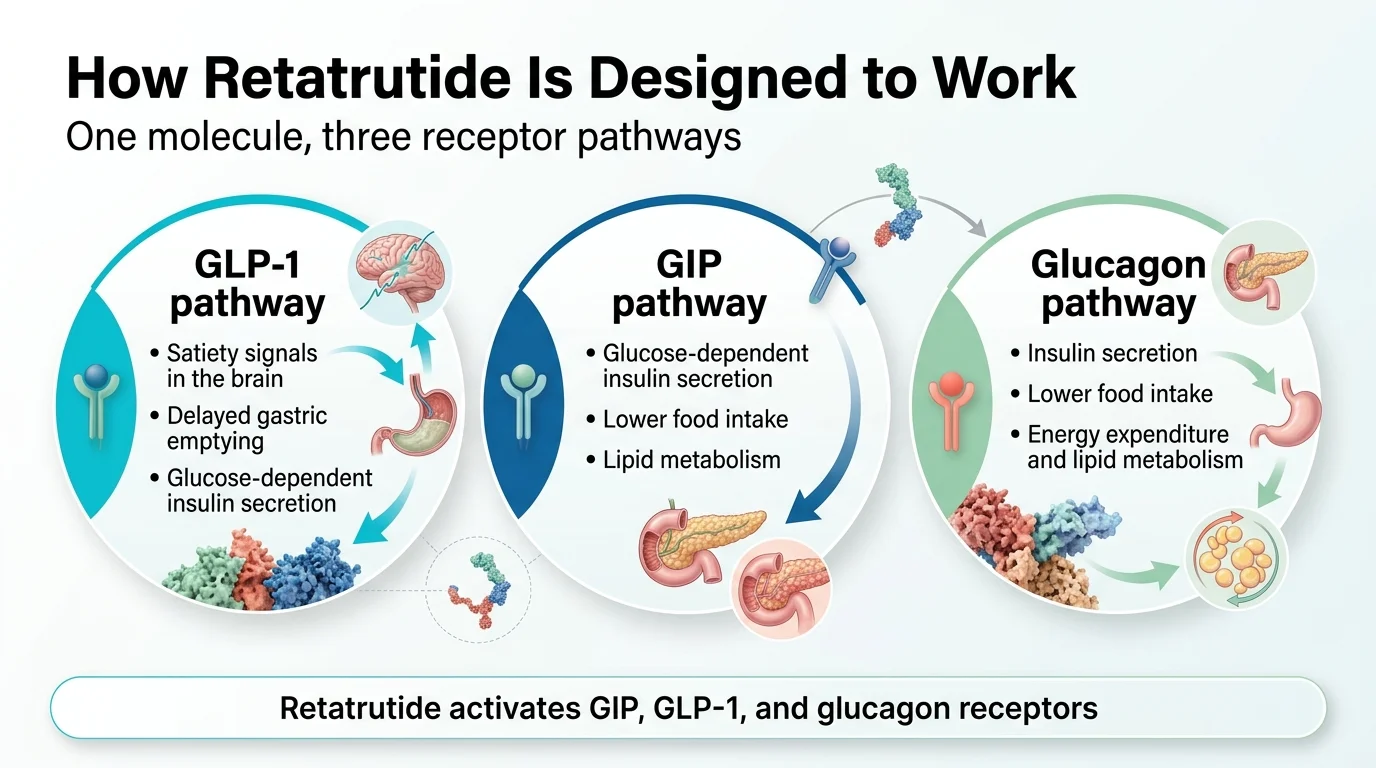
How Does Retatrutide Work in the Body?
Retatrutide works by mimicking three hormones your body already makes. Each one does something different, and the combination is what makes this drug unique.
GLP-1: The appetite brake
GLP-1 (glucagon-like peptide-1) is a gut hormone released after you eat. It tells your brain you're full, slows down how fast food leaves your stomach, and helps your pancreas release insulin. This is the hormone that semaglutide (Wegovy/Ozempic) mimics.
By activating GLP-1 receptors, retatrutide reduces appetite and food intake — the same core mechanism behind every GLP-1 drug on the market.
GIP: The metabolic amplifier
GIP (glucose-dependent insulinotropic polypeptide) is the second hormone. It enhances the satiety signal from GLP-1, improves insulin sensitivity, and appears to play a role in fat metabolism. This is what tirzepatide (Mounjaro/Zepbound) added on top of GLP-1 — and it's why tirzepatide outperforms semaglutide in trials.
Retatrutide activates GIP receptors too, but with notably high potency. Published data shows retatrutide is about 8.9 times more potent at the human GIP receptor than the body's own GIP hormone.
Glucagon: The calorie-burning engine
This is the new addition. Glucagon is traditionally seen as a “raise blood sugar” hormone — the opposite of insulin. So using it in a diabetes or obesity drug sounds counterintuitive. But here's what makes it work:
When combined with GLP-1 and GIP activation, the blood-sugar-raising effect of glucagon is counterbalanced. What remains is glucagon's other effects — and they're powerful:
- Increased energy expenditure. Your body burns more calories at rest.
