By The RX Index Editorial Team · Last verified: April 1, 2026 · Sources: NEJM, The Lancet, Eli Lilly IR, FDA.gov, Reuters
Medication Guide · April 1, 2026
Orforglipron Reviews: Honest 2026 Verdict on Eli Lilly's GLP-1 Weight Loss Pill
Our verdict in one sentence:
In the pivotal Phase 3 obesity trial, the highest dose produced 11.2% average body weight loss over 72 weeks (12.4% among patients who stayed on treatment) — and it outperformed oral semaglutide head-to-head on both weight loss and blood sugar. The pill is not yet FDA-approved as of April 1, 2026, with Lilly expecting a U.S. decision in Q2 2026. If you're ready to start now, Lilly's own ATTAIN-MAINTAIN trial showed that patients who switched from injectables to orforglipron maintained much of their weight loss.
Here's the reality of searching for “orforglipron reviews” right now: there are no legitimate customer reviews because nobody can buy this drug yet. What does exist is seven completed clinical trials, over 4,800 patients' worth of data, and a very clear picture of what this pill can and can't do.
We dug through all of it so you don't have to — the weight loss numbers separated by trial, side effects with real percentages, expected pricing Lilly has already committed to, head-to-head comparisons, and the question nobody else answers clearly: whether you should actually wait for it.

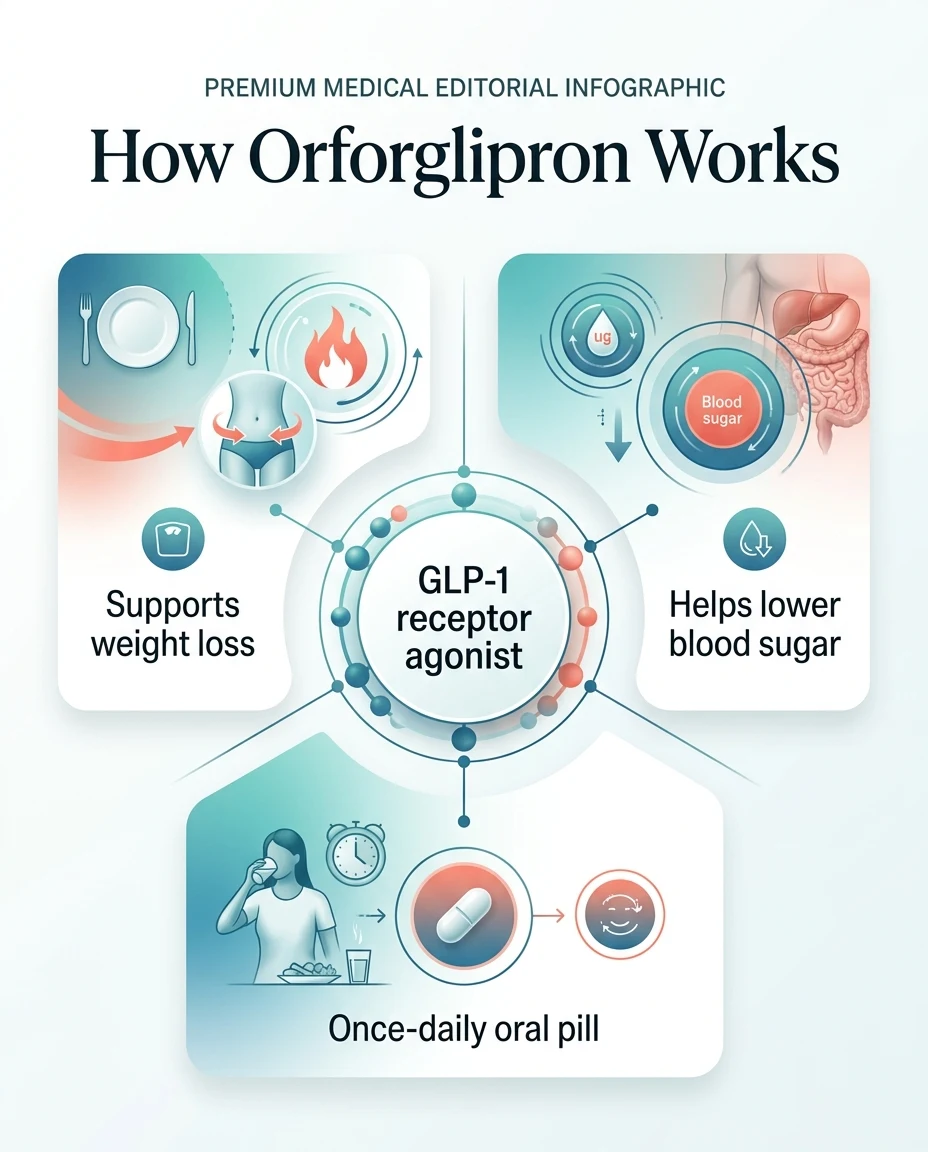
What Is Orforglipron, Exactly?
