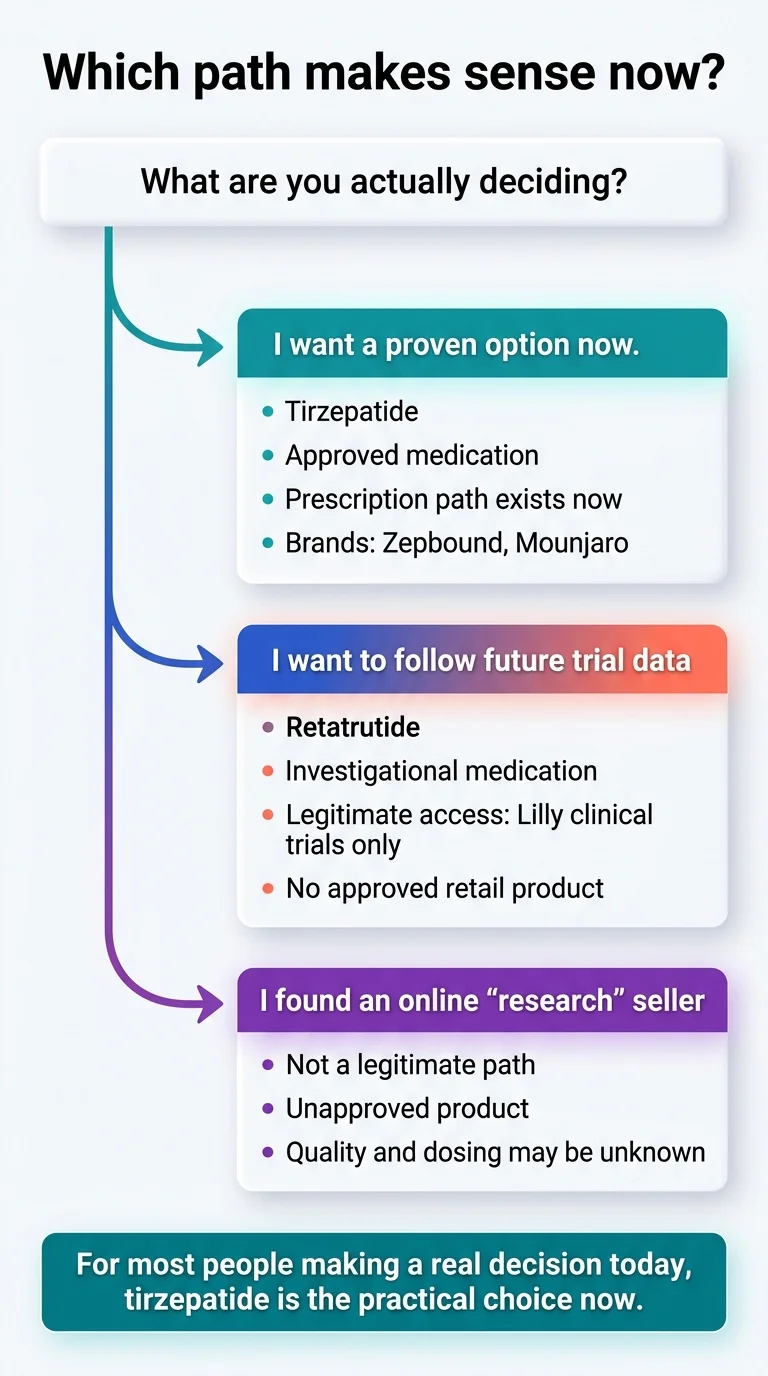
Tirzepatide (Zepbound for weight loss, Mounjaro for diabetes) is FDA-approved, available by prescription, and backed by real-world prescribing experience plus pivotal trial data showing 20.9% mean weight loss at 72 weeks in SURMOUNT-1.1 Retatrutide — a next-generation triple agonist — has posted the highest weight loss numbers ever recorded in obesity drug trials (up to 28.7% in TRIUMPH-4)2, but it is still investigational, still in Phase 3 clinical trials, and not available by prescription anywhere in the world.
Both drugs are made by Eli Lilly. Both are once-weekly injections. But one is a proven, prescribable medication and the other is a promising research compound that has never been FDA-reviewed — and that distinction changes everything about the decision you're actually making.

Most people deciding today: tirzepatide is the practical choice. Retatrutide is promising but still investigational.
Retatrutide vs Tirzepatide at a Glance
| Tirzepatide | Retatrutide | |
|---|---|---|
| Brand Names | Zepbound (obesity), Mounjaro (T2D) | None — investigational |
| Manufacturer | Eli Lilly | Eli Lilly |
| How It Works | Dual agonist (GLP-1 + GIP) | Triple agonist (GLP-1 + GIP + Glucagon) |
| FDA-Approved? | Yes (T2D: 2022, Obesity: 2023, Sleep apnea: 2024) | No |
| Available by Prescription? | Yes | No — clinical trials only |
| Pivotal Obesity Trial | 20.9% at 72 weeks (SURMOUNT-1, 15 mg); 25.3% total by week 88 (SURMOUNT-4)1 | 28.7% at 68 weeks (TRIUMPH-4, 12 mg)2 |
| Dosing | 2.5 mg → max 15 mg weekly (FDA-approved)4 | 2 mg → 9 or 12 mg weekly (trial protocol only)5 |
| Monthly Cost | From ~$299/mo (self-pay options)6 | Not commercially priced |
| Insurance Coverage | Yes, expanding by plan | None (not approved) |
| Key Side Effects | Nausea, diarrhea, vomiting | Nausea, diarrhea, vomiting + dysesthesia |
| Long-Term Safety Data | Years of real-world use | Limited to clinical trials |
| Head-to-Head Trial | — | TRIUMPH-5 (ongoing, ~89 weeks, 800 patients) |
Bottom line: If you need treatment now, tirzepatide is the proven path. If you're watching the future, retatrutide is the drug to follow — but not to buy from unregulated sources.
Most people spend hours researching medications and minutes choosing a provider. Skip the confusion.
