Retatrutide vs Semaglutide: Which Actually Makes Sense for You Right Now?
Retatrutide looks stronger for weight loss in early trials, but it is not FDA-approved and is legally available only inside Eli Lilly clinical trials. Semaglutide is approved, available now — including higher-dose and oral options new in 2026 — and has proven heart-health benefits.

Disclosure: Some links on this page are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you.
JUMP TO YOUR QUESTION
QUICK ANSWER
Retatrutide looks stronger for weight loss in early trials, but it is not FDA-approved and is legally available only inside Eli Lilly clinical trials. Semaglutide is approved, available now — including higher-dose and oral options new in 2026 — and has proven heart-health benefits. If you need treatment today, semaglutide or tirzepatide is the real-world decision.
Retatrutide vs semaglutide is one of the most searched GLP-1 comparisons on the internet right now — and most pages answering it get the story wrong. They compare excitement to availability. We’re going to compare evidence quality, availability reality, and what you should actually do next.
Retatrutide looks stronger on raw weight-loss potential. The strongest public retatrutide obesity result so far is 28.7% mean body weight loss in the TRIUMPH-4 Phase 3 trial (December 2025). Semaglutide’s data ranges from 14.9% at 68 weeks in the original STEP 1 trial to 20.7% at 72 weeks with the newly approved Wegovy HD 7.2 mg in STEP UP (approved March 19, 2026). The gap is real but narrower than headlines suggest — and the comparison is still mostly cross-trial, not a published head-to-head.
But here’s what changes everything: retatrutide is not FDA-approved and you cannot get it. Not legally. Not safely. Not from any pharmacy, telehealth platform, or legitimate provider. Semaglutide — the drug behind Wegovy and Ozempic — is approved, available today, backed by years of real-world safety data, proven heart-health benefits, and now comes in more options than ever.
We spent weeks pulling apart the clinical trial data, FDA filings, and Eli Lilly’s own statements to build the comparison we wish existed when we started researching this topic. Below you’ll find the actual numbers from both trial programs, a section explaining why the popular comparison is misleading, side effects that just changed this week, and a clear decision framework based on where you are right now.
If you already know you want to start a GLP-1 and just need help choosing a provider, you can skip straight to our matching tool. Otherwise, keep reading — this is the page that should have existed months ago.
Answer a few questions. See your clearest next step — no commitment.
What Changed in March 2026
Two major developments — March 19, 2026
FDA approved Wegovy HD (semaglutide 7.2 mg injection) — a higher dose that produced 20.7% mean weight loss in STEP UP. This narrows the gap with retatrutide and is expected to launch in U.S. pharmacies in April 2026. Dysesthesia (altered skin sensation) was reported in 22% of Wegovy HD patients — a new finding for semaglutide at this dose.
Eli Lilly released first Phase 3 retatrutide data in type 2 diabetes (TRANSCEND-T2D-1) — retatrutide produced up to 2.0% A1C reduction and 16.8% weight loss at 40 weeks in patients with type 2 diabetes. Weight loss had not plateaued at the end of the study.
Both updates are reflected throughout this page.
Retatrutide vs Semaglutide at a Glance
Every number in this table links back to a published trial or official press release — not a blog post, not a medspa landing page.
| Semaglutide | Retatrutide | |
|---|---|---|
| Drug class | GLP-1 receptor agonist (single) | GLP-1 + GIP + glucagon receptor agonist (triple) |
| Manufacturer | Novo Nordisk | Eli Lilly |
| Brand names | Wegovy pen and tablets, Wegovy HD 7.2 mg, Ozempic (T2D), Rybelsus (oral T2D) | None — investigational compound LY3437943 |
| FDA status | Approved since 2021; expanded in 2026 with Wegovy HD 7.2 mg, Wegovy tablets, MASH indication, and CV risk reduction. | Not approved. Phase 3 clinical trials (TRIUMPH + TRANSCEND programs) |
| Can you get it today? | Yes — widely available by prescription (injection and oral) | No. Only through Eli Lilly clinical trial enrollment |
| Best weight-loss evidence | 14.9% at 68 wks (Wegovy 2.4 mg, STEP 1); 20.7% at 72 wks (Wegovy HD 7.2 mg, STEP UP, approved March 2026) | 24.2% at 48 wks (Phase 2, NEJM 2023); 28.7% at 68 wks (TRIUMPH-4 Phase 3, Dec 2025) |
| Proven heart protection | Yes — 20% reduction in major CV events (SELECT trial, NEJM 2023) | No outcome data yet. Cardiovascular trial still enrolling |
| Common side effects | Nausea (~44%), diarrhea (~30%), vomiting (~24%); dysesthesia in 22% with Wegovy HD 7.2 mg | Nausea (~43%), diarrhea (~33%), vomiting (~21%); dysesthesia ~21% at 12 mg in TRIUMPH-4 |
| Long-term safety data | 5+ years of real-world evidence | Phase 2 and two Phase 3 trials only |
| How it's taken | Once-weekly injection; also available as oral Wegovy tablet (25 mg) | Once-weekly injection (in trials only) |
| Cost | Varies — see cost section below | Unknown — not commercially available |
| Availability | Now | Lilly says launch depends on trial completion and regulatory process |
Sources: Wilding et al., NEJM 2021 (STEP 1); Wharton et al., Lancet Diabetes Endocrinol 2025 (STEP UP); Jastreboff et al., NEJM 2023 (retatrutide Phase 2); Eli Lilly TRIUMPH-4 press release, Dec 11, 2025; Eli Lilly TRANSCEND-T2D-1 press release, March 19, 2026; Novo Nordisk Wegovy HD press release, March 19, 2026; Lincoff et al., NEJM 2023 (SELECT); FDA Wegovy prescribing information 2026. Table verified March 21, 2026.