- Enhanced fat breakdown (lipolysis). Stored fat gets mobilized for energy.
- Reduced liver fat. In a Phase 2a MASLD substudy, the 12 mg group had an 82.4% mean relative reduction in liver fat at 24 weeks, and 86% of participants in that group reached normal liver fat levels (below 5%). (Peer-reviewed: Hartman et al., published 2024, PMID 38858523)
This is the piece of the puzzle that semaglutide and tirzepatide don't have. Semaglutide reduces how much you eat. Tirzepatide reduces intake and improves metabolic efficiency. Retatrutide does both of those things and increases how much energy your body burns. Input goes down, output goes up. That combination is why the trial results have been so notable.
What scientists still don't fully understand
Retatrutide's half-life is about six days, which allows once-weekly dosing. But the precise interplay between all three receptors — why the glucagon component doesn't cause hyperglycemia in practice, exactly how much of the weight loss comes from reduced intake versus increased expenditure, and why the dysesthesia side effect appears at higher doses — is still under active investigation. Phase 3 data is filling in these gaps, but this drug is still being characterized. That matters.
What Do the Latest Retatrutide Clinical Trials Show?
We've compiled the evidence picture — what's been peer-reviewed, what's been released as company topline data, and what's still running. We label each source type so you know the level of scrutiny each finding has undergone.
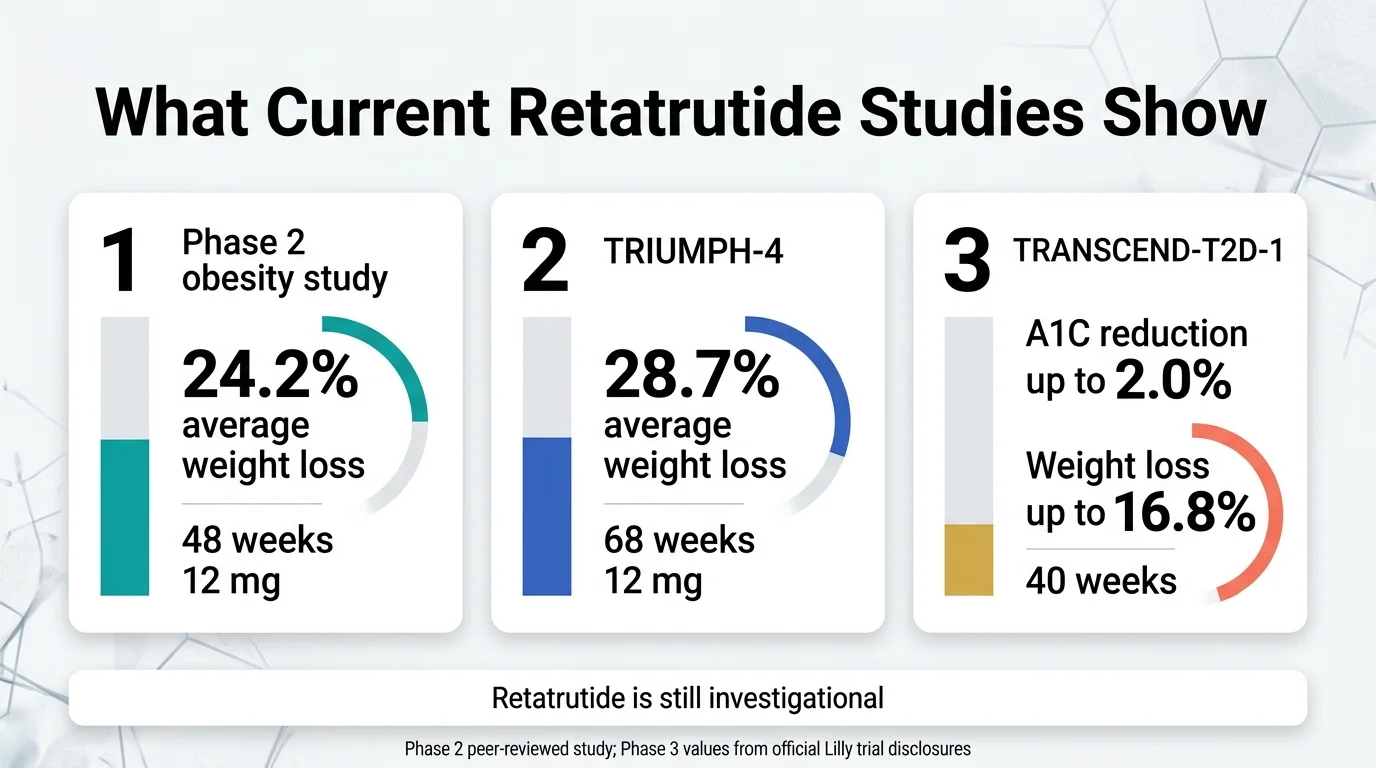
Phase 2 obesity trial — peer-reviewed (NEJM, 2023)
This was the study that put retatrutide on the map. Published in the New England Journal of Medicine, it enrolled 338 adults with obesity (BMI ≥30, or ≥27 with a weight-related condition) and tested multiple doses over 48 weeks.