What Changed in 2026?
If you're seeing “retatrutide vs tirzepatide” pop up everywhere, here's why.
On March 19, 2026, Eli Lilly announced the first Phase 3 diabetes results for retatrutide (TRANSCEND-T2D-1).5 The drug hit every endpoint — reducing A1C by up to 2.0% and producing weight loss up to 16.8% at just 40 weeks, with no weight loss plateau in sight. That came on top of the December 2025 TRIUMPH-4 results showing 28.7% weight loss in an obesity trial2 — the highest number ever recorded for any anti-obesity medication.
Those two data drops turned retatrutide from a “promising Phase 2 curiosity” into a serious next-generation contender. But — and this matters — none of that changed its availability. Retatrutide is still not FDA-approved. Eli Lilly has not submitted a New Drug Application.3 The drug is still only legally available through Lilly-sponsored clinical trials, and the FDA has specifically warned consumers about buying unapproved GLP-1 products online.8
So this page exists because the question just got more interesting, but the answer hasn't fundamentally changed: tirzepatide is still the real-world option, and retatrutide is still the investigational one.
Retatrutide vs Tirzepatide: Which Is Better Right Now?
This is the question you came here for. Let's answer it directly.
For most people, tirzepatide is the better choice today
Not because it's theoretically superior. Because it's real. You can get a prescription this week. Your doctor has experience dosing it. Insurance may cover it. Years of safety data exist. And the results — ~20% body weight loss in clinical trials, 47% greater weight loss than semaglutide in the SURMOUNT-5 head-to-head — are already exceptional by any historical standard.
Before tirzepatide existed, losing 20% of your body weight with medication was considered almost impossible. Now it's the baseline for a drug that's sitting on pharmacy shelves.
Retatrutide is the more exciting future drug — not the more practical current one
Retatrutide's numbers are remarkable. But remarkable numbers from a drug you can't access don't help you this month. The triple-agonist mechanism is genuinely novel. The Phase 3 data is genuinely impressive. The eventual head-to-head trial (TRIUMPH-5) will be genuinely informative. None of that changes the fact that you can't walk into a pharmacy and fill a retatrutide prescription today, tomorrow, or at any point while it remains in clinical development.
The real decision framework
Start tirzepatide now if…
- ✓Weight loss is a current health priority
- ✓You want FDA-approved medication with established safety data
- ✓You have type 2 diabetes (Mounjaro is approved)
- ✓You have obesity with sleep apnea (Zepbound has OSA indication)
- ✓You want a defined cost and insurance pathway
- ✓You have high cholesterol, high BP, or prediabetes
- ✓You've tried semaglutide and want stronger data
Consider watching retatrutide if…
- ·You've plateaued on current medications
- ·You're willing to wait until 2027+ for approval
- ·You're open to participating in a legitimate clinical trial now
- ·You have MASH/MASLD (fatty liver disease) and are interested in glucagon receptor benefits
- ·Delaying treatment 1–2 years doesn't pose significant health risks
Important: Do not do this
- Don't buy “retatrutide” from online peptide suppliers. It's not the same thing as using a drug studied in controlled trials with verified dosing. The FDA has issued warnings about unapproved GLP-1 products.
- Don't stop tirzepatide to “save your appetite” for retatrutide. That's not how these medications work, and you'd lose months of progress.
Talk to a clinician first if you have a history of pancreatitis, gallbladder issues, or medullary thyroid cancer; if you're planning pregnancy; if you have very low muscle mass; or if you're taking insulin or sulfonylureas.
Tell us your goals, insurance, and timeline. We match you with the right tirzepatide program.
What's the Difference Between Retatrutide and Tirzepatide?
The core difference is how many hormonal pathways each drug activates.
Think of it as generations of the same idea. Semaglutide (Ozempic, Wegovy) was the first generation — it mimics one hormone (GLP-1) to suppress appetite and slow digestion. Tirzepatide was the second generation — it mimics two hormones (GLP-1 + GIP), adding improved insulin response and metabolic efficiency. Retatrutide is the third generation — it mimics three hormones (GLP-1 + GIP + glucagon), adding a mechanism that tells your body to burn more fat and increase resting energy expenditure.
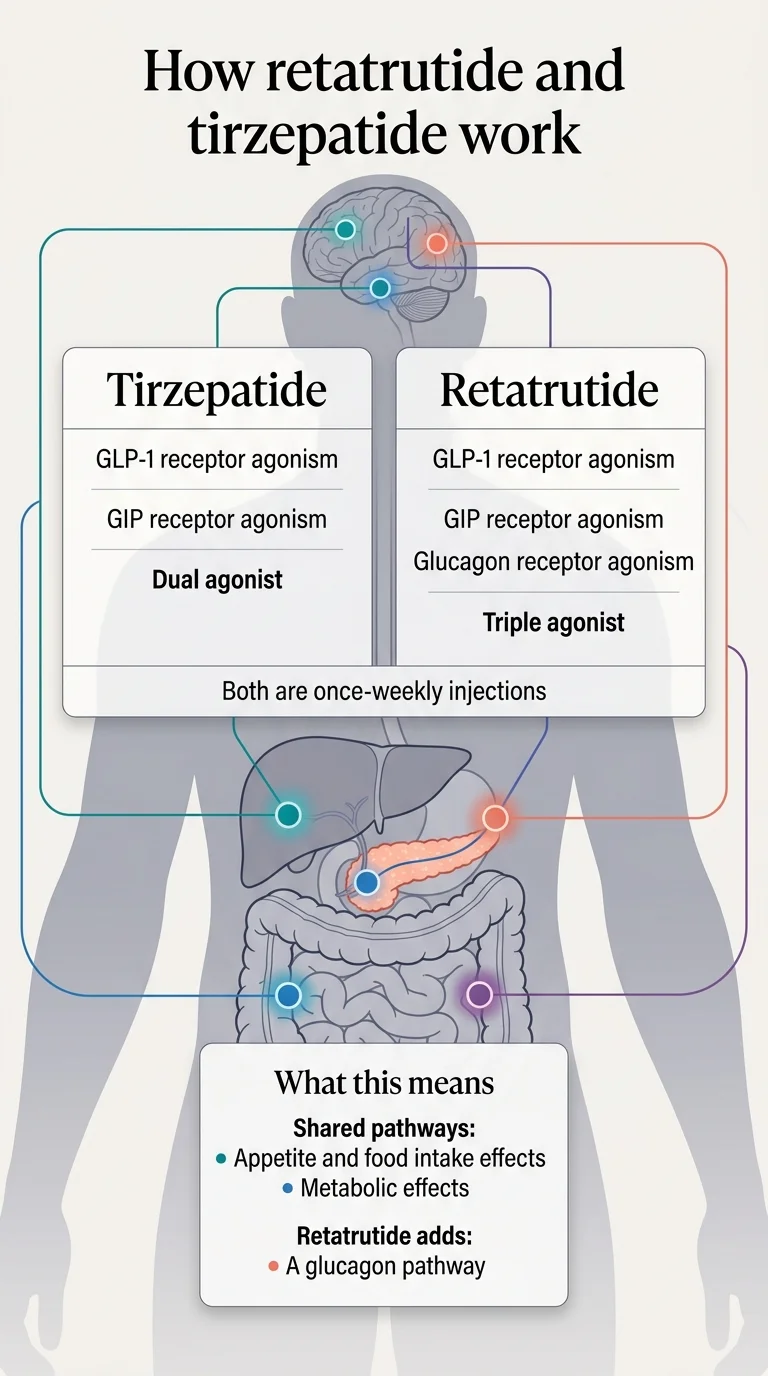
Shared pathways: appetite and food intake, metabolic effects. Retatrutide adds a glucagon pathway for additional fat oxidation.
What tirzepatide does
