FDA-Approved OSA Treatment · Pricing Verified April 4, 2026
Zepbound for Sleep Apnea: Does It Work, Who Qualifies, and How Do You Get It?
Written by The RX Index Editorial Team · Last verified April 4, 2026
Pricing checked · FDA status verified · Trial data verified · Our methodology
Disclosure: Some links on this page are affiliate links. If you check eligibility or purchase through these links, we may earn a commission at no extra cost to you. Our recommendation would be the same without those links — the data drives the verdict, not the commissions.
Bottom Line
Zepbound for sleep apnea is the first FDA-approved medication for moderate-to-severe obstructive sleep apnea in adults with obesity — and the clinical data behind it is unusually strong. In the SURMOUNT-OSA trials, participants averaged 25–29 fewer breathing interruptions per hour over one year. 42% of people not using CPAP reached remission or mild OSA with no symptoms. In a separate trial of participants already on PAP therapy, that figure was 50%.
If you have documented moderate-to-severe sleep apnea and a BMI of 30+, this page walks you through every real question: does it actually work, who qualifies, what it costs by insurance type, and exactly how to get started.

Which of these sounds like you?
| Your situation | Your best next step | Why this route |
|---|---|---|
| Diagnosed OSA + BMI 30+ + commercial insurance | Check Zepbound eligibility on Ro | Ro evaluates you online, can prescribe FDA-approved Zepbound, and helps navigate commercial insurance prior authorization |
| Diagnosed OSA + BMI 30+ + Medicare Part D | Talk to your doctor or sleep specialist | Medicare OSA coverage goes through Part D — Ro currently cannot coordinate government insurance for GLP-1s |
| Diagnosed OSA + BMI 30+ + no insurance / cash pay | Compare Ro vs. LillyDirect pricing | Both offer Zepbound KwikPen starting at $299 for the 2.5mg dose — Ro adds clinical support, LillyDirect is medication-only |
| Suspect OSA but no sleep study yet | Get tested first, then come back | A documented sleep study is the foundation for the FDA indication, insurance coverage, and knowing whether this is your move |
Have commercial insurance or paying cash?
Check If You Qualify for FDA-Approved Zepbound on Ro →Ro's medical team can evaluate your diagnosis online and help with commercial insurance paperwork. Takes about 10 minutes.
On Medicare? Your strongest path is through your doctor or sleep specialist, who can submit prior authorization directly to your Part D plan. Read our Medicare GLP-1 coverage guide →
Is Zepbound actually FDA-approved for sleep apnea?
Yes — and it's the only medication with this approval. The FDA approved Zepbound on December 20, 2024 for moderate-to-severe obstructive sleep apnea in adults with obesity. No other GLP-1 medication — not Ozempic, not Wegovy, not any compounded version — has this indication.
That distinction matters more than most people realize. It's not just a medical footnote. It's the reason insurance companies are building coverage pathways for Zepbound that don't exist for other weight loss drugs. It's why Medicare Part D may cover it. And it's why your doctor can submit a prior authorization specifically framed around OSA treatment — a framing that changes the entire coverage conversation.
Before this approval, obstructive sleep apnea had exactly zero FDA-approved medications. The standard of care was CPAP, oral appliances, surgery, or lifestyle changes. For millions of people whose sleep apnea is driven by excess weight around the airway, those options managed the symptom without addressing what was actually causing it.
Zepbound changes that equation. It's a dual GIP and GLP-1 receptor agonist — meaning it activates two hormone pathways that regulate appetite, blood sugar, and metabolism. The result is significant, sustained weight loss that reduces the fat pressing on your upper airway while you sleep.
We want to be precise here because other pages aren't: Zepbound is approved for moderate-to-severe OSA (that's an AHI of 15 or higher on a sleep study) in adults with obesity (BMI of 30 or higher). It's meant to be used alongside a reduced-calorie diet and increased physical activity — not as a standalone “do nothing and wait” solution.
It is not approved for mild sleep apnea. It is not approved for central sleep apnea. And compounded tirzepatide — no matter who sells it — is not Zepbound and does not carry this FDA approval.
Does Zepbound actually work for sleep apnea? (The clinical evidence)
It works — and the numbers are hard to argue with. The SURMOUNT-OSA phase 3 trials studied 469 adults with moderate-to-severe obstructive sleep apnea and obesity over 52 weeks. The results were strong enough that Zepbound received Fast Track, Priority Review, and Breakthrough Therapy designations from the FDA.
