Written by The RX Index Editorial Team · Last evidence review: March 28, 2026 · Sources: FDA prescribing label, NEJM (ESSENCE trial, SYNERGY-NASH trial), AASLD Practice Guidance (Nov 2025), CMS · Editorial standards · Updated monthly
GLP-1 FOR FATTY LIVER — 2026 COMPLETE GUIDE
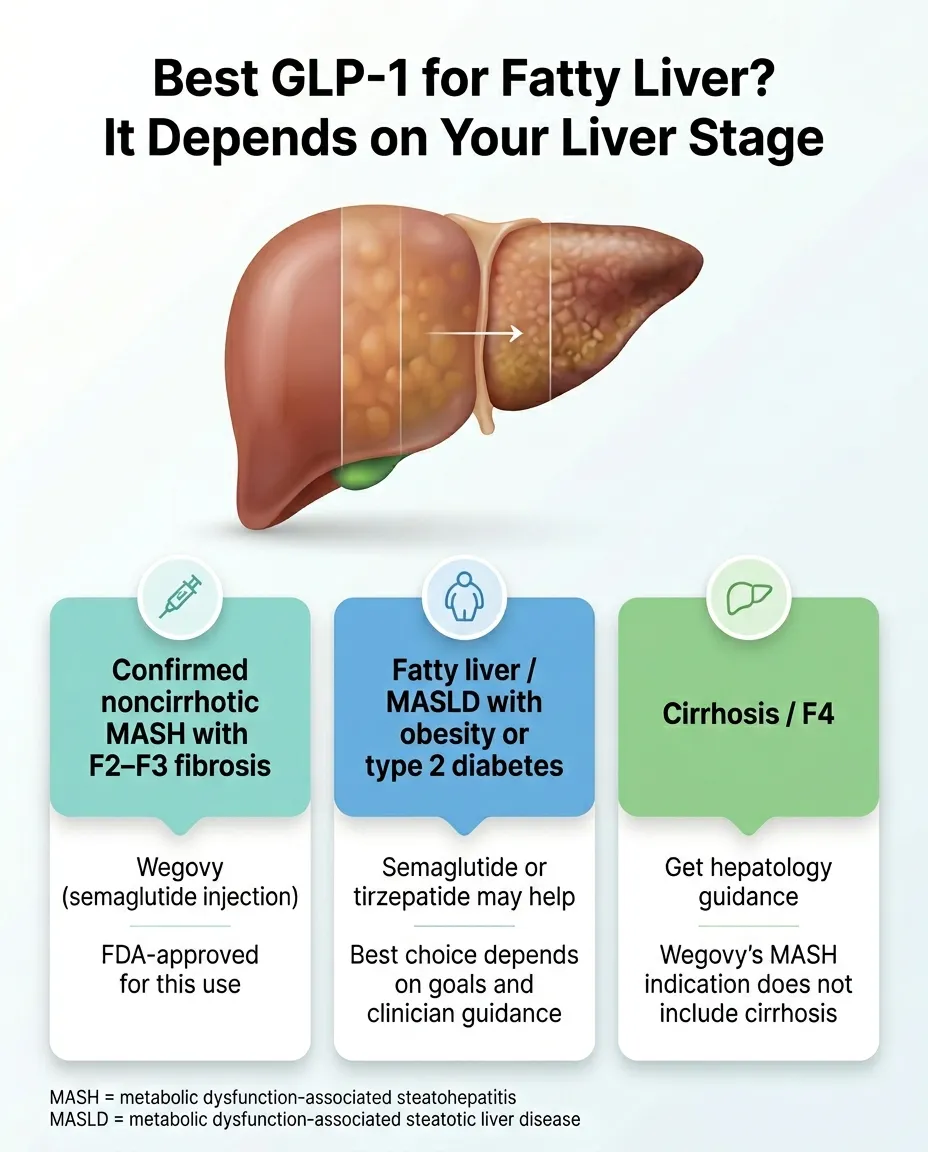
Best GLP-1 for Fatty Liver: The Right Answer Depends on Your Liver Stage
For adults with confirmed noncirrhotic MASH and F2–F3 fibrosis, Wegovy (semaglutide injection) is the strongest approval-based GLP-1 option — and it's the only one with FDA backing for a liver indication. In the Phase 3 ESSENCE trial's 72-week interim analysis, 62.9% of patients on Wegovy achieved steatohepatitis resolution without worsening fibrosis, versus 34.3% on placebo.