Orforglipron is a once-daily oral GLP-1 weight loss pill made by Eli Lilly — the same company behind Mounjaro and Zepbound. Lilly describes it as the first oral small-molecule GLP-1 receptor agonist to successfully complete a Phase 3 trial, which means it's built differently from every other GLP-1 on the market.
Why the Small-Molecule Structure Matters
Current GLP-1 medications like Ozempic, Wegovy, and Zepbound are peptides — modified proteins that mimic a natural gut hormone called GLP-1. Proteins get destroyed by stomach acid, which is why most GLP-1 drugs need to be injected. The Wegovy pill uses special absorption enhancers and requires an empty stomach to protect the protein during digestion.
Orforglipron takes a completely different approach. It's a synthetic chemical compound — small enough to survive your digestive system intact and absorb directly through the gut wall. No special protections needed. In clinical trials, orforglipron was taken once daily without food or water timing restrictions.
This small-molecule structure also makes orforglipron cheaper and simpler to manufacture. Eli Lilly has said it can launch without supply constraints — a notable claim given the Ozempic and Mounjaro shortages that frustrated millions of patients.
What It Does in Your Body
Reduces appetite
You feel satisfied sooner and experience less 'food noise' between meals
Slows digestion
Food stays in your stomach longer, keeping you full
Improves insulin response
Particularly valuable for people with type 2 diabetes
Supports weight loss
Through the combined effect of eating less and improved metabolic function
Should You Wait for Orforglipron or Start Something Now?
The short answer: If you can tolerate waiting a few more weeks and specifically want a daily pill with no fasting rules, waiting is reasonable. But if you've been putting off treatment while waiting for the “perfect” option, you're losing time you won't get back. The best GLP-1 is the one you actually start.
We've seen this pattern with every new GLP-1 launch. People wait for Mounjaro. Then Zepbound. Then the Wegovy pill. Then orforglipron. Meanwhile, people who started six months ago are already seeing real results on the scale.
Wait for orforglipron if all three are true:
- 1.You specifically want a daily pill (not injections)
- 2.The Wegovy pill's fasting requirement (empty stomach + 30-min wait) is a dealbreaker
- 3.You're okay waiting until at least mid-to-late April — possibly longer
Start an approved GLP-1 now if any of these are true:
- →You've been 'about to start' for more than a month
- →You want the strongest possible weight loss (injectables average 20%+ vs orforglipron's 11%)
- →You'd rather start making progress this month than wait for a slightly more convenient option
- →You're comfortable with weekly injections or the Wegovy pill's morning routine
The strongest argument against waiting:
Lilly's own ATTAIN-MAINTAIN trial showed that people who started on injectable Wegovy or Zepbound and then switched to orforglipron maintained much of their previously achieved weight loss — an average difference of just 0.9 kg after switching from Wegovy and 5.0 kg after switching from Zepbound over 52 weeks. You can start now and switch later. That's not a compromise — that's a strategy.
Start your GLP-1 journey today — switch to the pill when it launches.
FDA-approved programs available now. ATTAIN-MAINTAIN data supports switching when orforglipron launches.
Check Your Eligibility on Ro — FDA-Approved Programs →Or take our free 60-second matching quiz to find the right program for your situation.
Is Orforglipron Approved by the FDA?
No — not yet. As of April 1, 2026, orforglipron is under FDA review. Lilly's public guidance says potential U.S. action for obesity is expected in Q2 2026. Reuters has reported a target action date of April 10, 2026, based on internal regulatory documents, though the FDA has not published a public action date.
The probability of approval looks strong. Orforglipron has four positive Phase 3 trials, no major safety signals flagged publicly, and institutional forecasts estimate an 84–85% approval probability. Lilly has pre-manufactured approximately $1.5 billion worth of supply and has stated it can begin shipping within approximately one week of approval.
