Having a seizure right now, or with someone who is?
Call 911 if the seizure lasts more than 5 minutes, another follows quickly, there's injury or trouble breathing, it's a first-ever seizure, the person is pregnant, in water, or has diabetes with loss of consciousness. This page covers decisions you'll make tomorrow — not while a seizure is happening.
Safety Guide · Last verified: April 24, 2026
By the editorial team at The RX Index · 8 FDA/DailyMed labels audited row-by-row · 2 peer-reviewed cohort studies · 2 case reports · Current FDA, WHO, and MHRA counterfeit alerts reviewed · 18-min read
Can GLP-1 Cause Seizures?
What 2026 FDA Labels and Evidence Show
Quick verdict
| Question | What the evidence shows |
|---|---|
| Are seizures a listed FDA side effect of GLP-1s? | No — not in the labels audited on April 24, 2026 for any major FDA-approved GLP-1 or GIP/GLP-1 medication. Exception: Soliqua (lixisenatide + insulin combination) lists seizure under its hypoglycemia warning — an insulin-related risk, not a GLP-1 class risk. |
| Do GLP-1s make seizures more likely in clinical studies? | No. 2025–2026 cohort studies in type 2 diabetes populations show lower, not higher, seizure-related outcomes (recurrence HR 0.82; new-onset epilepsy HR 0.84). |
| Can a GLP-1 still contribute to a seizure in specific situations? | Yes, indirectly — through severe hypoglycemia (mainly with insulin or sulfonylureas), rare SIADH, disrupted anti-seizure medication absorption during GI side effects, or counterfeit product. |
| If I have epilepsy, am I disqualified? | No. Epilepsy is not a contraindication to any FDA-approved GLP-1. The real question is coordination with your neurologist, not whether you're allowed. |

Not sure which risk bucket you're in?
Most readers don't have the risk profile they're worried about — they just haven't had anyone sort it out. The free 60-second assessment asks 5 questions and gives you a personalized list of warning signs for your specific situation.
Take the free GLP-1 + seizure risk self-assessment →Can GLP-1 medications cause seizures?
The short answer: no — not directly, based on the FDA-approved labels and clinical trial data reviewed on April 24, 2026. The word "seizure" does not appear as an adverse reaction on the FDA labels for semaglutide (Ozempic, Wegovy, Rybelsus), tirzepatide (Mounjaro, Zepbound), liraglutide (Saxenda, Victoza), dulaglutide (Trulicity), or orforglipron (Foundayo). Clinical trials involving tens of thousands of participants have not identified seizures as a class-level safety signal.
The distinction most people miss:
- Direct causation means the medication itself triggers the seizure. The evidence does not support this for major FDA-approved GLP-1 medications.
- Indirect contribution means the medication causes something else (low blood sugar, dehydration, disrupted medication absorption) that can lower the seizure threshold in someone already susceptible. The evidence supports this — for specific, identifiable patient profiles.
Why "not on the label" doesn't mean "zero cases have ever happened"
Published case reports describe rare serious events in people taking GLP-1 medications. An October 2025 paper in JCEM Case Reports described a 63-year-old woman who developed tonic-clonic seizures four days after starting tirzepatide, caused by SIADH-induced hyponatremia. A 2026 paper in Neurological Sciences described a probable case of tirzepatide-associated limbic encephalitis with status epilepticus. Case reports generate hypotheses — they don't establish causation for the general population. But "rare" isn't "impossible," which is why we cover them in detail below.
What FDA labels actually say about GLP-1s and seizures
We audited the current DailyMed prescribing information for the major FDA-approved GLP-1 and GIP/GLP-1 medications on April 24, 2026. Every row links to its primary source.
| Medication | Active ingredient | "Seizure" listed as adverse reaction? | Label-supported seizure-relevant pathway |
|---|---|---|---|
| Wegovy | Semaglutide (injection) | No | Hypoglycemia risk increases with insulin or sulfonylurea; GI effects and volume depletion during titration; delayed gastric emptying may affect oral medications |
| Ozempic / Rybelsus | Semaglutide (injectable / oral) | No | Hypoglycemia risk higher with insulin or sulfonylurea; dehydration from GI effects; delayed gastric emptying may impact oral medications |
| Mounjaro | Tirzepatide | No | Hypoglycemia risk with insulin or sulfonylurea; dehydration from GI effects; delayed gastric emptying may affect oral medications |
| Zepbound | Tirzepatide | No | Label notes Zepbound lowers blood glucose; hypoglycemia reported in adults without type 2 diabetes; higher risk with insulin or sulfonylurea |
| Foundayo | Orforglipron (oral) | No | Hypoglycemia under 54 mg/dL reported, higher with sulfonylurea; CYP3A4-metabolized — avoid strong CYP3A4 inducers like carbamazepine; volume depletion monitoring |
| Saxenda | Liraglutide | No | Adult hypoglycemia risk with insulin/sulfonylurea; higher rate of symptomatic hypoglycemia in pediatric trial vs. placebo |
| Victoza | Liraglutide | No | Adult and pediatric hypoglycemia risk; dehydration-related acute kidney injury reported |
| Trulicity | Dulaglutide | No | Hypoglycemia rates higher in combination with insulin or sulfonylurea |
| Soliqua 100/33 (combination) | Lixisenatide + insulin glargine | Yes — under hypoglycemia warning | Because Soliqua contains insulin, its hypoglycemia warning explicitly states that severe hypoglycemia can cause seizures. This is a labeled insulin-related risk, not a GLP-1 class risk. |
Source: DailyMed prescribing information as published by the U.S. National Library of Medicine, verified April 24, 2026. We verify this audit quarterly.