But the right GLP-1 for your liver depends entirely on your liver stage, whether you have diabetes, and how you're paying for it. Someone with confirmed MASH and documented F2–F3 fibrosis has a fundamentally different best answer than someone whose doctor casually mentioned “a little fat on the liver” during a routine ultrasound.
Below is the head-to-head comparison, a stage-by-stage decision framework, the Ozempic-vs-Wegovy insurance strategy most pages ignore, and the actual steps to get started.
Sources: FDA prescribing label (2025), AASLD Practice Guidance update (Nov 2025), ESSENCE trial — NEJM (2025), SYNERGY-NASH trial — NEJM (2024). Last evidence review: March 28, 2026.
Disclosure: Some links on this page are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you.
At a Glance: GLP-1 Medications Compared for Fatty Liver Disease
| Wegovy (semaglutide inj.) | Tirzepatide (Mounjaro/Zepbound) | Ozempic (semaglutide) | Liraglutide (Saxenda) | |
|---|---|---|---|---|
| FDA-approved for noncirrhotic MASH with F2–F3? | ✅ Yes — Aug 2025 | ❌ No (Phase 3 underway) | ❌ No | ❌ No |
| Best for | Confirmed MASH with F2–F3 fibrosis | Obesity-first with fatty liver; strongest weight loss | Already on for diabetes; liver benefit is a bonus | Older generation; weaker liver data |
| MASH resolution rate | 62.9% vs 34.3% placebo (Phase 3) | 44–62% vs 10% placebo (Phase 2) | Not established for this indication | 39% vs 9% placebo (small Phase 2) |
| Fibrosis improvement | 36.8% vs 22.4% placebo | 51–55% vs 30% placebo (Phase 2) | Not established for this indication | Slowed progression vs placebo |
| Key trial | ESSENCE (n=800 interim, Phase 3) | SYNERGY-NASH (n=190, Phase 2) | N/A | LEAN (n=52, Phase 2) |
| Average weight loss | ~10.5% | ~10.7–15.6% by dose | ~10–15% (diabetes trials) | ~5–8% |
| Injection frequency | Once weekly | Once weekly | Once weekly | Once daily |
| Insurance path for liver? | Strongest — MASH is on the label | Off-label only | Not indicated for liver | Not indicated for liver |
Tirzepatide percentages are from the Phase 2 SYNERGY-NASH primary analysis, not a head-to-head comparison with semaglutide. These are cross-trial numbers from different patient populations. Pricing varies by formulation, dose, and insurance.
Quick read: If your liver is the primary concern and you have confirmed noncirrhotic MASH with F2–F3 fibrosis, Wegovy is the clear frontrunner — it has the FDA approval, the Phase 3 data, and the strongest insurance pathway. If your primary driver is maximum weight loss and metabolic improvement with liver benefits as a major bonus, tirzepatide deserves serious consideration despite lacking the liver-specific indication.
⚠️ If you have cirrhosis (stage F4): Wegovy is not approved for cirrhotic MASH. Get hepatology involvement before making medication decisions. The AASLD recommends careful monitoring for patients with compensated cirrhosis already receiving semaglutide for another approved indication — but that's a specialist conversation, not a telehealth decision.
Before You Pick a GLP-1: Which Type of Fatty Liver Do You Actually Have?
This is the question that changes everything, and it's the question most guides skip entirely. “Fatty liver” is an umbrella term. It can mean simple fat buildup with no damage, moderate scarring that needs treatment, or advanced disease headed toward transplant. The best GLP-1 — and whether you even need one specifically for your liver — depends on where you fall.
MASLD (formerly NAFLD)
The broad category. Excess fat in your liver tied to metabolic factors like obesity, diabetes, high triglycerides, or high cholesterol. Roughly a quarter to a third of U.S. adults are affected. Most people with MASLD will never develop serious liver problems, especially with lifestyle changes.
