Pipeline Report · April 2026
By The RX Index Editorial Team · Last verified: April 24, 2026 · Next re-verification: July 24, 2026
VK2735 Viking Therapeutics Weight Loss: Results, Side Effects, and FDA Timeline
⚠ VK2735 is NOT available. Here’s what that means:
- • Not FDA approved — investigational only
- • Cannot be prescribed by any doctor today
- • Not sold legitimately by any pharmacy or telehealth provider
- • Anything for sale online as "VK2735" is an unapproved research chemical — not the drug in trials
- • Earliest realistic FDA decision: late 2028–2029 (editorial projection)
This page is informational and not medical advice. Always consult a licensed clinician before starting, stopping, or changing any medication.
VK2735 Viking Therapeutics Weight Loss: The 30-Second Status Check
| Question | Direct answer |
|---|---|
| Is VK2735 FDA approved? | No. Investigational only. |
| Can a doctor prescribe it today? | No. Not available anywhere. |
| Pill or injection? | Both in development. |
| Best reported result so far? | Injection: up to 14.7% at 13 weeks. Pill: up to 12.2% at 13 weeks. |
| Where is it in development? | Injection: Phase 3 (fully enrolled). Pill: Phase 3 starting Q3 2026. |
| Earliest realistic availability? | Late 2028–2029 (editorial projection if Phase 3 succeeds). |
| Can I buy “VK2735” from a research site? | No. That's not the drug being tested. It's unsafe. |
| If I want real GLP-1 help now? | Talk to a clinician about a currently approved option. |
If waiting 3+ years isn’t your path, find out what’s available right now:
Check eligibility — find my GLP-1 path (free 60-sec quiz) →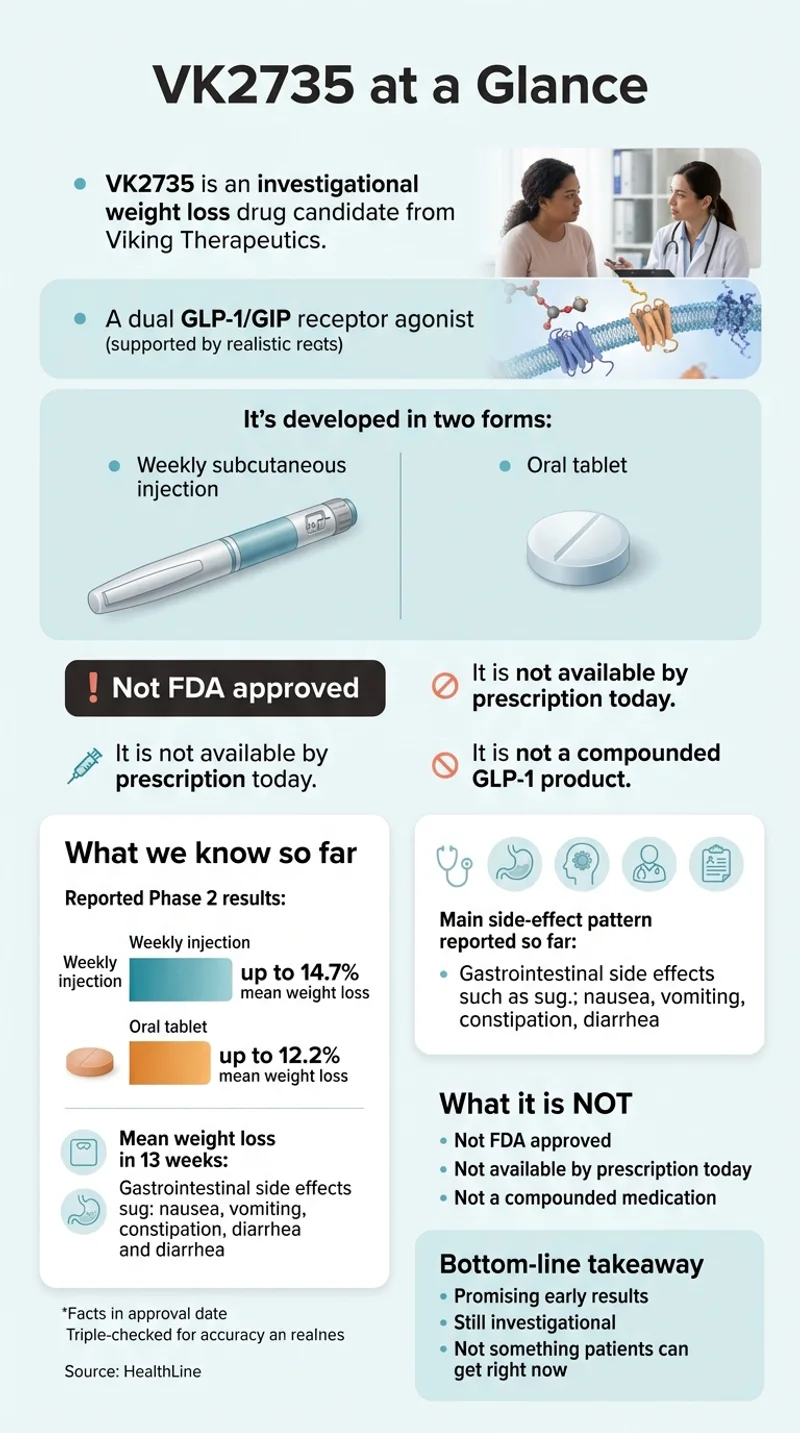
VK2735 overview — Viking Therapeutics investigational weight loss drug. Last verified April 24, 2026.