Here's what the data showed across two separate studies:
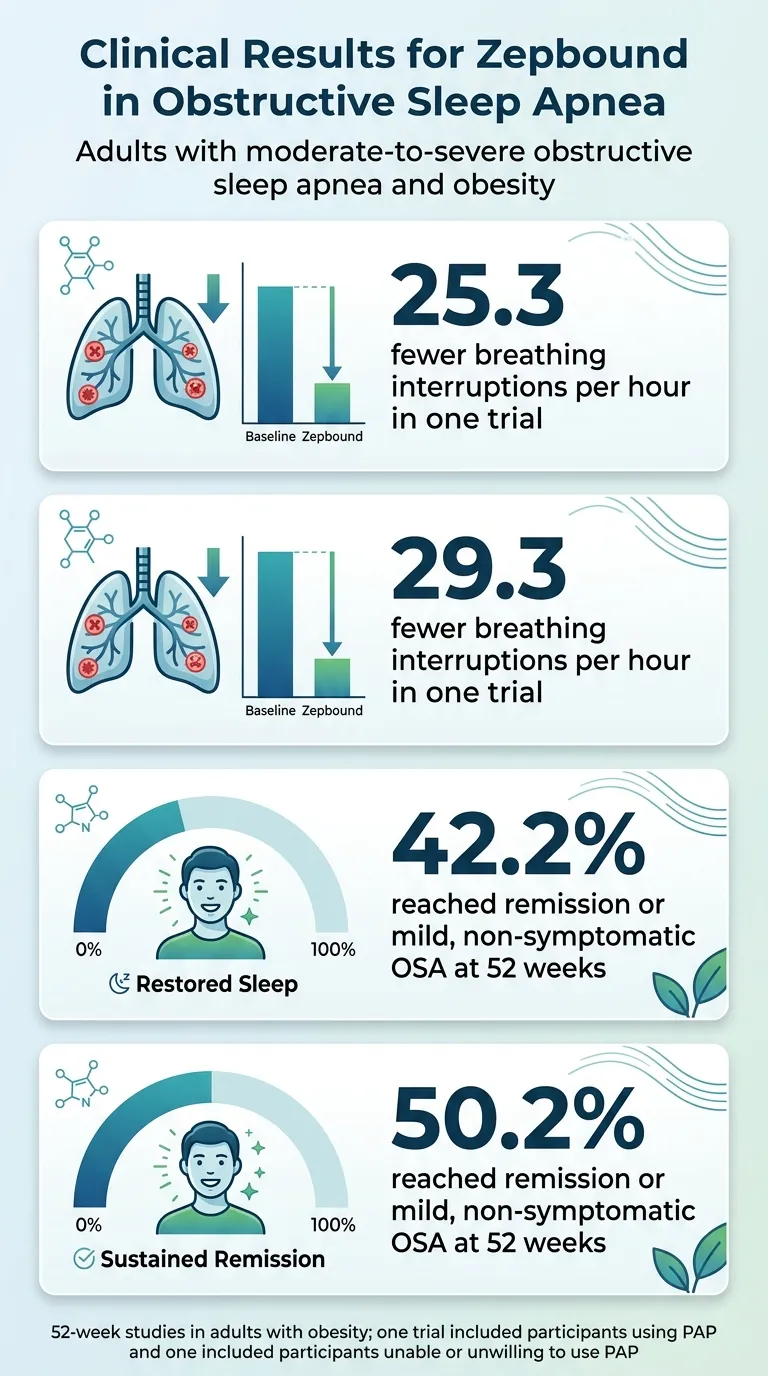
| Outcome | Study 1: Zepbound without PAP | Study 2: Zepbound with PAP | Placebo |
|---|---|---|---|
| Breathing interruptions reduced per hour (AHI) | −25.3/hr | −29.3/hr | −5.3 to −5.5/hr |
| Reached remission or mild OSA (no symptoms) | 42.2% | 50.2% | 14–16% |
| Average body weight loss | 18% (~45 lbs) | 20% (~50 lbs) | ~2% (~4–6 lbs) |
| Hypoxic burden improvement (oxygen levels) | −95.2% min/hr | −103.0% min/hr | −25.1 to −41.7% min/hr |
Source: SURMOUNT-OSA trials, Eli Lilly / FDA. These were two separate studies — not a head-to-head comparison. Study 1 enrolled people not using PAP. Study 2 enrolled people already using PAP. Baseline average AHI was 46–53 events/hour (severe). Baseline average BMI was ~39.
Let's translate that. Participants started with severe sleep apnea — averaging roughly 50 breathing interruptions per hour. After a year on Zepbound, that dropped by about 25 events per hour. In Study 1, 42% of people not using CPAP saw their sleep apnea either resolve entirely or drop to a level that no longer produced symptoms.
Those aren't marginal improvements. In sleep medicine, reducing AHI by 25 per hour is the kind of change that can move someone from “severe, symptomatic, and exhausted” to “mild or resolved.”
In the separate study of participants already on PAP therapy, 50.2% reached remission or mild non-symptomatic OSA, and weight loss averaged 50 pounds. Because these were separate study populations and not a direct head-to-head test, we can't say definitively that adding CPAP “boosted” the numbers — but the results are consistent with combined treatment being a reasonable approach.
What “remission” actually means
The trials defined remission or disease resolution as an AHI below 5 per hour, or an AHI between 5 and 14 with normal daytime sleepiness scores. In practical terms: the person's breathing normalized enough during sleep that they no longer met diagnostic criteria for clinically significant sleep apnea.
When results start showing up
Improvements built progressively over the 52-week treatment period, with the biggest gains after participants reached their maintenance dose and sustained weight loss. Some data suggests measurable changes may begin around 20 weeks, but this isn't a quick fix. It's a sustained biological shift that takes months to fully express.
What the data does NOT prove
We need to say this plainly: Zepbound did not produce remission in everyone. 58% of the non-PAP group and 50% of the PAP group did not reach remission at the one-year mark. If your sleep apnea is driven heavily by anatomical factors — a narrow jaw, large tonsils, a naturally narrow airway — weight loss may improve your numbers without fully resolving the condition.
But here's the reframe that matters: even “partial” improvement — going from severe to moderate, or moderate to mild — can meaningfully improve sleep quality, daytime energy, and long-term cardiovascular risk. You don't need full remission for Zepbound to change your life.
Can Zepbound replace your CPAP?
