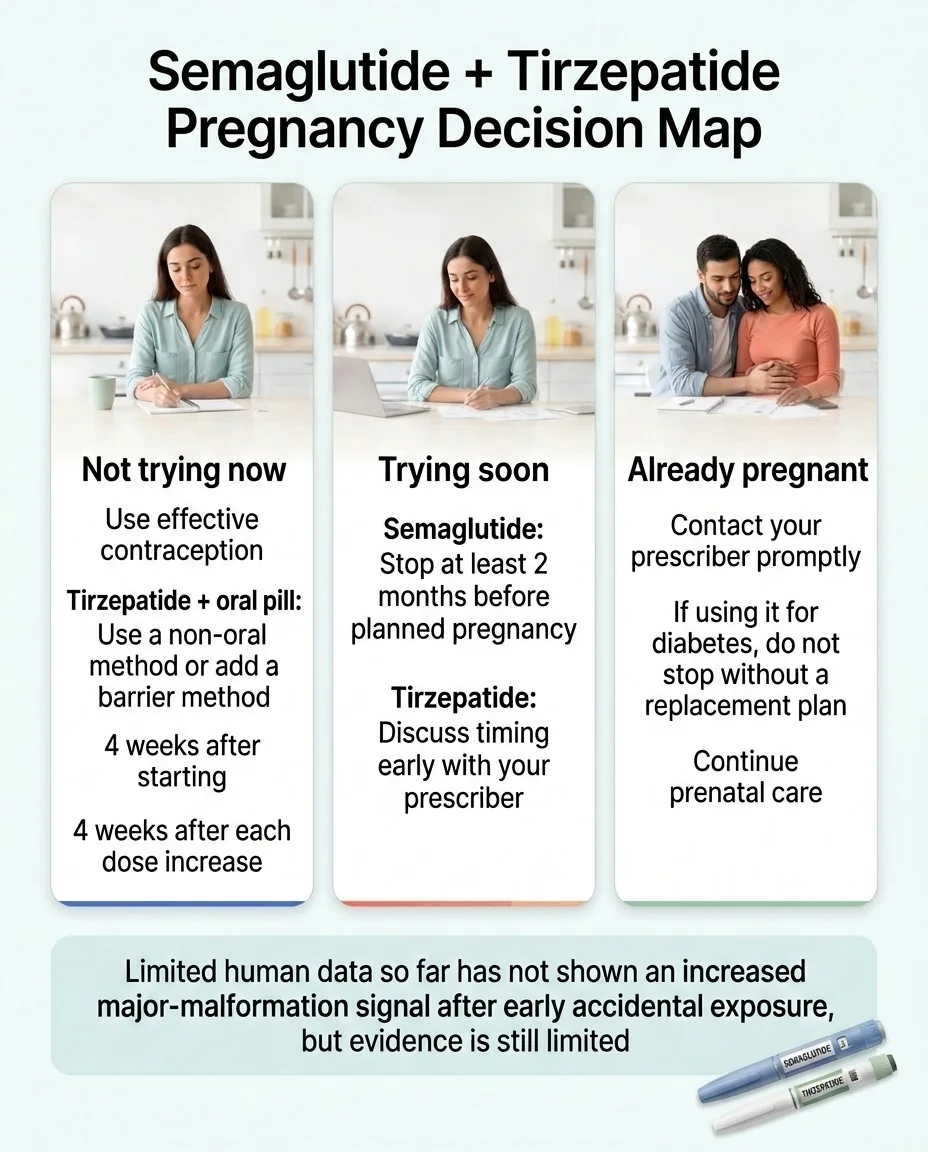
If you're taking a GLP-1 or GIP/GLP-1 medication — Ozempic, Wegovy, Mounjaro, Zepbound, or another — and pregnancy is anywhere on your radar, three things matter right now. First, GLP-1 and GIP/GLP-1 medicines are generally not recommended during pregnancy, and preconception planning should happen before conception. Second, tirzepatide (Mounjaro and Zepbound), a dual GIP/GLP-1 receptor agonist, is the main oral-birth-control exception — which most pages on this topic get wrong by lumping all GLP-1s together. Third, the timeline for stopping depends on which medication you take, and the difference between semaglutide and tirzepatide is weeks, not days.