What We Actually Verified on This Page
✅ What we verified
- • VK2735 is investigational, not FDA-approved, and not legally available. (Verified against Drugs@FDA, FDA shortage list, FDA 503B bulks list.)
- • Viking is developing both a weekly subcutaneous injection and a daily oral tablet.
- • Phase 2 VENTURE (injection): up to 14.7% mean body weight loss at 13 weeks. (Source: Viking IR, Feb 27, 2024; Obesity, Jan 2026.)
- • Phase 2 VENTURE-Oral (tablet): up to 12.2% mean body weight loss at 13 weeks. (Source: Viking IR, Aug 19, 2025.)
- • VANQUISH-1 Phase 3 enrollment completed November 19, 2025 (~4,650 adults). (Viking IR.)
- • VANQUISH-2 Phase 3 enrollment completed March 26, 2026 (~1,000 adults). (Viking IR.)
- • Maintenance dosing study enrollment completed January 8, 2026; results expected Q3 2026.
- • Oral VK2735 Phase 3 expected to begin Q3 2026. (Viking IR, Feb 11, 2026.)
- • VANQUISH study completion expected 2027. (Viking IR, Mar 26, 2026.)
❓ What we could not verify
- • Any FDA approval date (there is none)
- • Any launch price (Viking has not disclosed one)
- • Any legitimate pharmacy or telehealth provider selling VK2735 (there aren’t any)
- • Long-term (52+ week) efficacy or safety data (Phase 3 hasn’t reported yet)
- • Whether VK2735 will be approved at all
What Is VK2735? (The Plain-English Explainer)
VK2735 is an experimental weight loss drug from Viking Therapeutics (NASDAQ: VKTX), a clinical-stage biotech in San Diego. It activates two gut hormone receptors at the same time — GLP-1 and GIP — which is the same dual mechanism used by Eli Lilly’s already-approved Zepbound. Viking is developing it as both a once-weekly injection and a once-daily pill, and it is currently in Phase 3 testing for the injection.
Who is making it?
Viking Therapeutics is a small publicly traded biotech led by CEO Brian Lian, Ph.D. They don’t have any FDA-approved products on the market yet — VK2735 is their lead candidate. If Phase 3 succeeds, Viking will either need to scale up its own commercial operation or partner with (or be acquired by) a larger pharma company to actually launch.
What does "GLP-1/GIP dual agonist" actually mean?
GLP-1 and GIP are two gut hormones that regulate appetite, blood sugar, and how your body handles food. A "dual agonist" activates both receptors at once. GLP-1 is the same hormone pathway hit by Wegovy and Ozempic — curbs appetite, slows stomach emptying, improves insulin signaling. GIP enhances the weight loss effect and, in some studies, reduces nausea.
The only other FDA-approved drug in this exact class is tirzepatide (Zepbound/Mounjaro). VK2735 is aiming to play in that same neighborhood.
What VK2735 is NOT
- • Not the same molecule as tirzepatide, semaglutide, or any other approved drug
- • Not a compounded GLP-1 product — it’s a novel investigational compound
- • Not something a telehealth provider can prescribe
- • Not a "research peptide" you can buy online — anything marketed that way is mislabeled or unverified
Is VK2735 FDA Approved?
No. VK2735 is not FDA approved. As of April 2026, the weekly injection is in Phase 3 (both VANQUISH-1 and VANQUISH-2 are fully enrolled), the oral tablet has completed Phase 2 with Phase 3 expected Q3 2026, and a Phase 1 maintenance dosing study is fully enrolled with results expected Q3 2026. Our editorial projection: a realistic FDA decision window is late 2028 to 2029 if Phase 3 succeeds.
No shortcut exists. No "early access" program has been announced. No expanded access or compassionate use pathway has been publicly disclosed for VK2735 outside of clinical trial enrollment.