MASH (formerly NASH)
The serious subset. Fat has caused inflammation and cell damage in your liver, with or without scarring (fibrosis). Only a subset of people with MASLD progress to MASH — often estimated at about one-fifth to one-third. This is where things get urgent, because MASH can advance to cirrhosis, liver cancer, and liver failure.
Fibrosis staging: the critical number
Doctors rate liver scarring from F0 (none) to F4 (cirrhosis). Wegovy's FDA approval covers F2–F3 — moderate to advanced fibrosis that hasn't yet reached cirrhosis. Staging can be determined by a FibroScan (a painless ultrasound-like test), MRE imaging, an ELF blood test, or a liver biopsy.
Why this matters for your GLP-1 decision
Simple MASLD with no significant fibrosis (F0–F1): Any GLP-1 that helps you lose weight will reduce liver fat. You don't necessarily need the liver-specific indication. Weight loss of even 7–10% of body weight meaningfully improves fatty liver. Your decision is more about weight loss, diabetes management, and cost.
Confirmed noncirrhotic MASH with F2–F3 fibrosis: Wegovy is the strongest option — it's the only GLP-1 with FDA approval for your exact condition. This also gives you the best shot at insurance coverage specifically for liver disease.
Cirrhosis (F4): Wegovy is not approved for your stage. Talk to a hepatologist. There may still be a role for GLP-1 medications under specialist supervision, but this requires that conversation.
Don't know your stage? Your first step isn't picking a medication — it's getting a FibroScan or a blood-based fibrosis score (FIB-4 or ELF). FibroScan is quick; blood-based scores can often be calculated from existing labs.
Quick Terminology Note
The medical world renamed these conditions in 2023. NAFLD became MASLD. NASH became MASH. If your doctor uses the old terms, they're describing the same conditions. You'll see both sets of terms in this guide because patients and search engines still use both.
Why Wegovy Is the Best GLP-1 for Confirmed MASH With F2–F3 Fibrosis
If your doctor has confirmed noncirrhotic MASH with F2–F3 fibrosis, the evidence points squarely at Wegovy (semaglutide 2.4 mg weekly injection). Here's what makes it the clear frontrunner.
The ESSENCE Trial: What the Interim Data Showed
The FDA approved Wegovy for MASH based on the Phase 3 ESSENCE trial — an ongoing study that randomized 1,197 adults with biopsy-confirmed MASH and stage F2–F3 fibrosis across 37 countries. The published interim efficacy analysis covers the first 800 participants who completed 72 weeks, published in the New England Journal of Medicine in 2025, led by Dr. Arun Sanyal at VCU's Stravitz-Sanyal Institute.
ESSENCE Trial — 72-Week Interim Results
Phase 3 interim analysis (n=800). Source: New England Journal of Medicine, 2025 (ESSENCE trial).
About half the participants had Type 2 diabetes, and roughly 73% had obesity. Semaglutide showed benefit across both groups. No patients on semaglutide discontinued due to liver enzyme elevations.
Part 2 of ESSENCE is ongoing, following nearly 1,200 participants for up to 240 weeks to evaluate long-term clinical outcomes — progression to cirrhosis, liver transplant, and mortality. The AASLD updated its clinical practice guidance in November 2025 to formally recommend semaglutide as a treatment option for adults with MASH and F2–F3 fibrosis.
How Semaglutide Helps Your Liver
Weight loss reduces liver fat directly
When you lose visceral fat — the fat around your organs — less fat accumulates in the liver. The ESSENCE trial showed significant reductions in liver fat content.
Insulin sensitivity improves
Insulin resistance is one of the primary engines driving fatty liver disease. GLP-1s improve how your body handles insulin, which slows the cycle of fat accumulation and inflammation.
Systemic inflammation drops
GLP-1s reduce inflammatory markers throughout the body. Since chronic inflammation is what transforms simple fatty liver into MASH, this has direct clinical relevance.
Multiple mechanisms working together
Semaglutide's benefit in MASH is not fully explained by weight loss alone — emerging evidence suggests effects on fat metabolism and inflammation that go beyond the scale.