We built this page because the current information out there is scattered across dozens of hospital blogs, fertility clinic marketing pages, and social media posts — none of which put the drug-specific details, birth control facts, and preconception timeline in one place. This is that one place.
Below, you'll find the washout timeline for your medication, a comparison of which birth control methods are affected (and which aren't), what to do if you're already pregnant, and how to handle this if you take a GLP-1 for diabetes — not just weight loss.
Why Is Everyone Suddenly Talking About GLP-1s and Pregnancy?
You've probably seen the term “Ozempic babies” on social media. Here's what's actually happening — and what's hype.
GLP-1 medications are not fertility drugs. None of them are FDA-approved for anything related to reproduction. But they can change the conditions around fertility in ways that catch people off guard.
Weight loss can improve ovulation. In some women with obesity or PCOS (polycystic ovary syndrome), modest weight loss can improve cycle regularity and restore ovulation. [ASRM, ACOG, Endocrine Society] If you've been told you're unlikely to conceive and you've now lost 20 or 30 pounds on a GLP-1, your body may disagree with that old diagnosis. Women who weren't using birth control because they believed they couldn't get pregnant are suddenly ovulating again, and they don't always realize it.
Tirzepatide can reduce oral birth control effectiveness. This is the part most articles get wrong. Not all GLP-1s affect the pill equally — but tirzepatide (Mounjaro/Zepbound) does, and the labeling is specific about it. More on this below.
The online conversation is ahead of the evidence. A 2025 review in PLOS ONE found that positive public sentiment about GLP-1s and fertility exceeds what the clinical evidence currently supports, particularly outside of PCOS. [Vu P, et al., PLOS ONE, 2025] That doesn't mean GLP-1s can't help — it means the picture is still developing, and a careful approach matters more than enthusiasm.
How GLP-1s Actually Affect Fertility and Birth Control
Before we get to the tables, it helps to understand the two mechanisms at play. This isn't complicated, but the distinction matters.
Delayed gastric emptying. GLP-1s work partly by slowing how fast your stomach empties. That's what makes you feel full longer. But it also means oral medications — including birth control pills — sit in your stomach longer and may not be absorbed as efficiently. This effect is clinically significant for tirzepatide specifically.
GI side effects. Nausea, vomiting, and diarrhea are common with all GLP-1s, especially during dose escalation. If you vomit within a few hours of taking your oral contraceptive, that pill may not have been absorbed. This is a practical pill-failure risk across the entire drug class — even the ones that don't affect absorption pharmacokinetically.
Metabolic improvement. Weight loss, better insulin sensitivity, and lower androgen levels can restore ovulation in women who weren't ovulating regularly. This is an indirect fertility effect — the drug isn't stimulating your ovaries, it's removing the metabolic obstacles that were suppressing them.
Non-oral birth control methods — IUDs, implants, patches, rings, injections — bypass your GI tract entirely. They are not affected by delayed gastric emptying or GI side effects from any GLP-1.
Which GLP-1s Affect Birth Control Pills — and Which Don't?
This is the most important table on this page. Bookmark it, screenshot it, or bring it to your next appointment.
