Drug Comparison · FDA Labels + Pricing Verified April 7, 2026
Zepbound vs Wegovy for Weight Loss: Which One Is Better in 2026?
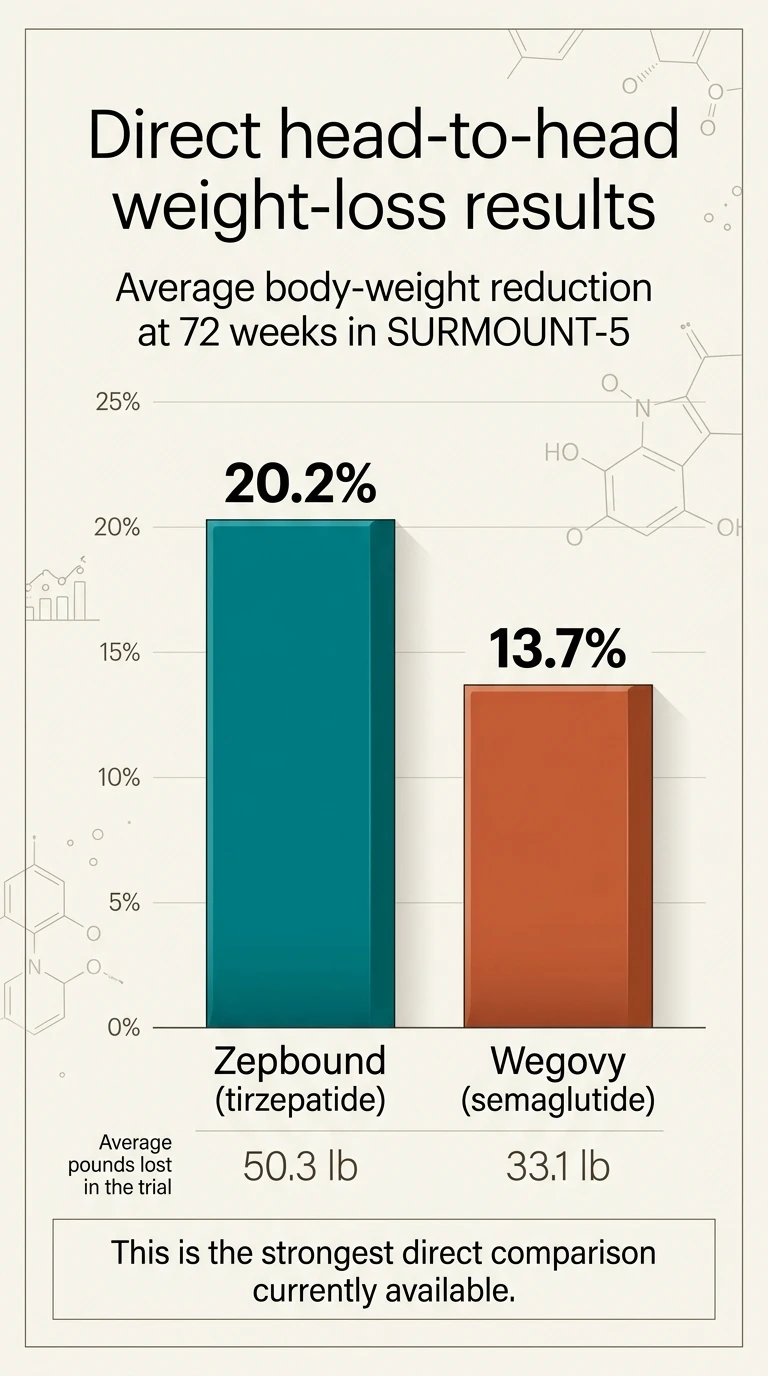
In the only direct head-to-head trial, Zepbound (tirzepatide) beat Wegovy (semaglutide) 20.2% vs 13.7% average body-weight loss at 72 weeks — roughly 50 pounds vs 33 pounds. That gap is real, significant, and the best direct evidence we have.
But Wegovy wins on four specific fronts that matter enormously for the right patient: an FDA-approved pill option, a cardiovascular risk-reduction indication, approval for teens ages 12+, and an FDA-approved MASH indication. None of those belong to Zepbound. If any of them apply to you, the weight-loss gap becomes secondary.
On cash-pay pricing (verified April 7, 2026): Wegovy tablet starts at $149/mo. Wegovy injection starts at $199/mo. Zepbound starts at $299/mo. Eligible commercially insured patients can pay as little as $25/mo for either.
By The RX Index Editorial Team · Last verified April 7, 2026 · Affiliate disclosure · Editorial standards
⚡ Update — April 7, 2026
Wegovy HD (semaglutide 7.2 mg) launched today after FDA approval on March 19, 2026. In the STEP UP trial, it delivered 20.7% average weight loss among patients who stayed on treatment — approaching Zepbound's 20.2% in a separate trial. We cover what this means for your decision below.
The Bottom Line
Zepbound is the better choice for most adults whose top priority is maximum weight loss. The only head-to-head trial (SURMOUNT-5, New England Journal of Medicine, 2025) showed 20.2% vs 13.7% average body-weight loss at 72 weeks. Choose Wegovy instead if you want an FDA-approved pill, are 12–17, have established cardiovascular disease or MASH, or your insurance covers semaglutide but not tirzepatide.