What this table tells you at a glance: Retatrutide posts the biggest weight-loss number. Semaglutide posts a bigger “things we actually know for certain” profile — and with Wegovy HD now approved at 20.7%, the raw gap is smaller than it was even a week ago. One is exciting. The other is available.
What Most Pages Get Wrong: This Is Not a Clean Head-to-Head
This is the section we’ve never seen on another comparison page — and it’s the most important part of this article.
When you read “retatrutide 28.7% vs semaglutide 14.9%,” it sounds like a direct matchup. Like someone ran both drugs in the same study, measured the same people, and retatrutide won. That didn’t happen.
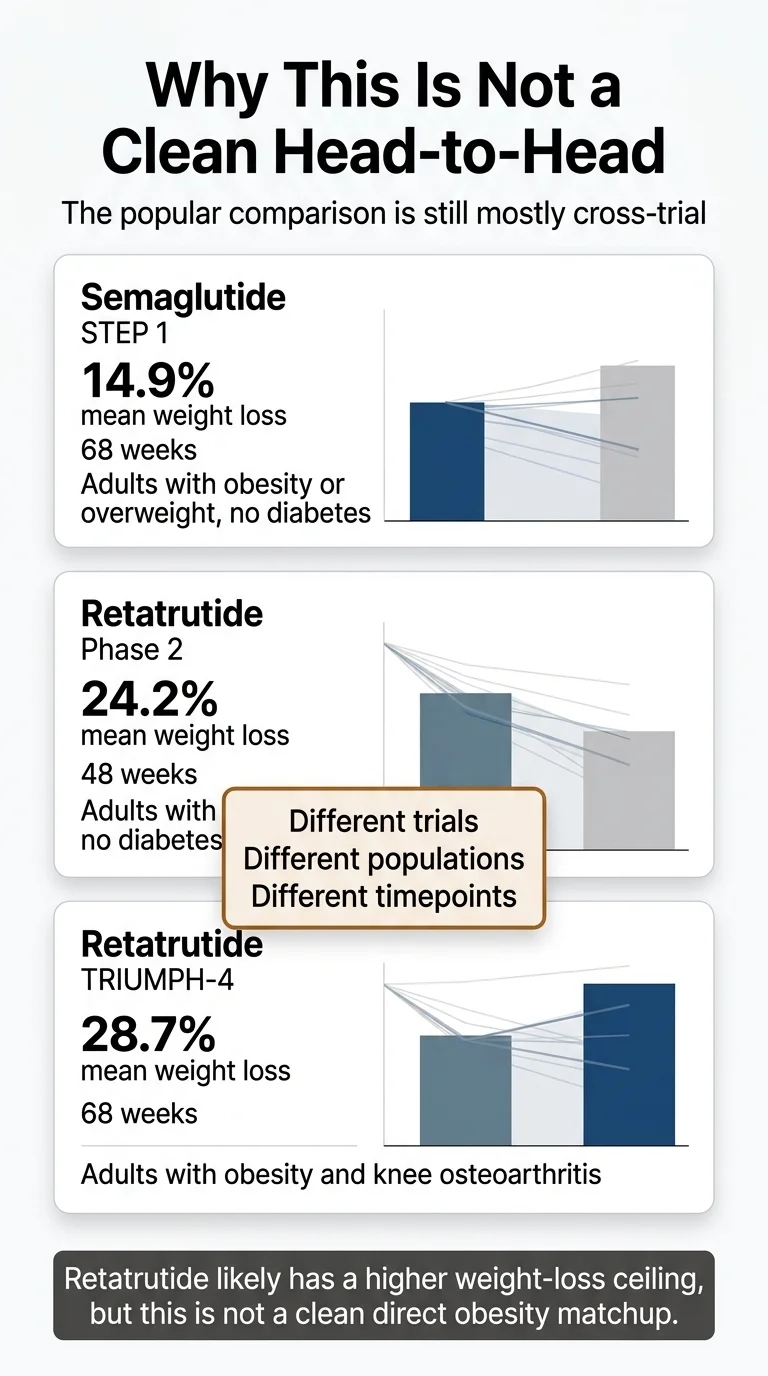
Here’s the reality:
- Semaglutide’s 14.9% comes from the STEP 1 trial — 1,961 adults with obesity, no diabetes, followed for 68 weeks. Published in the New England Journal of Medicine in 2021.
- Semaglutide’s 20.7% comes from STEP UP — 1,407 adults with obesity, no diabetes, followed for 72 weeks, using the new 7.2 mg dose. Published in Lancet Diabetes & Endocrinology in 2025.
- Retatrutide’s 24.2% comes from a Phase 2 trial — 338 adults with obesity, no diabetes, followed for 48 weeks. Published in the NEJM in 2023. Phase 2 trials are smaller and earlier-stage.
- Retatrutide’s 28.7% comes from TRIUMPH-4 — 445 adults with obesity and knee osteoarthritis, followed for 68 weeks. This was a sicker, heavier population (average BMI 40.4), which may have influenced the weight-loss numbers.
Different trials. Different populations. Different timepoints. Different phases of development.
Does retatrutide likely produce more weight loss than semaglutide? Almost certainly yes — the triple-agonist mechanism is genuinely more powerful. But the exact gap between them is something we won’t know precisely until a properly designed head-to-head obesity trial is published. There is a retatrutide-vs-semaglutide study underway, but it’s in type 2 diabetes — not the general obesity population.
Why this matters to you: If you’re making a decision based on cross-trial percentages, you’re comparing numbers from different studies with different designs. The directional conclusion — retatrutide is likely stronger — holds up. But treating these percentages like a final scoreboard is exactly the mistake most comparison pages make.