In plain English: if your fear is that a GLP-1 will directly fire off a seizure in your brain the way some older psychiatric medications are known to, the FDA-reviewed evidence does not support that fear for any of the major stand-alone GLP-1 medications. What you should pay attention to is whether your personal setup involves insulin, a sulfonylurea, severe vomiting, an oral seizure medication with a narrow therapeutic window, or a product from an unverified source.
What the 2025–2026 real-world evidence actually shows
Two large independent real-world cohort studies published in the past six months — one in Epilepsia (2026), one in Neurology (2026) — both found that starting a GLP-1 medication was associated with lower, not higher, seizure outcomes in adults with type 2 diabetes. These findings are consistent with a 2024 meta-analysis of 27 randomized trials.
| Study | Population | What it supports | What it does NOT prove |
|---|---|---|---|
| AbuAlrob 2026 Epilepsia (8,688 matched pairs) | Adults with established epilepsy + T2D | Lower seizure recurrence, hospitalization, mortality, status epilepticus in GLP-1 initiators | Causation; applicability to non-diabetic weight-only users |
| Cheng 2026 Neurology (452,766 patients) | Adults with T2D, no prior epilepsy | Lower incident epilepsy risk vs. DPP-4 inhibitors | Anything about tirzepatide; applicability to people who already have epilepsy |
| Sindhu 2024 Epilepsia Open meta-analysis (27 RCTs, ~200,000 participants) | Pooled RCT populations | 24% lower risk of new-onset seizure/epilepsy | Individual-patient risk outside the pooled average |
The 2026 Epilepsia cohort — 8,688 matched adults with epilepsy
AbuAlrob et al. pulled de-identified records from the TriNetX Research Network across 220+ healthcare organizations (January 2003 – August 2025). Adults with at least three documented epilepsy diagnoses, comparing 8,688 who newly started a GLP-1 against a propensity-matched group who started a different glucose-lowering medication.
| Outcome | GLP-1 group vs. comparator | Hazard ratio (95% CI) |
|---|---|---|
| Seizure recurrence | 18% lower risk | HR 0.82 (0.78–0.86) |
| Hospitalization | 65% lower risk | HR 0.35 (0.29–0.43) |
| All-cause mortality | 60% lower risk | HR 0.40 (0.34–0.47) |
| Status epilepticus | 25% lower risk | HR 0.75 (0.66–0.85) |
The 2026 Neurology cohort — 452,766 patients
Cheng et al. at Chung Shan Medical University used TriNetX to compare new-onset epilepsy risk in adults with T2D starting a GLP-1 vs. a DPP-4 inhibitor. After propensity matching, 226,383 in each group:
- Cumulative incidence of new epilepsy: 2.35% in GLP-1 users vs. 2.41% in DPP-4 users
- Hazard ratio for new epilepsy: 0.84 — a 16% lower risk in GLP-1 users
- Protective association held at 1, 3, and 5 years (HRs 0.71, 0.81, 0.82)
- Among individual agents, semaglutide showed the strongest association, HR 0.68
What's not settled
- Whether GLP-1s prevent seizures — FDA has not approved any GLP-1 for seizure prevention
- Whether the neuroprotective signal is real or confounding
- What happens for individual patients outside the pooled averages
- Whether newer oral small-molecule GLP-1s like Foundayo behave like injectable peptides long-term
- Tirzepatide-specific new-onset epilepsy risk in diabetes (not in the Cheng cohort)
The four indirect mechanisms that actually matter
Four real but narrow indirect mechanisms can connect GLP-1 use to seizure activity. Each applies to a specific subset of users. Each is preventable with the right preparation. If none of these four apply to you, your risk is very low.
| Mechanism | Who it applies to | Evidence level | Prevention step | Action threshold |
|---|---|---|---|---|
| Severe hypoglycemia | Users combining a GLP-1 with insulin or a sulfonylurea | FDA-labeled, established | Prescriber reduces insulin or sulfonylurea dose; carry fast-acting carbs | Treat with 15 g carbs; 911 for seizure or inability to self-treat |
| SIADH with severe hyponatremia | Rare; documented with tirzepatide; possible class effect | Case-report level | Baseline electrolytes recommended, especially off-label | 911 for seizure or severe confusion |
| Disrupted AED absorption | People with epilepsy on narrow-therapeutic-index oral AEDs | Pharmacologically plausible | Neurologist coordination; AED blood levels at baseline and around escalations | Same-day neurologist call for any pattern change |
| Severe vomiting / dehydration | Anyone with persistent GI side effects | Well-established GI mechanism | Antiemetic plan; written 'what to do if I vomit my AED' plan | Same-day prescriber call; ER for severe dehydration |
Mechanism 1: Hypoglycemia with insulin or sulfonylurea (labeled, established)
This is the FDA-labeled pathway. Every GLP-1 medication lowers blood glucose. If you're also taking insulin or a sulfonylurea (glipizide, glyburide, glimepiride), the combined glucose-lowering effect can drive blood sugar dangerously low. CDC identifies blood sugar below 54 mg/dL as severe low blood sugar and lists seizures among severe symptoms.