Check Zepbound or Wegovy Eligibility on Ro →Evidence confidence labels used on this page:
- 🟢 Direct head-to-head — from SURMOUNT-5 (Zepbound vs Wegovy injection, NEJM 2025)
- 🟡 Separate trial — from STEP UP (Wegovy HD vs placebo) or Study 7 (Wegovy tablet vs placebo)
- ⚪ Official pricing — from LillyDirect, NovoCare, or manufacturer press releases
- 🔵 Regulatory — from FDA prescribing information or FDA press releases
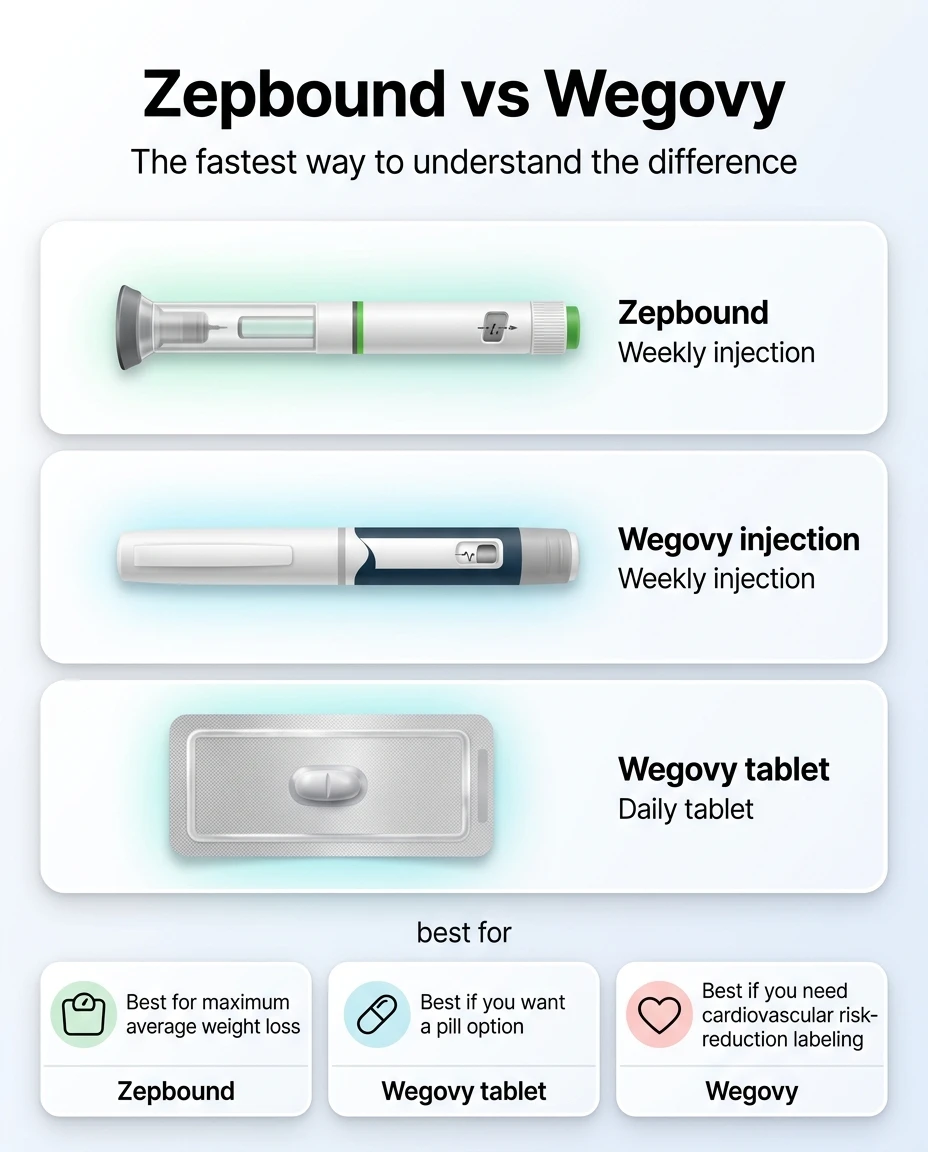
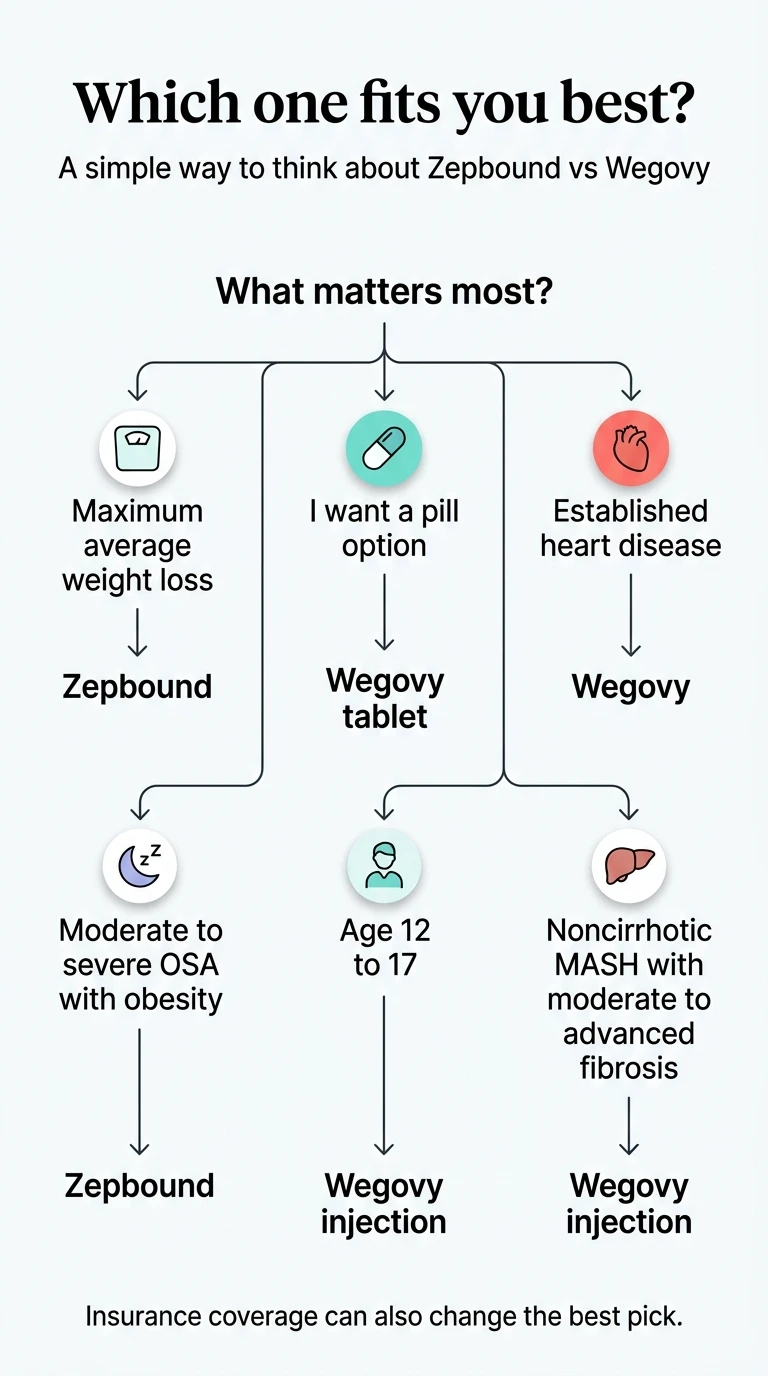
Quick Decision: Find Your Situation Below
Not sure which fits you? Match your primary concern to the right pick.