What has to happen before VK2735 could be approved
| # | Step | Status |
|---|---|---|
| 1 | Phase 3 (VANQUISH-1, VANQUISH-2) study completion | Expected 2027 |
| 2 | Data analysis and top-line readout | After completion |
| 3 | NDA submission to FDA | After data analysis |
| 4 | FDA acceptance for review | Typically 60 days after submission |
| 5 | FDA review (standard) | ~10 months |
| 6 | FDA approval decision | Earliest late 2028 |
| 7 | Manufacturing scale-up and launch | After approval |
When Will VK2735 Be Available? (The Verified Timeline)
The earliest VK2735 could realistically be available is late 2028, with 2029 being the more likely window. Phase 3 trials are 78 weeks long, Viking has guided completion in 2027, an NDA would follow, and FDA review typically takes around 10 months.
| Date | Milestone | Source |
|---|---|---|
| Q1 2023 | Phase 1 SAD/MAD trial initiated (subcutaneous) | Viking IR |
| Feb 27, 2024 | Phase 2 VENTURE top-line: up to 14.7% weight loss at 13 weeks | Viking press release |
| Jun 25, 2025 | Phase 3 VANQUISH program initiated | Viking press release |
| Aug 19, 2025 | Phase 2 VENTURE-Oral top-line: up to 12.2% weight loss at 13 weeks | Viking press release |
| Nov 19, 2025 | VANQUISH-1 enrollment completed (~4,650 patients, 78-week study) | Viking IR |
| Jan 8, 2026 | Maintenance dosing study enrollment completed (~180 adults) | Viking press release |
| Jan 12, 2026 | Phase 2 VENTURE results published in Obesity journal | Viking IR |
| Feb 11, 2026 | Viking corporate update confirms oral Phase 3 expected Q3 2026 | Viking IR |
| Mar 26, 2026 | VANQUISH-2 enrollment completed (~1,000 patients, 78-week study) | Viking press release |
| Q3 2026 (expected) | Maintenance dosing study readout; oral VK2735 Phase 3 initiation | Viking guidance |
| 2027 (expected) | VANQUISH-1 and VANQUISH-2 study completion | Viking guidance |
| Late 2027–2028 (editorial projection) | NDA submission to FDA | Editorial projection |
| 2028–2029 (editorial projection) | FDA decision on subcutaneous VK2735 | Editorial projection |
| 2029+ (editorial projection) | FDA decision on oral VK2735 | Editorial projection |
What could accelerate or delay this?
Accelerators
- FDA priority review designation (unlikely in this crowded category)
- Acquisition by a larger pharma with stronger regulatory muscle
- Cardiovascular outcome data positioning VK2735 as clinically distinct
Delays
- Phase 3 results falling short of Phase 2 numbers (a real risk)
- Safety signal emerging in 5,650-patient Phase 3
- Manufacturing scale-up challenges (Viking is small)
- FDA requests for additional data
The honest read: plan around 2028–2029 as the earliest, and don’t be surprised if it slips. Retatrutide (Eli Lilly’s triple agonist) is projected for 2027–2028. CagriSema (Novo Nordisk, filed already) is projected for 2026–2027. VK2735 is a later entrant.
How Much Will VK2735 Cost?
Unknown. Viking has not disclosed any pricing, and pricing won’t be set until closer to launch. For reference, Wegovy and Zepbound currently list at roughly $1,000–$1,400 per month at retail without coverage; with insurance or manufacturer programs, out-of-pocket can be much lower. If you want to compare what FDA-approved options actually cost today, our GLP-1 provider guides track current pricing across providers.
How Well Does VK2735 Work? (Phase 2 Data in Plain English)
In the Phase 2 VENTURE trial, the weekly injection produced up to 14.7% mean weight loss from baseline over 13 weeks, with up to 88% of patients at the highest dose hitting at least 10% weight loss compared to 4% on placebo. In the Phase 2 VENTURE-Oral trial, the daily pill produced up to 12.2% weight loss over 13 weeks. Both trials showed no plateau — weight loss was still trending down when the studies ended.
Those are big numbers for 13 weeks. But context matters: VK2735’s Phase 2 was a 13-week sprint, while Wegovy and Zepbound have data from 64–72-week marathons. Comparing them directly without acknowledging that gap is how people get misled.