Who it applies to: type 2 diabetes patients on insulin or sulfonylureas. Much less likely in non-diabetic users — but Foundayo's label reports hypoglycemia under 54 mg/dL even in adults without type 2 diabetes.
How it's prevented: your prescriber reduces your insulin or sulfonylurea dose when adding a GLP-1. This is standard practice, not optional. If your prescriber didn't mention this when starting you on a GLP-1 and you're on insulin or a sulfonylurea, call them today.
When to act: treat hypoglycemia immediately with 15 g fast-acting carbs (4 oz juice, glucose tablets, regular soda). Retest in 15 minutes. Call 911 for any seizure, loss of consciousness, or inability to self-treat.
Mechanism 2: SIADH and severe hyponatremia (rare, case-report level)
SIADH (syndrome of inappropriate antidiuretic hormone secretion) causes your body to hold onto too much water, diluting blood sodium. Severe hyponatremia (generally under 125 mmol/L) can cause seizures and brain swelling.
The most prominent published case: a 63-year-old woman with no significant medical history developed tonic-clonic seizures four days after starting tirzepatide for weight loss. Her serum sodium was 122 mmol/L. Discontinuing tirzepatide and restricting fluids restored her sodium to 134 mmol/L within 5 days and resolved the seizures. She remained seizure-free at two-week follow-up.
Watch for: new unexplained confusion, severe headache not relieved by usual remedies, nausea with altered mental state, seizure activity. When to act: 911 for seizure or severe confusion.
Mechanism 3: Disrupted absorption of anti-seizure medications
GLP-1s slow gastric emptying, particularly during the first weeks of therapy and after each dose increase. For most oral medications this is clinically irrelevant. For oral anti-seizure medications with a narrow therapeutic index — phenytoin, carbamazepine, lamotrigine, valproate — even modest absorption changes can move blood levels around.
Foundayo is the exception. Foundayo (orforglipron) is metabolized by CYP3A4, and its label instructs prescribers to avoid strong CYP3A4 inducers. Carbamazepine is a strong CYP3A4 inducer. If you're on carbamazepine and considering Foundayo, that's a specific conversation to have with your neurologist and prescriber.
How it's prevented: neurologist should be in the loop before you start. Ask about baseline AED blood levels and a re-check schedule — typically 4–6 weeks after starting and after each dose increase.
Mechanism 4: Severe vomiting that disrupts hydration and medication adherence
Severe, persistent vomiting is uncommon but known during early dose escalation. For someone with epilepsy, vomiting an oral anti-seizure medication within an hour of taking it creates an absorption-uncertainty window. The Epilepsy Foundation identifies missed medication as the most common cause of breakthrough seizures.
How it's prevented: have an antiemetic plan before you start. If you have epilepsy, ask your neurologist for a written "what to do if I vomit my AED" plan before you start the GLP-1 — not at 2 a.m. when it happens.
If none of the four applies to you
If you're a non-diabetic adult taking a legitimate FDA-approved GLP-1 from a licensed US pharmacy, not on insulin or sulfonylureas, without a seizure history, without severe GI side effects, and without new neurologic symptoms — the best current evidence puts your seizure risk in the "very low" category. That's not a medical guarantee — it's a practical risk tier based on the evidence framework on this page.
Can you take a GLP-1 if you have epilepsy?
Yes, in most cases. Epilepsy is not listed as a contraindication on the FDA-approved prescribing information for any stand-alone GLP-1 medication. The 2026 Epilepsia cohort study found GLP-1 initiation was associated with an 18% lower — not higher — risk of recurrent seizures in adults with epilepsy and type 2 diabetes. The practical requirements are coordination with your neurologist, monitoring anti-seizure drug levels during titration, and a clear plan for handling GI side effects.