But “strong probability” is not a guarantee. The FDA could issue a Complete Response Letter requesting additional data, flag a manufacturing concern, or require labeling changes. We'll update this page the moment news breaks.
| Status Detail | Current Information (April 1, 2026) |
|---|---|
| FDA approved? | No |
| Available to the public? | No |
| Lilly's public timeline | Potential U.S. obesity action in Q2 2026 |
| Reuters-reported target date | April 10, 2026 (based on internal documents) |
| Submitted for | Obesity / overweight with comorbidities |
| Diabetes submission | Planned later in 2026 |
| Shipping readiness | ~1 week after approval (per Reuters) |
| Pre-manufactured supply | ~$1.5 billion worth |
| Also being studied for | Sleep apnea, hypertension, osteoarthritis |
Sources: Eli Lilly official FAQ (Feb 2026), Reuters (Jan 2026), FDA.gov, BioSpace
Ready to start before the decision drops?
FDA-approved GLP-1 programs are available now through telehealth providers. Starting today doesn't lock you in — you can switch to orforglipron once it's available.
Check GLP-1 Eligibility on Ro — Available Now →How Much Weight Can You Actually Lose on Orforglipron?
Weight loss results vary by trial, dose, duration, and patient population. Here are all the numbers from every completed trial — separated so you can see exactly what was studied.
Orforglipron Weight Loss Results by Trial
| Trial | Patients | Duration | Best Result (36 mg) | Placebo | Published |
|---|---|---|---|---|---|
| ATTAIN-1 (Ph 3) | 3,127 — obesity, no diabetes | 72 wks | –11.2% / –12.4%* | –2.1% | NEJM, Sept 2025 |
| ATTAIN-2 (Ph 3) | Obesity + type 2 diabetes | 72 wks | –10.5% / –22.9 lbs | –2.2% | Lancet, Nov 2025 |
| Phase 2 (Obesity) | 272 — obesity/overweight | 36 wks | –14.7% (45 mg) | –2.3% | NEJM, 2023 |
| ACHIEVE-3 (Ph 3) | 1,698 — type 2 diabetes | 52 wks | –9.2% / –19.7 lbs | vs oral sema: –5.3% | Lancet, Feb 2026 |
| ACHIEVE-1 (Ph 3) | 559 — type 2 diabetes | 40 wks | –7.9% / –16 lbs | –1.6% | NEJM, Jun 2025 |
| ATTAIN-MAINTAIN | Switched from Wegovy/Zepbound | 52 wks | Maintained prior loss | Placebo regained ~9+ kg | Lilly PR, Dec 2025 |
*Treatment-regimen estimand includes all randomized patients regardless of whether they stayed on treatment. Efficacy estimand counts only those who completed treatment. Both are standard FDA reporting measures.
What these numbers mean in plain English:
If you weigh 220 pounds and take the highest dose for 72 weeks, you'd lose roughly 24–27 pounds on average. Some people in the trials lost significantly more — over half the patients on 36 mg lost at least 10%, and about a third lost 15% or more.
Those numbers are meaningful and comparable to what injectable GLP-1s achieved in their early trials. But they are lower than top-end results from injectable Zepbound (tirzepatide), which has shown 20%+ average weight loss in pivotal trials. That's an important distinction and we're not going to gloss over it.
The honest tradeoff:
Orforglipron does not deliver the highest weight loss numbers in the GLP-1 class. If raw maximum weight loss is your single top priority and you're fine with weekly injections, Zepbound is stronger. But orforglipron skips the needles, skips the fasting routine, and removes the barriers that keep millions of people from ever starting. A pill that people actually take consistently may produce better real-world outcomes than an injection they avoid.