The One Honest Limitation
Wegovy requires ongoing use. This isn't a 6-month course that fixes your liver and you stop. If you discontinue, weight typically returns, and liver fat can follow. But most patients on GLP-1s report the weight loss, improved energy, better blood sugar control, and reduced inflammation make them want to stay on the medication. And with Wegovy's MASH approval, insurance coverage pathways are expanding — making long-term use more financially realistic than it was even a year ago.
See if you qualify for Wegovy through Ro
Ro offers FDA-approved semaglutide with insurance verification and prior authorization support built into the process.
Check Eligibility on Ro →
When Tirzepatide Is Actually the Better Choice for Fatty Liver
Wegovy wins the liver-specific lane. That's accurate. But tirzepatide (Mounjaro for diabetes, Zepbound for weight loss) has trial data that's impossible to ignore — and for certain readers, it may be the smarter play.
The SYNERGY-NASH Trial Results
The Phase 2 SYNERGY-NASH trial, published in the New England Journal of Medicine in 2024, tested tirzepatide in 190 patients with biopsy-confirmed MASH and F2–F3 fibrosis:
SYNERGY-NASH Trial — Phase 2 Results
vs 10% MASH resolution and 30% fibrosis improvement on placebo. Phase 2 trial only (n=190). Source: NEJM, 2024.
So Why Isn't Tirzepatide #1 for Liver?
Two reasons. First, SYNERGY-NASH was a Phase 2 trial with 190 patients. ESSENCE was Phase 3 with 800 in the interim analysis. Phase 3 data is the gold standard for FDA approval decisions. Second, tirzepatide does not have FDA approval for MASH. Eli Lilly has a Phase 3 liver trial underway, with results expected around 2027.
This matters practically: without the liver indication on its label, insurance is far less likely to cover tirzepatide specifically for fatty liver, and clinicians prescribing it for liver disease are doing so off-label.
Who Should Seriously Consider Tirzepatide Anyway
If your primary driver is obesity or Type 2 diabetes, with fatty liver as a secondary concern — tirzepatide may be the stronger overall metabolic choice. It produces greater average weight loss than semaglutide.
If you need insurance coverage for weight loss or diabetes and your liver staging doesn't reach the F2–F3 threshold — tirzepatide via Zepbound (weight loss) or Mounjaro (diabetes) may be easier to get covered.
If your doctor is managing your full cardiometabolic picture — tirzepatide gives strong overall metabolic improvement. Observational data presented at ACG 2025 suggested tirzepatide may outperform semaglutide on some cardiometabolic and liver-related outcomes in real-world patients with MASLD, obesity, and Type 2 diabetes — though that's observational, not randomized proof.
The honest framing: tirzepatide will likely pursue FDA approval for MASH. The Phase 2 data supports it. But “likely to be approved” and “is approved” are different things in 2026, and they lead to very different insurance conversations.
Compare Wegovy vs Zepbound on Ro
Ro offers both FDA-approved options and helps you figure out which one fits your insurance and diagnosis.
Compare Options on Ro →Ozempic vs. Wegovy for Fatty Liver: Same Drug, Very Different Insurance Conversation
This confusion sends more people back to search than almost any other GLP-1 question. Let's put it to rest.
| Ozempic | Wegovy injection | |
|---|---|---|
| Molecule | Semaglutide | Semaglutide |
| Dose | Up to 2 mg weekly | 2.4 mg weekly |
| Approved for | Type 2 diabetes; CV/kidney risk (diabetics) | Weight management; CV risk; noncirrhotic MASH with F2–F3 fibrosis |
| MASH approved? | ❌ No | ✅ Yes — Aug 2025 |
| Insurance for liver? | Typically denied (off-label) | Strongest path with PA documentation |
Why the Brand Name Matters More Than the Molecule
When your insurer reviews a prior authorization request, the brand name determines the coverage path. If you're prescribed Ozempic for fatty liver, your insurer will likely deny it — Ozempic's label doesn't include any liver indication. If you're prescribed Wegovy injection for MASH, your insurer has a significantly stronger reason to approve — the FDA approved Wegovy specifically for that indication.