Tirzepatide activates GLP-1 and GIP receptors. GLP-1 slows stomach emptying, reduces appetite, and helps regulate blood sugar. GIP enhances insulin sensitivity and appears to play a role in how the body manages energy from food. Working together, these two pathways produce strong appetite suppression and meaningful metabolic improvements.
In practical terms, people on tirzepatide describe feeling genuinely less hungry — not white-knuckling through cravings, but simply not thinking about food as much. The effect on blood sugar is also significant, which is why tirzepatide was first approved as a diabetes medication (Mounjaro) before getting obesity approval (Zepbound).
This is not theoretical. In the SURMOUNT-1 trial published in the New England Journal of Medicine, tirzepatide at 15 mg produced an average 20.9% body weight reduction over 72 weeks. That's roughly 52 pounds for someone starting at 231 pounds. More than 90% of participants lost at least 5% of their weight. Over half lost 20% or more.1
What retatrutide adds
Retatrutide keeps everything tirzepatide does and adds glucagon receptor activation. Glucagon is a hormone that tells your liver to produce energy, increases fat oxidation (your body's ability to burn stored fat), and raises resting energy expenditure — essentially, your body burns more calories even at rest.
That third pathway is one plausible reason retatrutide may produce greater weight loss, though the precise contribution of each receptor pathway has not been established in a direct mechanistic comparison.7 It's also why the drug is being studied for liver disease (MASH) — the glucagon receptor has specific effects on liver fat metabolism that neither GLP-1 nor GIP provide. And it's why retatrutide has a side effect that tirzepatide doesn't: dysesthesia (more on that below).
One way to think about it: semaglutide turns down your appetite. Tirzepatide turns down your appetite and improves how your body handles the food you do eat. Retatrutide does both of those things and also turns up your body's fat-burning engine. Each step adds efficacy. Each step also adds biological complexity.
Why mechanism alone shouldn't decide the winner
It's tempting to think “three receptors is automatically better than two.” But pharmacology doesn't work that way. More targets means more physiological effects — wanted and unwanted. It also means more unknowns.
Tirzepatide has been prescribed to millions of people. Its safety profile is well-characterized. We know what happens when you take it, when you stop it, and what the long-term risks look like. Retatrutide has been studied in approximately 6,000 clinical trial participants. We don't yet know the full long-term picture.
The mechanism makes retatrutide promising. The evidence and access make tirzepatide practical. Those are two different things.
Weight Loss: What Do the Trials Actually Show?
Let's put real numbers on the table. Every figure below is cited to a specific published trial or official press release.
Tirzepatide weight loss data (proven)
SURMOUNT-1 (NEJM, 2022): 2,539 adults with obesity, no diabetes. At 72 weeks, the 15 mg dose produced an average 20.9% weight loss (~52 lbs from a ~231 lb baseline). 91% of participants lost ≥5% of body weight. 57% lost ≥20%.1
SURMOUNT-4 (JAMA, 2024): Maintenance study. Adults who continued tirzepatide after 36 weeks achieved 25.3% total weight loss by week 88. Those who stopped regained most of their lost weight — confirming that this is a long-term treatment, not a short course.10
SURMOUNT-5 (NEJM, 2025): Head-to-head against semaglutide (Wegovy). Tirzepatide: 20.2% weight loss. Semaglutide: 13.7%. That's 47% greater relative weight loss with tirzepatide.11 This is the strongest comparative evidence for any obesity drug vs. another.
Retatrutide weight loss data (emerging)
Phase 2 (NEJM, 2023): 338 adults with obesity. At 48 weeks, the 12 mg dose produced 24.2% average weight loss. The 8 mg dose: 22.8%. More than 8 in 10 participants at the highest doses lost ≥15%.7
TRIUMPH-4 Phase 3 (Eli Lilly, December 2025): 445 adults with obesity and knee osteoarthritis. At 68 weeks, the 12 mg dose produced 28.7% weight loss — an average of 71.2 pounds from a baseline of ~248.5 lbs. The 9 mg dose: 26.4%. These are the highest weight loss figures ever reported for any obesity medication.2
TRANSCEND-T2D-1 Phase 3 (Eli Lilly, March 19, 2026): Adults with type 2 diabetes. The 12 mg dose produced 16.8% weight loss at 40 weeks. A1C dropped by up to 2.0%. Notably, there was no weight loss plateau — participants were still losing weight when the study period ended.5
Why you can't directly compare 28.7% vs 20.9%
Those two numbers come from different trials, different populations, different durations, and different conditions. TRIUMPH-4 enrolled people with knee osteoarthritis (average baseline weight: ~248 lbs). SURMOUNT-1 enrolled a broader obesity population (average baseline: ~231 lbs). The trial durations were different (68 vs 72 weeks). The dosing escalation protocols were different.