VENTURE (weekly injection) — the headline numbers
- 176 adults, 13-week randomized placebo-controlled study
- Up to 14.7% mean weight loss from baseline
- Up to 13.1% placebo-adjusted weight loss
- Up to 88% of patients at the highest dose hit ≥10% weight loss (vs. 4% placebo)
- Discontinuation: 13% on VK2735 vs. 14% on placebo — balanced
- No plateau observed at 13 weeks
- Published in Obesity (January 2026)
VENTURE-Oral (daily tablet) — the headline numbers
- 280 adults, 13-week randomized placebo-controlled study
- Up to 12.2% mean weight loss from baseline (26.6 lbs at the highest dose)
- Up to 10.9% placebo-adjusted
- Up to 97% of participants hit ≥5% weight loss; up to 80% hit ≥10%
- Overall discontinuation across VK2735 groups: 28% vs. 18% placebo
- Exploratory maintenance arm: titrate to 90 mg, drop to 30 mg — weight loss held
The honest read
Stifel analyst Annabel Samimy called the oral results “among the upper echelon” of oral obesity medicines. But investors cut Viking’s market value roughly in half on August 19, 2025 — specifically because of the 38% discontinuation rate at the 120 mg dose. William Blair’s Andy Hsieh called the sell-off “extreme and unwarranted,” noting the 30 mg dose showed a “placebo-like” tolerability profile. The real story: strong efficacy signal, real tolerability questions at top doses, Phase 3 will settle it.
13-week sprint vs. 72-week marathon: why direct comparisons don’t work yet
| Drug | Best result | Trial length | N | Source |
|---|---|---|---|---|
| VK2735 injection (Phase 2) | Up to 14.7% | 13 weeks | 176 | Viking VENTURE press release |
| VK2735 oral (Phase 2) | Up to 12.2% | 13 weeks | 280 | Viking VENTURE-Oral press release |
| Wegovy pill (FDA label) | -13.6% | 64 weeks | ~1,600 | FDA Wegovy 25 mg tablet label |
| Zepbound (FDA label) | -20.9% (15 mg) | 72 weeks | ~2,539 | FDA Zepbound label, Study 1 |
| Foundayo (FDA label) | -11.1% (no T2D) | 72 weeks | ~3,000+ | FDA Foundayo label |
Different trial lengths, populations, endpoints, and baseline weights. The fair comparison comes after VANQUISH-1 reads out in 2027.
What Are the Side Effects of VK2735?
The most common side effects in Phase 2 were gastrointestinal — nausea, vomiting, constipation, and diarrhea — the same pattern seen across every GLP-1 class drug. In the injection trial, 43% had nausea (vs. 20% on placebo) and 18% had vomiting (vs. 0% on placebo). In the oral trial, 58% had nausea and 26% had vomiting. Viking reported the majority of these events as mild to moderate, and weekly rates dropped below 5% after the first few weeks.
Injection (VENTURE) side effect profile
| Side effect | VK2735 | Placebo |
|---|---|---|
| Nausea | 43% (mostly mild/moderate; none severe) | 20% |
| Vomiting | 18% | 0% |
| Constipation | 26% | 11% |
| Diarrhea | 20% | 9% |
| Serious adverse events | 1 case of drug-related dehydration | — |
| Discontinuation (all causes) | 13% | 14% |
Oral (VENTURE-Oral) side effect profile
| Side effect | VK2735 (across doses) | Placebo |
|---|---|---|
| Nausea | 58% (99% mild/moderate) | 48% |
| Vomiting | 26% | 10% |
| Adverse-event discontinuation | 20% | 13% |
| Overall discontinuation (15 mg) | 20% | 18% |
| Overall discontinuation (30 mg) | 20% | 18% |
| Overall discontinuation (60 mg) | 28% | 18% |
| Overall discontinuation (90 mg) | 25% | 18% |
| Overall discontinuation (120 mg) | 38% ⚠ | 18% |
Source: Viking IR, Aug 2025. Weekly nausea and vomiting rates dropped below 5% after week 3. Analysts expect Viking to carry the lower, better-tolerated doses forward into Phase 3.
The damaging admission (because you deserve the honest version)
Oral VK2735 looks exciting because it’s a pill, but the Phase 2 data showed meaningfully higher discontinuation than placebo at the most effective doses — 38% at 120 mg. If your priority is an approved, proven oral weight loss medication you can actually stay on — VK2735 isn’t the right answer today.
But as of 2026 there are already two FDA-approved oral weight loss medications available right now: Wegovy pill (oral semaglutide 25 mg, approved December 2025) and Foundayo (orforglipron, approved April 2026). Both come with established safety data and actual availability through licensed telehealth providers.
Is VK2735 a Pill or a Shot?
Viking is developing both — a once-weekly subcutaneous injection and a once-daily oral tablet. They use the same active molecule. The injection is further along (Phase 3 fully enrolled). The pill’s Phase 3 is expected to start Q3 2026.