Important 2026 note: Wegovy tablets now exist for weight management. However, the MASH indication is on the Wegovy injection label, not the tablet label. If your liver is the reason for the prescription, the injection is the correct formulation.
What This Means for You
Already on Ozempic for diabetes with fatty liver? Your liver is very likely benefiting from the semaglutide. Don't switch just because of this article. But if your doctor is willing, ask whether transitioning to Wegovy injection at the 2.4 mg dose — with the MASH indication driving the prescription — might give you better insurance support.
Starting fresh with liver as the primary reason? Push for Wegovy injection from the beginning. It avoids the off-label insurance battle entirely.
Get on the right prescription path
Ro's clinical team evaluates your conditions and routes you to the appropriate FDA-approved formulation, then handles prior authorization if insurance is in play.
Check Eligibility on Ro →Will Insurance Actually Cover a GLP-1 for Fatty Liver?
This is the question behind the question for most readers. The landscape shifted significantly in 2025 and keeps changing in 2026. If you have confirmed noncirrhotic MASH with F2–F3 fibrosis, Wegovy injection coverage is more accessible now than it has ever been.
How to Maximize Your Chances of Getting Covered
- 1
Get your liver staged
You need documentation. A FibroScan (VCTE) result in the 8–15 kPa range, an MRE in the 3.1–4.4 kPa range, or an ELF blood test score of 9.2–10.5 all help identify likely F2–F3 fibrosis per AASLD guidance. Ask your doctor for one of these assessments.
- 2
Get the diagnosis coded correctly
Your medical record needs to show MASH — ICD-10 code K75.81 — not just "fatty liver" or "elevated enzymes." The specificity of the diagnosis matters for prior authorization.
- 3
Submit prior authorization citing the FDA-approved indication
Your doctor (or Ro's clinical team) submits documentation showing: MASH diagnosis, fibrosis staging in the F2–F3 range, and a request for Wegovy injection under the MASH indication. The AASLD's November 2025 Practice Guidance update provides strong clinical support.
- 4
If denied, appeal
First denials are common with GLP-1 medications. Include the AASLD guideline, ESSENCE trial results summary, and your specific staging documentation. Many denials are overturned on appeal.
What Your Insurer Wants to See
| What they want | Where it comes from | Common mistake |
|---|---|---|
| Confirmed MASH diagnosis | FibroScan, MRE, ELF, or biopsy | Submitting only "fatty liver" — too vague |
| Fibrosis stage F2–F3 | FibroScan (8–15 kPa), MRE, or ELF score | No staging documentation at all |
| Prior lifestyle modification | Doctor's notes showing diet/exercise discussed | Not documenting lifestyle attempts |
| Comorbidity documentation | Labs showing diabetes, obesity, high triglycerides | Missing relevant metabolic comorbidities |
| Individualized clinician letter | Letter of medical necessity | Using a generic template |
Medicare and MASH: The 2026 Update
CMS announced the BALANCE model in December 2025. A separate Medicare GLP-1 payment demonstration is planned to begin in July 2026, with BALANCE Part D plan access slated for January 2027. Noncirrhotic MASH with F2–F3 fibrosis is a qualifying condition. Eligible Medicare beneficiaries under the bridge demonstration are expected to pay approximately $50 per month — though this is not blanket MASH coverage; specific eligibility criteria apply. If you're on Medicare with confirmed MASH: talk to your doctor now about getting staged and diagnosed so you're positioned when access opens.
Check current Wegovy coverage and availability on Ro
Insurance verification is part of Ro's intake process, and they walk you through alternatives if coverage isn't available yet.
Check Coverage on Ro →How to Actually Get a GLP-1 for Fatty Liver (Your Three Paths)
You've read the evidence. You understand your stage. Now: how do you go from this page to having medication in hand?
Path 1 — Our Top Recommendation
FDA-Approved Semaglutide Through Ro
Ro is our top recommendation for readers treating fatty liver because they focus exclusively on FDA-approved medications, offer insurance support, and provide ongoing clinical oversight — the combination that matters most for a liver-disease patient.