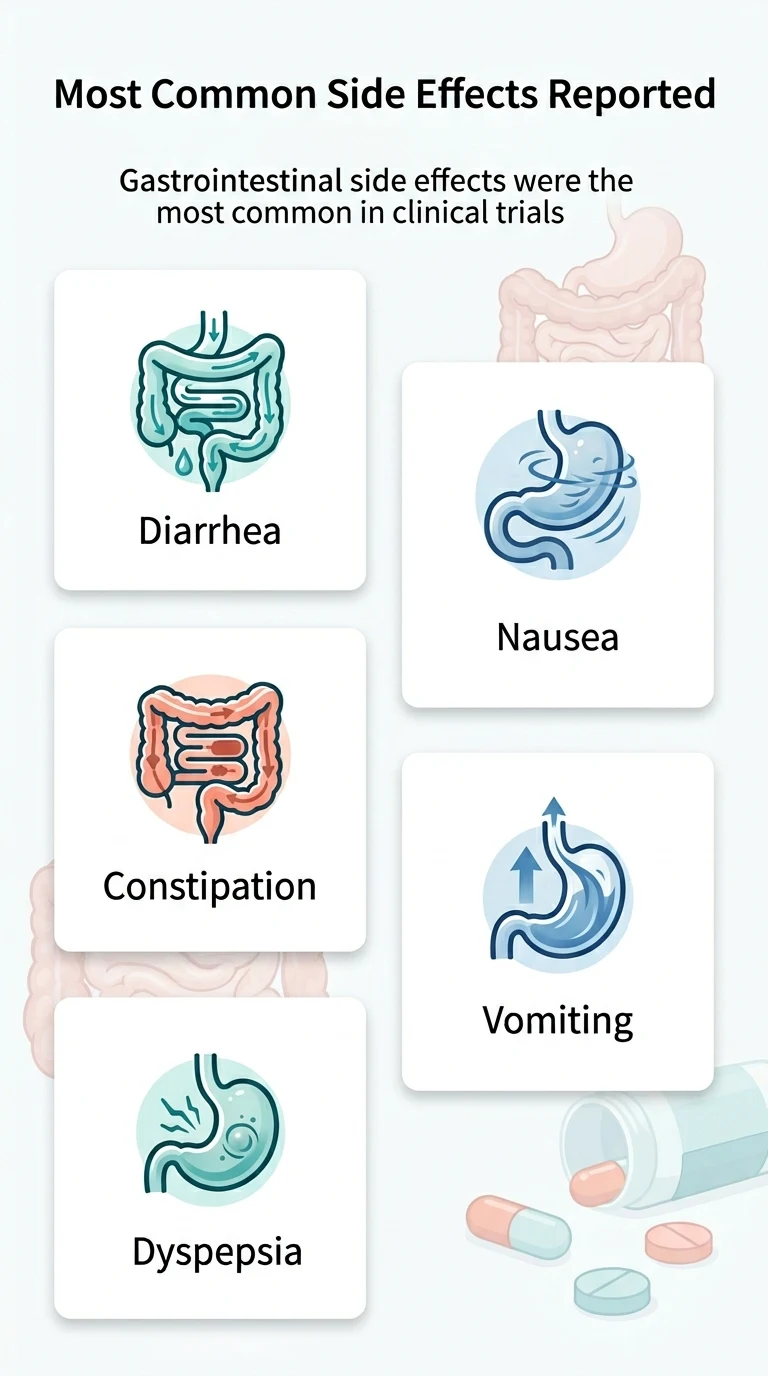
What Are the Side Effects of Orforglipron?
The side effects are GI-heavy — diarrhea, nausea, constipation, and vomiting — which is consistent with other GLP-1 medications. In head-to-head testing, orforglipron caused more GI symptoms than oral semaglutide, but the vast majority were mild to moderate and improved over time. Let's look at the actual numbers instead of vague warnings.
Side Effect Rates From Phase 3 Trials (ACHIEVE-1)
| Side Effect | Orforglipron 36 mg | Orforglipron 12 mg | Placebo |
|---|---|---|---|
| Diarrhea | 26% | 21% | 9% |
| Nausea | 16% | 18% | 2% |
| Dyspepsia | 15% | 20% | 7% |
| Constipation | 14% | 17% | 4% |
| Vomiting | 14% | 7% | 1% |
| Stopped due to side effects | 8% | 4% | 1% |
Source: Eli Lilly ACHIEVE-1 Phase 3 results, published NEJM June 2025
What this actually means for you:
In ACHIEVE-3, about 58–59% of orforglipron patients reported GI symptoms versus 37–45% on oral semaglutide. The discontinuation rate was roughly 9.7% for orforglipron versus 4.9% for oral semaglutide. But flip the framing: roughly 90% of people on orforglipron stayed on treatment. Most dropouts happened during early dose escalation. If you push through that initial period (which your prescribing doctor manages with gradual dose increases), the side effects typically settle down.