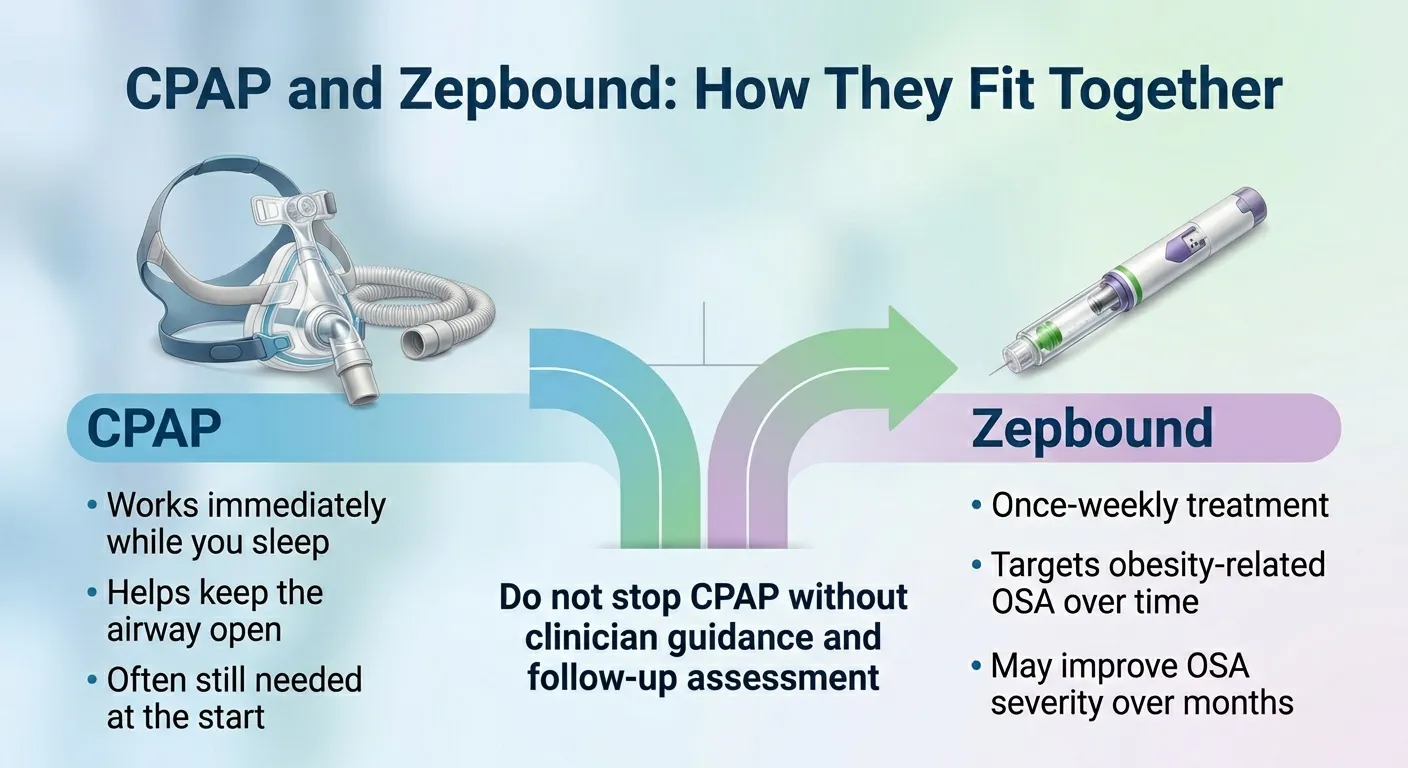
For some people, eventually — but not tonight. This is the question everyone wants answered, and the honest answer has layers.
Here's what the American Academy of Sleep Medicine says: the goal of tirzepatide therapy is not to replace traditional OSA treatments like CPAP. It's an additional tool that targets excess weight as a root cause. Some patients may do best with combined treatment, and any decision to reduce or stop CPAP should follow a repeat sleep study and a clinician's guidance.
The data supports that framing. In the study of participants already using PAP, 50.2% reached remission — the strongest remission rate across both trials. And in a recent survey, 88% of sleep medicine doctors supported combination therapy.
The practical framework
Think of it this way: CPAP protects you tonight. Zepbound changes the trajectory over the next 12 months.
Use CPAP to keep your airway open and your oxygen levels stable while Zepbound works in the background — reducing appetite, driving weight loss, shrinking the tissue that's collapsing your airway during sleep. After six to twelve months of treatment and significant weight loss, get a follow-up sleep study. Let the data tell you whether your AHI has dropped enough to safely step down from CPAP.
When CPAP is still the better first answer
If you have severe OSA with high cardiovascular risk, CPAP is non-negotiable while you're losing weight. CPAP works from night one. Zepbound takes months. For immediate airway protection, especially if your oxygen levels are dropping dangerously low during sleep, there is no substitute for positive airway pressure.
When Zepbound makes sense without CPAP
If you can't tolerate CPAP — and roughly 30–50% of people struggle with adherence — Zepbound offers a real treatment path where one barely existed before. Study 1 specifically enrolled people “unable or unwilling to use PAP,” and 42% of that group reached remission. That's not permission to skip CPAP carelessly, but it is evidence that Zepbound alone can produce meaningful results for people who've tried and failed with masks and machines.
How this compares to your other options
| Treatment | How it works | Time to results | AHI improvement | Cost | Best for |
|---|---|---|---|---|---|
| Zepbound | Weight loss reduces airway fat and obstruction | Months (gradual) | −25 to −29 events/hr | $25–$499/fill | OSA driven by obesity, CPAP-intolerant |
| CPAP | Pressurized air holds airway open | Night 1 | Near-complete when used | $50–$150/mo | All severity levels, immediate protection |
| Inspire | Implanted nerve stimulator moves tongue | Weeks post-surgery | Significant for select patients | $30K–$40K | Moderate-severe, CPAP-intolerant, BMI <40 |
| Oral appliance | Repositions jaw to widen airway | Days to weeks | Mild-to-moderate improvement | $1,500–$3,000 | Mild-moderate OSA, primary snoring |
| Zepbound + CPAP | Combined: root-cause + immediate protection | Night 1 + months | −29/hr, 50% remission | Combined costs | Severe OSA, strongest outcome |
We built this table because it doesn't exist on any other page covering this topic. You shouldn't have to visit five sites to compare your options.