Comparing them directly is like comparing a runner's 5K time at sea level to another runner's time at altitude. The numbers are real, but they're not from the same race.
The honest takeaway: both drugs produce extraordinary weight loss. Retatrutide's numbers are higher in the data we have. But the only way to truly compare them is a randomized head-to-head trial — and that's exactly what TRIUMPH-5 is doing right now.
The head-to-head trial that matters most
TRIUMPH-5 (NCT06662383) is an ongoing Eli Lilly trial directly comparing retatrutide to tirzepatide in 800 adults with obesity over 89 weeks.12 This is the study that will actually answer the question “which one causes more weight loss in the same population under the same conditions?” Results haven't been published yet. When they are, we'll update this page.
Weight loss evidence comparison
| Trial | Drug | Phase | Duration | Max Dose | Avg Loss | Status |
|---|---|---|---|---|---|---|
| SURMOUNT-1 | Tirzepatide | 3 | 72 wks | 15 mg | 20.9% | Published (NEJM) |
| SURMOUNT-4 | Tirzepatide | 3 | 88 wks | 15 mg | 25.3% total | Published (JAMA) |
| SURMOUNT-5 | Tirz vs Sema | 3b | 72 wks | 15 mg | 20.2% vs 13.7% | Published (NEJM) |
| Phase 2 | Retatrutide | 2 | 48 wks | 12 mg | 24.2% | Published (NEJM) |
| TRIUMPH-4 | Retatrutide | 3 | 68 wks | 12 mg | 28.7% | Topline released |
| TRANSCEND-T2D-1 | Retatrutide | 3 | 40 wks | 12 mg | 16.8% | Topline released |
| TRIUMPH-5 | Retat vs Tirz | 3 | 89 wks | TBD | Pending | Ongoing |
Sources: NEJM (2022, 2023, 2025), JAMA (2024), Eli Lilly press releases (Dec 2025, March 2026), ClinicalTrials.gov. Cross-trial comparisons have significant limitations; see explanation above.
Retatrutide vs Tirzepatide Side Effects: The Honest Comparison
Both drugs share a common set of gastrointestinal side effects because they both activate GLP-1 receptors, which slow stomach emptying. Nausea, diarrhea, vomiting, and constipation are the most frequently reported with both — typically worst during the first few weeks or when the dose increases, and improving as the body adjusts.
But there's an important difference that many comparison sites downplay or skip entirely.
What retatrutide has that tirzepatide doesn't: dysesthesia
In the TRIUMPH-4 Phase 3 trial, 20.9% of patients on the 12 mg retatrutide dose reported dysesthesia — an abnormal skin sensation that can feel like tingling, tenderness, or heightened sensitivity to touch. At the 9 mg dose, 8.8% reported it. In the placebo group, just 0.7%.2
That means roughly 1 in 5 people on the highest dose experienced this. It was generally described as mild and rarely led to stopping treatment. But it's clinically meaningful, dose-dependent, and new — it wasn't seen in the Phase 2 trial.
The mechanism behind retatrutide-associated dysesthesia has not been confirmed. Neither tirzepatide nor semaglutide activate the glucagon receptor, and neither produces dysesthesia at comparable rates — which has led researchers to investigate the glucagon component as a potential contributor, but this remains unproven.5
We're telling you about this not to scare you off retatrutide, but because a trustworthy comparison has to include the trade-offs. If a site compares these two drugs and doesn't mention dysesthesia, they're either not up to date or they're not being straight with you.
Side-by-side comparison
| Side Effect | Tirzepatide (15 mg) | Retatrutide (12 mg, Phase 3) |
|---|---|---|
| Nausea | ~25–30% | ~43% |
| Diarrhea | ~17–20% | ~33% |
| Vomiting | ~9–12% | ~21% |
| Constipation | ~10–12% | Reported (rate TBD) |
| Dysesthesia | Rare / not significant | 20.9% |
| Heart rate increase | Mild | Dose-dependent increases observed |
Sources: Zepbound prescribing information; TRIUMPH-4 topline data (Eli Lilly, Dec 2025)
Discontinuation rates
In TRIUMPH-4, 18.2% of patients on the 12 mg retatrutide dose discontinued due to adverse events, compared to 4.0% on placebo. Interestingly, Lilly noted that some discontinuations were due to “perceived excessive weight loss” in patients with lower BMI.
In pooled Zepbound weight-reduction studies, discontinuation due to gastrointestinal adverse reactions was 1.9% at 5 mg, 3.3% at 10 mg, and 4.3% at 15 mg, versus 0.5% with placebo.4
Serious side effects and contraindications
Tirzepatide carries an FDA boxed warning about thyroid C-cell tumors based on rodent findings.4 Retatrutide does not have an FDA-approved label or boxed warning because it is not approved. Both drug classes have been associated with rare cases of pancreatitis and gallbladder issues.
The main safety difference: tirzepatide has a well-characterized safety profile from years of commercial use. Retatrutide's full picture will emerge as more Phase 3 data comes in throughout 2026.
⚠️ Warning About “Research” Retatrutide Sold Online
We need to address this directly because it's a real safety issue affecting real people.
You may have seen websites selling “retatrutide” as a “research peptide” or “for research use only.” This is not the same thing as the drug being studied in Eli Lilly's clinical trials. These products are unregulated, unverified, and potentially dangerous.

If you want retatrutide before approval, the legitimate route is a clinical trial.