The Evidence We Actually Have (Trial-by-Trial)
| Trial | Drug | Population | Duration | Mean Weight Loss | Phase | Published |
|---|---|---|---|---|---|---|
| STEP 1 | Semaglutide 2.4 mg | Obesity, no diabetes (n=1,961) | 68 wks | 14.9% | Phase 3 | NEJM 2021 |
| STEP 5 | Semaglutide 2.4 mg | Obesity, no diabetes (n=304) | 104 wks | 15.2% | Phase 3 | Nature Med 2022 |
| STEP UP | Semaglutide 7.2 mg | Obesity, no diabetes (n=1,407) | 72 wks | 20.7% | Phase 3b | Lancet D&E 2025 |
| Phase 2 | Retatrutide 12 mg | Obesity, no diabetes (n=338) | 48 wks | 24.2% | Phase 2 | NEJM 2023 |
| TRIUMPH-4 | Retatrutide 12 mg | Obesity + knee OA (n=445) | 68 wks | 28.7% | Phase 3 | Eli Lilly Dec 2025 |
| TRANSCEND-T2D-1 | Retatrutide 12 mg | Type 2 diabetes (n=537) | 40 wks | 16.8% | Phase 3 | Eli Lilly Mar 2026 |
A fair reading of this data: Retatrutide almost certainly has a higher weight-loss ceiling than semaglutide. Semaglutide has the far more mature, more replicated, more decision-ready body of evidence — and the ceiling just got higher with Wegovy HD. Both things are true at the same time.
How Do These Drugs Actually Work?
Understanding why retatrutide produces more weight loss starts with one straightforward concept: it activates more systems in your body at once.
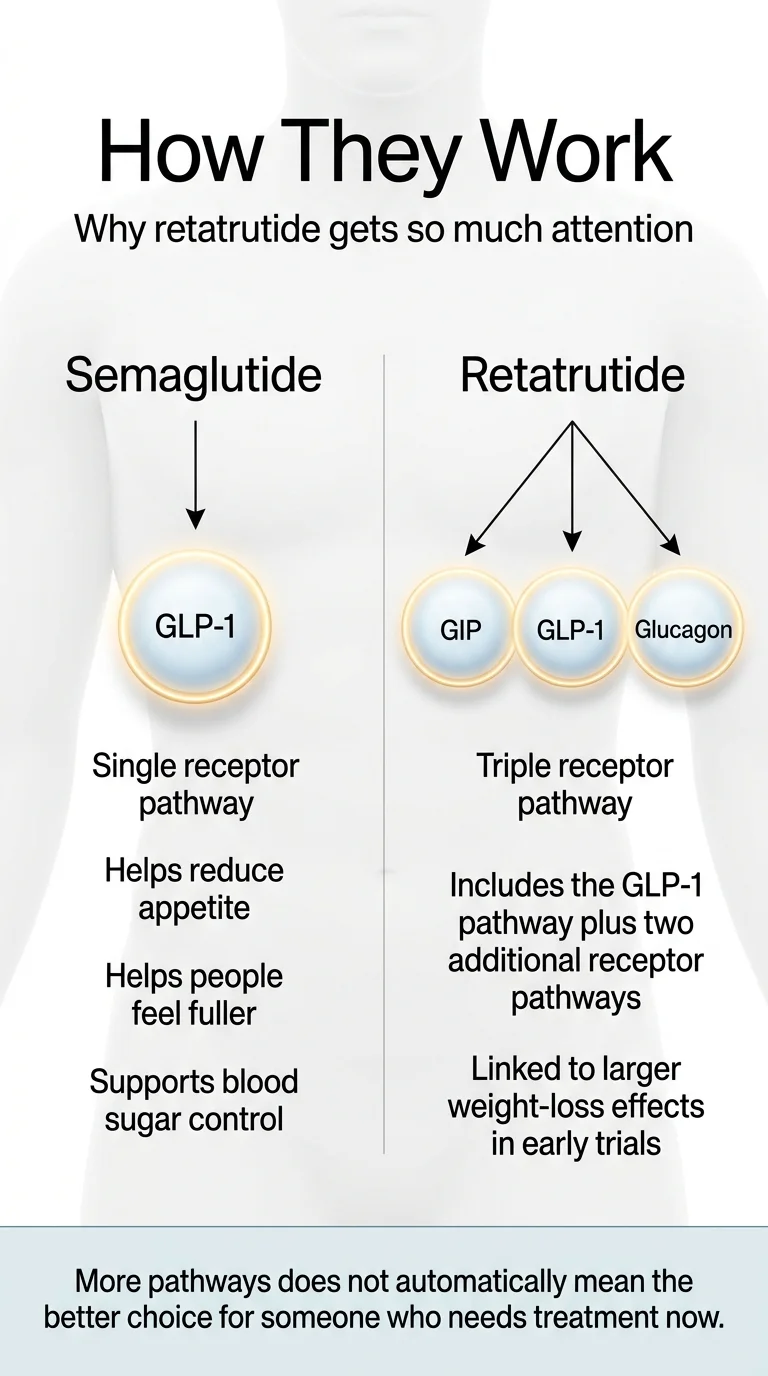
Semaglutide: One Pathway, Proven Results
Semaglutide mimics a hormone called GLP-1 that your gut naturally produces after you eat. When you inject semaglutide (or take the oral tablet), the GLP-1 signal is amplified and sustained. That does three main things: reduces appetite, slows stomach emptying so you feel full sooner, and improves insulin response to keep blood sugar stable.
This single-pathway approach has been enough to produce average weight loss of 15–21% depending on dose — results that seemed impossible from a medication just a few years ago.
Retatrutide: Three Pathways, Uncharted Territory
Retatrutide hits all three of those GLP-1 effects plus two additional receptor systems. GIP receptor activation enhances insulin signaling and improves how your body processes fat — the same second pathway that tirzepatide (Mounjaro/Zepbound) uses. Glucagon receptor activation increases energy expenditure and directly mobilizes fat stored in the liver.
Think of it this way: semaglutide reduces how much fuel you take in. Retatrutide does that and turns up how fast your body burns through stored fuel. That’s a fundamentally different metabolic equation.