If you've been fighting with your CPAP mask and wondering if there's another path — this is it. The data is real, the FDA approval is real, and checking whether you're a fit takes minutes, not weeks.
See If You Qualify for Zepbound — Check Eligibility on Ro →Who qualifies for Zepbound for sleep apnea?
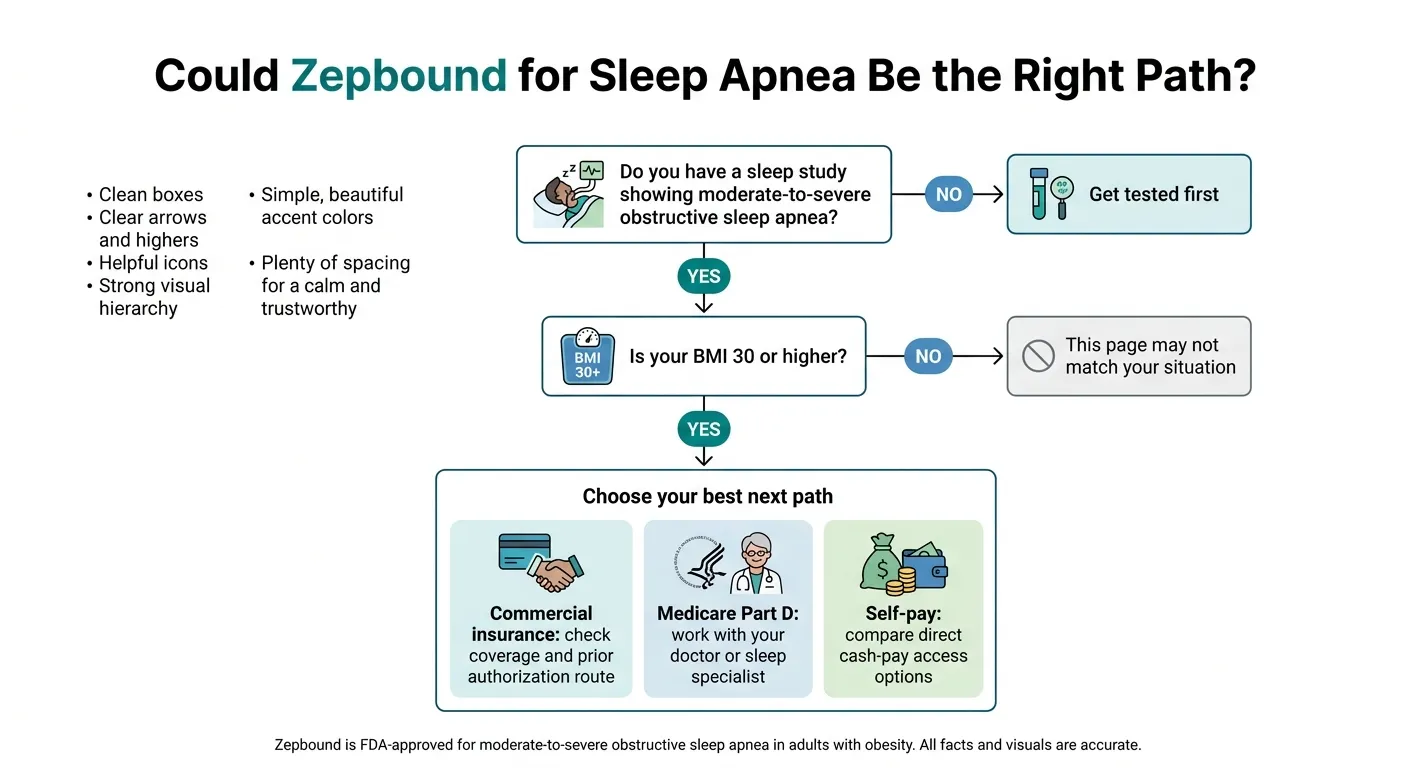
You need two things: a documented sleep apnea diagnosis and obesity. More specifically, the FDA approval covers adults with moderate-to-severe obstructive sleep apnea (AHI of 15 or higher) and a BMI of 30 or above.
That's not a suggestion — it's the label. Insurance companies, Medicare, and prescribers all work from these criteria. Here's what each piece means:
The sleep study requirement
You need a polysomnography (PSG) or home sleep apnea test (HSAT) showing moderate-to-severe OSA. This is non-negotiable for insurance coverage and, frankly, for knowing whether Zepbound is the right tool.
- Moderate OSA: AHI of 15–30 events per hour
- Severe OSA: AHI above 30 events per hour
If you suspect you have sleep apnea but haven't been formally tested, that's your first step — not Zepbound. Your doctor can order a sleep study, or a telehealth provider can coordinate a home sleep test in many cases.
The BMI requirement
The FDA label specifies adults with obesity — defined as a BMI of 30 or higher. For the OSA-specific Zepbound path, use BMI 30+ as the working requirement unless a specific plan document says otherwise.
For reference: a 5'10” person weighing 210 pounds has a BMI of about 30. A 5'6” person weighing 186 pounds is in the same range.
Who this is NOT for
We'd rather lose you to the right page than waste your time on the wrong one:
- Mild OSA (AHI under 15): Not covered by this approval. An oral appliance or positional therapy may be more appropriate.
- Central sleep apnea: Zepbound is only approved for obstructive sleep apnea. Different mechanism, different condition.
- Sleep apnea driven primarily by anatomy (narrow jaw, large tonsils, deviated septum) without significant obesity: Weight loss may help, but it may not resolve the structural issue. Talk to a sleep specialist.