| Your situation | Best pick | Why |
|---|---|---|
| You want the strongest average weight loss, no CV history | Zepbound | 20.2% in head-to-head trial, dual GLP-1/GIP mechanism |
| You want an FDA-approved pill (no needles) | Wegovy tablet | First oral GLP-1 approved for weight loss; ~13.6% avg weight loss |
| You have a history of heart attack, stroke, or heart disease | Wegovy | Only one with an FDA-approved cardiovascular risk-reduction indication |
| You have obesity-related sleep apnea | Zepbound | Only GLP-1 with FDA-approved OSA indication |
| You're 12–17 years old | Wegovy | Only option approved for adolescents |
| You have MASH (fatty liver with fibrosis) | Wegovy injection | FDA-approved (accelerated) for noncirrhotic MASH with moderate to advanced fibrosis |
| Your insurance covers one but not the other | Whichever is covered | A 14% reduction you can afford beats 20% you can't sustain |
Sources: SURMOUNT-5 (NEJM 2025); FDA prescribing information for Zepbound and Wegovy (2026 labels). Pricing verified April 7, 2026.
Zepbound vs Wegovy at a Glance: The Full 2026 Comparison
Every number sourced and verified against manufacturer sites and FDA labels on April 7, 2026.
| Zepbound | Wegovy Injection | Wegovy Tablet | |
|---|---|---|---|
| Active ingredient | Tirzepatide | Semaglutide | Semaglutide |
| How it works | GLP-1 + GIP (dual action) | GLP-1 only | GLP-1 only |
| Manufacturer | Eli Lilly | Novo Nordisk | Novo Nordisk |
| How you take it | Weekly injection (pen or vial) | Weekly injection (pre-filled pen) | Daily pill, morning, empty stomach |
| Maintenance dose | 5, 10, or 15 mg weekly | 1.7 or 2.4 mg weekly (+ 7.2 mg HD) | 25 mg daily |
| Avg weight loss (trials) | 20.2% SURMOUNT-5 🟢 | 13.7% SURMOUNT-5 / 20.7% HD 🟡 | 13.6% Study 7 🟡 |
| FDA-approved uses | Weight loss; OSA in obesity | Weight loss; CV risk reduction; MASH; ages 12+ | Weight loss; CV risk reduction (adults with established CV disease) |
| Age eligibility | Adults 18+ | Ages 12+ | Adults 18+ |
| Cash-pay price (starter) | $299/mo (LillyDirect) ⚪ | $199/mo intro (2 fills, through 6/30/26) ⚪ | $149/mo (1.5 mg, 4 mg) ⚪ |
| Cash-pay price (maintenance) | $449/mo program / $499–$699 standard ⚪ | $349/mo or $249/mo subscription ⚪ | $299/mo or $249/mo subscription ⚪ |
| With commercial insurance | As low as $25/mo | As low as $25/mo | As low as $25/mo |
| Biggest catch | No pill, no CV labeling, no teen approval | Less weight loss in head-to-head trial | Daily morning routine with food restrictions |
Sources: SURMOUNT-5 (NEJM 2025), STEP UP (Lancet Diabetes Endocrinol 2025), FDA prescribing information (2026 labels), LillyDirect, NovoCare. All prices verified April 7, 2026.
Which Is Better for Weight Loss: Zepbound or Wegovy?
For most adults whose primary goal is losing the most weight, Zepbound is the stronger pick. The head-to-head data is clear. But “most” is doing a lot of work in that sentence — the rest of this page exists because your situation might be one of the important exceptions.
The SURMOUNT-5 trial: the best evidence we have
Until 2025, comparing Zepbound and Wegovy meant looking at separate trials run by different companies with different patient groups — like comparing two restaurants based on different food critics on different nights. SURMOUNT-5 fixed that. Published in the New England Journal of Medicine in May 2025, it randomized 751 adults to receive either Zepbound or Wegovy at their highest tolerated doses for 72 weeks. Same patients. Same protocol. Direct comparison.
SURMOUNT-5 Results at 72 Weeks 🟢 Direct Head-to-Head
Zepbound (Tirzepatide)
20.2%
average body weight loss
~50 lbs on average
Wegovy (Semaglutide)
13.7%
average body weight loss
~33 lbs on average
Lost ≥25% of body weight: 31.6% of Zepbound patients vs 16.1% of Wegovy patients
Waist circumference reduced: 18.4 cm (7.2 in) vs 13.0 cm (5.1 in)
Systolic blood pressure: Greater improvement on Zepbound
Discontinued due to side effects: 6.1% (Zepbound) vs 8.0% (Wegovy)
Why Zepbound outperformed
Zepbound targets two hormones — GLP-1 and GIP — while Wegovy targets only GLP-1. GLP-1 suppresses appetite and slows digestion. GIP adds another layer by improving insulin sensitivity and potentially enhancing fat metabolism. Two signals working together produce a stronger effect than one alone. According to the study's lead author, Dr. Louis Aronne of Weill Cornell Medicine, this dual-hormone approach is likely why Zepbound consistently delivered more weight loss.