Why “More” Doesn’t Automatically Mean “Better for You”
More pathways also means more variables. We have much less data on what happens when you activate three hormonal systems simultaneously over years of use. Semaglutide’s simplicity is actually one of its strengths. We know what it does. We know what it doesn’t do. We know what happens after 2 years of continuous use (STEP 5), and we know what happens when you stop. Retatrutide’s complexity is exciting — and genuinely less well-understood. That’s not a criticism. It’s where the science stands today.
The Weight Loss Data: What the Trials Actually Show
Semaglutide Results (STEP Program)
The STEP trials are among the most rigorous weight-loss studies ever conducted:
- STEP 1 (n=1,961): 14.9% average weight loss at 68 weeks with 2.4 mg. 86% of participants lost at least 5% of body weight. About one-third lost 20% or more.
- STEP 5 (long-term): 15.2% sustained at 104 weeks (2 years), confirming durability.
- STEP UP (n=1,407): 20.7% average weight loss at 72 weeks with the new 7.2 mg dose. Roughly one in three participants lost 25% or more of their body weight. FDA-approved March 19, 2026.
An important reality check: the STEP 1 extension study showed that participants who stopped semaglutide regained about two-thirds of their lost weight within a year. This confirms that obesity is a chronic condition requiring ongoing management — not a “take it and stop” situation.
Retatrutide Results (TRIUMPH + TRANSCEND Programs)
Retatrutide’s numbers are, frankly, staggering:
- Phase 2 (n=338): Up to 24.2% average weight loss at 48 weeks at 12 mg. At week 48, the weight-loss curve was still trending downward.
- TRIUMPH-4 Phase 3 (n=445): 28.7% average body weight loss at 68 weeks at 12 mg. That’s an average of 71.2 pounds lost. Nearly half of participants lost 25% or more.
- TRANSCEND-T2D-1 Phase 3 (n=537): Up to 16.8% weight loss at 40 weeks in patients with type 2 diabetes. Weight loss had not plateaued — Lilly noted the trajectory was still declining at week 40.
To put 28.7% in context: that’s the highest weight loss ever recorded in a Phase 3 obesity trial. Before GLP-1 drugs, that kind of result came only from bariatric surgery.
But TRIUMPH-4 was specifically in patients with obesity and knee osteoarthritis — a population with average BMI of 40.4. Heavier patients tend to lose a larger percentage. The upcoming TRIUMPH-1 trial (general obesity, 80-week duration) will give us the clearest picture.
What These Numbers Mean for a Real Person
If you weigh 230 pounds:
Semaglutide 2.4 mg
14.9%
~34 lbs lost
~196 lbs
Semaglutide HD 7.2 mg
20.7%
~48 lbs lost
~182 lbs
Retatrutide 12 mg
28.7%
~66 lbs lost
~164 lbs
Those are averages. Outcomes vary by genetics, starting weight, adherence, diet, activity, and factors we don’t fully understand yet. But the directional difference is real.
The semaglutide numbers — especially with Wegovy HD — are no longer as far behind as people think. That 20.7% is available now. The 28.7% is not.
60-second quiz — see which FDA-approved program fits your goals and budget.
Is Retatrutide Actually “Better” — or Just Newer?
“Better” is doing a lot of heavy lifting in that sentence. Better at what? For whom?
Retatrutide is likely better at:
- Producing raw weight loss
- Reducing liver fat (via glucagon pathway)
- Potentially broader metabolic improvements through its triple-receptor mechanism
Semaglutide is better at:
- Being a proven, available treatment you can access today
- Protecting your heart — proven 20% CV event reduction (SELECT)
- Having 5+ years of real-world safety data
- Multiple formats (injection 2.4 mg or 7.2 mg, oral tablet)
- Established insurance pathways and multiple price points
The honest framing: retatrutide looks like the stronger molecule. Semaglutide is the stronger choice — right now, today, for someone who wants to take action rather than wait.
If your real question is “what’s the strongest approved option I can start this week?”, the answer is tirzepatide (Zepbound/Mounjaro) at ~20–22% weight loss, or now Wegovy HD at 20.7%. Both are FDA-approved and available.
Side Effects: The Honest Comparison
Both drugs cause similar GI side effects — that’s true across the entire GLP-1 class. But the side-effect picture just changed significantly this week.
What Both Drugs Share
Nausea, diarrhea, vomiting, constipation, and decreased appetite are common with both. These are worst during dose escalation and typically improve as your body adjusts. Slow, gradual titration makes a significant difference.
Dysesthesia: No Longer Unique to Retatrutide
Until this week, we would have told you dysesthesia (skin tingling, sensitivity, tenderness) was essentially a retatrutide-specific finding. That is no longer accurate.
- Retatrutide: Dysesthesia reported in approximately 21% of patients at the 12 mg dose in TRIUMPH-4. In the TRANSCEND-T2D-1 diabetes trial, dysesthesia rates were lower (2.3–4.5% across doses).
- Semaglutide: The March 19, 2026 Wegovy HD approval revealed that altered skin sensation occurred in 22% of Wegovy HD (7.2 mg) patients — compared to 6% at 2.4 mg and 0.3% with placebo. The FDA noted this is under further investigation. Current Wegovy labeling now lists dysesthesia among common adverse reactions.
This is a significant development. Dysesthesia appears to be dose-related across both drugs and may be a broader incretin-class phenomenon at higher doses rather than something unique to retatrutide’s mechanism. We’ll update this section as more data emerges.
The Semaglutide Safety Advantage: Maturity and Heart Data
Semaglutide has been on the market since 2017 (diabetes) and 2021 (weight management). Years of real-world safety data from millions of prescriptions means we understand its risk profile well.
The SELECT trial — 17,604 patients followed for nearly 40 months — showed a 20% reduction in major adverse cardiovascular events (heart attack, stroke, cardiovascular death). That’s a proven, direct cardioprotective effect that led the FDA to expand Wegovy’s approved indications. No other weight-loss drug has this.