- Already taking another GLP-1 or tirzepatide product: You cannot take Zepbound with Mounjaro, Ozempic, Wegovy, or any other GLP-1 receptor agonist.
| Requirement | What it means | Where to find it | If you're missing it |
|---|---|---|---|
| Moderate-to-severe OSA | AHI ≥ 15 on a sleep study | Your sleep study report | Ask your doctor or get a home sleep test |
| Obesity | BMI ≥ 30 | Calculate at any BMI tool online | This is the FDA label threshold for the OSA indication |
| Age 18+ | Adults only | — | — |
| No other GLP-1/tirzepatide use | Can't combine with Mounjaro, Ozempic, etc. | Check your current medications | Discuss switching with your provider |
| Diet + activity commitment | FDA label requires use with lifestyle changes | — | Your provider will discuss this at your visit |
If you checked every box — you're likely a candidate. The next question is what it costs and how to access it.
How much does Zepbound cost for sleep apnea in 2026?
The “$1,000+ per month” narrative is outdated. The single-dose pen list price is about $1,086 per fill, but almost nobody should be paying that. Between manufacturer savings programs, insurance coverage, and the newer KwikPen self-pay option, most people pay significantly less. Here's every realistic price point:
| How you pay | What you'll pay | Who this works for | How to access |
|---|---|---|---|
| Commercial insurance + Lilly Savings Card | As low as $25/fill | Employer or marketplace insurance that covers Zepbound | Doctor submits prior auth → activate savings card at zepbound.lilly.com |
| Commercial insurance does NOT cover + Savings Card | As low as $499/fill | Commercial insurance that excludes Zepbound | Savings card still provides some offset |
| Lilly KwikPen self-pay | $299 (2.5mg) · $399 (5mg) · $449 (7.5–15mg) | No insurance, or insurance that doesn't cover | Order through LillyDirect or Ro |
| Ro (evaluation + prescription + KwikPen) | Lilly self-pay prices + Ro membership ($45 first month, then $145/mo) | Cash-pay or commercially insured readers who want one-stop access | Start evaluation on Ro |
| Medicare Part D | Varies by plan | Medicare enrollees with documented OSA + obesity | Doctor submits Rx for OSA to Part D plan |
| Retail pharmacy, no savings | ~$1,086/fill (list price) | Not recommended — explore the options above first | — |
Lilly's KwikPen self-pay offer pricing ($299/$399/$449) requires meeting specific refill timing rules. Regular self-pay pricing is higher if those terms aren't met. Verify current pricing at zepbound.lilly.com/savings.
A few things worth knowing about these numbers
The Lilly Savings Card is only for commercial insurance. If your plan covers Zepbound for its approved uses, you may pay as little as $25 per fill. Maximum savings of $100 per monthly fill, $1,300 per year. Expires 12/31/2026. Not available to government-insured patients (Medicare, Medicaid, Tricare).
Medicare Part D coverage for Zepbound only applies when prescribed for OSA — not for weight loss alone. Your out-of-pocket cost depends on your specific Part D plan's formulary, tier placement, and cost-sharing structure. The 2026 annual out-of-pocket cap for Part D prescriptions is $2,100. Note: the Medicare GLP-1 Bridge program (launching July 2026) is a separate weight-management demonstration — Zepbound prescribed for OSA goes through regular Part D, not the Bridge.
Ro's pricing includes both the medication cost and a membership fee. Ro matches Lilly's self-pay KwikPen medication pricing, but the membership adds $45 for the first month and $145 per month ongoing (if you remain eligible). What you get for that fee: clinical evaluation, prescription, commercial insurance prior authorization support, and for cash-pay KwikPen orders, direct shipping to your door. If you have commercial insurance and Ro fills through a pharmacy, you'd pick up at a local pharmacy instead.
LillyDirect offers the same KwikPen self-pay medication pricing without a membership fee — but you need to already have a prescription and handle the clinical logistics separately.
The honest take on cost
Zepbound is not cheap. Even on the self-pay KwikPen at $299–$449 per month, it's a real ongoing commitment. And if your commercial insurance doesn't cover it, the savings card offset still leaves you paying $499 per fill for the single-dose pen.
Here's why that cost still makes sense for the right person: untreated moderate-to-severe sleep apnea carries its own price — higher cardiovascular risk, cognitive decline, increased accident risk, and the ongoing cost of CPAP supplies ($50–$150/month). If Zepbound meaningfully improves your sleep apnea while also producing 45–50 pounds of weight loss, the health improvements can be substantial. We're not going to make a blanket claim that “it pays for itself” — that depends on your situation. But the cost of doing nothing isn't zero either.
You've seen the real numbers. No inflated list prices, no vague “call for pricing.” If you have commercial insurance, Ro can tell you exactly what you'd pay after coverage. If you're self-paying, the KwikPen pricing is transparent.
Check Your Zepbound Coverage and Pricing on Ro →Ro shows what you'd actually pay — not the list price that scares everyone off.
Does insurance cover Zepbound for sleep apnea?
More often than you'd expect — and the OSA approval is the key that unlocks it. Before December 2024, getting any GLP-1 covered for weight loss was an uphill battle. The sleep apnea FDA approval changed the game because it gave insurers a clear, non-weight-loss medical indication to build coverage around.
Commercial insurance
Most major insurance companies have coverage pathways for Zepbound, but nearly all require prior authorization. That means your doctor submits a request explaining why Zepbound is medically necessary for your OSA — and the insurer reviews it before approving.
The strongest framing is not “my patient wants a weight loss drug.” It's: “My patient has documented moderate-to-severe obstructive sleep apnea, obesity with a BMI of [X], and Zepbound is the only FDA-approved medication for this indication.”