What Ro offers
- → Wegovy injection, Wegovy pill, Ozempic, Zepbound pen and vial
- → Insurance verification and prior authorization support
- → Ongoing licensed provider care and dosing management
- → Ro Body: $45 first month, $145/mo thereafter (medication separate)
Best for
- → Confirmed MASH patients wanting the FDA-approved liver treatment pathway
- → Readers with insurance wanting prior authorization help
- → Patients wanting clinician-managed ongoing care
“You actually get to talk to people. It makes you feel supported and like you can do it.” — Ro memberStart the FDA-Approved Path on Ro — Check Eligibility →
Path 2
Through Your Own Doctor
If you already have a gastroenterologist, hepatologist, or primary care physician managing your fatty liver, bring this information to your next appointment:
- •Ask about your current fibrosis stage (request a FibroScan if you haven't had one)
- •Reference the AASLD Practice Guidance update from November 2025 recommending semaglutide for MASH with F2–F3 fibrosis
- •Ask specifically whether Wegovy injection under the MASH indication is appropriate for you
- •Discuss whether tirzepatide might be stronger if your primary driver is obesity or diabetes with liver as secondary
Path 3 — If Insurance Fails
The Compounded Cash-Pay Backup
Important: Compounded GLP-1 medications are not FDA-approved and do not undergo the same regulatory review for safety, effectiveness, or manufacturing quality. For a progressive liver condition like MASH, we recommend the FDA-approved path when accessible. Discuss this with your doctor.
If coverage is denied and FDA-approved options don't fit your budget, compounded semaglutide through providers like MEDVi offers a lower price point — advertised starting around $179 for the first month and $299 for refills.
See MEDVi's Current Cash-Pay Pricing →Does GLP-1 Hurt Your Liver? Safety Concerns Addressed
If you searched “GLP-1 liver toxicity” or “does GLP-1 hurt your liver” — you're asking the right question. You're trying to help your liver, not damage it further. Here's what the evidence says.
What Typically Happens
In the relevant trial populations, semaglutide showed liver-related benefit and a favorable hepatic safety profile. In the ESSENCE trial, no patients on semaglutide discontinued due to liver enzyme elevations. Liver enzymes (ALT and AST) typically improve on GLP-1 therapy. The SYNERGY-NASH trial showed significant reductions across all tirzepatide doses as well.
The Rare Exception
Rare case reports of drug-induced liver injury have been published for both semaglutide and tirzepatide. Michigan Medicine published a case of tirzepatide-associated hepatitis with jaundice — the first known report. The NIH's LiverTox database notes isolated reports with semaglutide. These are uncommon, but they mean your doctor should monitor liver enzymes periodically, especially with pre-existing liver disease. That's standard medical practice.
The Gallstone Connection
One indirect concern worth knowing: rapid weight loss from any cause — GLP-1 medications, bariatric surgery, aggressive dieting — can trigger gallstone formation. Gallstones can occasionally obstruct bile ducts and affect liver function. This is a monitoring issue during the first 6–12 months when weight loss is fastest, not a reason to avoid treatment.
The Risk of Waiting
Untreated MASH progresses. It can advance to cirrhosis, liver cancer, and liver failure requiring transplant. The risk of treating fatty liver with a well-studied, FDA-approved medication in the appropriate patient population is meaningfully lower than the risk of watching the disease advance without intervention.
What to Ask Your Doctor Before Starting a GLP-1 for Fatty Liver
Print this or screenshot it for your next appointment:
About your diagnosis
- ☐Do I have MASLD, MASH, or something else? What's the specific diagnosis?
- ☐What is my fibrosis stage? Do I have a FibroScan, FIB-4, ELF, or biopsy result?
- ☐Am I in the F2–F3 range where Wegovy injection is FDA-indicated for MASH?
About the prescription
- ☐If we're treating my liver specifically, should the prescription be Wegovy injection rather than Ozempic?
- ☐Would tirzepatide (Mounjaro/Zepbound) be a better fit given my overall metabolic picture?
- ☐What dose will we target and how will we titrate up?
About monitoring
- ☐What liver labs do you want before I start?
- ☐How often should we recheck liver enzymes?
- ☐When should we repeat imaging to assess liver response?