Retatrutide has no cardiovascular outcome data yet. Its TRIUMPH CVOT is still enrolling.
Side Effect Comparison
| Side Effect | Semaglutide 2.4 mg | Semaglutide HD 7.2 mg | Retatrutide 12 mg (TRIUMPH-4) |
|---|---|---|---|
| Nausea | ~44% | Similar | ~43% |
| Diarrhea | ~30% | Similar | ~33% |
| Vomiting | ~24% | Similar | ~21% |
| Dysesthesia | ~6% | ~22% (new finding) | ~21% |
| Discontinuation (AEs) | ~7% | Higher than 2.4 mg | 12–18% |
| CV outcome data | Positive (SELECT) | Covered under Wegovy label | Not yet available |
| Long-term safety | Well-established | New dose, monitoring ongoing | Emerging |
Is Retatrutide FDA-Approved? Can You Actually Get It?
No. As of March 2026, retatrutide is not FDA-approved for any indication. It has no brand name. It is available only to participants enrolled in Eli Lilly’s clinical trials. No NDA has been filed with the FDA yet.
Lilly has stated this clearly: retatrutide is an investigational molecule that is legally available only to participants in Lilly’s clinical trials, and launch will depend on the completion of trials and the regulatory approval process.
Any website or provider claiming to sell retatrutide for weight loss is either selling an unapproved, unregulated product or outright lying. The FDA has specifically warned against purchasing non-FDA-approved GLP-1 products marketed online. In March 2026, the FDA issued warning letters to 30 telehealth companies for illegal marketing of compounded GLP-1 medications.
Where Things Stand
- Two Phase 3 trials have reported results: TRIUMPH-4 (obesity + knee OA, Dec 2025) and TRANSCEND-T2D-1 (type 2 diabetes, March 2026). Both met all primary endpoints.
- Additional Phase 3 trials are expected to read out throughout 2026, covering obesity, sleep apnea, chronic low back pain, liver disease, and cardiovascular outcomes.
- No NDA has been filed. Lilly says launch depends on trial completion and the regulatory approval process.
What this means for you: If waiting for retatrutide is your plan, you’re looking at a significant wait — and every month spent waiting is a month you could be making progress with a proven, available medication. There’s a real cost to waiting. Not just financial — but the health impact of carrying excess weight for another year or two while a drug completes regulatory review.
Ro offers Wegovy and oral semaglutide with insurance billing support. Licensed providers.
Cost: What Will You Actually Pay?
You can price-compare semaglutide options right now. You cannot meaningfully price retatrutide because it doesn’t exist as a consumer product.
Semaglutide Costs (Current, Verified March 2026)
- Wegovy list price: Approximately $1,349/month — but almost no one pays this.
- Novo Nordisk self-pay programs (current): Wegovy pen offers start at $199 for the first two lower-dose fills, then $349/month for ongoing fills. Wegovy tablets start at $149/month for lower doses, with higher doses listed at $299/month.
- With insurance: Varies — $0 to $500+/month depending on plan, copay cards, and prior authorization.
- Telehealth GLP-1 programs: $199–499/month depending on provider, dose level, and specific medication.
- Wegovy HD (7.2 mg) pricing: Expected to be announced at or before the April 2026 U.S. launch.
Note: Novo Nordisk has announced it will lower the list price of Wegovy effective January 1, 2027. Current self-pay programs offer significantly lower prices than the list price.
Retatrutide Cost Reality
No pricing has been announced — the drug isn’t approved yet. Analysts project it in the $1,000–1,500/month range based on the precedent set by Eli Lilly’s tirzepatide (Zepbound), but these are projections, not confirmed prices.
The practical implication: Right now, semaglutide has multiple access pathways at various price points, including official manufacturer programs well below list price. Retatrutide has zero legitimate access pathways at any price.
For a deeper look at insurance coverage for semaglutide, see our guide: How to Get Semaglutide Covered by Insurance.
Dosing: What Does Treatment Actually Look Like?
Both drugs are once-weekly subcutaneous injections. Semaglutide now also comes in a daily oral tablet.
Semaglutide Dosing (FDA-Approved — Wegovy)
Classic Wegovy 2.4 mg injection titration:
| Weeks | Dose |
|---|---|
| Weeks 1–4 | 0.25 mg |
| Weeks 5–8 | 0.5 mg |
| Weeks 9–12 | 1.0 mg |
| Weeks 13–16 | 1.7 mg |
| Week 17+ | 2.4 mg (maintenance) |
Wegovy HD 7.2 mg (approved March 2026): For adults who have tolerated 2.4 mg for at least 4 weeks and need additional weight reduction. Titration continues upward from 2.4 mg to 3.6 mg, then 7.2 mg. Available in U.S. pharmacies from April 2026.
Wegovy tablets (25 mg oral): Once-daily pill, launched January 2026. An alternative for patients who prefer oral dosing. See: How to Get the Wegovy Pill Online.
Retatrutide Dosing (Investigational — Trial Protocol Only)
In trials, retatrutide titration followed this general pattern: 2 mg → 4 mg → 6 mg → 9 mg or 12 mg, with increases every 4 weeks. A 4 mg maintenance dose is also being studied.
A critical distinction: There is no FDA-approved dose of retatrutide. These are clinical trial protocols, not prescribing guidance. Dosage charts on peptide websites or forums are not equivalent to the carefully monitored titration used in registered clinical trials. If someone is offering you a “retatrutide dosing guide” online, that’s a signal to walk away.
Can You Take Semaglutide and Retatrutide Together?
No. This is not an approved or studied combination. Wegovy labeling states that semaglutide should not be used with other semaglutide-containing products or other GLP-1 receptor agonists. Retatrutide includes GLP-1 activity as one of its three mechanisms.