What insurers typically want to see:
- Sleep study confirming moderate-to-severe OSA (AHI ≥ 15)
- BMI ≥ 30
- Documentation of current or attempted OSA treatment (CPAP trial, oral appliance, etc.)
- Provider notes supporting medical necessity for Zepbound specifically
- Some plans require a “step therapy” history showing prior treatment attempts
The CVS Caremark situation
CVS Caremark dropped Zepbound from its standard formulary in mid-2025 after striking a rebate deal with Wegovy's manufacturer. If your pharmacy benefit manager is CVS Caremark, expect a denial on the initial claim.
Here's why you should appeal: Wegovy is NOT FDA-approved for sleep apnea. Zepbound is the only GLP-1 with that indication. That gives you strong grounds for a formulary exception — there is no on-formulary therapeutic equivalent for OSA. Multiple class-action lawsuits have been filed challenging these denials. Don't accept the first “no” as the final answer.
What to do if you're denied
- Get the denial letter and identify the specific reason (missing documentation, “not medically necessary,” formulary exclusion, step therapy requirement)
- Have your doctor submit an appeal with a letter of medical necessity that specifically references the FDA-approved OSA indication
- Include your sleep study, BMI documentation, and any prior treatment history
- If the denial is a formulary exclusion, file for a formulary exception — emphasize that no on-formulary alternative has the OSA indication
- If your internal appeal is denied, most plans have an external review process
One real patient on an insurance forum described it this way: trying to get Zepbound approved felt like “a nightmare” that dragged on for weeks. But another patient with a different plan said the process was “pleasantly simple” — the doctor's office called with sleep test results and weight info, and coverage was approved quickly. The difference usually comes down to documentation and framing.
Medicare Part D
Medicare Part D may cover Zepbound when prescribed specifically for moderate-to-severe obstructive sleep apnea in adults with obesity — not for weight loss alone. This is a critical distinction.
Important: The Medicare GLP-1 Bridge program launching in July 2026 is a separate weight-management demonstration. CMS has stated that Zepbound prescribed for its FDA-approved OSA indication goes through your regular Part D plan's utilization-management process — not through the Bridge. Don't confuse the two pathways.
To pursue Medicare coverage for Zepbound for sleep apnea:
- You must have Medicare Part D (standalone or through Medicare Advantage)
- Your Rx must be written specifically for moderate-to-severe OSA
- Prior authorization is almost always required
- BMI of 30+ with documented moderate-to-severe OSA
- Your out-of-pocket cost depends on your plan — the 2026 Part D annual out-of-pocket cap is $2,100
Note for Medicare readers: Ro currently states that it cannot coordinate GLP-1 coverage for government insurance plans. If you're on Medicare, your best route is through your personal physician or a sleep specialist who can submit the prior authorization directly to your Part D plan.
No insurance / self-pay
If you don't have insurance that covers Zepbound, you're not locked out. The Lilly KwikPen self-pay savings program starts at $299 for the 2.5mg dose, available through LillyDirect or Ro. Ro provides the clinical evaluation and prescription along with the medication (with a membership fee). LillyDirect offers the medication at the same pricing if you already have a prescription.
The insurance path is navigable — especially when the prior auth is framed around the OSA indication. If you have commercial insurance, Ro can handle the paperwork for you.
Start the Commercial Insurance Coverage Check for Zepbound on Ro →How to actually get Zepbound for sleep apnea (step by step)
The path is simpler than most pages make it sound. But it depends on your insurance situation, so here are the real-world routes:
Path 1: Commercial insurance or cash pay → Ro
This is the most streamlined online path for most non-Medicare readers.
- Start an online evaluation on Ro (takes about 10 minutes)
- Ro's medical team reviews your sleep apnea diagnosis, BMI, and medical history
- If appropriate, a provider prescribes FDA-approved Zepbound
- For commercial insurance: Ro helps with prior authorization — if approved, your Zepbound pen prescription is typically filled at a local pharmacy
- For cash pay: Zepbound KwikPen can ship directly to you at Lilly self-pay pricing, plus Ro's membership fee
- If insurance doesn't cover it, you can switch to the cash-pay KwikPen route
Why Ro for this? You can absolutely use your own doctor — and for Medicare patients, you should. But for commercially insured or cash-pay readers, Ro consolidates the evaluation, prescription, insurance paperwork, and access into one workflow. Particularly useful if your primary care doctor isn't experienced with GLP-1 prior authorizations for OSA.
Path 2: Medicare → Your doctor + Part D
- Schedule an appointment with your doctor or a sleep medicine specialist
- Bring your sleep study results, BMI documentation, and current treatment history
- Ask your doctor to write the Zepbound prescription specifically for obstructive sleep apnea — not generic weight loss
- Your doctor submits prior authorization to your Part D plan
- If approved, fill at a Part D in-network pharmacy
- Your copay depends on your specific plan's cost-sharing structure
Path 3: No diagnosis yet → Get tested first
If you suspect sleep apnea but haven't had a sleep study, don't try to jump straight to Zepbound. The diagnosis is the foundation — for the FDA indication, for insurance coverage, and for knowing whether this medication makes sense for your specific situation.
Ask your doctor about a polysomnography (overnight lab study) or a home sleep apnea test (HSAT). Many telehealth platforms can also coordinate home sleep testing.
Dosing timeline once you start
The recommended starting dose is 2.5mg once weekly for 4 weeks. After that, the dose increases in 2.5mg steps — after at least 4 weeks on the current dose — until your maintenance dose is reached. For the OSA indication, the recommended maintenance doses are 10mg or 15mg once weekly.
The full dose ladder: 2.5mg → 5mg → 7.5mg → 10mg → 12.5mg → 15mg
You inject once per week in the stomach, thigh, or upper arm. Rotate injection sites each week. Most people reach their maintenance dose around months 4–5.