What the trial did NOT prove
Important limitations of SURMOUNT-5
- • Excluded patients with type 2 diabetes
- • Compared standard Wegovy doses (up to 2.4 mg), not the new Wegovy HD 7.2 mg dose
- • Did not measure long-term cardiovascular outcomes — one of Wegovy's biggest advantages
- • “Average” results hide wide individual variation — some people lose more, some lose less
The honest admission: Zepbound does not give you a pill option, teen approval, cardiovascular risk-reduction labeling, or a MASH indication. If any of those is your priority, Wegovy is the smarter pick — full stop.
Want the strongest average weight loss? Ro carries FDA-approved Zepbound with access to new pricing and eligibility verification online.
Check Zepbound Eligibility and Pricing on Ro →Who Should Choose Zepbound?
Zepbound is the right call when maximum weight loss is the priority and you don't have a specific condition that steers you toward Wegovy. Here's who benefits most.
Your #1 goal is losing the most weight
The 20.2% average body-weight loss in SURMOUNT-5 is the strongest head-to-head result any GLP-1 has posted against another GLP-1. If you're starting this journey primarily to lose weight and your doctor agrees you're a candidate, Zepbound gives you the highest average expectation on direct evidence.
You have obesity-related sleep apnea 🔵
Zepbound carries a specific FDA indication for moderate to severe obstructive sleep apnea in adults with obesity. Wegovy does not. If sleep apnea is part of your health picture, this is a meaningful differentiator — and it may open insurance coverage pathways that weight loss alone won't.
You've plateaued on Wegovy
This is one of the most common reasons people switch. If you've reached the highest dose of Wegovy (2.4 mg) and your weight loss has stalled, Zepbound's dual-hormone mechanism offers a genuinely different approach. Always discuss with your prescriber before switching — never self-convert doses between brands.
You're comfortable with a weekly injection
Zepbound is injection-only. There is no Zepbound pill. Eli Lilly's new oral GLP-1, Foundayo (orforglipron), was approved April 1, 2026 — but it's a different drug with lower weight-loss results (~11% average). If you need a pill, Zepbound isn't your option.
When Wegovy Is the Smarter Choice
Wegovy's strength isn't just weight loss — it's versatility. No other GLP-1 covers as many FDA-approved indications or delivery formats. Here's when it wins.
You want an FDA-approved pill 🔵
The Wegovy tablet was approved in December 2025 and launched in January 2026. In the FDA label's Study 7, the 25 mg daily tablet produced a 13.6% average body-weight reduction at week 64, with 27.9% of participants achieving 20% or more weight loss.
The catch: you take it first thing on an empty stomach with no more than 4 ounces of plain water, then wait at least 30 minutes before eating, drinking anything else, or taking other oral medications. For most people this becomes routine. For shift workers or people with chaotic mornings, it's a real friction point.
You have established cardiovascular disease 🔵
This is Wegovy's biggest clinical advantage and it's not close. Wegovy is the only medication of the two with an FDA-approved indication to reduce the risk of major cardiovascular events — heart attack, stroke, and cardiovascular death — in adults with established cardiovascular disease who also have obesity or overweight. Zepbound does not have this indication. If you have a history of heart problems, this isn't a marginal consideration — it's potentially the most important factor in your decision.
You have MASH (metabolic dysfunction-associated steatohepatitis) 🔵
Wegovy injection carries an FDA indication — granted under accelerated approval — for noncirrhotic MASH with moderate to advanced liver fibrosis in adults. Continued approval may depend on confirmatory benefit. If your doctor has flagged fatty liver disease progressing toward MASH, Zepbound doesn't match this.
You're 12–17 years old 🔵
Wegovy is approved for adolescents ages 12 and older. Zepbound is currently approved for adults 18+ only. (Eli Lilly is studying Zepbound in adolescents, but no approval yet.)
Your insurance favors semaglutide
Insurance coverage can make the entire decision for you. Some pharmacy benefit managers — CVS Caremark made headlines in 2025 by dropping Zepbound from its preferred list — now require trying Wegovy first. A 14% weight reduction you can afford and sustain beats 20% you abandon after three months because you can't afford it.
Prefer a pill or need Wegovy's label benefits? Ro carries both FDA-approved Wegovy injection and Wegovy tablet, including the new subscription pricing.
See Wegovy Availability and Pricing on Ro →How Much Do Zepbound and Wegovy Cost in 2026?
Real prices verified directly from manufacturer pricing pages on April 7, 2026.
List prices for both medications are over $1,000/month — but almost nobody pays list price anymore. Manufacturers slashed self-pay pricing dramatically in 2025–2026 to compete for the cash-pay market.