Eli Lilly has stated that retatrutide is investigational and available only through their sponsored clinical trials.3 The FDA has issued warnings about unapproved products containing GLP-1 receptor agonists being sold online, noting risks including wrong dosages, contamination, and dangerous interactions.8
The version of “retatrutide” being sold online may not contain what it claims, may not be dosed correctly, and has not been manufactured under the quality controls required for human use. If you're genuinely interested in accessing retatrutide before FDA approval, the only legitimate path is through a clinical trial. You can search active trials at ClinicalTrials.gov.
Retatrutide vs Tirzepatide Dosage Chart
One of the most searched topics around this comparison is dosing. Here's what you need to know.
Tirzepatide (FDA-approved dosing)
Tirzepatide follows a well-established titration schedule from its FDA-approved prescribing information:4
| Phase | Dose | Notes |
|---|---|---|
| Starting | 2.5 mg once weekly | Starting dose — not a maintenance dose |
| First increase | 5 mg once weekly | After at least 4 weeks at 2.5 mg |
| Further increases | +2.5 mg increments | After at least 4 weeks on each dose |
| Maintenance (weight) | 5, 10, or 15 mg | Based on response and tolerability |
| Maintenance (OSA) | 10 mg or 15 mg | Per the sleep apnea indication |
| Maximum | 15 mg once weekly | FDA-approved maximum |
Retatrutide (trial protocol dosing — NOT an approved schedule)
There is no FDA-approved retatrutide dosing guide. The following reflects the titration used in Phase 3 clinical trials only — this is informational context, not consumer guidance:5
| Weeks | 9 mg Target Arm | 12 mg Target Arm |
|---|---|---|
| Weeks 1–4 | 2 mg | 2 mg |
| Weeks 5–8 | 4 mg | 4 mg |
| Weeks 9–12 | 6 mg | 6 mg |
| Weeks 13–16 | 9 mg (maintenance) | 9 mg |
| Weeks 17+ | — | 12 mg (maintenance) |
⚠️ No approved consumer dosing exists for retatrutide. These are trial schedules for informational context only.
Can you switch from tirzepatide to retatrutide?
Not right now, because retatrutide isn't available by prescription. When it's eventually approved, switching protocols will be part of the clinical guidance and labeling. Since both drugs target overlapping pathways, switching would almost certainly require starting retatrutide's titration from the beginning rather than picking up mid-dose.
What if tirzepatide stopped working for you?
This is a common reason people start searching “retatrutide vs tirzepatide.” You were losing weight, the progress slowed, and now you're wondering if the next drug would be better.
First, some context: weight loss plateaus on GLP-1 medications are common and don't necessarily mean the drug stopped working. Before assuming you need retatrutide, work through this checklist with your provider:
- Are you on the maximum tolerated dose? Many patients are maintained at 10 mg when 15 mg might restart progress.
- Is your protein intake adequate? Aim for 1.0–1.2 grams per kilogram of body weight daily.
- Are you resistance training? Even 2–3 resistance sessions per week make a meaningful difference.
- How's your sleep? Poor sleep directly affects hunger hormones and can undermine the appetite suppression these drugs provide.
- Is there a medication interaction? Certain antidepressants, steroids, and beta-blockers can promote weight retention.
- Have expectations been recalibrated? A 20% weight loss from your starting weight is an exceptional medical outcome.
Retatrutide vs Tirzepatide Cost and Access
Tirzepatide cost today
Zepbound pricing varies depending on insurance, format, and savings programs. Lilly's official self-pay options start at approximately $299/month for some Zepbound presentations, while the standard list price is higher.6 What you actually pay depends heavily on your situation — many commercial insurance plans now cover Zepbound or Mounjaro, though prior authorization is often required. Eli Lilly offers savings programs for eligible commercially insured patients, potentially reducing medication cost to as low as $25/month.
Retatrutide cost (not yet established)
Retatrutide has no official retail price because it is not commercially available.3 If you're enrolled in a clinical trial, the medication is provided at no cost.
The access reality
| Access Path | What It Is | Cost | Insurance? | Legal? |
|---|---|---|---|---|
| Tirzepatide (insurance) | Prescription via your plan | Varies ($0–$300+ copay) | Yes | Yes ✓ |
| Tirzepatide (self-pay/savings) | Lilly savings or cash | From ~$299/mo6 | No | Yes ✓ |
| Tirzepatide (telehealth) | Online provider prescription | Varies by provider | Sometimes | Yes ✓ |
| Retatrutide (clinical trial) | Eli Lilly-sponsored trial | $0 (medication provided) | N/A | Yes ✓ |
| Retatrutide (online sellers) | Unregulated “research” product | Varies | No | No — unsafe ✗ |
Your next step doesn't have to be complicated. We've already done the provider comparison work. See which tirzepatide program fits your budget and goals:
Dozens of providers exist. Most people overpay or get the wrong fit. Our 60-second path finder fixes that.
When Will Retatrutide Be Available?
Here's the realistic timeline based on what Eli Lilly has publicly communicated and what we know about the FDA process.
Where retatrutide stands today (March 2026):
- Phase 3 clinical trials are running across three programs: TRIUMPH (obesity, 4+ trials, 5,800+ participants), TRANSCEND (type 2 diabetes, 3 trials, 2,050+ participants), and SYNERGY (MASH liver disease)
- Two Phase 3 trials have reported positive topline results: TRIUMPH-4 (Dec 2025) and TRANSCEND-T2D-1 (March 2026)
- Lilly says additional Phase 3 results are expected over the next year5
- Eli Lilly has not submitted a New Drug Application (NDA) to the FDA
- TRIUMPH-5 (head-to-head vs. tirzepatide) is ongoing
- No FDA approval date has been announced3
Waiting might make sense if you're specifically interested in trial participation or if your medical situation is uniquely suited to retatrutide's triple-agonist profile. For most people, starting a proven treatment now and switching later if something better becomes available is the more practical path.