This is Viking’s unique pitch: most current GLP-1 programs require switching drugs if you want to change forms. Viking is arguing VK2735 could let a patient start on the injection to lose weight quickly, then transition to the oral tablet for long-term maintenance — staying on the exact same compound the whole time.
| Weekly Injection | Oral Tablet | |
|---|---|---|
| Phase | Phase 3 (fully enrolled) | Phase 2 complete; Phase 3 expected Q3 2026 |
| Best Phase 2 result | 14.7% at 13 weeks | 12.2% at 13 weeks |
| Tolerability | Discontinuation balanced with placebo (13% vs 14%) | Discontinuation higher than placebo (28% vs 18%; 38% at 120 mg) |
| Earliest FDA decision | 2028–2029 (editorial projection) | 2029+ (editorial projection) |
Maintenance dosing concepts (the wildcard worth watching)
Viking’s Phase 1 maintenance study is evaluating six maintenance regimens after an initial 19 weeks of weekly subcutaneous induction: weekly subcutaneous, every-other-week subcutaneous, monthly subcutaneous, daily oral, weekly oral, and placebo. Results expected Q3 2026. If monthly injection or weekly oral dosing holds weight loss, VK2735 would be the first dual-agonist to enable that schedule — a real differentiator against Zepbound and Wegovy.
VK2735 vs. Wegovy, Zepbound, Foundayo, and the Full Pipeline
VK2735’s Phase 2 numbers put it in the conversation with tirzepatide (Zepbound) — which shares the same dual GLP-1/GIP mechanism. But Zepbound has 72 weeks of Phase 3 data across 2,500+ patients per its FDA label. VK2735 has 13 weeks of Phase 2 data across 176 patients. Anyone saying VK2735 is "as good as Zepbound" or "better than Wegovy" is comparing a sprint to a marathon.
| Drug | Company | Form | Stage | Best result | Available? |
|---|---|---|---|---|---|
| Wegovy (injection) | Novo Nordisk | Weekly SC | Approved | -14.9% (FDA label) | Yes |
| Wegovy pill | Novo Nordisk | Daily oral | Approved Dec 2025 | -13.6% (FDA label) | Yes |
| Zepbound | Eli Lilly | Weekly SC | Approved | -20.9% at 15 mg (FDA label) | Yes |
| Foundayo (orforglipron) | Eli Lilly | Daily oral | Approved Apr 2026 | -11.1% (FDA label) | Yes |
| VK2735 (injection) | Viking | Weekly SC | Phase 3 | Up to 14.7% (Phase 2) | No |
| VK2735 (oral) | Viking | Daily oral | Phase 2 (Ph3 Q3 '26) | Up to 12.2% (Phase 2) | No |
| Retatrutide | Eli Lilly | Weekly SC | Phase 3 | Up to -28.7% (TRIUMPH-4) | No |
| CagriSema | Novo Nordisk | Weekly SC | Filed/Phase 3 | ~20.4% | No |
| Aleniglipron | Structure Tx | Daily oral | Phase 3 exp. 2H 2026 | 15.3% placebo-adj. at 36 wks | No |
Clinical Readiness Score (editorial framework)
| Path | Evidence | Data maturity | Available? | Our conclusion |
|---|---|---|---|---|
| VK2735 injection | 4/5 | 4/5 | 0/5 | Strongest VK2735 path, but 3+ years from any realistic access. |
| VK2735 oral | 4/5 | 3/5 | 0/5 | Exciting pill play — tolerability at top doses is the wildcard. Phase 3 hasn't started. |
| VK2735 maintenance dosing | 3/5 | 1/5 | 0/5 | Potentially category-defining if monthly dosing works. Too early for any patient decision. |
| FDA-approved GLP-1s (Wegovy, Zepbound, Foundayo) | 5/5 | 5/5 | 5/5 | The only path that actually lets you do something about your weight this week. |
Track VK2735 if you…
- ✓ Are interested in the future of obesity drug development
- ✓ Specifically want an oral GLP-1/GIP combination
- ✓ Are not under medical time pressure
- ✓ Might realistically enroll in a future clinical trial
Don’t wait for VK2735 if you…
- ✗ Are actively trying to lose weight for health reasons now
- ✗ Have diabetes, CV risk, or another condition needing intervention
- ✗ Have insurance coverage for FDA-approved medications
- ✗ Want proven, FDA-labeled prescribing information
Can You Buy VK2735 Right Now?
No. Full stop.
VK2735 is not legally available for purchase anywhere. It is not FDA approved, not on FDA’s drug shortage list, not on FDA’s 503B bulks list, and not sold by any licensed pharmacy or telehealth provider. Any website offering "VK2735 for sale" or "research-grade VK2735" is an unapproved research chemical — not the drug Viking is testing.