Zepbound Pricing (April 2026) ⚪
| How you pay | What to expect |
|---|---|
| Commercial insurance + Lilly savings card | As low as $25/mo (eligible patients) |
| Commercial insurance without coverage | As low as $499/mo (single-dose pen) |
| Self-pay / LillyDirect vials (2.5 mg) | $299/mo |
| Self-pay / LillyDirect vials (5 mg) | $399/mo |
| Self-pay Journey Program (7.5–15 mg) | $449/mo (45-day refill program) |
| Standard price (7.5–10 mg) | $499–$699/mo (without program) |
| Medicare/Medicaid | Not eligible for Lilly savings card; may be covered for OSA indication |
| Medicare Bridge (July–Dec 2026) | $50/mo for eligible Part D beneficiaries |
Wegovy Pricing (April 2026) ⚪
| How you pay | Wegovy Injection | Wegovy Tablet |
|---|---|---|
| Commercial insurance + Novo savings card | As low as $25/mo | As low as $25/mo |
| Self-pay intro | $199/mo (0.25/0.5 mg, first 2 fills, through June 30, 2026) | $149/mo (1.5 mg and 4 mg; 4 mg at $149 through Aug 31, 2026) |
| Self-pay maintenance | $349/mo (1 mg+ doses) | $299/mo (9 mg and 25 mg) |
| Subscription — 12-month | $249/mo | $249/mo |
| Subscription — 6-month | $299/mo | $269/mo |
| Subscription — 3-month | $329/mo | $289/mo |
| Wegovy HD (7.2 mg) | $399/mo self-pay; $25/mo with eligible commercial insurance | — |
| Medicare Bridge (July–Dec 2026) | $50/mo for eligible Part D beneficiaries | $50/mo for eligible Part D beneficiaries |
Subscription program launched March 31, 2026 through Ro, WeightWatchers, and LifeMD. Longer commitments = lower monthly pricing. Telehealth platforms charge separate membership/consultation fees above drug-only pricing. All prices verified April 7, 2026.
The promo rules most pages don't tell you
- • Wegovy injection $199/mo: Only the two lowest doses (0.25 mg and 0.5 mg), first two fills, through June 30, 2026. After that: $349/mo unless on a subscription.
- • Wegovy tablet 4 mg at $149: Limited-time offer through August 31, 2026. After that: $199/mo.
- • Zepbound $449 for higher doses: Requires the Self Pay Journey Program with 45-day refill rules. Outside the program: $499–$699 depending on dose.
- • The “$25/mo with insurance” offer: Only for commercially insured patients. Medicare, Medicaid, and other government insurance generally do not qualify for manufacturer savings cards.
Are the Side Effects Different?
Both medications share the same basic GI side-effect profile — nausea, vomiting, diarrhea, constipation, and abdominal pain — especially during the first weeks and during dose escalation. What you may not know is how the two drugs compare directly.
What SURMOUNT-5 found on side effects 🟢 Direct Head-to-Head
• Nausea: 44% in both groups — essentially identical
• Abdominal pain: ~25% in both groups
• Vomiting: Zepbound users reported less — the GIP hormone may have a nausea-suppressing effect in the brain
• Discontinuation due to adverse events: 6.1% (Zepbound) vs 8.0% (Wegovy)
Neither difference was large enough for researchers to declare one drug “safer.” Individual tolerance varies widely.
Wegovy HD (7.2 mg): one new side effect to know
The higher dose adds a new signal: dysesthesia — an altered skin sensation described as sensitivity, pain, or burning. This occurred more frequently at 7.2 mg than at lower doses. The FDA noted it generally resolved on its own or with a dose reduction and is investigating further.
Boxed warning — both medications carry it
Both Zepbound and Wegovy carry the same boxed warning (FDA's most serious label alert) about potential thyroid C-cell tumor risk based on rodent studies. Neither should be used by anyone with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2).
Other serious-but-rare risks for both: pancreatitis, gallbladder problems, kidney issues from dehydration, and severe allergic reactions.
“From the moment I took the first injection, the food noise was gone.” — Wegovy reviewer, Drugs.com
“I can really tell the difference in my eating and cravings. Down 25 lbs in two months with little to no side effects.” — Zepbound reviewer, Drugs.com
“It works, but the side effects are too much for me.” — Zepbound reviewer, Drugs.com
Patient reviews are anecdotal and not the basis of our recommendations. Individual response is genuinely unpredictable.
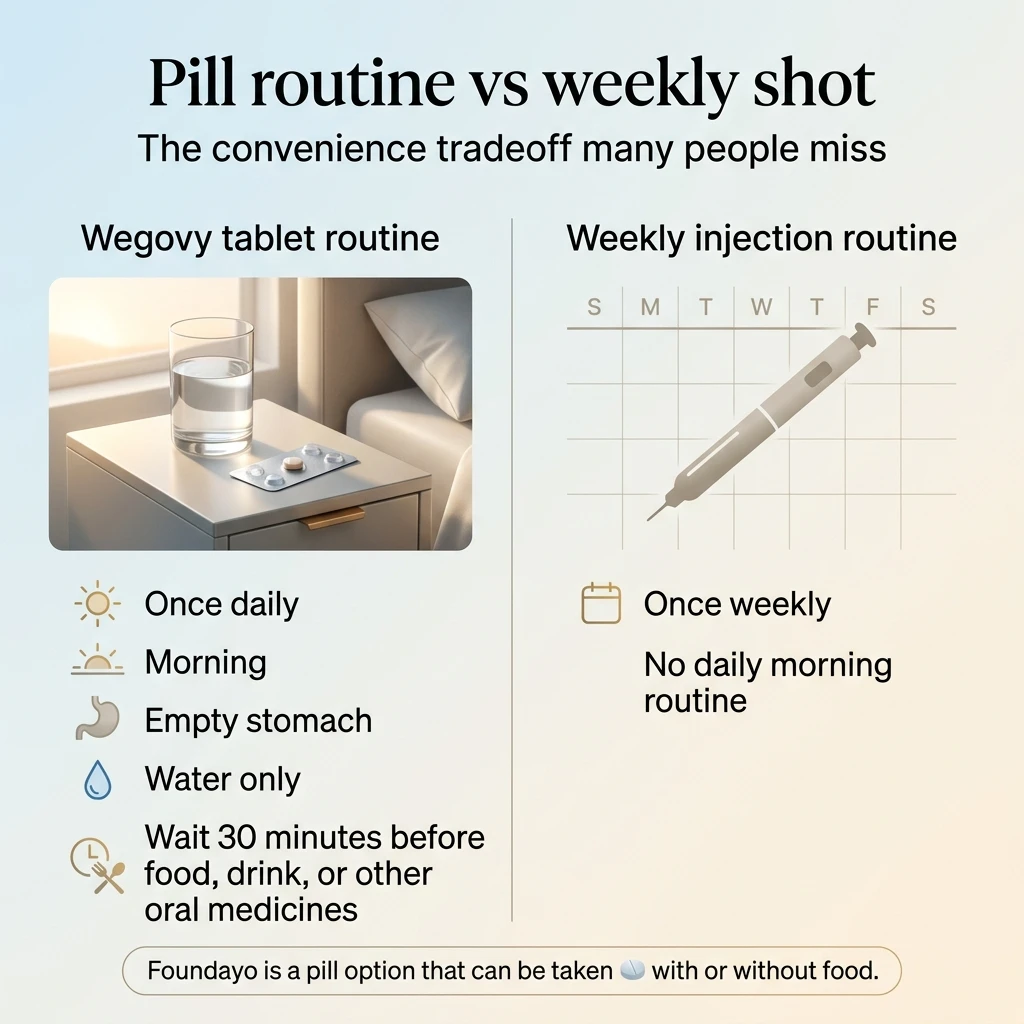
Is the Wegovy Pill Really Easier Than a Weekly Shot?