What are the side effects of Zepbound?
The side effects are real, they're mostly GI-related, and for most people, they're manageable and temporary. Being upfront about this is important because the people who quit early usually do so because nobody prepared them for the first few weeks.
Common side effects
These are the ones most people experience, especially during dose escalation:
- Nausea (most common — tends to peak during the first 2–3 months)
- Diarrhea
- Constipation
- Stomach pain or indigestion
- Reduced appetite (this is partly how it works, but can feel uncomfortable at first)
- Injection site reactions (redness, itching)
Zepbound is not recommended for people with severe gastroparesis (severely delayed stomach emptying).
Serious but less common side effects
- Pancreatitis (stop taking and call your doctor immediately if you have severe abdominal pain that won't go away)
- Gallbladder problems (gallstones, cholecystitis)
- Acute kidney injury (usually from dehydration caused by vomiting/diarrhea — stay hydrated)
- Serious hypersensitivity / allergic reactions
- Low blood sugar (hypoglycemia, especially if you're also taking insulin or sulfonylureas)
- Pulmonary aspiration risk during general anesthesia or deep sedation — tell your healthcare provider about Zepbound before any planned surgery
The boxed warning
Zepbound carries a thyroid C-cell tumor warning based on animal studies. It is contraindicated in people with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2). Your provider will screen for this before prescribing.
The honest take on side effects
Let's be direct: the first 4–8 weeks on Zepbound can be rough. Nausea is common. Some people deal with constipation that requires management. Eating feels different — your hunger drops significantly, and meals that used to feel normal can feel like too much.
But here's what the data tells us: the vast majority of trial participants stayed on Zepbound for the full 52 weeks despite side effects. The GI issues tend to plateau and then fade as your body adjusts to each dose. Starting low (2.5mg) and increasing gradually every four weeks is specifically designed to minimize this.
Real patients manage it with practical strategies: smaller meals, staying hydrated, MiraLAX or fiber gummies for constipation, eating slowly, avoiding high-fat or heavy meals during dose transitions. It's not glamorous. But the weight loss and sleep apnea improvements don't fade the way the nausea does.
Zepbound vs. Ozempic vs. Wegovy: Why the OSA approval only belongs to Zepbound
Only Zepbound is FDA-approved for sleep apnea. This isn't a technicality — it determines everything about coverage, access, and how your doctor frames the prescription.
| Feature | Zepbound (tirzepatide) | Wegovy (semaglutide) | Ozempic (semaglutide) |
|---|---|---|---|
| FDA-approved for OSA | Yes (Dec 2024) | No | No |
| Drug class | GIP + GLP-1 agonist | GLP-1 only | GLP-1 only |
| Average weight loss in trials | 18–20% | ~15% | ~12–15% |
| Medicare coverage for sleep apnea | Yes (Part D) | No | No |
| Commercial insurance path for OSA | OSA-specific prior auth | Weight loss only | Diabetes only |
If you're currently on Ozempic or Wegovy and you also have sleep apnea, this is worth a conversation with your provider. Switching to Zepbound doesn't just change your medication — it can change your insurance coverage story entirely. A prescription framed around OSA treatment may unlock coverage that was denied when framed around weight loss.
What about Mounjaro? Same active ingredient (tirzepatide) as Zepbound, but approved for type 2 diabetes — not OSA and not weight loss. If you're already on Mounjaro for diabetes and also have sleep apnea, talk to your doctor about whether a Zepbound prescription for the OSA indication makes sense alongside or instead of your current regimen.
What about compounded tirzepatide for sleep apnea?
We need to draw a clear line here. Compounded tirzepatide is not FDA-approved Zepbound. The OSA indication, the clinical trial data, the insurance coverage pathways — all of that belongs to the brand-name product manufactured by Eli Lilly.
This matters for practical reasons:
First, no compounded version has been tested for sleep apnea in the way Zepbound has. The SURMOUNT-OSA data is specific to Eli Lilly's product.
Second, insurance won't cover compounded tirzepatide for OSA. The coverage pathway depends on the FDA-approved indication, which only Zepbound carries.
For this specific situation — getting FDA-approved treatment for a diagnosed medical condition with a real coverage pathway — the authentic route is the right one.
If you're currently on compounded tirzepatide and want to explore the FDA-approved route for sleep apnea coverage, we've written a guide to transitioning from compounded to brand-name GLP-1s that walks through the process.
What happens if you stop taking Zepbound?
Most people regain weight after stopping GLP-1 medications, and sleep apnea symptoms are likely to return with the weight. This is the part nobody loves hearing, but it's important to understand upfront.
Zepbound works by changing your appetite signaling. When you stop the medication, that signaling reverts to its previous patterns. Weight regain follows in most cases, and with it, the fat that was pressing on your airway returns.
This doesn't mean you're “trapped” on Zepbound forever. Some people lose enough weight and successfully build eating and exercise habits that partially sustain their progress. Others use Zepbound to reduce their OSA to a manageable level and then transition to CPAP alone or a lower-intensity maintenance approach.
But the honest expectation is: Zepbound for OSA is more like blood pressure medication than a course of antibiotics. It's managing an ongoing condition, not curing it with a fixed treatment course. Your provider will help you figure out the right long-term plan — which might mean staying on it, reducing the dose, or transitioning to a different maintenance strategy.
Any decision to stop should involve a follow-up sleep study to see where your AHI stands without the medication. Don't guess. Let the data guide it.
Real experiences: What people are actually saying about Zepbound for sleep apnea
We read through forums, patient communities, and public discussions to find what real people are experiencing. These are not clinical evidence — they're individual stories that reflect the emotional reality of this decision.