Key results at the highest dose (12 mg):
- Average weight loss: 24.2% of body weight (vs. 2.1% with placebo)
- 26% of participants lost ≥30% of their starting weight
- Weight loss was still accelerating at 48 weeks — no plateau
- Adverse events were mostly GI (nausea, diarrhea, vomiting), dose-related, and mostly mild to moderate; a lower 2 mg starting dose partly mitigated them
This was a Phase 2 trial — smaller, earlier-stage — but the numbers were striking enough to generate significant interest in the obesity medicine community.
Source: Jastreboff AM et al., NEJM 2023 (PMID 37366315) — peer-reviewed
Phase 3: TRIUMPH-4 — Obesity + knee osteoarthritis (December 2025)
This was the first Phase 3 readout. It enrolled 445 adults with obesity or overweight plus knee osteoarthritis — a population dealing with both excess weight and chronic joint pain.
Key results at 68 weeks (efficacy estimand — participants who stayed on treatment):
| Measure | Retatrutide 9 mg | Retatrutide 12 mg | Placebo |
|---|---|---|---|
| Body weight loss | -26.4% (-64.2 lbs) | -28.7% (-71.2 lbs) | -2.1% |
| WOMAC pain reduction | -4.5 points (75%) | -4.4 points (75.8%) | -2.4 points (40.3%) |
| Completely pain-free at end | 14.1% | 12.0% | 4.2% |
Beyond weight and pain, retatrutide also improved cardiovascular risk markers: non-HDL cholesterol, triglycerides, hsCRP, and systolic blood pressure all declined.
The catch: Discontinuation rates were higher than typical for GLP-1 drugs — 12.2% (9 mg) and 18.2% (12 mg), compared to 4% for placebo. Eli Lilly attributed some of this to “perceived excessive weight loss” rather than adverse events.
The new safety signal: Dysesthesia — an abnormal skin sensation like tingling, burning, or sunburn-like sensitivity — appeared in 8.8% of the 9 mg group and 20.9% of the 12 mg group, versus just 0.7% on placebo. This was not seen prominently in Phase 2 and is now being closely monitored across all ongoing trials. We cover this in detail in the side effects section below.
Source: Eli Lilly press release, Dec. 11, 2025 — company-reported topline data; detailed results not yet published in a peer-reviewed journal. Eli Lilly says detailed results will be presented at a future medical meeting and published later.
Phase 3: TRANSCEND-T2D-1 — Type 2 diabetes (March 19, 2026)
This is the first Phase 3 trial of retatrutide specifically in people with Type 2 diabetes who hadn't been on other diabetes drugs.
Key results at 40 weeks:
- A1C reduction: up to 2.0% (efficacy estimand, from a baseline of 7.9%)
- Weight loss: up to 16.8% of body weight (efficacy estimand); 15.3% under the treatment-regimen estimand, which includes all randomized participants regardless of whether they stayed on the drug
- No weight-loss plateau was observed through 40 weeks
- Cardiovascular risk factors (non-HDL cholesterol, triglycerides, blood pressure) improved
- Discontinuation due to side effects: up to 5%
- Dysesthesia: 2.3–4.5% across dose groups (lower than TRIUMPH-4)
- GI side effects: nausea (26.5%), diarrhea (22.8%), vomiting (17.6%) at highest dose
For context, people with Type 2 diabetes typically lose less weight on GLP-1 drugs than people without diabetes. The fact that retatrutide still showed 15–17% weight loss in this harder-to-treat population is noteworthy.
Source: Eli Lilly press release / PRNewswire, March 19, 2026 — company-reported topline data; detailed results to be presented at the American Diabetes Association Scientific Sessions in June 2026.
What's still running: Selected major trials
Retatrutide is being studied across three large Phase 3 programs. Here's a snapshot of where things stand. Trial descriptions are based on Eli Lilly and ClinicalTrials.gov registry entries.
| Trial | Population | Status |
|---|---|---|
| TRIUMPH-1 | Adults with obesity or overweight (primary weight management; includes knee OA and OSA substudies) | Ongoing |
| TRIUMPH-2 | Adults with Type 2 diabetes and obesity or overweight (includes an OSA substudy) | Ongoing |
| TRIUMPH-3 | Adults with obesity or overweight and established atherosclerotic CVD and/or CKD | Ongoing |
| TRIUMPH-4 | Adults with obesity or overweight and knee osteoarthritis | ✓ Complete (Dec 2025) |
| TRANSCEND-T2D-1 | Adults with Type 2 diabetes (diet/exercise only) | ✓ Complete (Mar 2026) |
| TRANSCEND-T2D-2 | Adults with Type 2 diabetes inadequately controlled on metformin | Ongoing |
| TRANSCEND-T2D-3 | Adults with Type 2 diabetes on insulin | Ongoing |
| TRIUMPH-Outcomes | Cardiovascular and renal outcomes (~10,000 participants) | Ongoing |
| SYNERGY | MASLD/MASH liver disease | Ongoing |
What this means: Multiple Phase 3 readouts are anticipated over the coming months and beyond. Each will fill in a piece of the safety and efficacy puzzle. The cardiovascular outcomes trial (TRIUMPH-Outcomes) won't report for several more years, which means long-term cardiovascular safety data is still well in the future. Eli Lilly says public availability depends on the completion of these trials and the regulatory review process.