Retatrutide vs Tirzepatide Muscle Loss: What Do We Actually Know?
This is the #1 concern we see in search data for this comparison, and it deserves a direct answer.
All significant weight loss — from any cause — involves some loss of lean mass alongside fat mass. This is true for medication, surgery, dieting, and even illness-related weight loss. It is a physiological reality, not a medication-specific failure.
What the data shows
For tirzepatide, the SURMOUNT trials included body composition measurements. The majority of weight lost was fat mass, but lean mass declined as well — consistent with what's seen across all obesity interventions at this magnitude.
For retatrutide, Phase 2 data indicated that the proportion of lean mass loss relative to total weight loss was similar to other GLP-1 class medications. The glucagon receptor component theoretically promotes fat oxidation, which has led to speculation that retatrutide might preserve more muscle. But this hasn't been demonstrated in a head-to-head comparison.
The honest bottom line on muscle loss
Neither drug has been proven to “spare muscle” better than the other. The best evidence for mitigating muscle loss during GLP-1 treatment comes from lifestyle factors, not drug selection:
- Protein intake: Aim for roughly 1.0–1.2 grams per kilogram of body weight daily
- Resistance training: Even 2–3 sessions per week significantly reduces lean mass loss
- Adequate hydration and sleep
- Working with a provider who monitors body composition, not just the scale
Which Is Better for Diabetes, Sleep Apnea, and Metabolic Health?
For type 2 diabetes
Tirzepatide is FDA-approved for type 2 diabetes as Mounjaro. The SURPASS clinical program demonstrated significant A1C reduction and weight loss in diabetic populations. This is a proven, prescribable, insurance-covered option for diabetes management today.
Retatrutide just posted strong Phase 3 diabetes data (TRANSCEND-T2D-1, March 2026): A1C reduction up to 2.0% and weight loss up to 16.8% at 40 weeks. But retatrutide isn't approved for diabetes or anything else.
Today's answer: Mounjaro (tirzepatide) is the clear choice for diabetes because it's approved, available, and effective.
For sleep apnea
Tirzepatide (Zepbound) received FDA approval for moderate-to-severe obstructive sleep apnea in 202413 — making it the first medication approved for this indication. Retatrutide is being studied for OSA in the TRIUMPH program, but no results have been released yet.
If you have obesity with sleep apnea, tirzepatide currently has no peer in this space.
For liver health (MASH/MASLD)
This is where retatrutide's third pathway gets particularly interesting. The glucagon receptor plays a role in liver metabolism, and Lilly is running the SYNERGY trial program specifically evaluating retatrutide for metabolic dysfunction-associated steatohepatitis (MASH). Note that this space has evolved — the FDA approved Rezdiffra (resmetirom) in 2024 for adults with noncirrhotic MASH with moderate to advanced fibrosis9 — but retatrutide's triple-agonist mechanism could still offer a differentiated approach. That's future potential, not current reality.
For cardiovascular and metabolic markers
Both drugs improve cardiovascular risk factors. TRIUMPH-4 showed retatrutide produced clinically meaningful improvements in non-HDL cholesterol, hs-CRP (a marker of inflammation), triglycerides, and systolic blood pressure. Tirzepatide's SURMOUNT trials showed comparable metabolic benefits. Weight loss itself is the primary driver of these improvements.
Does Retatrutide or Tirzepatide Better Reduce Inflammation?
Both medications reduce markers of systemic inflammation — and the primary reason is straightforward: significant weight loss improves inflammation. Excess body fat, especially visceral fat, is metabolically active and produces inflammatory compounds. When you lose 20–28% of your body weight, that inflammatory load drops substantially. Both drugs accomplish this.
Retatrutide is being studied for potential liver-related advantages through its glucagon receptor pathway, and early data shows improvements in hs-CRP, non-HDL cholesterol, and triglycerides.2 Tirzepatide has shown comparable metabolic improvements in its own trials.1 But superiority for inflammation has not been established in a head-to-head trial.
The honest answer: Both drugs can improve inflammatory markers, largely through weight loss and metabolic improvement. If your concern is specifically liver-related inflammation, retatrutide's mechanism is more targeted — but the SYNERGY trial data hasn't been released yet. For general metabolic inflammation, the best intervention available today is the weight loss itself — and tirzepatide can deliver that right now.
How Does Semaglutide (Ozempic/Wegovy) Compare to Both?
Semaglutide was the drug that started the modern GLP-1 weight loss revolution. Sold as Ozempic for type 2 diabetes and Wegovy for weight loss (both by Novo Nordisk), it targets a single receptor (GLP-1) and produces roughly 15–17% weight loss at the highest dose.
Tirzepatide added a second receptor (GIP) and pushed weight loss to ~20%. Retatrutide added a third (glucagon) and pushed the number higher still.
| Semaglutide | Tirzepatide | Retatrutide | |
|---|---|---|---|
| Brand Names | Ozempic (T2D), Wegovy (obesity) | Mounjaro (T2D), Zepbound (obesity) | None (investigational) |
| Manufacturer | Novo Nordisk | Eli Lilly | Eli Lilly |
| Mechanism | Single agonist (GLP-1) | Dual agonist (GLP-1 + GIP) | Triple agonist (GLP-1 + GIP + Glucagon) |
| Best Weight Loss | ~15–17% (STEP trials) | ~20.9% (SURMOUNT-1) | ~28.7% (TRIUMPH-4) |
| FDA Status | Approved | Approved | Phase 3 trials |
| Head-to-Head Result | Lost to tirzepatide in SURMOUNT-5 | Beat semaglutide by 47% | vs tirzepatide: TRIUMPH-5 ongoing |
| Monthly Cost (list) | ~$1,350 (Wegovy) | ~$1,059 (Zepbound) | Not priced |
For someone starting fresh, tirzepatide generally offers the strongest combination of proven efficacy and current availability. If cost is the primary concern and your insurance covers Wegovy but not Zepbound, semaglutide remains a highly effective option. See our full comparison: Best Semaglutide Online: Legit Providers, Real Prices (2026)
Who Should NOT Take These Medications?