The pill changes the comparison for a specific group of people — but it's not the free pass some expect.
Why the pill is a real advantage
If injecting yourself weekly makes you anxious or embarrassed — the Wegovy tablet removes that barrier entirely. That's a genuine clinical benefit for millions of people. Studies show about half of injectable GLP-1 users stop treatment at some point — the needle is a real factor for many.
The morning routine most people underestimate
The Wegovy tablet must be taken:
- First thing in the morning, on a completely empty stomach
- With no more than 4 ounces (half a cup) of plain water
- Then wait at least 30 minutes before eating, drinking anything else, or taking any other oral medication
That 30-minute window matters because the pill uses a special absorption enhancer that temporarily changes your stomach environment. Skip the rules and you may absorb significantly less medication.
For comparison: Foundayo (Eli Lilly's new pill)
Foundayo (orforglipron), approved April 1, 2026, can be taken any time of day with or without food. But Foundayo delivers less weight loss (~11% average) and is a completely different drug — not a pill version of Zepbound.
For the deeper breakdown of oral vs injection tradeoffs: Wegovy Pill vs Zepbound →
Can You Switch From Wegovy to Zepbound (or Vice Versa)?
Yes — and it's more common than you might think. The three biggest triggers are weight-loss plateaus, side-effect intolerance, and insurance changes.
Why there's no simple dose conversion chart
Zepbound and Wegovy use different active ingredients with different dose ranges. Neither manufacturer publishes a cross-brand conversion guide. Most prescribers start you at a low dose of the new medication and titrate up from there — even if you were on a high dose of the previous one. Yes, this can feel like starting over. But it reduces the risk of severe GI side effects from the new drug's mechanism.
What to tell your prescriber
- • Am I switching because of a plateau, side effects, or coverage?
- • Should I re-titrate from the lowest dose, or start at a moderate dose given my GLP-1 history?
- • Is there a washout period between stopping one and starting the other?
- • What side effects should I watch for during the transition?
Never take both at the same time
Do not take Zepbound and Wegovy simultaneously. They work through overlapping mechanisms, and combining them increases side-effect risk without proven benefit. No clinical study has tested dual GLP-1 therapy for weight loss.
Does Wegovy HD (7.2 mg) Change the Answer?
It changes the landscape, but it doesn't change our verdict — yet.
⚡ What happened in March and April 2026
On March 19, 2026, the FDA approved Wegovy HD — a 7.2 mg injectable dose of semaglutide, three times the previous highest dose of 2.4 mg. Novo Nordisk launched it on April 7, 2026 in a single-dose pen at $399/month self-pay.
Avg weight loss (on-treatment)
20.7%
STEP UP trial, efficacy estimand
Avg weight loss (all patients)
18.8%
Includes doses missed or discontinued
Lost ≥25% body weight
~1 in 3
Patients who stayed on treatment
Why it doesn't replace the direct evidence
There is no head-to-head trial of Wegovy HD vs Zepbound. The 20.7% comes from STEP UP (Wegovy 7.2 mg vs 2.4 mg vs placebo). The 20.2% comes from SURMOUNT-5 (Zepbound vs standard Wegovy). Different trials, different patient populations, different protocols. Comparing results across different studies is a weaker form of evidence than a direct head-to-head. Our position: Zepbound retains the strongest direct evidence for weight loss against Wegovy until someone runs a head-to-head trial of Zepbound vs Wegovy 7.2 mg.
Who Wegovy HD is actually for: It's not a starting dose. It's specifically approved for adults who have already tolerated Wegovy 2.4 mg for at least four weeks and need additional weight reduction — a step-up for people already on Wegovy who want to push further.
How Much Weight Can You Actually Lose? Setting Real Expectations
Clinical trial averages are useful, but they can set you up for disappointment if you treat them as guarantees. Here's what to actually expect over time.
Appetite changes, modest scale movement
Both medications start at low doses to let your body adjust. Most people notice reduced hunger and smaller portions feeling satisfying. Actual scale movement in month one is typically modest — a few pounds, sometimes more.
Acceleration as doses increase
This is where weight loss accelerates as you titrate to higher doses. Most of the dramatic before-and-after results come from this window. Expect the most visible changes here.
Continued loss, then plateau
Weight loss typically continues but slows. By 12–18 months on a stable maintenance dose, most people have reached or are approaching their maximum response. SURMOUNT-5 measured at 72 weeks: Zepbound ~50 lbs (20.2%), Wegovy ~33 lbs (13.7%).
Weight regain is common — plan for the long term
Weight regain is common after stopping GLP-1 medications. These drugs treat obesity as a chronic condition, similar to how blood pressure medication treats hypertension. For most people, long-term use is the plan, not a short-term fix.
What About Insurance, Medicare, and Medicaid?