No hepatic safety signal has been observed in Lilly's trial readouts to date. Overall safety has looked broadly consistent with the GLP-1 class.
Tips That Actually Help With GLP-1 Side Effects
Orforglipron vs. Current GLP-1 Options: The Full Picture
Here's how orforglipron stacks up against every approved GLP-1 option available right now — so you can make an honest comparison.
| Option | Format | Avg. Weight Loss | Key Advantage | Key Limitation |
|---|---|---|---|---|
| Orforglipron | Daily pill | ~11–12.4% (72 wks) | No food rules, pill format | Not yet available |
| Wegovy pill (semaglutide 25mg) | Daily pill | ~14–16.6% | Available NOW, strong loss | Empty stomach + 30-min wait |
| Wegovy injection | Weekly injection | ~15% | Proven CV benefit, 8+ years data | Weekly injection |
| Ozempic (diabetes) | Weekly injection | ~12–14% | Strong real-world track record | Not approved for weight loss |
| Zepbound (tirzepatide) | Weekly injection | ~18–22% | Highest weight loss in class | Weekly injection |
| Compounded GLP-1 | Injection or tablet | Varies | Lowest cost entry point | Quality and sourcing varies |
Which Option Fits Your Situation?
You hate needles but want to start NOW →
The Wegovy pill
FDA-approved and available today. Yes, it has fasting requirements, but many people build the routine quickly — take it first thing with a small sip of water, wait 30 minutes, then eat breakfast.
You want the most weight loss possible →
Zepbound (tirzepatide)
The strongest option available. Injectable, weekly, but averaging over 20% weight loss in clinical trials — roughly 40–50 lbs for many patients. If results matter more than convenience, this is the clear choice.
You want to wait for orforglipron specifically →
Valid, but set a decision deadline.
If you can wait a few weeks for a no-fasting daily pill, that's reasonable. But have your plan ready — know which provider you'll use, know your budget, don't let 'waiting' become 'never starting.'
You're already on a GLP-1 and want to switch to a pill later →
ATTAIN-MAINTAIN supports this pathway.
Talk to your current prescriber about your interest in orforglipron when it becomes available. In the meantime, keep taking your current medication — stopping without a bridge leads to weight regain.
Orforglipron vs. The Wegovy Pill: Head-to-Head
These are the two oral GLP-1 pills competing for the same patients. In a head-to-head trial, orforglipron won on efficacy but had higher side effects and discontinuation. The Wegovy pill is available now. Orforglipron is not.
Here's what the ACHIEVE-3 trial showed, with a critical caveat most sites skip:
| Measure | Orforglipron 36 mg | Oral Semaglutide 14 mg* |
|---|---|---|
| A1C reduction | –2.2% | –1.4% |
| Weight loss | –19.7 lbs (9.2%) | –11.0 lbs (5.3%) |
| GI side effects | ~58–59% | ~37–45% |
| Discontinuation (adverse events) | ~9.7% | ~4.9% |
| Food restrictions | None in trials | Empty stomach + 30-min wait |
*Source: ACHIEVE-3, published in The Lancet, February 2026. *Critical caveat: semaglutide doses in this trial (7 mg and 14 mg) were Rybelsus diabetes doses, NOT the Wegovy pill's approved 25 mg obesity dose. We don't yet have a head-to-head trial vs. full-strength Wegovy pill. In separate trials, Wegovy pill at 25 mg showed ~14–16.6% weight loss vs. orforglipron's ~11.2–12.4%.