- 18% lower seizure recurrence in GLP-1 initiators (HR 0.82, 95% CI 0.78–0.86)
- 25% lower risk of status epilepticus (HR 0.75, 95% CI 0.66–0.85)
- 65% lower hospitalization risk, 60% lower all-cause mortality
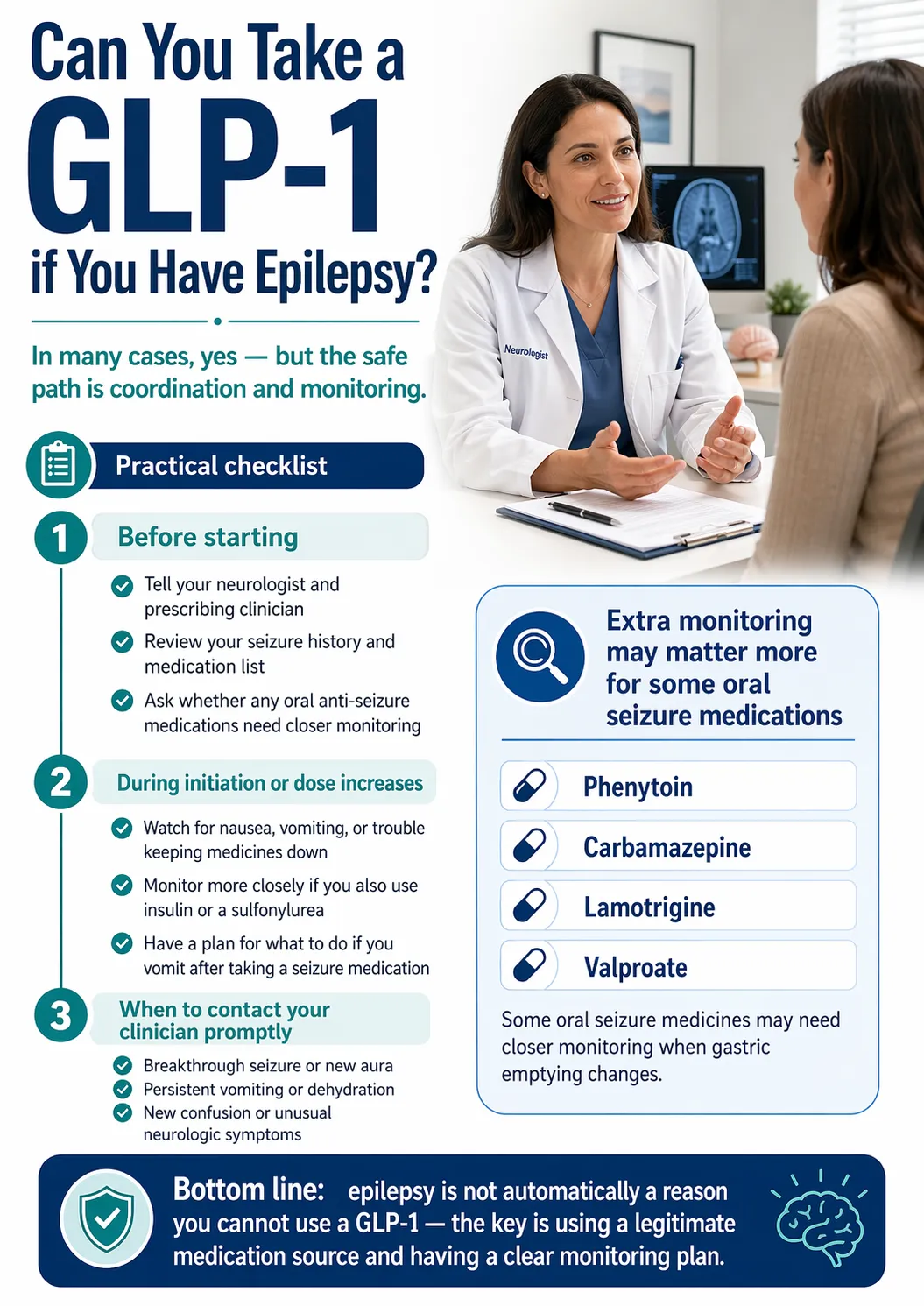
Eight specific questions to bring to your appointment
- Given my current AED regimen, are any of my medications in the narrow-therapeutic-index group where absorption changes matter?
- Do you want baseline AED blood levels before I start, and which specific levels?
- After initiation and after each GLP-1 dose increase, what's your recheck schedule?
- If I vomit within an hour of taking my AED, what exactly do you want me to do? (Ask for written instructions.)
- At what threshold of GI symptoms do you want me to pause the GLP-1 escalation?
- Am I on insulin or a sulfonylurea, and if so, how will we adjust those doses?
- What signs would make you want to stop the GLP-1 entirely?
- If I'm on carbamazepine and considering Foundayo, is that combination okay for me? (Foundayo's label instructs avoidance of strong CYP3A4 inducers; carbamazepine is one.)
Contraception note for women with epilepsy of reproductive age
GLP-1 provider pages often bury this. Foundayo's label specifically instructs females using oral contraceptives to switch to a non-oral contraceptive method or add a barrier method for 30 days after initiation and for 30 days after each dose escalation. For injectable peptide GLP-1s, similar guidance exists in some labels — ask your prescriber about your specific medication.
When a GLP-1 might not be the right move right now
- If your seizures aren't currently well-controlled
- If you're mid-titration on a new AED
- If you've had recent breakthrough seizures you and your neurologist haven't made sense of
- If you had a seizure within the past 90 days and you're still in the workup phase
In those cases, the answer isn't "no, never." It's "not yet, let's stabilize first."
For neurologist-cleared readers only
If your neurologist gives you the green light
Ro is our lead recommendation for confirmed-epilepsy, neurologist-cleared readers. Ro has a dedicated insurance concierge that handles prior-authorization paperwork, a free GLP-1 Insurance Coverage Checker, and access to Zepbound® (tirzepatide) and Foundayo™ (orforglipron). For medically complex readers with seizure concerns, FDA-approved brand-name options are the cleaner path — compounded products carry dosing-error risks you don't need layered on top of AED-absorption complexity.
$39 first month, then as low as $74/month with annual plan paid upfront (or $149/month standard). Membership and medication billed separately.
Check your insurance coverage on Ro →Low-pressure eligibility check — no commitment, no obligation to start. Use only after neurologist clearance. Affiliate disclosure: we may earn a commission.