No study has evaluated this combination. Combining them could increase side effect risk with no proven benefit. If you’ve seen online forums or peptide websites suggesting otherwise, treat that as a red flag about the source.
Can You Switch From Semaglutide to Retatrutide?
Not today. Retatrutide isn’t available to switch to.
But we understand why you’re asking. The real question is usually one of these: My weight loss slowed down. Is there something stronger? Or: Should I wait for the better drug instead of starting now?
If semaglutide isn’t working as well as you’d hoped, the most productive next steps — available today:
- Check your dose. Many people haven’t titrated to full dose. With Wegovy HD now approved, there’s a new option above 2.4 mg for those who need more.
- Audit the basics. Protein intake, resistance training, sleep, and stress all meaningfully affect GLP-1 response.
- Ask about tirzepatide. Zepbound is FDA-approved and produces approximately 20–22% weight loss.
- Consider a program change. Different providers offer different levels of clinical support and dose flexibility.
When retatrutide eventually reaches the market, switching protocols will be developed by clinicians. But the best move is almost never “wait and do nothing while a drug completes regulatory review.”
Answer a few questions — get a personalized next step based on your goals and history.
Where Does Tirzepatide Fit?
Tirzepatide is the available middle ground — and it deserves a serious look. See our full Retatrutide vs Tirzepatide comparison for the complete picture.
| Semaglutide 2.4 mg | Semaglutide HD 7.2 mg | Tirzepatide | Retatrutide | |
|---|---|---|---|---|
| Receptors | GLP-1 (1) | GLP-1 (1) | GLP-1 + GIP (2) | GLP-1 + GIP + Glucagon (3) |
| Avg weight loss | ~14.9% | ~20.7% | ~20–22% | ~24–28.7% |
| FDA approved | Yes | Yes (March 2026) | Yes (2023) | No |
| Available now | Yes | April 2026 | Yes | No |
| Brand names | Wegovy, Ozempic | Wegovy HD | Zepbound, Mounjaro | None |
| CV outcome data | Yes — positive | Under Wegovy label | Expected 2027 | Not yet |
If your core desire is “the most weight loss from a drug I can actually take today,” tirzepatide and Wegovy HD are remarkably close in efficacy — and both are available.
Heart Health: Which One Protects Your Heart?
This isn’t close right now — semaglutide wins clearly.
The SELECT trial (17,604 patients, ~40 months follow-up) showed semaglutide produced a 20% reduction in major adverse cardiovascular events — heart attack, stroke, cardiovascular death — in people with obesity and established heart disease. This is a proven, clinically meaningful result. No other weight-loss drug has this.
Retatrutide has shown improvements in cardiovascular markers (cholesterol, triglycerides, blood pressure) across its trials. Promising, but improved biomarkers are not the same as proven outcomes. The dedicated cardiovascular outcomes trial is still enrolling.
If heart health is a primary concern: semaglutide is the clear choice.
Liver and MASH: Promising Retatrutide Data, but Semaglutide Now Has an Approved MASH Indication
Retatrutide produced striking liver-fat reductions in Phase 2 substudies — up to 86%. That’s driven by its glucagon receptor activation, which directly mobilizes fat stored in the liver. A dedicated TRIUMPH liver disease trial is underway.
But semaglutide is no longer just a hypothetical comparator in liver disease. The current Wegovy injection label now includes an FDA-approved indication for treating MASH (metabolic dysfunction-associated steatohepatitis) in adults with moderate-to-advanced liver scarring (fibrosis). This makes semaglutide the only GLP-1 with an approved MASH use.
Retatrutide’s liver-fat data are very impressive and may eventually prove superior. But today, semaglutide is the approved option for patients with liver disease — and it’s available now.
Does Either Drug Cause Muscle Loss?
Yes — both can, to some degree. But the picture is more nuanced than headlines suggest.
All significant weight loss — whether from medication, surgery, or calorie restriction — involves some lean mass loss alongside fat loss. This is a biological reality, not a drug-specific failure.
- What we know: Both semaglutide and retatrutide can reduce lean mass as part of overall weight loss. A 2025 retatrutide body-composition substudy in type 2 diabetes found that the proportion of lean-mass loss relative to total weight loss was similar to other obesity treatments. Semaglutide STEP 1 body-composition data showed lean mass decreased but the lean-mass proportion of total body mass actually improved.
- What we don’t know yet: Non-diabetic Phase 3 retatrutide body-composition data are still limited.
The practical guidance is the same for both drugs:
- Aim for 0.7–1.0 grams of protein per pound of your target body weight daily
- Resistance train 2–3 times per week throughout treatment
- Work with a provider who monitors body composition, not just scale weight
- The net health benefit of significant fat loss far outweighs lean mass reduction when exercise and protein are prioritized
Who Should Choose Semaglutide, Who Should Watch Retatrutide, and Who Should Do Something Else?