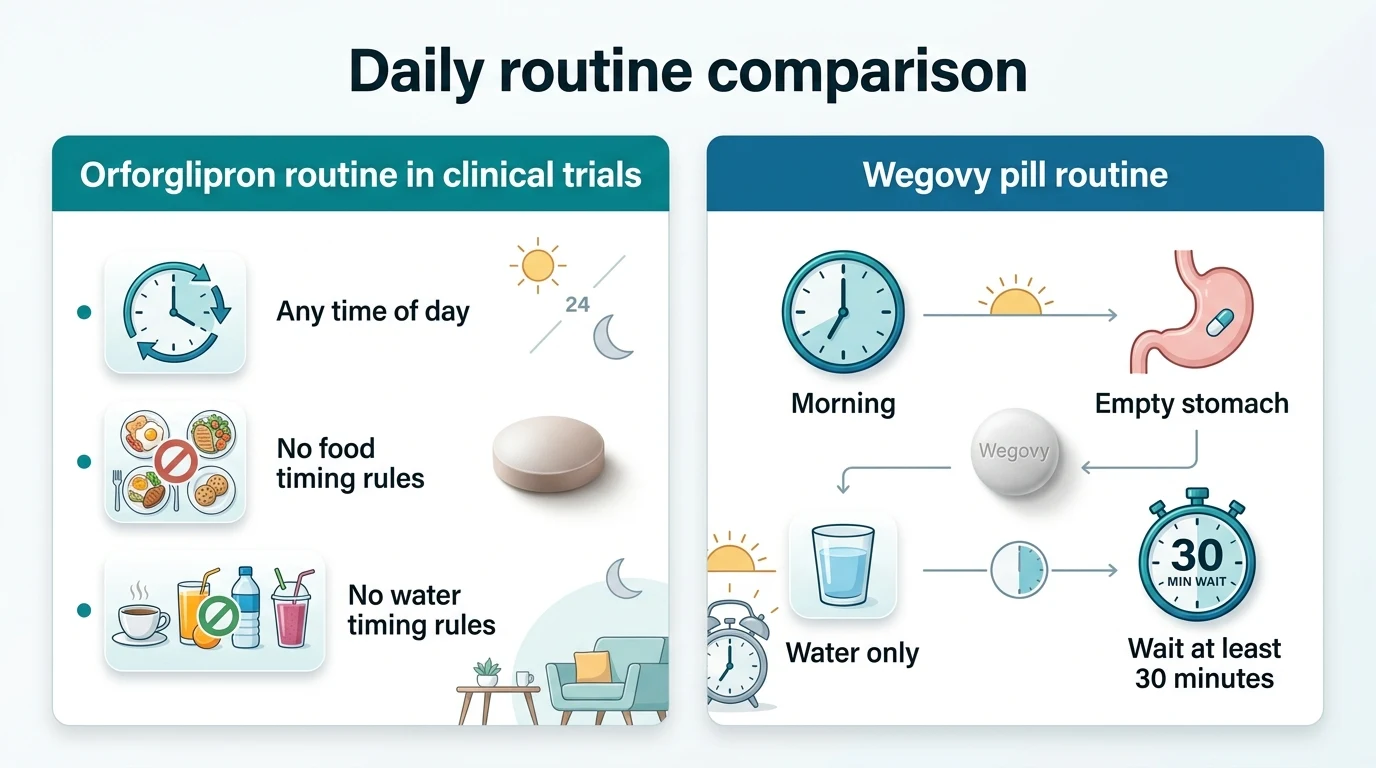
Where orforglipron clearly wins: convenience.
No empty stomach. No 30-minute wait. No worrying about whether your morning routine will interfere with absorption. For parents with chaotic mornings, shift workers, travelers, or people with multiple daily medications, that fasting requirement is a daily annoyance that erodes adherence over time.
“The dosage requirements for oral Wegovy are not attractive to me, so orforglipron already has a leg up.”
— Public Reddit commenter, r/RoBody
How Much Will Orforglipron Cost?
Self-pay through LillyDirect: $149/month for the lowest dose, up to $399/month for higher doses — contingent on FDA approval. Under Lilly's November 2025 government agreement, Medicare beneficiaries would pay no more than $50/month, though the actual rollout has moving parts.
Expected Orforglipron Pricing vs. Current GLP-1 Costs
| Option | Self-Pay Monthly | Medicare | Notes |
|---|---|---|---|
| Orforglipron (if approved) | $149–$399 | $50/mo framework | Via LillyDirect; bridge demo Jul 2026, BALANCE Part D Jan 2027 |
| Wegovy pill | $149–$299 | Varies by plan | Available now; via NovoCare/Ro |
| Wegovy injection | ~$199 intro, then ~$349 | Covered | Intro pricing for new self-pay patients |
| Zepbound | $299–$699 | Covered | Via LillyDirect; lower doses more affordable |
| Compounded GLP-1 via MEDVi | Starting ~$179/mo | Not covered | Cash-pay; includes clinical support |
Last verified: April 1, 2026. Pricing may change. Sources: Eli Lilly (Nov 6, 2025 agreement), NovoCare, provider websites.
What Do People Actually Say About Orforglipron?
There are no legitimate retail customer reviews for orforglipron because nobody can buy it yet. Any site showing star ratings or “customer testimonials” for this drug is fabricating them or conflating clinical trial reports with consumer reviews. We're not going to do that.