What we actually checked
| Database / source | What we looked for | Result |
|---|---|---|
| Drugs@FDA | Any approved VK2735 product | None |
| FDA drug shortage list | VK2735 as a listed shortage drug | Not listed |
| FDA 503B bulks list | VK2735 as approved bulk substance | Not listed |
| ClinicalTrials.gov | Active VK2735 trial enrollment | NCT07104500 and NCT07104383 fully enrolled |
| FDA warning letter database | Enforcement against compounded GLP-1 pipeline drugs | Yes — FDA warned compounders of retatrutide (same situation) |
| Viking Therapeutics IR | Any commercial availability disclosure | None — VK2735 remains investigational |
Why "research chemical" VK2735 is dangerous
A vial labeled "VK2735, 5 mg" purchased from a research-chemical site may contain a different amount of active compound than the label claims, may not have been made under sterile conditions, may not even contain VK2735 at all, and offers no recourse if something goes wrong. No prescribing information, no pharmacy consultation, no liability insurance, no meaningful consumer protection. According to FDA, poor compounding practices can cause contamination, too much or too little active ingredient, and serious injury or death.
Red flags that a site is selling unapproved VK2735 illegally
- “Research use only” disclaimer but marketing targeted at weight loss consumers
- “No prescription needed”
- Claims of being “the same as” an approved medication
- Before-and-after photos
- Crypto-only or anonymous payment
- No company name, business address, or pharmacy license displayed
- No licensed prescriber involved
The only legal way to access VK2735 today
Clinical trial participation. As of April 2026, VANQUISH-1, VANQUISH-2, and the Phase 1 maintenance study are all fully enrolled. The oral Phase 3 program expected to start Q3 2026 will be the next realistic enrollment opportunity. Check ClinicalTrials.gov directly for the latest status.
NCT IDs to track: NCT07104500 (VANQUISH-1), NCT07104383 (VANQUISH-2).
VK2735 Clinical Trial Status (Current as of April 24, 2026)
| Trial | NCT ID | Phase | N | Status | Next milestone |
|---|---|---|---|---|---|
| VANQUISH-1 | NCT07104500 | 3 | ~4,650 | Fully enrolled (Nov 19, 2025) | Study completion 2027 |
| VANQUISH-2 | NCT07104383 | 3 | ~1,000 | Fully enrolled (Mar 26, 2026) | Study completion 2027 |
| Maintenance dosing | Phase 1 | 1 | ~180 | Fully enrolled (Jan 8, 2026) | Results Q3 2026 |
| Oral VK2735 Phase 3 | TBD | 3 (planned) | TBD | Not yet initiated | Expected Q3 2026 |
Should You Wait for VK2735 or Start Something Available Now?

It depends on your situation. Waiting three-plus years for a drug that may or may not be approved is a reasonable choice for some people. Starting an FDA-approved option this month — while VK2735 finishes Phase 3 — is the right choice for most.
| Your situation | The better next step |
|---|---|
| “I just want to understand VK2735.” | You're in the right place. Bookmark this page and check back quarterly. |
| “I want medication I can discuss with a clinician this month.” | Compare current FDA-approved GLP-1s. Don't wait for an unapproved drug. |
| “I only want FDA-approved medication.” | Wegovy, Zepbound, Foundayo, or Wegovy pill — all available today. |
| “I specifically want an oral GLP-1.” | Wegovy pill (oral semaglutide) or Foundayo (orforglipron) are both FDA-approved and available now. |
| “I want to participate in research.” | Monitor ClinicalTrials.gov for VK2735 oral Phase 3 (expected Q3 2026). |
| “Cost is the main thing blocking me.” | HSA/FSA covers most FDA-approved options. Some providers offer free GLP-1 insurance coverage checks. |
| “Someone online is selling VK2735 cheap.” | Close the tab. That's not the drug. It's unsafe. |
If you’ve decided waiting isn’t your path:
Get my personalized GLP-1 action plan (60-sec quiz) →Sorts oral vs injection, insurance vs cash-pay. No email required.