The key distinction: tirzepatide is the only GLP-1/GIP agonist with a clinically significant effect on oral contraceptive absorption. The UK's Faculty of Sexual and Reproductive Healthcare (FSRH) confirmed this in their clinical statement. Semaglutide, liraglutide, dulaglutide, exenatide, and lixisenatide do not require additional barrier contraception for this pharmacokinetic reason. [FSRH CEU Clinical Statement]
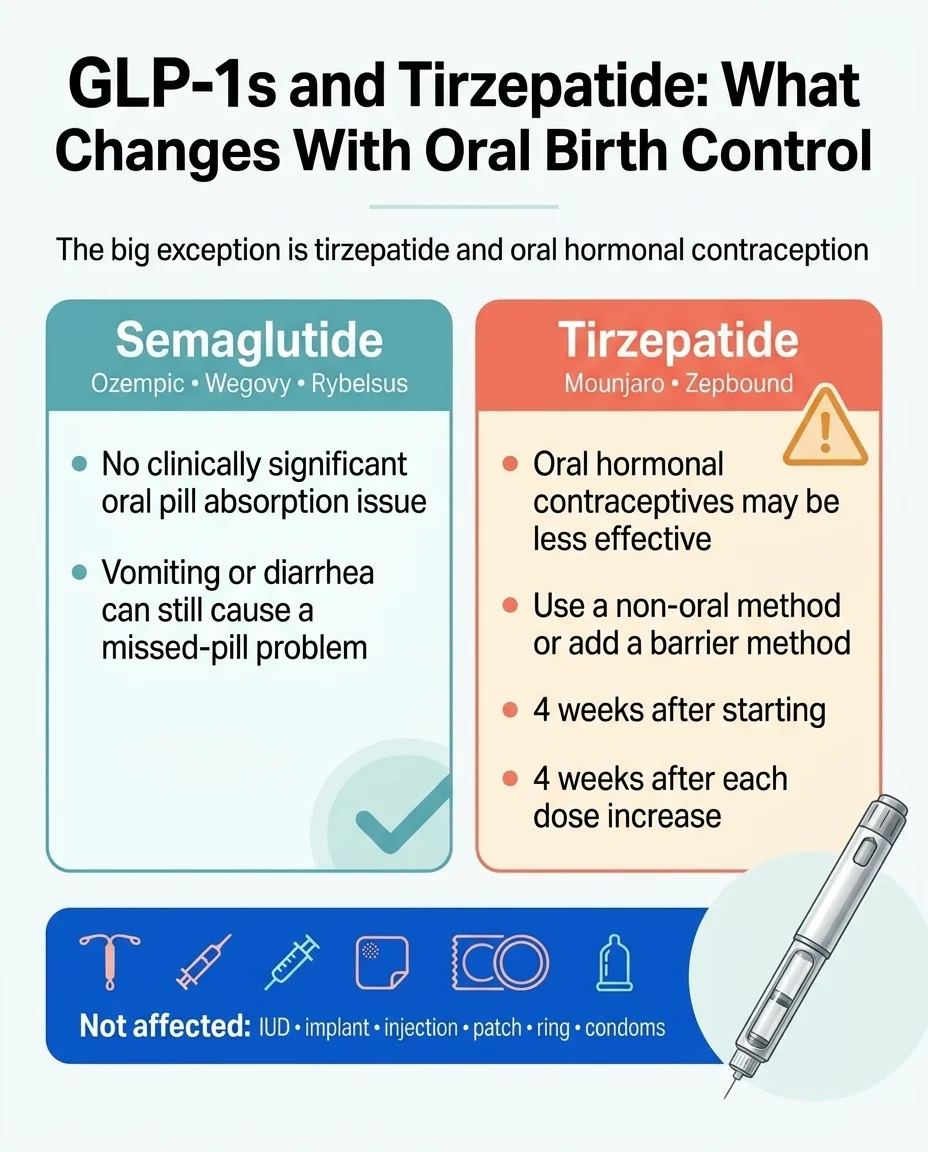
Most-Prescribed GLP-1/GIP-GLP-1 Drugs + Birth Control
| Your Birth Control | Semaglutide Ozempic, Wegovy, Rybelsus (all formulations) | Tirzepatide Mounjaro, Zepbound | Liraglutide Saxenda, Victoza | Dulaglutide Trulicity |
|---|---|---|---|---|
| Combined pill or mini-pill | ⚠️ No absorption issue — vomiting/diarrhea can cause a missed dose | ❌ May be less effective. Use a non-oral method OR add condoms for 4 wks after starting + each dose increase | ⚠️ No absorption issue — vomiting/diarrhea risk applies | ⚠️ No absorption issue — vomiting/diarrhea risk applies |
| Hormonal IUD | ✅ Not affected | ✅ Not affected | ✅ Not affected | ✅ Not affected |
| Copper IUD | ✅ Not affected | ✅ Not affected | ✅ Not affected | ✅ Not affected |
| Implant (Nexplanon) | ✅ Not affected | ✅ Not affected | ✅ Not affected | ✅ Not affected |
| Injection (Depo-Provera) | ✅ Not affected | ✅ Not affected | ✅ Not affected | ✅ Not affected |
| Patch or ring | ✅ Not affected | ✅ Not affected | ✅ Not affected | ✅ Not affected |
| Condoms | ✅ Not affected | ✅ Not affected | ✅ Not affected | ✅ Not affected |
Note: Exenatide and lixisenatide do not require added barrier contraception for this reason, but oral-contraceptive timing still matters: take oral contraceptives at least 1 hour before exenatide; with lixisenatide, take oral contraceptives at least 1 hour before or 11 hours after the dose. [FSRH CEU Clinical Statement]
Sources: Zepbound FDA prescribing information; FSRH clinical statement on GLP-1 agonists and contraception; semaglutide pharmacokinetic data (Kapitza C, et al., J Clin Pharmacol, 2015;55(5):497-504, PMC4418331)
The bottom line: If you're on tirzepatide and using the pill, talk to your prescriber about switching to a non-oral method or adding a barrier method. If you're on semaglutide and the pill, the absorption issue doesn't apply to you — but vomiting during dose escalation can still mean a missed dose. Treat it accordingly.