“I've been on it for several months and have lost about 15 pounds. I am sleeping better — better oxygen level. I have no significant side effects. I don't know if I will ever be able to stop the CPAP machine, but am thankful for the weight loss and increased energy.”
— Medicare patient on Mayo Clinic Connect
The realistic middle ground. Not a miracle, not a failure. Steady weight loss, better sleep, improved oxygen — with CPAP still in the picture. For many people, this is exactly what success looks like.
“Coverage just started this year. The doctor's office called and provided results of my sleep test and info on my weight. It was pleasantly simple. I feel that this is making a huge difference in my health.”
— Medicare patient, Mayo Clinic Connect
The insurance process isn't always a fight. When the documentation is clean and the diagnosis is clear, approvals can happen smoothly. That's why we keep emphasizing: get your sleep study and BMI documentation ready before you start.
“I did a sleep study recently and the sleep apnea is gone!!”
— Reddit user, r/Zepbound
Not everyone gets here. But enough people do that these posts show up regularly in patient communities. This is what the 42% remission rate looks like in real life.
These stories paint a consistent picture: Zepbound is working for real people with real sleep apnea. The insurance path is navigable. The combination of weight loss plus sleep improvement feels life-changing for those it works for.
Does this sound like your situation? The people in these stories all started where you are right now — wondering if this was real, if they'd qualify, if the process would be worth it. They took the step. Most of them are glad they did.
Questions to ask your doctor before starting Zepbound for sleep apnea
Screenshot this section and bring it to your appointment. These are the questions that will make the conversation productive and help your doctor submit the strongest possible prior authorization.
- “Do I meet the FDA-approved criteria for Zepbound for OSA?” — You need documented moderate-to-severe OSA plus a BMI of 30+. Your doctor can confirm based on your sleep study and current measurements.
- “Is my sleep apnea primarily obesity-related?” — This is the mechanism Zepbound targets. If your OSA has significant anatomical drivers (narrow jaw, large tonsils), Zepbound may help but may not fully resolve it.
- “Can you submit the prescription specifically for OSA, not generic weight loss?” — This one sentence can be the difference between insurance approval and denial. The FDA indication is for sleep apnea. The prior auth should reflect that.
- “What documentation will insurance want?” — Sleep study results, BMI, diagnosis codes, notes on prior treatment attempts (CPAP trial, lifestyle changes), letter of medical necessity.
- “Should I keep using CPAP while on Zepbound?” — In almost all cases, yes. CPAP provides immediate nightly protection. Zepbound's benefits build over months.
- “When should I repeat my sleep study?” — Typically after 6–12 months of treatment and significant weight loss, to objectively measure how your AHI has changed.
- “What if my insurance denies it?” — Have a plan. Ask your doctor about the appeal process and whether they'll submit a letter of medical necessity.
Walking into your appointment with these questions shows your provider you've done your homework — and it helps them build the strongest possible case for your coverage.
How we verified this page
Every factual claim on this page traces back to a specific, verifiable source. We don't guess, and we don't round up.
Sources used:
- FDA approval announcement (December 20, 2024) — fda.gov
- Zepbound U.S. prescribing information (revised 02/2026) — pi.lilly.com
- SURMOUNT-OSA trial data — published outcomes via Eli Lilly
- Eli Lilly savings and pricing pages — zepbound.lilly.com/savings
- Ro pricing, insurance, and logistics pages — ro.co
- CMS Medicare GLP-1 Bridge documentation — cms.gov
- American Academy of Sleep Medicine clinical guidance — aasm.org
- UnitedHealthcare and South Carolina Blues prior authorization policies (representative examples)
- Mayo Clinic Connect patient forum (public patient experiences)
- Reddit r/Zepbound and r/SleepApnea (public discussion)
What we verified and when:
- FDA approval language and labeled indication: Verified April 2026
- Lilly KwikPen self-pay pricing ($299/$399/$449 offer terms): Verified April 2026
- Ro pricing, membership fees, and insurance limitations: Verified April 2026
- Medicare Part D coverage rules for OSA vs. GLP-1 Bridge distinction: Verified April 2026
- Prescribing information warnings and dosing (02/2026 label): Verified April 2026
Medical review: Dr. Diana Isaacs, PharmD, BCPS, BCACP, BC-ADM, CDCES.
Affiliate disclosure: The RX Index earns a commission when you use certain links on this page, including links to Ro. This does not affect our clinical reporting, and our editorial team independently verifies all claims. If we removed every affiliate link, this page would still be the most complete Zepbound-for-sleep-apnea resource on the internet — and that's by design. Read our full disclosure →
Zepbound for sleep apnea FAQ
Is Zepbound FDA-approved for sleep apnea?▾
How much does Zepbound cost for sleep apnea?▾
Does Medicare cover Zepbound for sleep apnea?▾
Can Zepbound replace CPAP?▾
Do I need a sleep study before getting Zepbound for sleep apnea?▾
What BMI do I need to qualify?▾
How soon can I expect sleep apnea improvement?▾
What are the most common side effects?▾
Is Zepbound the same as Mounjaro?▾
What if my insurance denies Zepbound for sleep apnea?▾
Can I get Zepbound for sleep apnea online?▾
What happens if I stop taking Zepbound?▾
What's your next step?
You've read the data. You've seen the real costs. You know whether you meet the criteria. The only thing left is to find out if a provider agrees you're a fit.
If you have commercial insurance or plan to self-pay:
Check Your Zepbound Eligibility on Ro — Takes About 10 Minutes →On Medicare? Your strongest path is through your doctor or sleep specialist, who can submit prior authorization directly to your Part D plan. Read our Medicare GLP-1 coverage guide →