GLP-1s and anti-seizure medications: what to actually monitor
Injectable peptide GLP-1s (semaglutide, tirzepatide, liraglutide, dulaglutide) don't significantly inhibit the liver enzymes that metabolize most anti-seizure medications. The practical concern is absorption timing — GLP-1s slow gastric emptying during the first weeks of therapy and after each dose increase. Foundayo (orforglipron) is different: it's CYP3A4-metabolized, so its label instructs avoidance of strong CYP3A4 inducers — and carbamazepine is one of those.
| AED | Generic name | Absorption concern (all GLP-1s) | Foundayo-specific concern |
|---|---|---|---|
| Phenytoin | Dilantin | High — narrow therapeutic index | Not a strong CYP3A4 inducer in typical use, but discuss |
| Carbamazepine | Tegretol, Carbatrol | High — narrow therapeutic index | Yes — strong CYP3A4 inducer; Foundayo label instructs avoidance |
| Lamotrigine | Lamictal | High — small changes can affect seizure control | Primarily glucuronidation — different pathway |
| Valproate / valproic acid | Depakote | High — narrow therapeutic index | Limited direct interaction data |
| Oxcarbazepine | Trileptal | Moderate | Moderate CYP3A4 inducer — ask prescriber about Foundayo monitoring |
| Topiramate | Topamax | Moderate | — |
| Levetiracetam | Keppra | Usually lower priority | — |
| Lacosamide | Vimpat | Usually lower priority | — |
| Zonisamide | Zonegran | Usually lower priority | — |
General guidance from clinical literature and Foundayo's prescribing information. Not a substitute for individualized advice — ask your neurologist about your specific regimen.
When to request AED blood level checks
- Baseline: before starting the GLP-1
- 4–6 weeks after starting: after you've completed the first dose level
- After each dose escalation: the gastric-emptying effect is strongest early and after each bump
- Any time you have persistent GI symptoms: vomiting or severe nausea that might be affecting absorption
Tirzepatide and seizures: what the 2025–2026 case reports describe
Two seizure-focused tirzepatide case reports we reviewed describe rare but serious events. Both remain rare against millions of prescriptions, and neither establishes a class-level seizure risk.
The October 2025 SIADH case (Shah et al., JCEM Case Reports)
Patient: 63-year-old woman, no significant past medical history. Started tirzepatide four days earlier via an online pharmacy. Presentation: tonic-clonic seizures; serum sodium 122 mmol/L (severe hyponatremia); consistent with SIADH.
Outcome: tirzepatide discontinued; sodium restored to 134 mmol/L within 5 days; seizure-free at 2-week follow-up.
Authors' conclusion: tirzepatide may induce SIADH even without predisposing factors. Electrolyte monitoring recommended when initiating GLP-1 receptor agonists, particularly off-label.
The 2026 limbic encephalitis case (Mazzeo et al., Neurological Sciences)
Patient: adult with extreme obesity on tirzepatide. Presentation: probable tirzepatide-associated autoimmune limbic encephalitis with status epilepticus; Naranjo score of 8 (probable adverse drug reaction).
Authors' conclusion: emerging reports describe tirzepatide-associated autoimmune encephalitis. Serious neurological adverse events — although rare — warrant neurological vigilance when using dual GLP-1/GIP receptor agonists.
How to read case reports without panicking
Case reports identify signals — they don't establish population-level risk. One case, or two, in a drug taken by millions means "this has happened to someone, ever" — not "this will happen to you." But it does mean two things:
- Don't dismiss new neurologic or psychiatric symptoms during initiation. Confusion, severe headache, behavior changes, seizure activity — worth an urgent call.
- Baseline electrolytes are a reasonable ask. Especially for off-label weight-loss use without diabetes.
Foundayo (orforglipron) and seizures
Foundayo (orforglipron) was FDA-approved on April 1, 2026 as the first oral small-molecule GLP-1 receptor agonist. Its prescribing information does not list seizures as an adverse reaction. Because Foundayo is metabolized by CYP3A4, its drug-interaction profile differs from injectable peptide GLP-1s — and that matters if you're on carbamazepine.
- Foundayo lowers blood glucose and can cause hypoglycemia under 54 mg/dL (2% of patients in Trial 2; 7% with sulfonylurea)
- Strong CYP3A4 inducers to be avoided — carbamazepine is one. The Foundayo label explicitly instructs avoidance
- Moderate CYP3A4 inducers (like oxcarbazepine) require monitoring and possibly dose escalation of Foundayo
- Delayed gastric emptying still applies during titration — same absorption-timing caution as all GLP-1s
- Oral contraceptive interactions: switch to non-oral method or add barrier method for 30 days after initiation and each dose escalation
Foundayo is available through providers including Ro. Because this page focuses on safety, not provider comparison, pricing details are on our dedicated Foundayo guide.
Counterfeit and gray-market GLP-1s: the preventable risk nobody talks about enough
Some of the most severe adverse events linked publicly to GLP-1 medications have involved counterfeit or unapproved products. The evidence differs by country:
What the MHRA documented (UK)
The UK's MHRA confirmed falsified Ozempic and Saxenda pens containing insulin. Multiple hospitalizations occurred. Because insulin was in the pens instead of semaglutide or liraglutide, non-diabetic users experienced severe hypoglycemic shock. The MHRA explicitly warns that severe low blood sugar can cause seizures or loss of consciousness. This is the most clearly documented counterfeit-to-seizure pathway in the public record.
What the FDA documented (US)
The FDA issued counterfeit Ozempic alerts including lot number PAR1229 (December 2024). Six adverse event reports were associated with the counterfeit lot; the FDA stated the events were consistent with authentic Ozempic and testing was underway. The FDA has not specifically attributed the US counterfeit lots to insulin content or to seizures.