When to Stop Your GLP-1 Before Trying to Conceive
Every GLP-1 has a different half-life, which means they leave your body at different speeds. The standard pharmacological rule: it takes roughly 5 half-lives for a drug to clear your system.

Washout Periods by Medication
| Medication | Brand Names | Half-Life | ~Time to Clear | Labeling/Guidance Before Planned Pregnancy |
|---|---|---|---|---|
| Semaglutide (injectable) | Ozempic injection, Wegovy injection | ~7 days | ~5 weeks | At least 2 months before planned pregnancy (FDA) |
| Semaglutide (oral tablets) | Rybelsus, Ozempic tablets, Wegovy tablets | ~7 days | ~5 weeks | At least 2 months (FDA) |
| Tirzepatide | Mounjaro, Zepbound | ~5 days | ~3.5 weeks | Zepbound: discontinue when pregnancy is recognized; no numeric preconception washout stated. Mounjaro: use during pregnancy only if benefit justifies risk. UK FSRH/MHRA: about 1 month before planned pregnancy. |
| Liraglutide | Saxenda, Victoza | ~13 hours | ~3 days | Saxenda: discontinue if patient wishes to become pregnant or pregnancy occurs. Victoza: no numeric preconception washout stated. |
| Dulaglutide | Trulicity | ~5 days | ~3.5 weeks | No numeric U.S. preconception washout stated; use during pregnancy only if benefit justifies risk. |
| Exenatide ER | Bydureon | ~6 days | ~4 weeks | FSRH notes 12-week washout for ER formulation |
Sources: FDA prescribing information; FSRH clinical statement; UK MHRA guidance; MotherToBaby semaglutide and tirzepatide fact sheets
If you take a GLP-1 for type 2 diabetes, do not stop without a replacement plan. See the diabetes section below.
What If You Got Pregnant While on a GLP-1?
Accidental early exposure happens. The limited human data available so far has not shown an increased major-malformation signal in the available cohorts. But the evidence is still too limited to establish safety in pregnancy.
What to do right now:
- Contact your prescriber promptly. Weight-loss labels say to discontinue semaglutide or tirzepatide when pregnancy is recognized. However, if you use one of these drugs for diabetes, you need prompt clinician-guided replacement planning rather than an abrupt stop without a glycemic plan — see the diabetes section below.
- Report to the pregnancy registry. If the exposure was to Wegovy, the label lists a pregnancy registry: Novo Nordisk at 1-877-390-2760 or wegovypregnancyregistry.com. If the exposure was to Zepbound, the label lists Eli Lilly's pregnancy exposure registry at 1-800-LillyRx (1-800-545-5979). [Zepbound FDA PI]
- Continue standard prenatal care. Early accidental exposure, based on current evidence, has not shown elevated birth-defect risk in available studies — but monitoring is important.
What the evidence actually says:
The animal data is the reason these drugs carry pregnancy warnings — in rats and rabbits, GLP-1 exposure during development was linked to skeletal abnormalities, reduced fetal growth, and pregnancy loss. [Wegovy FDA PI; Zepbound FDA PI] That's concerning, and it's why these drugs are not recommended during pregnancy.