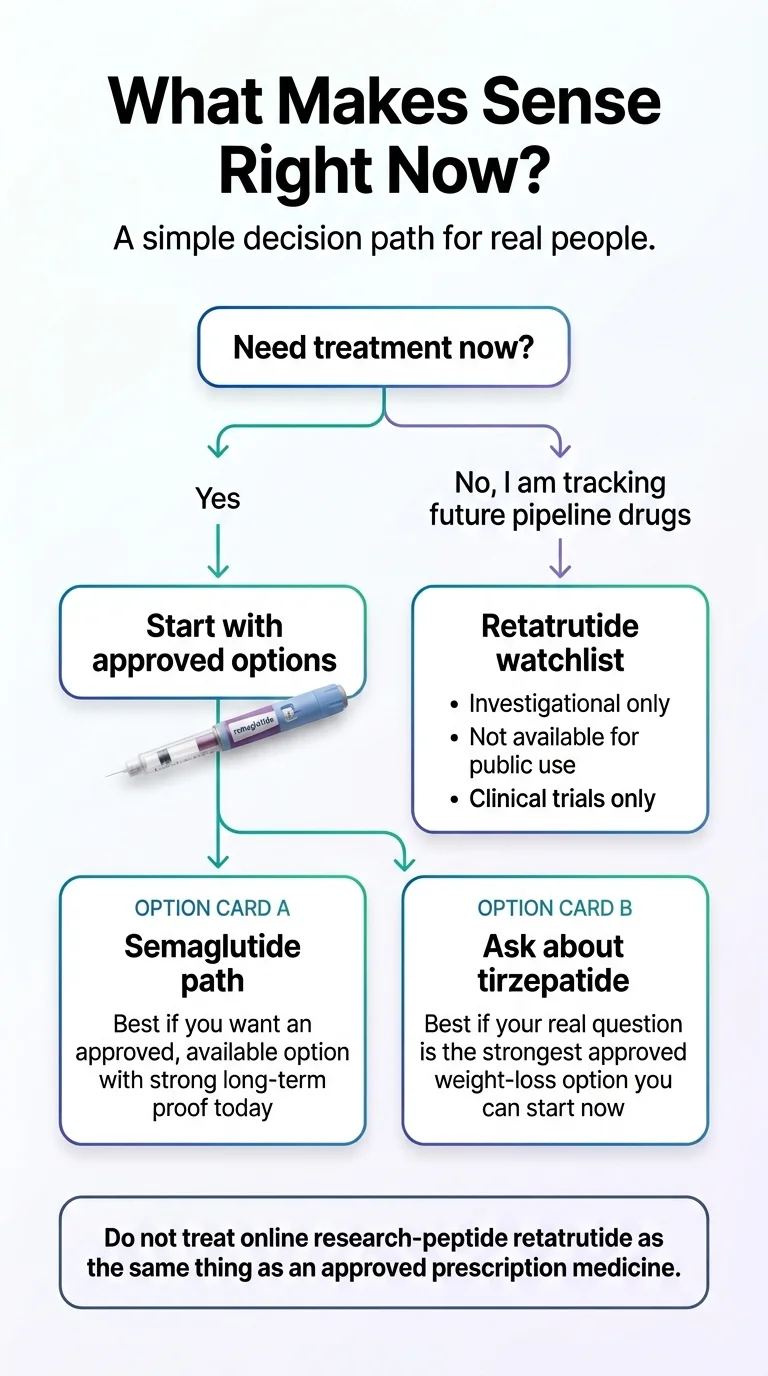
Start semaglutide (or tirzepatide) now if:
- You have a BMI of 30+ (or 27+ with weight-related health conditions) and want to begin treatment this month
- Heart health is a priority — semaglutide has proven cardiovascular benefits no other weight-loss drug matches
- You want the confidence that comes with years of real-world safety data
- You want multiple price points and format options (injection or pill)
- You’d rather make 12–18 months of real progress than spend that time waiting
This is most people reading this page. 15–21% weight loss over the next year is infinitely more valuable than 28% weight loss starting at some unknown future date.
Keep retatrutide on your radar if:
- You’ve tried semaglutide or tirzepatide and genuinely plateaued despite optimized dose, diet, and exercise
- You have a very high BMI (40+) and may eventually benefit from the most aggressive pharmaceutical option
- You have significant liver disease and are interested in the triple-agonist approach
- You’re comfortable being an early adopter when the drug becomes available
Even in this group, the smart play is usually: start an available GLP-1 now, make progress, and switch later if retatrutide delivers on its promise.
Be extra cautious if:
- You’re tempted to buy “retatrutide” from an online peptide website → don’t. The FDA has warned against this and issued warning letters to 30 telehealth companies in March 2026 for illegal GLP-1 marketing. Independent testing of some compounded products has revealed impurities as high as 86%.
- You have contraindications to GLP-1 drugs → personal or family history of medullary thyroid carcinoma, MEN type 2, or severe GI conditions. Talk to your doctor.
THE ONE THING WE KEEP HEARING
In the GLP-1 community, the most common regret isn’t “I chose the wrong drug.” It’s “I wish I’d started sooner.”
A year from now, you’ll be glad you started today.
Free, 60 seconds. No commitment. Matched to your goals, budget, and health profile.
Weight Regain: What Happens When You Stop?
Both drugs share this reality: this isn’t a “take it for six months and you’re done” situation.
The STEP 1 extension study showed participants regained approximately two-thirds of their lost weight within 12 months of stopping semaglutide. Cardiometabolic improvements also reverted toward baseline. We don’t have discontinuation data for retatrutide yet, but there’s no reason to expect it would differ.
This isn’t a failure — it’s confirmation that obesity is a chronic condition that typically requires ongoing treatment, similar to blood pressure medication. Plan for long-term commitment with whichever medication you choose.
How We Built This Comparison
Our Source Hierarchy
| Evidence Tier | What It Is | How We Use It |
|---|---|---|
| Tier 1 | FDA prescribing information, peer-reviewed randomized controlled trials (NEJM, Lancet, Nature Medicine) | Primary proof for all efficacy and safety claims |
| Tier 2 | ClinicalTrials.gov registrations, official trial design documents | Trial design, enrollment, timeline verification |
| Tier 3 | Manufacturer press releases, investor communications (Eli Lilly, Novo Nordisk) | Topline results for trials not yet published in journals |
| Tier 4 | Community discussions, forum language, patient experience | Understanding real questions — never used as proof |
Every clinical number on this page traces to Tier 1 or Tier 3 sources. We explicitly note when a data point comes from a press release rather than a peer-reviewed publication.
Affiliate Relationship
We earn commissions when readers use our links to GLP-1 providers. This does not influence our analysis. We would recommend starting an available GLP-1 over waiting for retatrutide regardless of any affiliate relationship, because it’s the honest answer based on the evidence. If you stripped every affiliate link from this page, the recommendations would be identical.