What Are the Side Effects and Risks of Retatrutide?
We're going to be straightforward about this because it's one of the areas where most coverage falls short. Retatrutide's side effect profile overlaps with other GLP-1 drugs, but it's not identical — and there's one finding that deserves more attention than it's getting.
The usual GI side effects
Like semaglutide and tirzepatide, retatrutide causes gastrointestinal side effects in a significant portion of users. In the Phase 2 trial, GI adverse events were dose-related, mostly mild to moderate, and occurred primarily during dose escalation. A lower 2 mg starting dose partly mitigated them (Jastreboff et al., NEJM 2023).
| Side effect | Retatrutide 12 mg (TRIUMPH-4) | For context: Tirzepatide 15 mg | For context: Semaglutide 2.4 mg |
|---|---|---|---|
| Nausea | Up to 43% | ~29% | ~44% |
| Diarrhea | ~33% | ~23% | ~30% |
| Vomiting | ~21% | ~12% | ~24% |
| Constipation | ~25% | ~12% | ~24% |
Important: These numbers come from different trials with different populations and durations. They're useful for general context, not as a direct head-to-head comparison.
Gradual dose escalation significantly reduces GI symptom severity. Participants who follow the standard titration (starting at 2 mg, increasing every 4 weeks) experience considerably fewer issues than those who escalate too quickly.
The dysesthesia signal — this is new
Dysesthesia is an abnormal skin sensation — tingling, burning, or a sunburn-like sensitivity to touch. It's the side effect that made headlines after TRIUMPH-4 and that most consumer-facing websites are either skipping or burying.
What the data shows:
- TRIUMPH-4 (Eli Lilly press release, Dec. 2025): 8.8% at 9 mg, 20.9% at 12 mg, vs. 0.7% on placebo
- TRANSCEND-T2D-1 (Eli Lilly press release, Mar. 2026): 2.3–4.5% across doses (lower rates)
- Phase 2 (NEJM 2023): Not prominently reported (which is why the Phase 3 signal caught analysts off guard)
What trial data indicates:
- In TRIUMPH-4, dysesthesia events were generally mild and rarely led to discontinuation (Eli Lilly press release)
- In TRANSCEND-T2D-1, Eli Lilly reported that most dysesthesia events resolved during treatment
- It appears to be dose-dependent — higher dose, higher incidence
What we don't know:
- Why it happens. No confirmed mechanism has been established.
- Whether it stabilizes, worsens, or resolves with longer-term use.
- Whether it has any implications beyond discomfort.
Our take: This is something to watch, not something to panic about. But it's also something to know about before making treatment decisions. Any site that doesn't mention it is doing you a disservice.
Other safety considerations
Heart rate increase: The Phase 2 trial reported dose-dependent heart-rate increases that peaked around week 24 and then declined (Jastreboff et al., NEJM 2023). The mechanism is not fully established but may be related to glucagon receptor activation.
Serious adverse events: In Phase 2, serious adverse event rates were 4% in both the retatrutide and placebo groups — a reassuring finding, though longer-term data is needed (NEJM 2023).
What we still don't know: Long-term safety beyond 68 weeks. Cancer risk with multi-year use. Weight regain patterns after stopping. Safety in elderly patients, adolescents, and those with significant kidney disease. The ongoing Phase 3 trials — especially the cardiovascular outcomes study — will address some of these gaps, but full answers are years away.
Is Retatrutide FDA Approved? Can You Buy It Online?
No and no. This section exists because the confusion here is real and the stakes are high.
FDA status: Investigational
As of March 2026, retatrutide has not been submitted to the FDA for approval. Eli Lilly is still collecting Phase 3 data. “Investigational” means the drug is being studied in controlled clinical trials. It has not gone through the full regulatory review process. It is not approved for any use — not for obesity, not for diabetes, not for anything. Eli Lilly says public availability depends on remaining Phase 3 results and regulatory review, and the company has not committed to a specific filing or approval date.
Can a compounding pharmacy make retatrutide?
No. The FDA says retatrutide cannot be used in compounding under federal law. It is not a component of an FDA-approved drug and has not been found safe and effective for any condition. The FDA has warned consumers about unapproved GLP-1 drugs used for weight loss, stating these products have not undergone the agency's evaluation for safety, effectiveness, or quality.
(Source: FDA, “FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss”)
What about “retatrutide peptide” sold online?