But human data tells a different story so far. A multicenter cohort study of 168 pregnancies with first-trimester GLP-1 exposure found no increased risk of major birth defects compared to reference groups. [Kim Dao K, et al., BMJ Open, 2024] MotherToBaby's summary reached a similar conclusion. A larger population-based cohort across 6 countries also did not find higher malformation risk. [Cesta CE, et al., JAMA Intern Med, 2024]
The honest take: The available human cohorts have not found an increased major-malformation signal after early accidental exposure. But neither study proves safety, and the evidence base is still small. The precautionary principle is the right approach — contact your prescriber, get care, and let your provider monitor things closely.
What If You Take a GLP-1 for Diabetes, Not Just Weight Loss?
Most articles skip this entirely, and it's one of the most important sections on this page.
If your GLP-1 manages your blood sugar — not just your weight — stopping cold is a bad idea. Abrupt discontinuation can spike blood glucose and cause rapid weight regain, both of which carry real risks during pregnancy. [Wyckoff JA, et al., J Clin Endocrinol Metab, 2025]
What needs to happen: Your prescriber (or an endocrinologist) and your OB/GYN need to coordinate a medication transition before you stop the GLP-1. The Endocrine Society specifically recommends discontinuing GLP-1 receptor agonists before conception — not at the point of a positive test — with timely transition to alternative therapy. [Wyckoff JA, et al., J Clin Endocrinol Metab, 2025]
What typically replaces a GLP-1 in pregnancy planning:
Insulin is the preferred medication for blood glucose management during pregnancy, per the American Diabetes Association. [ADA] The replacement plan should be individualized by the clinician managing diabetes and pregnancy planning; whether metformin has a role depends on the indication and the rest of the regimen.
Start this conversation 3–6 months before you want to try conceiving, so there's time to stabilize on a new regimen and confirm your blood sugar is well-controlled before pregnancy adds its own metabolic demands.
Can GLP-1s Improve Fertility If You Have PCOS?
For some women — yes, meaningfully. But not for the reasons social media suggests.
PCOS is the most common cause of ovulatory infertility, affecting an estimated 6–20% of reproductive-age women. [Szczesnowicz A, et al., J Clin Med, 2023] It's driven by insulin resistance, elevated androgens, and disrupted ovulation. GLP-1s address several of these root causes simultaneously: they reduce insulin resistance, promote weight loss, and lower circulating testosterone. Studies show women with PCOS on GLP-1 therapy saw greater improvements in menstrual regularity and ovulation rates compared to lifestyle changes alone. [Szczesnowicz A, et al., J Clin Med, 2023]
One study found that liraglutide + metformin was superior to metformin alone for IVF pregnancy rates in women with PCOS. Another showed significantly more spontaneous pregnancies in an exenatide group compared to metformin after 24 weeks. [Salamun V, et al., Eur J Endocrinol, 2018; Li R, et al., Arch Gynecol Obstet, 2023]
But the evidence is strongest for PCOS specifically. For women with obesity but without PCOS or diabetes, the fertility picture is less clear. [Vu P, et al., PLOS ONE, 2025] And the ASRM acknowledges GLP-1s as an option for weight management in PCOS while warning that effective contraception must be in place because pregnancy safety data remain insufficient. [ASRM, 2023]
The tension for women with PCOS: The drug that's finally working for your insulin resistance, cycles, and weight is the one you'll need to stop to try conceiving. There's no universal right answer — it depends on your age, how long you've been trying, your ovarian reserve, and your metabolic health. A reproductive endocrinologist can help you navigate the tradeoffs.
If you're 35+: Time is a factor that weight loss doesn't fix. Spending months optimizing on a GLP-1 means months not trying to conceive while age-related fertility decline continues. Some clinicians suggest moving to fertility treatment sooner rather than chasing a target weight indefinitely.
What Should You Do Next?
Trying now: Stop your GLP-1 per the washout timeline above. If it's for diabetes, switch medications first — do not stop without a glycemic plan. Start prenatal vitamins. If periods don't return within 2–3 months after stopping, see a reproductive endocrinologist.