What we do have: public comments from self-described participants and GLP-1 community discussions on Reddit. These give genuine insight into real experience and what people are thinking about orforglipron right now.
“I do seem to feel full sooner and haven't really been craving food like I normally would.”
— Self-described orforglipron user, r/orforglipron
“It worked for me for 6 months (slow and steady weight loss) with minimal side effects.”
— Public Reddit commenter, r/orforglipron
“There's no fasting requirement with orforglipron so that seems easier to be on... but also who knows how soon it will actually be available once approved and maybe it's better to just get started on my weight loss journey.”
— Public Reddit commenter, r/WegovyPillWeightLoss
Our read on the forum chatter:
The tension is real — people want the convenience of this pill, but recognize that waiting has a cost. The ATTAIN-MAINTAIN data shows that starting an injectable GLP-1 now and switching to the pill later is a supported strategy. The “waiting vs. starting” question is a false dilemma. The sooner you see it that way, the sooner you make progress.
Can You Switch From Injections to Orforglipron Later?
Lilly's ATTAIN-MAINTAIN trial specifically tested this — and the topline results are encouraging. Patients who hit their weight loss plateau on injectable Wegovy or Zepbound switched to daily orforglipron for 52 weeks.
Wegovy → Orforglipron
+0.9 kg average regain over 52 weeks
Zepbound → Orforglipron
+5.0 kg average regain over 52 weeks
Placebo group
Regained ~9+ kg — confirming the drug effect
These are topline results from Lilly's press release (December 2025). Full peer-reviewed data will provide more detail on individual variation and long-term durability.
Think of it like training for a marathon.
You don't skip months of running waiting for better shoes. You start training now and upgrade when the shoes drop. The injectables are your training plan: powerful, proven, available now. Orforglipron is your long-term maintenance strategy: convenient, simple, and coming soon. One leads naturally into the other.
Start Your GLP-1 Program Now on Ro →The Bottom Line
Orforglipron is real. The data is strong. The FDA decision is approaching. If approved, it will be the most convenient oral GLP-1 available — a once-daily pill with no food restrictions in trials, priced competitively, backed by seven clinical trials and thousands of patients' worth of evidence.
But “real and coming soon” is not the same as “available today.” And every week you spend waiting is a week you could have spent making progress.
The most common thing we hear from GLP-1 users across every platform is some version of: “I wish I had started sooner.” The regret is always about delay, never about action.
If you're ready to start now:
- →Pick the GLP-1 option that fits your life today
- →You can switch to orforglipron once it launches
- →ATTAIN-MAINTAIN data says switching works — starting now doesn't lock you in
If you're specifically holding out for orforglipron:
- →Make it an active decision, not a passive one
- →Have your plan ready — know which provider you'll use
- →Know your budget; don't let 'waiting' become 'never starting'
Orforglipron: Frequently Asked Questions
Is orforglipron approved by the FDA?
No. As of April 1, 2026, orforglipron is still under FDA review. Lilly says potential U.S. action for obesity is expected in Q2 2026. Reuters has reported a target action date of April 10, 2026, based on internal regulatory documents. It is not yet available to the public.
When will orforglipron be available?
Eli Lilly expects potential U.S. FDA action for obesity in Q2 2026. Reuters has reported an April 10, 2026 target action date. If approved, Lilly has stated it can begin shipping within approximately one week, having pre-manufactured approximately $1.5 billion worth of supply.
How much will orforglipron cost?
Eli Lilly announced that self-pay patients would access orforglipron through LillyDirect starting at $149/month for the lowest dose and up to $399/month for higher doses, contingent on FDA approval. Under Lilly's government agreement, Medicare beneficiaries would pay no more than $50/month, though CMS has indicated Medicare Part D access through the BALANCE model would begin in January 2027, with a bridge demonstration expected in July 2026.
What are the side effects of orforglipron?
The most common side effects are gastrointestinal: diarrhea, nausea, constipation, vomiting, and dyspepsia. These are consistent with the GLP-1 class and were mostly mild to moderate. In Phase 3 trials, treatment discontinuation due to adverse events ranged from about 4% to 10% depending on dose.
How much weight can you lose on orforglipron?