About coverage
- ☐Do I have any contraindications — history of medullary thyroid carcinoma, MEN2, pancreatitis, or pregnancy plans?
- ☐Can we submit prior authorization under the MASH indication specifically?
- ☐What documentation do you need from me to support the request?
GLP-1 for Fatty Liver vs. Rezdiffra: How Do They Compare?
If you've heard about Rezdiffra (resmetirom), you're probably wondering how it stacks up. Both are FDA-approved for adults with noncirrhotic MASH and F2–F3 fibrosis, but they work through completely different mechanisms and were never compared head-to-head.
| Wegovy (semaglutide inj.) | Rezdiffra (resmetirom) | |
|---|---|---|
| Drug class | GLP-1 receptor agonist | THR-β agonist (thyroid hormone receptor) |
| How it works | Systemic: weight loss, insulin sensitivity, reduced inflammation | Direct: reactivates fat metabolism specifically in liver cells |
| FDA-approved for MASH? | ✅ Yes (Aug 2025) | ✅ Yes (Mar 2024) |
| Route | Weekly injection | Daily oral pill |
| MASH resolution rate | ~63% (ESSENCE) | ~26–30% (MAESTRO-NASH) |
| Weight loss | ~10.5% | Minimal |
| Also addresses diabetes? | ✅ Yes | ❌ No |
| Also reduces cardiovascular risk? | ✅ Yes | ❌ Not demonstrated |
Cross-trial comparisons from different studies and patient populations — not head-to-head data.
When Rezdiffra may make more sense
- → You can't tolerate GLP-1 side effects (nausea, GI symptoms)
- → You're at a healthy weight but still have MASH (genetics play a role)
- → You prefer a daily pill over a weekly injection
When Wegovy makes more sense
- → Obesity or metabolic disease is driving your MASH
- → You want systemic metabolic benefits: weight, insulin, cardiovascular
- → That describes the majority of MASH patients
What's in the Pipeline
The MASH treatment landscape is evolving fast. The key development to watch: tirzepatide's Phase 3 MASH trial is underway, with results expected around 2027. If approved, it would join Wegovy and Rezdiffra as a third FDA-approved option. Several other investigational agents — including survodutide (dual glucagon/GLP-1) and retatrutide (triple agonist) — are in clinical development with encouraging early-phase results.
This page focuses on what is approved and available today. We update it as the evidence, pricing, and coverage landscape changes.
Frequently Asked Questions About GLP-1 Medications for Fatty Liver
What is the best GLP-1 for fatty liver disease?
Semaglutide via the Wegovy injection is the best GLP-1 for fatty liver disease in adults with confirmed noncirrhotic MASH and F2–F3 fibrosis. It's the only GLP-1 with FDA approval for a liver indication, backed by the Phase 3 ESSENCE trial showing 62.9% steatohepatitis resolution. Tirzepatide has strong Phase 2 data but lacks the liver-specific FDA approval.
Is any GLP-1 FDA-approved for fatty liver?
Yes. On August 15, 2025, the FDA granted accelerated approval to Wegovy (semaglutide injection, 2.4 mg weekly) for adults with noncirrhotic MASH and moderate-to-advanced fibrosis (F2–F3). It's the first GLP-1 ever approved for a liver indication.
Is the Wegovy pill approved for fatty liver?
No. The MASH indication is on the Wegovy injection label specifically. Wegovy tablets are currently labeled for weight management and cardiovascular-risk-related uses, not MASH. If your liver is the reason for the prescription, the injection is the correct formulation.
Is Zepbound (tirzepatide) approved for fatty liver?
No. Zepbound is FDA-approved for weight loss and obstructive sleep apnea, not MASH. Eli Lilly has a Phase 3 MASH trial underway. Some doctors prescribe it off-label, but the liver-specific approval belongs to Wegovy.
Is Ozempic approved for fatty liver?
No. Ozempic contains semaglutide but is approved only for Type 2 diabetes and related cardiovascular/kidney risk reduction. For liver-specific treatment and the strongest insurance case, Wegovy injection is the correct prescription.
Will insurance cover Wegovy for fatty liver?