If you've seen websites selling “retatrutide” labeled as a “research peptide” or “for research use only” — that is not legitimate medical access. Eli Lilly says no one should consider taking anything claiming to be retatrutide outside a Lilly-sponsored clinical trial. These online products:
- Are not manufactured to pharmaceutical standards
- Are not FDA-evaluated for purity, potency, or safety
- Are explicitly labeled “not for human consumption”
- Are not the same product being studied in Eli Lilly's clinical trials
Our strong recommendation: Do not buy retatrutide online. The versions being sold outside of clinical trials are not the same product that's producing results in Lilly's controlled studies. The risk-to-benefit calculation doesn't work when you can't trust the product.
(Source: Eli Lilly, “What to Know About Retatrutide,” lilly.com)

How to tell if an access path is legitimate
Here's a simple checklist:
| Signal | Legitimate | Risky |
|---|---|---|
| Prescribed by a licensed physician? | ✓ | ✗ |
| Sourced from a licensed pharmacy? | ✓ | ✗ |
| Part of a registered clinical trial? | ✓ | ✗ |
| FDA-approved for this indication? | ✓ (not yet for retatrutide) | ✗ |
| Labeled “for research use only”? | ✗ | 🚩 |
| Promises “retatrutide for sale” before FDA approval? | ✗ | 🚩 |
| Offers COA but no prescriber relationship? | ✗ | 🚩 |
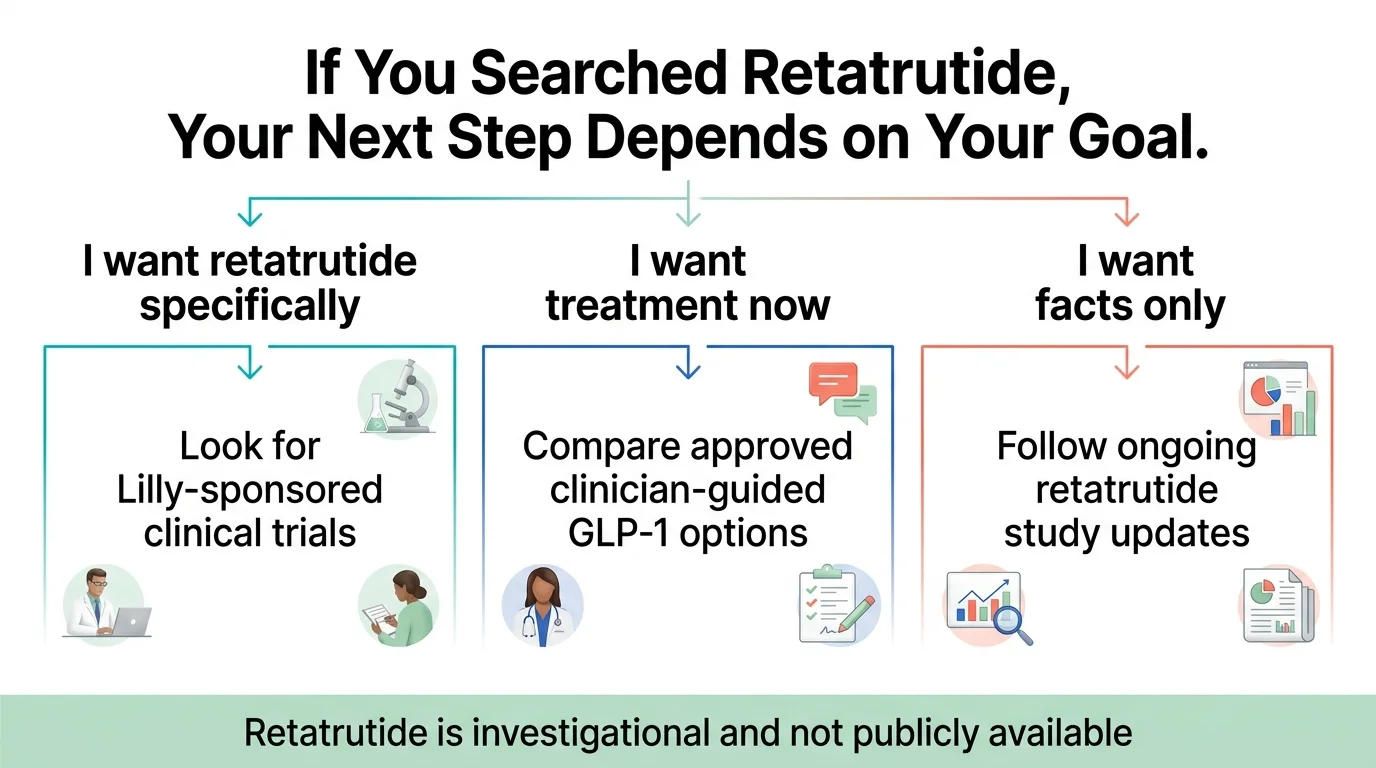
How Can You Access Retatrutide Legitimately Right Now?
There is currently one path: clinical trial enrollment.
Eli Lilly is running multiple Phase 3 trials under the TRIUMPH and TRANSCEND programs, with hundreds of sites across the United States and internationally. If you enroll and are randomized to the treatment arm, you receive pharmaceutical-grade retatrutide at no cost, with full medical supervision.