Pregnancy in 1–3 months: Schedule your preconception conversation now. Review washout timing. Start prenatal vitamins (400–800 mcg folate daily). If on tirzepatide + the pill, switch contraception methods during the transition.
Pregnancy in 3–6+ months: Continue your GLP-1 with proper birth control. Build sustainable habits that don't depend on the medication — some weight regain after stopping is expected. Plan the stop date in advance.
Not trying, but worried about birth control: Check the compatibility table above. On tirzepatide + oral contraceptive? Add condoms or switch. On semaglutide + oral contraceptive? Your main risk is from GI side effects, not absorption.
Doing IVF or egg retrieval: Tell your fertility clinic and anesthesia team that you are taking a GLP-1/GIP-GLP-1 medicine. Delayed gastric emptying raises aspiration risk under sedation. [Wegovy FDA PI] Protocols vary by clinic and procedure — coordinate the timeline with your reproductive endocrinologist.
GLP-1 Conception Planner
Use this tool to get a personalized preconception checklist based on your medication, birth control, timeline, and health situation. Select your inputs below, and the tool will generate your specific washout timeline, birth control guidance, and a step-by-step checklist.
GLP-1 Conception Planner
Answer 5 questions to get your personalized preconception checklist.
What this tool provides:
- • Your medication-specific washout timeline based on FDA labeling and FSRH guidance
- • Whether your birth control method is affected by your GLP-1 medication
- • A personalized preconception checklist including prenatal vitamins, stop date planning, and prescriber coordination
- • Diabetes-specific medication transition guidance if applicable
- • Urgency level based on your conception timeline
Select your medication, birth control, timeline, reason for treatment, and pregnancy status above to generate your personalized results.
Frequently Asked Questions
Does Ozempic affect birth control pills?
Semaglutide does not reduce oral contraceptive absorption pharmacokinetically. But if Ozempic causes vomiting or diarrhea — common during dose escalation — your pill may not absorb fully. If you vomit soon after taking your pill or have severe diarrhea for more than 24 hours, follow your pill's missed-pill instructions and use backup as directed. Persistent GI side effects are a reason to consider a non-oral method.
Does Mounjaro or Zepbound make birth control pills less effective?
Yes. The manufacturer warns that oral hormonal contraceptives may be less effective when taken with tirzepatide. The recommendation is to use a non-oral contraceptive method or add a barrier method for 4 weeks after starting and after each dose increase.
How long before pregnancy should I stop semaglutide?
FDA labeling says at least 2 months. Pharmacokinetic clearance takes about 5 weeks — the 2-month recommendation adds a buffer.
How long before pregnancy should I stop tirzepatide?
U.S. labeling says discontinue when pregnancy is recognized but does not give a numeric preconception washout. UK FSRH/MHRA guidance uses approximately 1 month before planned pregnancy. Discuss your timeline with your prescriber.
What if I vomit after taking my pill while on a GLP-1?
If you vomit soon after taking your oral contraceptive or have severe diarrhea for more than 24 hours, follow your pill's missed-pill instructions/package insert and use backup contraception as directed. Persistent GI side effects are a reason to consider switching to a non-oral method.
Can I breastfeed while on a GLP-1?
Breastfeeding data are still limited and differ by product. Injectable semaglutide was not detected in a small milk study, with a worst-case calculated infant exposure of 1.26% — well below the 10% safety threshold. Oral semaglutide tablets should not be used during breastfeeding. Current FDA labels state that breastfeeding is not recommended during treatment with RYBELSUS or OZEMPIC tablets and with WEGOVY tablets. Tirzepatide concentrations in milk were undetectable or very low in a recent lactation study, but infant-safety data remain limited. Restart timing should be individualized with your clinician. Insulin and metformin are established safe alternatives during lactation.
Does my male partner's GLP-1 use matter?
Human data on male fertility effects are limited. Current evidence does not show a pregnancy-risk signal from paternal exposure, and MotherToBaby says paternal exposure is unlikely to increase pregnancy risk.
Can I use a GLP-1 during IVF?