Sources Cited
- Wilding JPH, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384(11):989-1002.
- Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity. N Engl J Med. 2023;389(6):514-526.
- Lincoff AM, et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. N Engl J Med. 2023;389(24):2221-2232.
- Garvey WT, et al. Two-year effects of semaglutide: the STEP 5 trial. Nature Medicine. 2022;28:2083-2091.
- Wharton S, et al. Once-weekly semaglutide 7.2 mg in adults with obesity (STEP UP). Lancet Diabetes Endocrinol. 2025;13(11):949-963.
- Eli Lilly. TRIUMPH-4 topline results. Press release, Dec 11, 2025.
- Eli Lilly. TRANSCEND-T2D-1 topline results. Press release, March 19, 2026.
- Novo Nordisk. Wegovy HD (semaglutide 7.2 mg) approved in the US. Press release, March 19, 2026.
- FDA Prescribing Information: Wegovy (semaglutide) injection, 2026 label.
- FDA. “FDA Intends to Take Action Against Non-FDA-Approved GLP-1 Drugs.” fda.gov.
- Eli Lilly. “What to know about retatrutide.” lilly.com (updated March 2026).
- ClinicalTrials.gov: NCT05869903 (TRIUMPH-4), NCT06354660 (TRANSCEND-T2D-1), NCT03548935 (STEP 1).
Frequently Asked Questions
Is retatrutide better than semaglutide for weight loss?
On raw numbers, retatrutide shows greater weight loss in trials — 28.7% vs semaglutide's range of 14.9–20.7% depending on dose. But 'better' depends on more than one metric. Semaglutide has proven cardiovascular benefits, years of safety data, an approved MASH indication, and is available right now. Retatrutide is not approved, not available, and its long-term safety profile is still being established.
Is retatrutide FDA-approved?
No. As of March 2026, retatrutide is an investigational drug in Phase 3 clinical trials. It has not been approved by the FDA or any regulatory body. Eli Lilly says launch depends on trial completion and the regulatory approval process.
When will retatrutide be available?
Eli Lilly has not announced a specific date. The company says public availability depends on completion of clinical trials and the regulatory approval process. Additional Phase 3 results are expected throughout 2026.
Can you switch from semaglutide to retatrutide?
Not currently — retatrutide isn't available. When eventually approved, switching protocols will be developed. If you're unsatisfied with semaglutide results, discuss dose optimization (including the new 7.2 mg HD option) or tirzepatide with your provider.
Can you take semaglutide and retatrutide together?
No. Wegovy labeling states semaglutide should not be used with other GLP-1 receptor agonists. No clinical trial has studied this combination. Don't do it.
What is the difference between Ozempic, Wegovy, and retatrutide?
Ozempic and Wegovy are both semaglutide, made by Novo Nordisk. Ozempic is approved for type 2 diabetes. Wegovy (including HD and tablets) is approved for weight management, cardiovascular risk reduction, and MASH. Retatrutide is a completely different drug — a triple agonist from Eli Lilly — not approved for anything yet.
Does retatrutide cause more nausea than semaglutide?
No — nausea rates are similar (~43% vs ~44%). The notable difference was dysesthesia (skin tingling), but the March 2026 Wegovy HD data shows dysesthesia is also common at higher semaglutide doses (22%). This appears to be a dose-related phenomenon across the drug class.
What is the approved dose of retatrutide?
There is no approved dose. Retatrutide is investigational. In trials, doses of 4 mg, 9 mg, and 12 mg were studied. Never attempt to self-dose using products sold online.
Is there a legitimate way to buy retatrutide online?
No. The only legal access is through enrollment in an active Eli Lilly clinical trial. Products sold online as 'retatrutide' or 'research peptides' are not FDA-regulated and may be dangerous. The FDA issued warning letters to 30 telehealth companies in March 2026 for illegal GLP-1 marketing.
Does retatrutide cause muscle loss?
All significant weight loss involves some lean mass reduction. Available body-composition data suggests the ratio of lean-mass loss to total weight loss with retatrutide is similar to other obesity treatments. Resistance training and high protein intake remain important with any GLP-1 medication.
Is retatrutide better than tirzepatide?
Retatrutide appears to produce more weight loss than tirzepatide based on cross-trial comparisons (28.7% vs ~20–22%). But tirzepatide is FDA-approved and available now. If your goal is the strongest available option today, tirzepatide is worth discussing with your provider.
Which option makes more sense if I need treatment now?
Semaglutide or tirzepatide. Both are FDA-approved, widely available, and backed by extensive trial data. Semaglutide now offers more options than ever — including Wegovy HD at 20.7% weight loss and an oral tablet. Choosing between them depends on your health profile, insurance, and goals.
What if semaglutide stopped working for me?
Weight-loss plateaus on semaglutide are common and don't necessarily mean the drug stopped working. First steps: confirm you're at full dose (and ask about Wegovy HD 7.2 mg if you've been on 2.4 mg), review protein and activity, and discuss adherence honestly with your provider. If you've optimized everything, tirzepatide may be a productive next conversation.
What should you do next?
You’ve read the data. You understand the difference. You know what’s available and what isn’t. Here’s where you go from here:
If you’re ready to start
Our GLP-1 matching tool takes about 60 seconds and pairs you with a licensed provider based on your health profile, goals, budget, and insurance situation. No commitment, no pressure — just clarity on your best realistic option.
If semaglutide isn’t working well enough
Don’t assume you’ve hit your ceiling. Wegovy HD 7.2 mg just became available. A provider switch, dose adjustment, or conversation about tirzepatide could change your trajectory.
If you want to track retatrutide
Bookmark this page. We update it with every trial readout and every regulatory milestone. You’ll know the moment anything changes.
Still not sure? Answer a few questions — we’ll show you the clearest next step based on your goals and what’s actually available today.