What to Do If You Don’t Want to Wait Until 2028+
If you’ve decided that waiting three-plus years isn’t your path, the two telehealth providers most relevant to the clinical territory VK2735 is trying to enter are Ro and Eden. Neither sells VK2735 — nothing does — but both prescribe currently-approved medications that occupy the same space.
| Provider | What they publicly state | What we verified | What they do NOT offer |
|---|---|---|---|
| Ro | $39 first month / $149 ongoing / as low as $74/mo with annual prepay; carries Zepbound® and Foundayo™; insurance concierge handles prior auth; free GLP-1 insurance coverage checker | Pricing structure, Zepbound and Foundayo lineup, insurance concierge confirmed (April 24, 2026) | Compounded GLP-1s; VK2735 (no provider sells it) |
| Eden | No membership fees; no insurance required; HSA/FSA eligible; online care; free shipping | No-membership-fee structure, HSA/FSA eligibility confirmed (April 24, 2026) | VK2735 (no provider sells it) |
For FDA-approved brand-name GLP-1s (Zepbound, Foundayo)
Ro is the most relevant option. Ro carries Zepbound® (tirzepatide, the closest currently-approved analog to VK2735’s dual GLP-1/GIP mechanism) and Foundayo™ (orforglipron, the new FDA-approved daily pill). Ro’s cash-pay pricing matches LillyDirect/NovoCare, and a dedicated insurance concierge handles prior-authorization paperwork. Ro Body: $39 for the first month, then $149/mo (or as low as $74/mo with annual prepay). Medication cost is separate and depends on insurance.
Check eligibility for Ro’s GLP-1 program → (sponsored affiliate link, opens in a new tab)For broad cash-pay access or HSA/FSA
Eden is a strong broad cash-pay default. No membership fees, accepts no-insurance patients, HSA/FSA eligible, free shipping. If you don’t have insurance coverage for GLP-1s, Eden is a reasonable starting point to see what’s available at what price.
See current Eden GLP-1 pricing and availability → (sponsored affiliate link, opens in a new tab)Not sure which fits? The 60-second quiz covers the full market →
The Expert Perspective
Two public statements worth knowing, both from the oral Phase 2 readout in August 2025:
On the oral VK2735 Phase 2 data, Lian emphasized the progressive weight loss curves across all doses and the absence of a plateau at 13 weeks, positioning the drug as a candidate for longer-duration efficacy in Phase 3.
Placed VK2735’s weight loss results “among the upper echelon” of oral obesity medications following the VENTURE-Oral readout, while flagging the discontinuation rates at higher doses as a tolerability concern investors were watching.
What’s Changing Next (Put These on Your Calendar)
- Q3 2026 Maintenance dosing study readout — will tell us whether weekly oral or monthly injection maintenance is viable
- Q3 2026 Oral VK2735 Phase 3 program initiation — will reopen enrollment windows
- 2027 VANQUISH-1 and VANQUISH-2 study completion — the real efficacy picture arrives here
- Late 2027/2028 Expected NDA submission if Phase 3 succeeds
- 2028–2029 FDA decision on subcutaneous VK2735 (editorial projection)
We’ll update this page within 7 days of any major Viking press release, FDA action, or trial readout. Last verified: April 24, 2026. Next scheduled verification: July 24, 2026.
The Bottom Line
VK2735 is real. The Phase 2 data is promising. The Phase 3 trials are fully enrolled and reading out in 2027. A 2028–2029 FDA decision is plausible if the data holds.
It’s also not something you can access today, not something a pharmacy can dispense, and not something being sold legitimately online right now.
If you’ve been reading about VK2735 because you actually want to lose weight, the honest advice is: don’t wait. Three years is a long time, Phase 3 isn’t a guarantee, and there are four FDA-approved GLP-1 options available right now that have more data behind them than VK2735 will have at launch.
VK2735 Frequently Asked Questions
Is VK2735 FDA approved?+
No. VK2735 is an investigational GLP-1/GIP dual agonist from Viking Therapeutics. The weekly injection is in Phase 3 (VANQUISH-1 and VANQUISH-2, both fully enrolled), and the oral tablet completed Phase 2 with Phase 3 expected to begin Q3 2026. The RX Index editorial projection is a 2028–2029 FDA decision window if Phase 3 succeeds; FDA has not announced an approval date.
Can I buy VK2735 online?+
No. VK2735 is not legally available for purchase. It is not FDA approved, not on the FDA drug shortage list, not on the FDA 503B bulks list, and not eligible for legal compounding. Anything sold online as 'VK2735' is an unapproved research chemical, not the drug being tested in clinical trials.
How much weight did people lose on VK2735?+
In Phase 2 trials, the weekly injection produced up to 14.7% mean body weight loss at 13 weeks, and the daily pill produced up to 12.2% mean body weight loss at 13 weeks. Phase 3 trials over 78 weeks are ongoing.
Is VK2735 a pill or a shot?+
Both. Viking Therapeutics is developing VK2735 as a once-weekly subcutaneous injection and a once-daily oral tablet. Both use the same active molecule. The injection is further along (Phase 3 fully enrolled); the oral tablet's Phase 3 is expected to start Q3 2026.