Coverage for GLP-1 medications is expanding in 2026 — but it's still maddeningly inconsistent.
Commercial insurance
Coverage varies by plan, employer, and pharmacy benefit manager (PBM). According to KFF, 19% of firms with 200+ workers and 43% of firms with 5,000+ workers covered GLP-1s for weight loss in 2025. The trend is expanding, but your specific plan may still exclude weight-loss medications entirely.
Step therapy is increasingly common. Some plans now require you to try Wegovy before they'll approve Zepbound. Your best first move: call the number on the back of your insurance card and ask specifically whether Zepbound and Wegovy are on your formulary and what prior authorization requirements exist.
Medicare Bridge Program (July 1 – December 31, 2026)
CMS's Medicare GLP-1 Bridge program covers Wegovy (tablets and injection) and Zepbound for eligible Part D beneficiaries using them for weight management at $50/month under the bridge. The program runs July 1–December 31, 2026. Existing Part D coverage for other labeled uses (such as Zepbound for OSA) remains separate. The Medicare landscape is evolving rapidly — check with your Part D plan for the latest.
Medicaid
13 state Medicaid programs covered GLP-1s for obesity under fee-for-service as of January 2026. North Carolina reinstated coverage in December 2025. Most states still don't cover weight-loss medications through Medicaid. Contact your state Medicaid program directly. See our 50-state Medicaid GLP-1 coverage tracker →
What to do if you're denied
- • Prior authorization: Most plans require it. Your prescriber's office usually handles this.
- • Step therapy documentation: If you've tried other weight-management approaches, documenting this strengthens your case.
- • Appeal: If denied, you have the right to appeal. Many successful appeals include a letter from your prescriber documenting medical necessity.
Who Should NOT Choose Either Medication
Do not use Zepbound or Wegovy if:
- • You or a family member has had medullary thyroid carcinoma (MTC)
- • You have Multiple Endocrine Neoplasia syndrome type 2 (MEN2)
- • You are pregnant or planning to become pregnant — Wegovy labeling advises stopping at least 2 months before a planned pregnancy; Zepbound labeling advises discontinuing when pregnancy is recognized
- • You are allergic to tirzepatide, semaglutide, or any ingredient in either medication
- • You have a history of pancreatitis — discuss risks carefully with your doctor
If your situation sounds complicated — multiple conditions, multiple medications, uncertainty about eligibility — stop reading comparison content and talk to a clinician. A comparison page is a starting point, not a substitute for medical evaluation.
Not sure you're a candidate? Our free matching tool takes 60 seconds and gives you a personalized next step based on your health, insurance, and preferences.
Take the Free GLP-1 Matching Quiz →The New GLP-1 Pills: Where Do They Fit?
Two oral GLP-1 medications are now FDA-approved for weight loss. Understanding where they land relative to the injectables helps complete the picture.
| Medication | Form | Avg Weight Loss | Trial |
|---|---|---|---|
| Wegovy HD 7.2 mg* | Weekly injection | ~20.7%* | STEP UP 🟡 |
| Zepbound (tirzepatide) | Weekly injection | ~20.2% | SURMOUNT-5 🟢 |
| Wegovy 2.4 mg | Weekly injection | ~15% | STEP 1 🟡 |
| Wegovy tablet 25 mg | Daily pill | ~13.6% | Study 7 (FDA label) 🟡 |
| Foundayo (orforglipron) 36 mg | Daily pill | ~11% | ATTAIN-1 🟡 |
*Wegovy HD efficacy estimand (patients who stayed on treatment); treatment-regimen analysis showed 18.8%. Wegovy HD and Zepbound have NOT been compared head-to-head. These numbers come from different studies with different patient populations — useful for context but not direct comparisons.
The takeaway: injectables still produce more weight loss than pills. If convenience matters more than maximizing the number on the scale, the pills are legitimate options — but they're not potency equivalents.
For a broader look at all your options: Best GLP-1 for Weight Loss →
How We Verified This Comparison
We want you to know exactly how we built this page — and what we deliberately didn't claim.
| Evidence type | Source | How used |
|---|---|---|
| Direct head-to-head trial | SURMOUNT-5 (NEJM, May 2025) | Primary basis for Zepbound vs Wegovy verdict |
| Phase 3 trial | STEP UP (Lancet Diabetes Endocrinol, 2025) | Wegovy HD efficacy data |
| FDA prescribing information | Wegovy PI, Zepbound PI (2026 labels) | Approved indications, dosing, warnings, tablet efficacy |
| FDA press announcements | March 19 and April 1, 2026 | Approval details for Wegovy HD and Foundayo |
| Manufacturer pricing pages | LillyDirect, NovoCare | All pricing claims (verified April 7, 2026) |
| Press release | Novo Nordisk, March 31, 2026 | Wegovy subscription program details |
| Launch reporting | Reuters, April 7, 2026 | Wegovy HD launch pricing |
| Patient reviews | Drugs.com (user submissions) | Anecdotal context only, clearly labeled |
What we deliberately did NOT claim
- • We did not claim Wegovy HD and Zepbound deliver “the same” weight loss. They've never been compared directly.
- • We did not claim one drug is “safer” than the other. Side-effect profiles are similar and individual response varies.
- • We did not use star ratings or review scores. There's no standardized review methodology to justify them.
- • We did not claim compounded medications are equivalent to FDA-approved brands. They are not interchangeable and are regulated differently.
Frequently Asked Questions
Is Zepbound or Wegovy better for weight loss?
For most adults, Zepbound produces more weight loss. The SURMOUNT-5 head-to-head trial showed 20.2% average body-weight loss with Zepbound vs 13.7% with Wegovy at 72 weeks. However, Wegovy may be the better fit if you have cardiovascular disease, want a pill, are under 18, have MASH, or if your insurance covers it and not Zepbound.
Is Wegovy better if I want a pill instead of a shot?