How to find an open trial:
- Go to ClinicalTrials.gov and search “retatrutide”
- Filter by recruiting status and location
- Contact the listed site coordinator to ask about eligibility screening
What to know before you apply:
- You may be randomized to placebo (that's how controlled trials work)
- Eligibility criteria are specific — typically BMI ≥30, or ≥27 with a weight-related condition
- You'll need to commit to regular visits, monitoring, and follow-up
- Trial participation is genuinely valuable — you get access to a next-generation drug under medical supervision, and you contribute to the data that will determine whether it gets approved
If you don't qualify for a trial — or if you'd rather start with something that's available now — that brings us to the next section.
Retatrutide vs. Semaglutide vs. Tirzepatide: The Full Comparison
This is the comparison most people searching this term are ultimately trying to make. We compiled data from published trials and official sources into one table. An important caveat upfront: no head-to-head trial between retatrutide and semaglutide or tirzepatide has been completed. A direct comparison trial of retatrutide vs. tirzepatide is currently underway (NCT06662383), but results aren't expected yet. Until that data exists, all comparisons are cross-trial — useful for general context, not definitive.
| Semaglutide (Wegovy) | Tirzepatide (Zepbound) | Retatrutide | |
|---|---|---|---|
| What it targets | GLP-1 (1 receptor) | GLP-1 + GIP (2 receptors) | GLP-1 + GIP + Glucagon (3 receptors) |
| Manufacturer | Novo Nordisk | Eli Lilly | Eli Lilly |
| FDA approved for weight loss | ✓ Yes | ✓ Yes | ✗ No |
| Available at pharmacies | ✓ Yes | ✓ Yes | ✗ No |
| Avg weight loss (best dose, cross-trial) | ~15% at 68 weeks | ~21% at 72 weeks | ~28.7% at 68 weeks (obesity + knee OA population) |
| A1C reduction (T2D) | ~1.5% | ~2.0%+ | ~2.0% |
| Administration | Weekly injection | Weekly injection | Weekly injection |
| Nausea rate | ~44% | ~29% | ~43% |
| Dysesthesia | Reported in case series at lower rates | Reported in case series at lower rates | 8.8–20.9% in Phase 3 (dose-dependent) |
| Liver fat reduction | Moderate | Significant | 82.4% mean relative reduction in Phase 2a MASLD substudy |
| Monthly cost | Check manufacturer sites for current pricing | No official price — not approved | |
| Insurance coverage | Variable, declining | Variable, declining | N/A (not approved) |
| Evidence maturity | Extensive (thousands of patients, years of real-world data) | Strong (large Phase 3 trials + real-world use since 2022) | Promising but early (Phase 3 in progress) |
What this comparison actually tells you
If you're looking at the raw weight loss data, retatrutide leads — and by a meaningful margin. But these results come from different trials in different populations and should be interpreted as cross-trial context, not head-to-head proof.
Semaglutide has years of real-world safety data, cardiovascular outcome trial data showing heart attack and stroke reduction, and is available right now at your pharmacy. Tirzepatide has been on the market since 2022, with strong efficacy data and growing clinical experience. Retatrutide has impressive early results — and a list of unknowns that won't be resolved until more trials report and the drug goes through FDA review.
The practical question isn't “which drug has the highest number.” It's: Which drug can I actually access, afford, and use under medical supervision right now?
For most people reading this page in 2026, that answer is semaglutide or tirzepatide.
How Much Will Retatrutide Cost?
There is no official retatrutide price because the drug is not approved. Eli Lilly has not announced pricing. Any specific dollar figure right now is speculative.
What we can say: if retatrutide is eventually approved, its pricing will likely be influenced by the existing GLP-1 market (where list prices for branded weight-loss injectables currently range from roughly $1,000–$1,350 per month before insurance). But the final number will depend on Eli Lilly's market strategy, the competitive landscape at time of launch, and payer negotiations — none of which are knowable today.
If you're looking for current cost information on approved GLP-1 medications, check the manufacturer sites directly — both Wegovy's official pricing page and Zepbound's official pricing page list up-to-date self-pay offers and savings programs, and these change frequently.
On compounded versions: Some sites quote monthly prices for compounded retatrutide. As we explained above, the FDA says retatrutide cannot be legally compounded under current federal law.
When Will Retatrutide Be Available?
Eli Lilly has not committed to a specific approval filing date or public launch timeline. What we know:
Where things stand now (March 2026): Multiple Phase 3 trials are ongoing across the TRIUMPH and TRANSCEND programs. Two Phase 3 readouts are complete (TRIUMPH-4, TRANSCEND-T2D-1). Additional results are anticipated over the coming months, according to Eli Lilly.
What has to happen before public availability: Eli Lilly must accumulate sufficient Phase 3 data, file a New Drug Application (NDA) with the FDA, and receive regulatory approval. The FDA review process typically takes 6–10 months after an application is accepted. After approval, manufacturing scale-up and distribution add further time before the drug reaches pharmacy shelves.