In the months before IVF to improve metabolic health — often yes, under your clinic's guidance. Tell your fertility clinic and anesthesia team that you are on a GLP-1/GIP-GLP-1 medicine. Protocols for when to stop vary by clinic and procedure — coordinate directly with your reproductive endocrinologist.
What about emergency contraception on a GLP-1?
No direct evidence exists on GLP-1s and emergency hormonal contraception. The copper IUD is the most effective emergency contraception option and is unaffected by any GLP-1.
Is weight loss safe during pregnancy?
No. FDA labeling states weight loss offers no benefit during pregnancy and may cause fetal harm. This is a primary reason GLP-1s must be stopped.
Sources
[1] ASRM, ACOG, and Endocrine Society guidance note that modest weight loss (5–10% of body weight) can improve cycle regularity and ovulation in women with obesity and PCOS. See also: Thessaloniki ESHRE/ASRM PCOS Consensus Workshop Group, 2008; Kiddy DS et al., BMJ, 1992.
[2] Vu P, et al. “Public sentiment about GLP-1 receptor agonists and fertility.” PLOS ONE, 2025. doi:10.1371/journal.pone.0326210
[3] Faculty of Sexual and Reproductive Healthcare (FSRH). CEU Clinical Statement: GLP-1 receptor agonists and contraception.
[4] Zepbound (tirzepatide) prescribing information. U.S. FDA, 2026.
[5] Kapitza C, et al. “Semaglutide, a once-weekly human GLP-1 analog, does not reduce the bioavailability of the combined oral contraceptive ethinylestradiol/levonorgestrel.” J Clin Pharmacol, 2015;55(5):497-504. PMC4418331.
[6] Semaglutide prescribing information. U.S. FDA, 2026. Includes: Wegovy injection, Wegovy tablets, Ozempic injection, Ozempic tablets, Rybelsus.
[7] UK Medicines and Healthcare products Regulatory Agency (MHRA). “GLP-1 medicines for weight loss and diabetes: what you need to know.” GOV.UK, 2025.
[8] MotherToBaby. Semaglutide fact sheet.
[9] MotherToBaby. Tirzepatide fact sheet.
[10] Kim Dao K, et al. “Use of GLP1 receptor agonists in early pregnancy and reproductive safety: a multicentre, observational, prospective cohort study.” BMJ Open, 2024. PMID: 38663923.
[11] Cesta CE, Rotem R, Bateman BT, et al. “Safety of GLP-1 Receptor Agonists and Other Second-Line Antidiabetics in Early Pregnancy.” JAMA Intern Med. 2024;184(2):144–152.
[12] Wyckoff JA, et al. “Preexisting Diabetes and Pregnancy.” J Clin Endocrinol Metab. 2025;110(9):2405–2452.
[13] American Diabetes Association. “Pregnancy and diabetes.”
[14] Szczesnowicz A, Szeliga A, Niwczyk O, et al. “Do GLP-1 Analogs Have a Place in the Treatment of PCOS?” J Clin Med. 2023;12(18):5915. PMCID: PMC10532286.
[15] Salamun V, et al. “Liraglutide increases IVF pregnancy rates in obese PCOS women.” Eur J Endocrinol. 2018;179(1):1–11. Also: Li R, et al. Arch Gynecol Obstet. 2023;307(3):839–849.
[16] ASRM. Recommendations from the 2023 International Evidence-Based Guideline for PCOS.
[17] Diab H, et al. “Subcutaneous semaglutide during breastfeeding.” Nutrients, 2024;16:2886. PMC11397063.
[18] Oral semaglutide tablet prescribing information. U.S. FDA, 2026. Includes Rybelsus, Ozempic tablets, and Wegovy tablets. See Section 8.2 (Lactation): breastfeeding is not recommended during treatment with oral semaglutide tablets.
This content is for informational purposes only and does not constitute medical advice. Always consult your healthcare provider before changing your medication, especially when planning pregnancy. We are not affiliated with Novo Nordisk, Eli Lilly, or any pharmaceutical manufacturer. This page does not replace the guidance of your OB/GYN, endocrinologist, or prescriber.
We update this page when FDA labeling, FSRH/MHRA guidance, or significant new research changes the clinical picture.