How to verify your product is legitimate
- Source: licensed US pharmacy, operating under a prescriber's supervision
- Packaging: sealed, tamper-evident, with a legitimate lot number and manufacturer information
- Red flags: product shipped from outside the US, offered without a prescription process, sold as "powder" for user reconstitution, or priced at a fraction of normal US prices
Compounded ≠ counterfeit (important distinction)
A legitimately compounded GLP-1 — prepared by a licensed US compounding pharmacy under a prescriber's supervision — is not the same as a counterfeit product. Compounding operates within a regulated framework. Counterfeits do not. That said, the FDA has issued alerts about dosing errors with compounded injectable semaglutide and tirzepatide — errors where patients received doses many times higher than intended because vial concentrations were confused with pen doses. For a reader with seizure concerns, that extra layer of dosing-variability risk argues for FDA-approved brand-name options.
What to do if you suspect counterfeit exposure
- Call 911 for any acute symptoms (seizure, severe hypoglycemia, loss of consciousness)
- Stop using the product immediately
- Preserve the vial, pen, packaging, lot number, and any receipts — don't discard
- Notify your prescriber
- Report to FDA via MedWatch (fda.gov/medwatch or 1-800-FDA-1088)
- Report to your state board of pharmacy
What to do if you had a seizure while taking a GLP-1
Treat the seizure as a medical event first. A clinician needs to evaluate glucose, hydration, electrolytes, medication levels or adherence, recent dose changes, vomiting, sleep loss, illness, and other triggers. No article can tell you whether a specific seizure was caused by a GLP-1 — that takes testing and clinical judgment.
| Detail to document | Why it matters |
|---|---|
| GLP-1 name, brand, and dose | Helps assess drug class and dose escalation |
| Date and time of last GLP-1 dose | Seizure timing relative to medication exposure |
| Recent dose increase (when and by how much) | Side effects intensify during escalation |
| Food intake in the 24 hours before the seizure | Low intake worsens hypoglycemia risk |
| Vomiting or diarrhea in the past 48 hours | Can affect hydration and oral medication absorption |
| Blood glucose reading if available | Helps evaluate hypoglycemia as a trigger |
| Seizure medication name, dose, and last dose timing | Missed or vomited doses matter |
| Sleep, alcohol, illness, stress in the past week | Common seizure triggers unrelated to medication |
| Product source (licensed US pharmacy, compounded, other) | Affects counterfeit evaluation |
Tell your prescriber — not just the emergency department. The prescriber is the one who decides whether to continue, hold, or discontinue. Also report to FDA MedWatch regardless of cause — reports are part of how safety signals are identified.
When to call 911 vs. your doctor vs. hold the next dose
| Call 911 immediately | Call your doctor today | Hold dose and contact tomorrow |
|---|---|---|
| Seizure lasts more than 5 minutes | Any seizure that resolved (if known epilepsy, follow seizure action plan) | Persistent mild nausea or mild dehydration |
| Another seizure follows quickly | New severe confusion or memory changes | Mild fatigue or dizziness |
| Trouble breathing or waking after a seizure | Inability to keep fluids down for 24+ hours | — |
| Injury from a seizure | Severe headache that's new or different | Bruising or injection-site irritation |
| First-ever seizure | Signs of severe low blood sugar not relieved by fast carbs | Slight change in taste or appetite |
| Pregnancy with seizure | Aura or unusual neurologic sensation | — |
| Seizure in water | Missed or vomited AED dose — call neurologist/pharmacist today; missed doses are the most common cause of breakthrough seizures | — |
| Diabetes with loss of consciousness | New behavioral or psychiatric symptoms during tirzepatide use | — |
| Chest pain or irregular heartbeat | — | — |
| Severe dehydration with altered mental state | — | — |
Aligned to CDC seizure first-aid guidance. Rule of thumb: if you're reading this table trying to decide whether something is "bad enough," that uncertainty is itself a reason to call.
Questions to bring to your prescriber and neurologist
For your prescribing clinician (GLP-1 prescriber)
- Given my medication list, is there any absorption interaction I should know about?
- Do you want baseline labs before I start — specifically a baseline sodium level?
- What's your titration schedule, and when should I expect the highest GI-side-effect window?
- If I'm on insulin or a sulfonylurea, how will you adjust those doses?
- What's your after-hours contact process for severe vomiting, confusion, or neurologic symptoms?
- What specific signs would make you want me to pause the GLP-1?
- Is my product from an FDA-approved source or compounded? If compounded, from what pharmacy?
For your neurologist (if you have epilepsy)
- Are any of my AEDs in the narrow-therapeutic-index category?
- Do you want baseline AED blood levels before I start?
- What's your recheck schedule after initiation and each dose escalation?
- If I vomit within an hour of taking my AED, what exactly do I do? May I have that in writing?
- At what GI threshold do you want me to pause escalation?
- If I'm on carbamazepine and considering Foundayo, is that combination okay? (Foundayo is CYP3A4-metabolized.)
- Are you aware of the 2026 Epilepsia cohort data showing lower seizure recurrence in GLP-1 users?