The bottom line on timing: Retatrutide is not available for prescription use today, and Eli Lilly says it is legally available only through Lilly-sponsored clinical trials. Drug development timelines are genuinely unpredictable. Planning your health decisions around a specific future date for an unapproved drug is not a strategy we'd recommend.
Should You Wait for Retatrutide or Start Treatment Now?
This is the question underneath all the other questions. You've read the data. You know retatrutide has produced the strongest weight loss results of any drug in this class so far. So should you hold off on treatment until it's available?
We've thought about this carefully, and here's our framework.
Start GLP-1 treatment now if:
- You have a BMI ≥30 (or ≥27 with weight-related health issues) — every month you wait is a month without the metabolic, cardiovascular, and quality-of-life benefits that proven GLP-1 drugs deliver
- You haven't tried GLP-1 therapy yet — semaglutide and tirzepatide are effective, FDA-approved, and available today
- You have obesity-related health conditions that need treatment now — Type 2 diabetes, sleep apnea, joint pain, fatty liver, cardiovascular risk
- You're losing time — retatrutide is not available for prescription use, and the timeline to availability remains uncertain
Consider watching and waiting if:
- You're already on a GLP-1 and it's working well — there's no reason to change something that's producing results
- You don't currently meet eligibility criteria — keep an eye on the landscape as it evolves
- You want to participate in a clinical trial — check ClinicalTrials.gov for TRIUMPH and TRANSCEND enrollment sites
Do not do this:
- ❌ Buy “retatrutide” from online peptide vendors. The FDA and Eli Lilly have both warned against this. These are not the same product being studied in clinical trials.
- ❌ Stop a working GLP-1 to “wait” for retatrutide. The health benefits you're getting now are real and cumulative.
- ❌ Assume any specific approval date. Drug development timelines shift. Plan around what's available, not what might be.
The honest bottom line
The best weight loss medication is the one you can access, afford, and take consistently under medical supervision. In 2026, for most people, that's semaglutide or tirzepatide. When retatrutide eventually reaches pharmacies — if and when it's approved — it may expand what's possible for people who need it. But waiting in limbo while effective treatments exist today is not a strategy — it's a delay.
How We Verified This Guide
We take accuracy seriously, especially on health topics. Here's how this page was built and how we keep it current.
Primary sources we use:
- FDA official communications and drug databases
- Eli Lilly corporate press releases and investor communications
- Peer-reviewed publications (NEJM, Diabetes Care, Obesity, PubMed-indexed journals)
- ClinicalTrials.gov registry entries
What gets rechecked on every update:
- Clinical trial status (active, completed, results available)
- FDA approval status
- Side-effect data as new trials report
- Legal/compounding status
How we label claims:
- Peer-reviewed data — cited to specific published study with PMID
- Company-reported topline data — from Eli Lilly press releases, labeled as such, with a note on whether detailed peer-reviewed publication is pending
- Unknown — stated clearly when the evidence doesn't exist yet
Affiliate transparency: The RX Index earns commissions when readers take action through our eligibility quiz and partner links. This does not affect our editorial recommendations, which are based on clinical evidence and independent analysis. We recommend options we'd recommend without the commission — and we say “don't do this” when the evidence warrants it.
Frequently Asked Questions About GLP-3 Peptide Retatrutide
Is "GLP-3" a real medical term?
No. "GLP-3" is a scientifically inaccurate nickname, not a formal classification. Eli Lilly says the nickname arose because retatrutide targets three receptors — GLP-1, GIP, and glucagon — not because "GLP-3" is a recognized scientific class. The accurate description is "triple hormone receptor agonist."
Is retatrutide FDA approved?
No. As of March 2026, retatrutide is an investigational drug in Phase 3 clinical trials. It has not been approved by the FDA or any other regulatory body worldwide. No New Drug Application has been filed.
Is retatrutide available outside of clinical trials?
Not through any legitimate medical channel. Eli Lilly says retatrutide is legally available only through Lilly-sponsored clinical trials. Products sold online as "retatrutide peptide" are unregulated and not equivalent to the pharmaceutical-grade drug being studied.
Can a compounding pharmacy make retatrutide?
No. The FDA says retatrutide cannot be used in compounding under federal law. It is not a component of any FDA-approved drug and does not appear on the FDA's bulk drug substances list.
How much weight can retatrutide help people lose?
In the Phase 3 TRIUMPH-4 trial — which enrolled adults with obesity or overweight and knee osteoarthritis — participants on the highest dose (12 mg) lost an average of 28.7% of their body weight (about 71 pounds from a starting weight of approximately 249 pounds) over 68 weeks (Eli Lilly press release, Dec. 2025; detailed peer-reviewed publication pending).
What are the most common side effects?
In TRIUMPH-4, the most common side effects included nausea (up to 43%), diarrhea (33%), vomiting (21%), and constipation (25%). A new signal — dysesthesia (abnormal skin sensations like tingling or burning) — affected up to 20.9% of participants at the highest dose. Most adverse events were dose-related and occurred primarily during dose escalation.
What is dysesthesia?