Contraindications (do not use)
- Personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2). This is a boxed warning on tirzepatide's FDA label.4
- Known serious hypersensitivity to tirzepatide or any component of the formulation. Anaphylaxis and angioedema have been reported.
Special populations (requires clinician guidance)
- Pregnancy or plans to become pregnant. Tirzepatide is not recommended during pregnancy; discontinue if pregnancy is recognized.4
- History of pancreatitis. Both GLP-1 class drugs have been associated with rare cases of acute pancreatitis.
- Gallbladder disease. Rapid weight loss from any cause increases the risk of gallstones and gallbladder inflammation.
- Severe GI disorders including gastroparesis — GLP-1 drugs further slow gastric emptying.
- Low baseline muscle mass or frailty (particularly relevant for older adults).
- Type 1 diabetes — these drugs are not indicated for type 1 diabetes.
Retatrutide-specific cautions
- Existing neuropathy or nerve sensitivity conditions — the dysesthesia signal (20.9% at 12 mg) may be particularly problematic.
- Heart rhythm concerns — dose-dependent heart rate increases have been observed in retatrutide studies.
- Comfort with uncertainty — if knowing the full long-term safety profile matters to you, tirzepatide's established track record may be more appropriate.
For a comprehensive look at absolute disqualifiers, see our guide: GLP-1 Absolute Disqualifiers: Hard Contraindications
What's Coming Next in Weight Loss Medications? (2026–2027)
Understanding where retatrutide and tirzepatide fit in the broader landscape helps put this comparison in context.
Retatrutide remaining Phase 3 data
Seven more Phase 3 readouts are expected in 2026, including TRIUMPH-1 (the pivotal obesity trial, 80 weeks, which analysts believe could show 30%+ weight loss), TRIUMPH-2 (obesity with sleep apnea), and additional TRANSCEND diabetes studies. These will define whether retatrutide's profile holds across diverse populations.
The broader GLP-1 pipeline
- Orforglipron (Eli Lilly): An oral GLP-1 receptor agonist — a pill version of GLP-1 treatment. Lilly has submitted the NDA to the FDA, with a decision expected in 2026. Could be a game-changer for people who dislike injections.
- Survodutide (Boehringer Ingelheim/Zealand): Another dual agonist (GLP-1 + glucagon) in late-stage trials for obesity and MASH liver disease.
- Amycretin (Novo Nordisk): An oral GLP-1/amylin dual agonist with impressive early results.
- CagriSema (Novo Nordisk): Fixed-ratio combination of semaglutide and cagrilintide. Phase 3 data showed ~22.7% weight loss.
Our advice: make the best decision you can with what's available now. Don't let the promise of a future drug keep you from starting a proven treatment today. You can always switch if something better becomes available and is right for your situation.
What Most Comparison Sites Get Wrong
We're going to be transparent about what frustrates us about the current search results for this query, because it's part of why we built this page.
They compare these drugs as if both are buyable options. Most comparison articles list retatrutide and tirzepatide side by side as if you're choosing between two items on a menu. You're not. One is a prescription medication. The other is a research compound in clinical trials. That distinction changes everything about the decision framework.
They skip the dysesthesia signal. A side effect that affects ~1 in 5 people at the highest dose is not a footnote. It's information you need to make an informed decision. Sites that minimize it are either outdated or prioritizing enthusiasm over accuracy.
They use misleading cross-trial comparisons. Calling retatrutide “better” because 28.7% > 20.9% without explaining that these numbers come from different trials with different populations is misleading. We've been careful to flag this above, and we'll continue updating when TRIUMPH-5 head-to-head results arrive.
They ignore the gray-market safety problem. Real people are buying unverified “retatrutide” from unregulated online sellers. A responsible comparison page addresses this.
They're usually not current. Most competitor pages still primarily cite Phase 2 data. We built this page around March 2026 Phase 3 results because that's what's actually current.
How We Compared Retatrutide and Tirzepatide
Transparency matters on a health topic. Here's exactly how we built this comparison.
Primary sources used: FDA prescribing information for Zepbound and Mounjaro; published trial data in the New England Journal of Medicine (SURMOUNT-1, Phase 2 retatrutide, SURMOUNT-5) and JAMA (SURMOUNT-4); Eli Lilly investor press releases for topline Phase 3 data (TRIUMPH-4, TRANSCEND-T2D-1); ClinicalTrials.gov for trial registry information; FDA safety communications on unapproved GLP-1 products.
What we did NOT do: We did not rely on clinic marketing pages, medspa blogs, or social media claims. We did not treat data from different trials as directly comparable without flagging limitations. We did not imply expert medical review beyond what we've actually done.
Evidence labeling: Throughout this page, we've distinguished between FDA-labeled data, published peer-reviewed trial data, and company topline releases. These are different levels of evidence and are treated as such.
Affiliate disclosure: Our provider matching tools may generate revenue through affiliate partnerships. This never influences our clinical analysis or drug comparisons.
Read our full research methodology.
Frequently Asked Questions
Is retatrutide FDA-approved?
No. As of March 2026, retatrutide is investigational and only available through Eli Lilly-sponsored clinical trials. No New Drug Application has been submitted to the FDA.
Is retatrutide better than tirzepatide for weight loss?
In indirect comparisons from separate trials, retatrutide has shown higher average weight loss (28.7% vs 20.9%). But these numbers come from different studies with different populations and conditions. The ongoing TRIUMPH-5 head-to-head trial will provide the first direct comparison. Until then, 'better' depends on the full picture: availability, safety data, access, and individual goals.
Which one has more side effects?
Retatrutide shows higher rates of GI side effects (nausea, diarrhea, vomiting) in trials and a unique side effect — dysesthesia (abnormal skin sensations) — reported in up to 20.9% of patients at the highest dose. Tirzepatide's side effect profile is well-established and generally milder.
Can I buy retatrutide online?