In the Phase 3 ATTAIN-1 trial (3,127 patients with obesity, 72 weeks), the highest dose (36 mg) produced 11.2% average body weight loss using the treatment-regimen estimand (12.4% using the efficacy estimand). In the earlier Phase 2 trial (272 patients, 36 weeks), weight loss reached up to 14.7%. Results vary by dose, duration, and population.
Is orforglipron better than the Wegovy pill?
In the ACHIEVE-3 head-to-head trial, orforglipron outperformed oral semaglutide on both blood sugar reduction and weight loss. However, that trial used lower semaglutide doses (7–14 mg) than the Wegovy pill's approved 25 mg obesity dose. Orforglipron also had higher GI side effects and discontinuation rates. Its major advantage is no food or water timing restrictions in trials.
Does orforglipron have to be taken on an empty stomach?
No. In clinical trials, orforglipron was taken once daily without food or water timing restrictions. This is a key advantage over the Wegovy pill, which requires an empty stomach and a 30-minute wait before eating or drinking.
Can you switch from injectable GLP-1s to orforglipron later?
Lilly's Phase 3 ATTAIN-MAINTAIN topline results showed that participants who switched from injectable Wegovy or Zepbound to orforglipron maintained much of their previously achieved weight loss over 52 weeks, with average regain of 0.9 kg (from Wegovy) and 5.0 kg (from Zepbound). This suggests starting injectables now and switching to the pill later is a viable strategy.
Will insurance cover orforglipron?
Lilly's government agreement includes a $50/month framework for Medicare beneficiaries, though CMS has indicated the BALANCE Part D model begins January 2027 with a bridge demonstration in July 2026. Coverage depends on participating plans and is not guaranteed for every individual. Commercial insurance formulary decisions typically take months after approval.
Is orforglipron the same as tirzepatide or semaglutide?
No. Orforglipron is a different molecule entirely — a small-molecule, non-peptide GLP-1 receptor agonist. Semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound) are peptide-based incretin drugs. Semaglutide is available as both injections and tablets; tirzepatide is currently injectable only.
What is the brand name for orforglipron?
Eli Lilly has not publicly announced a brand name for orforglipron.
Should I wait for orforglipron or start a GLP-1 now?
It depends on your priorities. If you strongly prefer a pill with no fasting rules and can wait a few weeks, orforglipron may be worth waiting for. If you want to start losing weight now, FDA-approved GLP-1 programs are available today — and Lilly's ATTAIN-MAINTAIN trial showed that participants who switched from injectables to orforglipron maintained much of their weight loss over 52 weeks.
Related guides
Sources
- Eli Lilly. “Lilly's oral GLP-1, orforglipron, demonstrated meaningful weight loss” — ATTAIN-1 Phase 3 results. Published NEJM, September 2025.
- Eli Lilly. “What to Know About Orforglipron Oral GLP-1.” Last updated February 2026. lilly.com
- Eli Lilly. “Lilly's orforglipron helped people maintain weight loss after switching from injectable incretins.” ATTAIN-MAINTAIN press release, December 18, 2025.
- Eli Lilly. ACHIEVE-1 press release / NEJM. June 2025.
- Reuters. “FDA delays decision for Lilly's obesity pill orforglipron.” January 2026.
- FDA. “FDA awards second batch of national priority vouchers.” November 2025.
- Jastreboff AM et al. “Daily Oral GLP-1 Receptor Agonist Orforglipron for Adults with Obesity.” NEJM, 2023.
- Rosenstock J et al. “Efficacy and safety of once-daily oral orforglipron compared with oral semaglutide in adults with type 2 diabetes (ACHIEVE-3).” The Lancet, February 2026.
- Eli Lilly / U.S. Government agreement press release. November 6, 2025.
Still not sure which GLP-1 program is right for you?
Take our free 60-second matching quiz. We'll ask about your goals, budget, insurance status, and preferences — and show you the 1–2 best options for your specific situation, whether that's waiting for orforglipron or starting an approved program today.
Take the Free GLP-1 Matching Quiz →Free · No signup · 60 seconds
This page is updated within 24 hours of any FDA action on orforglipron. Last verified: April 1, 2026.
The RX Index is an independent GLP-1 medication research platform. We are not affiliated with Eli Lilly, Novo Nordisk, or any pharmaceutical manufacturer. This page contains affiliate links to telehealth providers. Our editorial recommendations are based on independent research and are not influenced by affiliate compensation. See our full disclosure.