An abnormal sensation where normal touch feels unusual — tingling, burning, or sunburn-like sensitivity. In TRIUMPH-4, dysesthesia events were generally mild and rarely led to discontinuation. In TRANSCEND-T2D-1, Eli Lilly reported that most events resolved during treatment. The cause is not fully understood and is being monitored in ongoing studies.
Is retatrutide better than tirzepatide?
The weight loss data for retatrutide (28.7% in TRIUMPH-4) is numerically higher than tirzepatide (~21%), but these come from different trials with different patients. A direct head-to-head trial is currently underway (NCT06662383) but hasn't reported results. Tirzepatide has more mature evidence and is FDA-approved and available now.
Is retatrutide better than semaglutide (Wegovy/Ozempic)?
Cross-trial data suggests retatrutide produces substantially more weight loss than semaglutide (~28.7% vs ~15%). However, semaglutide has years of real-world safety data and proven cardiovascular benefits. They're at different stages of the evidence landscape.
When might retatrutide be available by prescription?
Eli Lilly has not committed to a specific filing or approval date. Public availability depends on remaining Phase 3 results and regulatory review. Drug development timelines are unpredictable, and we would not recommend planning health decisions around a specific date for an unapproved drug.
How much will retatrutide cost?
There is no official price because the drug is not approved. Any specific dollar figure at this point is speculative. Pricing will depend on Eli Lilly's strategy, the competitive landscape, and payer negotiations at the time of launch.
Is there a head-to-head trial comparing retatrutide and tirzepatide?
Yes. A Phase 3 trial (NCT06662383) is currently underway comparing retatrutide directly to tirzepatide in adults with obesity. Results have not been reported yet.
What does "research use only" mean on retatrutide sold online?
It means the product is explicitly not intended for human use. These are chemical compounds sold to laboratories, not medications manufactured for patients. Eli Lilly says no one should consider taking anything claiming to be retatrutide outside a Lilly-sponsored clinical trial.
Can retatrutide treat conditions other than obesity?
It's being studied for Type 2 diabetes (TRANSCEND program), knee osteoarthritis, obstructive sleep apnea, chronic low back pain, cardiovascular outcomes, and MASLD/MASH liver disease. None of these uses are approved.
Are retatrutide reviews reliable?
There are no normal post-market consumer prescription reviews for retatrutide because the drug is not publicly available. Eli Lilly says it is legally available only through clinical trials. Anecdotal reports from people using unregulated online products are not the same as approved-drug real-world evidence and should not be treated as such.
What is the TRIUMPH trial program?
TRIUMPH is Eli Lilly's Phase 3 clinical trial program for retatrutide in obesity and weight-related conditions. It includes multiple trials enrolling over 5,800 participants across populations with obesity, sleep apnea, osteoarthritis, and cardiovascular disease.
What should I do if I want weight-loss treatment right now?
FDA-approved options — semaglutide (Wegovy) and tirzepatide (Zepbound) — are available today and have strong evidence for safety and efficacy. A licensed healthcare provider can help determine which is right for your situation. Our eligibility quiz can also help narrow the options.
The Bottom Line
Retatrutide is an investigational triple agonist that has produced the strongest weight loss results of any drug in this class in clinical trials to date. The Phase 3 data — 28.7% body weight loss in adults with obesity and knee osteoarthritis, meaningful improvements in diabetes and cardiovascular risk markers — is noteworthy by any measure.
But it's not there yet. And in the space between promising clinical data and real-world access, there's noise: misinformation, gray-market vendors selling unregulated products, and a lot of websites that leave you more confused than when you arrived.
We built this page to be the antidote to that noise. One page, the most complete and honest picture of where retatrutide stands right now, updated as the story develops. Bookmark it. We'll keep it current as new Phase 3 data is released.
In the meantime — your weight loss journey doesn't have to wait for retatrutide. The drugs available today are effective, FDA-approved, and helping people right now.
Sources Cited in This Article
- Jastreboff AM et al., “Triple–Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial,” NEJM 2023 (PMID 37366315) — peer-reviewed
- Hartman ML et al., “Effects of Retatrutide on Liver Fat in Adults With Metabolic Dysfunction–Associated Steatotic Liver Disease,” NEJM 2024 (PMID 38858523) — peer-reviewed
- Eli Lilly Press Release: TRIUMPH-4 Phase 3 Topline Results, December 11, 2025 — company-reported
- Eli Lilly Press Release: TRANSCEND-T2D-1 Phase 3 Topline Results, March 19, 2026 — company-reported
- FDA: “FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss”
- Eli Lilly: “What to Know About Retatrutide” (lilly.com)
- ClinicalTrials.gov: NCT05931367 (TRIUMPH-4), NCT06354660 (TRANSCEND-T2D-1), NCT06662383 (Retatrutide vs Tirzepatide)
- Eli Lilly Medical: Retatrutide Clinical Trials Information (medical.lilly.com)
Medical Disclaimer: This content is for informational purposes only and does not constitute medical advice. Always consult a licensed healthcare provider before starting any medication.