Products sold as 'retatrutide' online are unregulated and not the same as the drug studied in clinical trials. The FDA has warned consumers about unapproved GLP-1 products. The only legitimate way to access retatrutide is through a Lilly-sponsored clinical trial.
How much will retatrutide cost when it's approved?
No official price exists because retatrutide is not commercially available. Pricing will depend on Eli Lilly's market strategy, insurance negotiations, and the competitive landscape at the time of approval.
Can I switch from tirzepatide to retatrutide?
Not currently, since retatrutide isn't available by prescription. If approved, switching protocols will be part of clinical guidance. If you've plateaued on tirzepatide, talk to your doctor about dose optimization and lifestyle factors first.
Does retatrutide cause more muscle loss?
Current data doesn't show a meaningful difference in lean mass preservation between the two drugs. Both produce significant weight loss that includes some lean mass. Protein intake and resistance training are the most important factors for preserving muscle regardless of which medication you use.
What is dysesthesia?
An abnormal skin sensation — tingling, tenderness, or heightened sensitivity to touch. It was reported in up to 20.9% of retatrutide patients at the 12 mg dose in Phase 3 trials. It's generally mild and rarely led to treatment discontinuation, but it's a new signal being closely monitored.
What is the difference between retatrutide and Zepbound?
Zepbound is the brand name for tirzepatide (FDA-approved for weight loss). Retatrutide is a different, investigational drug. Both are made by Eli Lilly, both are weekly injections, but retatrutide targets three hormonal pathways (GLP-1, GIP, and glucagon) while tirzepatide targets two (GLP-1 and GIP).
What is the difference between retatrutide and Mounjaro?
Mounjaro is tirzepatide (FDA-approved for type 2 diabetes). Retatrutide is a separate, unapproved investigational drug with a different mechanism (triple vs dual agonist). Both are made by Eli Lilly.
Is there a retatrutide vs tirzepatide dosage chart?
Tirzepatide has an FDA-approved dosing schedule (2.5 mg up to 15 mg weekly). Retatrutide has trial protocols (2 mg up to 12 mg weekly), but no consumer-facing approved dosage chart exists. No approved consumer dosing exists for retatrutide — trial schedules are for informational context only.
Which is better for type 2 diabetes?
Tirzepatide (Mounjaro) is FDA-approved for type 2 diabetes and is the clear choice today. Retatrutide showed strong Phase 3 diabetes results in TRANSCEND-T2D-1 (March 2026), but isn't approved for this or any indication.
Should I wait for retatrutide or start tirzepatide now?
For most people, starting tirzepatide now is the better decision. It's available, proven, and effective. Retatrutide still has to complete Phase 3 development and FDA review before it reaches pharmacies. Waiting means leaving months or years of potential progress on the table. You can always switch if retatrutide is approved and proves to be a better fit.
How do I find a legitimate retatrutide clinical trial?
Search ClinicalTrials.gov for 'retatrutide' and filter by recruiting status and location. The TRIUMPH and TRANSCEND programs have trial sites across the U.S. and internationally.
Are retatrutide and tirzepatide made by the same company?
Yes. Both are developed and manufactured by Eli Lilly and Company.
Is retatrutide the same as semaglutide?
No. Semaglutide (Ozempic/Wegovy) is a single-receptor GLP-1 agonist made by Novo Nordisk. Retatrutide is a triple-receptor agonist made by Eli Lilly. Different drugs, different companies, different mechanisms.
What should I do if tirzepatide stopped working?
Plateaus are common with any weight loss treatment. Before assuming you need a different drug, work with your doctor on: dose optimization (are you on the maximum tolerated dose?), protein intake and resistance training, sleep quality, and medication adherence. If those are optimized and you're still stuck, your provider may have additional strategies.
When will the retatrutide vs tirzepatide head-to-head trial results be available?
TRIUMPH-5 (NCT06662383) is an ongoing 89-week trial comparing retatrutide directly to tirzepatide in 800 adults with obesity. Results are expected in 2027. This page will be updated when they are released.
What does 'triple agonist' mean?
An agonist is a molecule that activates a receptor. A 'single agonist' like semaglutide activates one receptor (GLP-1). A 'dual agonist' like tirzepatide activates two (GLP-1 + GIP). A 'triple agonist' like retatrutide activates three (GLP-1 + GIP + glucagon). More receptors means broader physiological effect — more hunger suppression, metabolic improvement, and fat burning — but also more complexity and potential side effects.
Is retatrutide safe long-term?
We don't know yet. Retatrutide's longest published trial data is from 68-week (TRIUMPH-4) and 48-week (Phase 2) studies. Long-term safety over years of use — the kind of data tirzepatide now has — simply doesn't exist for retatrutide. The ongoing Phase 3 program will provide more data, but true long-term safety understanding comes from years of real-world prescribing experience.
The Bottom Line
If you came here wondering whether to start tirzepatide now or hold out for retatrutide, here's our take:
Start now. Not because tirzepatide is the final word in weight loss medication — it's not. But because waiting for a drug that still needs to complete Phase 3 development, regulatory review, and commercial launch — and that may or may not be covered by your insurance, and may or may not work better for you personally — is a gamble you don't need to make. Tirzepatide is an extraordinarily effective medication that's available today.
Retatrutide is worth following closely. The Phase 3 data is remarkable. The triple-agonist mechanism is genuinely novel. The TRIUMPH-5 head-to-head results will be one of the most important data readouts in obesity medicine when they arrive. But “worth following” is different from “worth waiting for,” especially when a proven option is already available.
We'll keep this page updated as new data emerges — and in 2026, there's a lot coming. Seven more Phase 3 retatrutide readouts are expected this year. The comparison is evolving, and we'll evolve with it.
Still not sure which GLP-1 program is right for you? Tell us your goals, insurance situation, and timeline — we'll match you with the specific providers and programs that fit. Over 10,000 people have used this to skip the confusion and start the right path.
Find My GLP-1 Path — 60 Seconds, Free, No Signup →You just read the most detailed retatrutide vs tirzepatide comparison available. Now make it personal.