When will VK2735 be available?+
The earliest realistic availability is late 2028 to 2029 per The RX Index editorial projection, assuming Phase 3 succeeds and FDA approval follows standard timelines. Phase 3 trials are 78 weeks long, Viking has guided completion in 2027, and FDA review typically adds approximately 10 months.
What are the main side effects of VK2735?+
Gastrointestinal — primarily nausea and vomiting. In Phase 2, the injection showed 43% nausea (vs 20% placebo) with discontinuation balanced at 13% vs 14%. The oral tablet showed 58% nausea (vs 48% placebo) with discontinuation ranging 20% to 38% by dose (vs 18% placebo). Most events were mild to moderate.
Is VK2735 the same as Ozempic?+
No. Ozempic is semaglutide, a GLP-1 receptor agonist made by Novo Nordisk and FDA-approved for type 2 diabetes. VK2735 is an investigational GLP-1/GIP dual agonist from Viking Therapeutics — a different molecule, a different mechanism (dual vs. single receptor), and not approved.
Is VK2735 the same as Zepbound?+
No. They share the same drug class (dual GLP-1/GIP agonists), but Zepbound is tirzepatide — a specific molecule approved by the FDA in 2023. VK2735 is a separate, investigational molecule. Zepbound has 72 weeks of Phase 3 data in thousands of patients; VK2735 has 13 weeks of Phase 2 data in fewer than 200 patients for the injection.
Is VK2735 a compounded drug?+
No. VK2735 is a novel investigational compound being developed by Viking Therapeutics. It is not on the FDA shortage list and is not eligible for legal compounding. Do not confuse it with compounded semaglutide or compounded tirzepatide programs.
Will VK2735 be a monthly injection?+
Possibly at maintenance stage. Viking's Phase 1 maintenance study is evaluating monthly subcutaneous dosing alongside every-other-week, weekly subcutaneous, daily oral, and weekly oral dosing. Results are expected Q3 2026.
When will VK2735 Phase 3 results come out?+
VANQUISH-1 and VANQUISH-2 are 78-week studies. Viking has guided study completion in 2027. Full data readout would follow that completion.
How much will VK2735 cost?+
Unknown. Viking has not disclosed pricing. Post-approval pricing will depend on Phase 3 results, label, competitive dynamics, and insurance negotiations.
Sources
Primary sources (Viking Therapeutics)
- Viking IR, Feb 27, 2024 — Phase 2 VENTURE top-line results
- Viking IR, Aug 19, 2025 — Phase 2 VENTURE-Oral top-line results
- Viking IR, Jun 25, 2025 — Phase 3 VANQUISH program initiation
- Viking IR, Nov 19, 2025 — VANQUISH-1 enrollment completion
- Viking IR, Jan 8, 2026 — Maintenance dosing study enrollment completion
- Viking IR, Feb 11, 2026 — Corporate update, oral Phase 3 expected Q3 2026
- Viking IR, Mar 26, 2026 — VANQUISH-2 enrollment completion
Trial registries
- ClinicalTrials.gov: NCT07104500 (VANQUISH-1), NCT07104383 (VANQUISH-2), NCT06828055 (VENTURE-Oral), NCT06068946 (VENTURE)
Peer-reviewed publication
- Phase 2 VENTURE trial results published in Obesity, January 2026
Regulatory sources (all verified April 24, 2026)
- FDA Drugs@FDA database — no VK2735 approval found
- FDA drug shortage list — VK2735 not listed
- FDA 503B bulks list — VK2735 not listed
- FDA warning letter database — ASN-LABS re: compounded retatrutide (September 9, 2025)
- FDA. "Compounding and the FDA: Questions and Answers."
FDA prescribing information (comparison table)
- Wegovy (injection) FDA label; Wegovy oral semaglutide 25 mg tablet FDA label, NDA 218316
- Zepbound (tirzepatide) FDA label, NDA 217806; Foundayo (orforglipron) FDA label, NDA 220934
Industry and clinical coverage
- BioPharma Dive — analyst coverage of VK2735 oral data (August 19, 2025)
- Eli Lilly TRIUMPH-4 retatrutide Phase 3 (December 2025)
- Novo Nordisk amycretin Phase 3 announcement (June 12, 2025)
- Structure Therapeutics aleniglipron Phase 3 data
Last verified: April 24, 2026 · Next scheduled re-verification: July 24, 2026
About this guide: Written and verified by The RX Index Editorial Team — a pricing intelligence and comparison resource for GLP-1 telehealth providers. Editorial conclusions are labeled as such throughout this page.
Medical disclaimer: This page is for informational purposes and is not medical advice. GLP-1 medications are prescription-only and carry potential risks and side effects. Always consult a licensed clinician before starting, stopping, or changing any medication.