Yes. The Wegovy tablet is one of two FDA-approved oral GLP-1 options for weight loss. In clinical trials, the Wegovy tablet produced a 13.6% average body-weight reduction. Foundayo (orforglipron) produces less weight loss (~11%) but can be taken any time without food restrictions. There is no Zepbound pill.
Which is cheaper without insurance in 2026?
Wegovy tablet starts at $149/mo for lower doses. Wegovy injection starts at $199/mo introductory for first two fills of the lowest doses. Zepbound starts at $299/mo for 2.5 mg vials via LillyDirect. All prices verified April 7, 2026.
Which is more likely to be covered by insurance?
It depends entirely on your plan and pharmacy benefit manager. Wegovy has a slight edge because it has been on the market longer and has more established formulary positions. Some PBMs now require trying Wegovy before covering Zepbound. Check your specific plan.
Can you switch from Wegovy to Zepbound?
Yes, switching is a common clinician-managed conversation after a plateau, side effects, or insurance changes. Your prescriber will typically start you at a lower dose of the new medication and titrate up. Do not self-convert doses between brands.
Can you switch from Zepbound to Wegovy?
Yes, the same principles apply. This became more common after CVS Caremark dropped Zepbound from its preferred list in 2025, requiring many patients to transition to Wegovy. Your prescriber will manage the dose adjustment.
Can you take Zepbound and Wegovy together?
No. Never combine two GLP-1 medications. They work through overlapping mechanisms and combining them increases side-effect risk with no proven benefit.
Does Wegovy HD change which one is best?
Wegovy HD (7.2 mg, approved March 2026) delivered 20.7% weight loss in the STEP UP trial (efficacy estimand), approaching Zepbound's 20.2% in SURMOUNT-5. However, these results come from different trials and have not been compared head-to-head. The direct evidence still favors Zepbound.
Which is better if I have sleep apnea?
Zepbound. It is the only GLP-1 with a specific FDA indication for moderate to severe obstructive sleep apnea in adults with obesity.
Which is better if I have heart disease?
Wegovy. Among these two medications, it is the one with an FDA-approved indication to reduce the risk of heart attack, stroke, and cardiovascular death in adults with established heart disease and obesity or overweight.
Which is better if I have MASH or fatty liver?
Wegovy injection. It carries an FDA indication (granted under accelerated approval) for noncirrhotic MASH with moderate to advanced fibrosis in adults. Zepbound does not have this indication.
Which GLP-1 is approved for teens?
Wegovy only. It is approved for adolescents ages 12 and older. Zepbound is currently approved for adults 18+ only.
Are the side effects worse on Zepbound or Wegovy?
Overall side-effect profiles are similar. In the SURMOUNT-5 head-to-head trial, Zepbound users reported slightly less vomiting and fewer discontinued due to side effects (6.1% vs 8.0%). Individual tolerance varies and neither drug is categorically easier to tolerate.
How much weight can you lose in the first month?
Most people notice appetite changes within the first few weeks but significant scale movement is modest in month one because both drugs start at low doses. Meaningful weight loss typically becomes visible by months 2 to 3 as you titrate to higher doses.
Ready to take the next step?
Still Not Sure Which GLP-1 Is Right for You?
Choosing between Zepbound and Wegovy comes down to your health history, your insurance, your feelings about injections vs pills, and which FDA-approved indications matter for your specific situation. If this page gave you the clarity you needed, here's your next step.
Related guides
This page is for informational purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider before starting, stopping, or switching any medication. The RX Index may receive compensation from providers linked on this page, which supports our ability to maintain and verify this content. Our editorial recommendations are based on published evidence and are not influenced by affiliate relationships.
Last verified: April 7, 2026 · Next scheduled update: May 2026
References
- Aronne LJ, et al. “Tirzepatide as Compared with Semaglutide for the Treatment of Obesity.” New England Journal of Medicine. 2025;393(1):26–36. DOI: 10.1056/NEJMoa2416394
- Eli Lilly and Company. “Savings options for Zepbound.” pricinginfo.lilly.com/zepbound. Accessed April 7, 2026.
- Novo Nordisk. “NovoCare: Wegovy.” novocare.com/patient/medicines/wegovy.html. Accessed April 7, 2026.
- U.S. Food and Drug Administration. “FDA Approves Higher Dose Semaglutide.” March 19, 2026.
- Wharton S, et al. “Once-weekly semaglutide 7.2 mg in adults with obesity (STEP UP).” Lancet Diabetes Endocrinol. 2025;13(11):949–963.
- U.S. FDA. Wegovy prescribing information. 2026. accessdata.fda.gov
- Novo Nordisk. “Novo Nordisk launches first and only multi-month subscription program for FDA-approved Wegovy.” PR Newswire, March 31, 2026.
- U.S. FDA. Zepbound prescribing information. 2026. accessdata.fda.gov
- U.S. FDA. “FDA Approves First New Molecular Entity Under National Priority Voucher Program.” April 1, 2026.
- U.S. FDA. Wegovy tablets prescribing information. 2026. accessdata.fda.gov
- Reuters. “Novo Nordisk launches higher-dose Wegovy in the United States.” April 7, 2026.
- Patient reviews from Drugs.com (Wegovy) and Drugs.com (Zepbound). Accessed April 7, 2026.
- KFF. “Perspectives from Employers on GLP-1 Agonists.” healthsystemtracker.org, 2025.
- CMS Medicare GLP-1 Bridge Program. July 1–December 31, 2026.
Related guides
- Wegovy Pill vs Zepbound: 9 Real Differences in 2026 (Actual Prices)
- How to Get GLP-1 Without Insurance in 2026: Real Prices [Verified]
- Wegovy Pill vs Injection: Cost & Results 2026
- Ro vs Eden for GLP-1: Honest Comparison With Actual 2026 Prices
- Mochi vs Ro for GLP-1: 9 Real Differences in 2026 (Actual Prices)