It depends on your plan, but the August 2025 FDA approval significantly improved coverage prospects. Many commercial plans now cover Wegovy injection under the MASH indication with prior authorization. You'll need documented MASH diagnosis with F2–F3 fibrosis staging. Medicare GLP-1 access for eligible patients is planned to begin through a bridge demonstration in July 2026.
Will insurance cover Ozempic for fatty liver?
Unlikely for a liver-specific claim. Since Ozempic's label doesn't include any liver indication, insurers typically deny coverage for that purpose. If you have Type 2 diabetes, Ozempic may be covered for that reason — and your liver will benefit. But for liver-specific coverage, Wegovy injection is the right ask.
Does GLP-1 help fatty liver if I don't have diabetes?
Yes. The ESSENCE trial included patients with and without Type 2 diabetes, and semaglutide showed benefit in both groups. Wegovy's MASH indication does not require a diabetes diagnosis. However, if you don't have diabetes, obesity, or confirmed MASH with fibrosis, the clinical case for starting a GLP-1 specifically for simple fatty liver is weaker — lifestyle changes may be the appropriate first step.
Can GLP-1 medications raise liver enzymes?
Typically the opposite. GLP-1 therapy consistently reduces liver enzymes (ALT, AST) in clinical trials. Temporary mild elevations can occur during rapid weight loss but are usually self-limiting. Rare isolated cases of drug-induced liver injury have been reported. Standard practice: periodic liver enzyme monitoring during treatment.
How long until a GLP-1 improves fatty liver?
Liver enzyme improvements often appear within 3–6 months. Measurable reduction in liver fat via imaging can emerge by 6 months. Fibrosis improvement (structural change in scarring) typically requires 12 or more months. The ESSENCE trial measured its primary endpoints at 72 weeks.
Can I use a GLP-1 if I have cirrhosis?
Wegovy is not approved for cirrhotic (F4) MASH. The AASLD recommends careful monitoring for patients with compensated cirrhosis who are already receiving semaglutide for another approved indication — rather than automatic discontinuation — but this requires specialist supervision, not self-directed telehealth prescribing.
What if my doctor mentions Rezdiffra instead?
Rezdiffra (resmetirom) is the other FDA-approved MASH treatment (approved March 2024). It works through a different mechanism — activating thyroid hormone receptors in liver cells. It's a daily pill, doesn't cause significant weight loss, and doesn't address diabetes or cardiovascular risk. Your doctor may recommend it if GLP-1 side effects are intolerable, if you're at a healthy weight, or if combination therapy is being considered.
Is compounded semaglutide a good option for fatty liver?
Compounded semaglutide is more affordable but is not FDA-approved and does not undergo the same regulatory review for safety, effectiveness, or manufacturing quality. For a progressive liver condition like MASH, we recommend the FDA-approved path when accessible. Compounded options exist as a backup for readers who cannot access or afford branded versions. Discuss the tradeoffs with your doctor.
How We Researched This Guide
We reviewed primary sources: the FDA prescribing label for Wegovy (2025 update), the Phase 3 ESSENCE trial published in the New England Journal of Medicine (2025), the Phase 2 SYNERGY-NASH trial published in NEJM (2024), the AASLD Practice Guidance update (November 2025), and published pricing and coverage information from Novo Nordisk (NovoCare), Ro, and CMS.
We do not treat compounded medications as equivalent to FDA-approved products. We do not use fabricated reviews, star ratings, or schema markup for content that doesn't exist on the page. All pricing references are from public sources verified as of March 2026.
This guide is for informational purposes only and is not medical advice. Consult your doctor for treatment decisions specific to your situation.
Still Not Sure Which GLP-1 Program Is Right for You?
Take our free 60-second matching quiz. We'll ask about your diagnosis, insurance situation, budget, and goals — then give you a personalized recommendation with the exact next step.
The RX Index is an independent research platform. We earn referral fees from some providers featured on this page, which helps fund our research and verification process. Our recommendations are based on clinical evidence, FDA status, access, and cost — not commission rates. If we removed every affiliate link, this page would still be the most thorough GLP-1 fatty liver resource we could find online.
Author: The RX Index Editorial Team · Last evidence review: March 28, 2026 · More GLP-1 guides →