What the March 2026 FDA warning letter to Novo Nordisk actually means
In March 2026, the FDA issued a warning letter to Novo Nordisk (maker of Ozempic and Wegovy) citing postmarketing adverse drug experience reporting violations — meaning the company didn't report certain serious adverse events within required timeframes. The letter cited specific examples including a disabling stroke in a patient on liraglutide and a death case associated with semaglutide.
| What it IS | What it is NOT |
|---|---|
| A pharmacovigilance compliance issue — Novo Nordisk didn't report certain events within required timeframes | A recall of Ozempic, Wegovy, or any other Novo Nordisk product |
| A requirement that Novo Nordisk submit corrective actions | A new finding that GLP-1s cause seizures |
| A reminder that FAERS data may be incomplete for Novo Nordisk products — independent EHR-based cohort data becomes relatively more valuable | A finding that Novo Nordisk caused the cited events |
Your GLP-1 + seizure risk self-assessment
A 60-second tool that takes your medication profile, seizure history, and product source into account and outputs a personalized risk tier, specific warning signs for your situation, and the exact questions to bring to your prescriber.
| Tier | Description | Recommended action |
|---|---|---|
| Emergency now | Active symptoms requiring immediate evaluation | Call 911. Do not wait. |
| Pre-start clinician review required | Confirmed epilepsy, AED on narrow-therapeutic-index list, carbamazepine + Foundayo, or insulin/sulfonylurea use | Pause; coordinate with neurologist and/or prescriber first |
| Use a monitored pathway | Risk factors present but manageable with proper oversight | Start only with a provider that screens history and supports side-effect management |
| Standard vigilance | No active risk factors beyond baseline | Proceed with standard monitoring |
Frequently asked questions
Does Ozempic cause seizures?
Ozempic's FDA-approved prescribing information does not list seizures as an adverse reaction. Hypoglycemia is a labeled risk, and it increases when Ozempic is combined with insulin or sulfonylureas — severe hypoglycemia (below 54 mg/dL) can trigger seizures. Most public reports of seizure events linked to Ozempic have involved either counterfeit product or severe hypoglycemia in specific medication combinations.
Can Wegovy cause seizures?
Wegovy contains the same active ingredient as Ozempic (semaglutide). Its FDA label does not list seizures as an adverse reaction. The practical seizure-relevant concern is hypoglycemia, which is uncommon among non-diabetic Wegovy users but can occur in people also taking insulin or sulfonylureas.
Can people with epilepsy take Ozempic, Wegovy, or Mounjaro?
Yes, in most cases. Epilepsy is not listed as a contraindication to any of these medications. The 2026 Epilepsia cohort study of 8,688 matched adults found an 18% lower risk of seizure recurrence in people with epilepsy who started a GLP-1 versus other glucose-lowering drugs. The practical requirements are neurologist coordination, monitoring anti-seizure medication blood levels during GLP-1 titration, and a clear plan for handling GI side effects that could disrupt AED absorption.
Can Mounjaro or Zepbound cause seizures?
Tirzepatide (the active ingredient in Mounjaro and Zepbound) is not listed as a cause of seizures on its FDA label. Two published case reports describe rare serious events: a 2025 JCEM Case Reports paper on tirzepatide-associated SIADH with hyponatremia-induced seizures, and a 2026 Neurological Sciences paper on probable tirzepatide-associated limbic encephalitis with status epilepticus. These cases remain rare against millions of prescriptions and do not establish a class-level seizure risk.
Can low blood sugar from a GLP-1 cause a seizure?
Yes, severe hypoglycemia can cause seizures. CDC identifies blood sugar below 54 mg/dL as severe low blood sugar and lists seizures among severe symptoms. GLP-1s alone rarely cause severe hypoglycemia. The main risk comes when a GLP-1 is combined with insulin or a sulfonylurea without appropriate dose adjustment of those glucose-lowering drugs.
Do GLP-1 medications interact with anti-seizure drugs?
Injectable peptide GLP-1s (semaglutide, tirzepatide, liraglutide, dulaglutide) don't significantly affect the liver enzymes that metabolize most anti-seizure medications, so classical pharmacokinetic interactions are not expected. The practical concern is absorption — GLP-1s slow gastric emptying, which can shift how quickly oral AEDs are absorbed. For medications with a narrow therapeutic index (phenytoin, carbamazepine, lamotrigine, valproate), blood level monitoring during GLP-1 initiation is a reasonable precaution. Foundayo is different — it's metabolized by CYP3A4, so its label instructs avoidance of strong CYP3A4 inducers like carbamazepine.
Does Foundayo (orforglipron) interact differently with carbamazepine?
Yes. Foundayo is metabolized by CYP3A4, and its FDA-approved prescribing information instructs avoidance of strong CYP3A4 inducers. Carbamazepine is a strong CYP3A4 inducer. That makes the Foundayo plus carbamazepine combination a specific conversation to have with your prescriber and neurologist before starting. Moderate CYP3A4 inducers like oxcarbazepine require monitoring and possible dose escalation of Foundayo.
Does Foundayo cause seizures?
Foundayo was FDA-approved on April 1, 2026, and its prescribing information does not list seizures as an adverse reaction. The same four indirect mechanisms apply (hypoglycemia with insulin or sulfonylurea, rare SIADH, AED absorption timing, severe dehydration from vomiting), plus the CYP3A4 metabolism concern specific to Foundayo.
Are seizures a side effect of counterfeit Ozempic?
Documented cases exist primarily from the UK, where the MHRA confirmed falsified Ozempic and Saxenda pens containing insulin and reported hospitalizations from severe hypoglycemia — including that severe low blood sugar can cause seizures or loss of consciousness. In the US, the FDA has issued counterfeit Ozempic alerts including lot number PAR1229 in December 2024, but has not specifically attributed the US counterfeit lots to insulin content or to seizures.
What if I missed or vomited my seizure medication while on a GLP-1?
Don't improvise a re-dose. Follow your neurologist or pharmacist's written plan — and if you don't have one yet, call today to get one before you need it. The Epilepsy Foundation identifies missed medication as the most common cause of breakthrough seizures. For AEDs with a narrow therapeutic index (phenytoin, lamotrigine, carbamazepine, valproate), individual guidance matters more than general advice.
What should I do if I have a seizure while on a GLP-1?
Call 911 immediately if the seizure lasts more than 5 minutes, another seizure follows quickly, there's injury or trouble breathing, it's your first seizure, you're pregnant, you're in water, or you have diabetes with loss of consciousness. Afterward, preserve the medication and packaging, notify your prescribing clinician (not just the emergency department), and report to FDA MedWatch if you suspect the product is counterfeit. Do not restart the medication without explicit clinical direction.
What we actually verified for this page
Because this is a safety page, you should know exactly what we checked and when. Last verified: April 24, 2026.
- Current DailyMed prescribing information for Wegovy, Ozempic, Rybelsus, Mounjaro, Zepbound, Foundayo, Saxenda, Victoza, Trulicity, and Soliqua 100/33 — searched each label for the word "seizure" and recorded seizure-relevant warnings
- The 2026 Epilepsia cohort study (AbuAlrob et al., Epilepsia 2026;67:1246–1255, doi:10.1111/epi.70022)
- The 2026 Neurology cohort study (Cheng et al., doi:10.1212/WNL.0000000000214509)
- The October 2025 JCEM Case Reports tirzepatide-SIADH paper (Shah et al., doi:10.1210/jcemcr/luaf261)
- The 2026 Neurological Sciences tirzepatide-limbic-encephalitis paper (Mazzeo et al., doi:10.1007/s10072-026-08977-7)
- FDA counterfeit Ozempic alerts, including lot number PAR1229
- MHRA falsified GLP-1 pen reports (UK) — insulin-containing counterfeits and documented hospitalizations
- FDA warning letter to Novo Nordisk (March 5, 2026, Letter ID 717576)
- Ro's current program pricing and formulary (verified April 24, 2026)
- CDC first aid for seizures and hypoglycemia guidance
- Epilepsy Foundation guidance on missed medications as a seizure trigger
We will re-verify the FDA labels, cohort study updates, Ro pricing, and FDA counterfeit alerts on a quarterly cadence (next review: July 2026) and update the "Last verified" date at the top of the page.
Still not sure which GLP-1 program is right for you?
Answer a few questions about your medical history, goals, and preferences, and get a personalized shortlist of legitimate US telehealth programs that fit your profile — not a generic top-10 list designed to sell you the highest-paying option.
Take the free GLP-1 matching quiz →No email required to see results.
Primary sources
- AbuAlrob MA, et al. "Seizure recurrence after GLP-1 receptor agonist initiation in adults with epilepsy." Epilepsia 2026;67:1246–1255. doi:10.1111/epi.70022
- Cheng CY, et al. "Association Between GLP-1 Receptor Agonist Use and Epilepsy Risk in Type 2 Diabetes." Neurology 2026. doi:10.1212/WNL.0000000000214509
- Sindhu U, et al. "Newer glucose-lowering drugs reduce the risk of late-onset seizure and epilepsy: A meta-analysis." Epilepsia Open 2024;9(6):2528–2536.
- Shah I, Sennik D, Veettil FAV. "Tirzepatide-Induced SIADH Presenting With Seizures." JCEM Case Reports 2025;3(12):luaf261. doi:10.1210/jcemcr/luaf261
- Mazzeo A, Alaimo V, Grimaldi LM. "Probable tirzepatide-associated limbic encephalitis with status epilepticus: a case report." Neurological Sciences 2026;47:373. doi:10.1007/s10072-026-08977-7
- FDA-approved prescribing information for Ozempic, Wegovy, Rybelsus, Mounjaro, Zepbound, Foundayo, Saxenda, Victoza, Trulicity, and Soliqua 100/33 (DailyMed, current as of April 24, 2026).
- FDA. "FDA warns consumers not to use counterfeit Ozempic found in U.S. drug supply chain." December 2024.
- FDA. "FDA alerts health care providers and patients of dosing errors associated with compounded injectable semaglutide products."
- FDA Warning Letter to Novo Nordisk. March 5, 2026. Letter ID 717576.
- MHRA. Alert on falsified Ozempic and Saxenda pens.
- CDC. Seizure First Aid. Hypoglycemia guidance.
- Epilepsy Foundation. "Missed Medication."
About this page
The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We don't manufacture, prescribe, or sell medications. This page was researched and written by The RX Index Editorial Team. It is educational, not medical advice. Prescription decisions belong with a licensed clinician. Affiliate disclosure: we may earn a commission when you use links on this page. Our recommendations are based on verified evidence and reader fit, not affiliate payout.
Last verified: April 24, 2026 by The RX Index Editorial Team. FDA labels, cohort study updates, Ro pricing, and FDA counterfeit alerts re-verified quarterly (next review: July 2026).
This page is for educational purposes only. It is not medical, legal, or insurance advice. Seek care from a licensed clinician for any medical emergency or prescription decision.