FDA-Approved Cardiovascular Indication · Pricing Verified April 4, 2026
Wegovy for Heart Disease
Who qualifies, what the evidence actually shows, and how coverage works in the U.S. in 2026.
Written by The RX Index Editorial Team · Last verified April 4, 2026
Reviewed against FDA prescribing information, CMS guidance, and major payer prior-authorization documents · Our methodology
Disclosure: Some links on this page are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you. Our recommendation would be the same without those links — the data drives the verdict, not the commissions.
The Bottom Line
If you're looking for reliable information on Wegovy for heart disease, here it is: Wegovy is FDA-approved to reduce the risk of cardiovascular death, heart attack, and stroke in adults with established cardiovascular disease and overweight or obesity. In the SELECT trial, major cardiovascular events occurred in 6.5% of people on Wegovy versus 8.0% on placebo over a mean follow-up of 39.8 months — a 20% relative reduction.
But the approval is only half the story. The questions that actually keep people stuck — do I specifically qualify, what does my insurer need to see, and what's my cheapest path to get this — are what most pages never answer. That's exactly what this guide was built for.
If you don't have established heart disease and want Wegovy for weight loss only, this isn't the right page — head to our GLP-1 comparison guide instead.
Wegovy for Heart Disease at a Glance
| Question | Short Answer | Source |
|---|---|---|
| FDA-approved for heart disease? | Yes — since March 2024, for cardiovascular risk reduction | FDA.gov |
| Who qualifies? | Adults with established CVD (prior heart attack, stroke, or symptomatic PAD) + BMI ≥ 27 | FDA label |
| What did the study show? | 20% fewer heart attacks, strokes, and cardiovascular deaths vs. placebo | SELECT trial, NEJM |
| Does high cholesterol alone qualify? | Usually not — most payers require a prior cardiovascular event | Payer PA criteria |
| Does Medicare cover it? | Part D may cover it under the CV indication, with prior authorization | CMS.gov |
| Pill or injection? | Both carry the CV indication — self-pay pill starts at $149/mo for lower doses | Wegovy.com |
| Best first step? | Gather your cardiac records and BMI, then ask your cardiologist or run a coverage check | — |
Is Wegovy FDA-Approved for Heart Disease?
Yes — but the exact wording matters, because it determines whether your insurance covers it and how your doctor prescribes it.
In March 2024, the FDA approved Wegovy (semaglutide) to reduce the risk of major adverse cardiovascular events — specifically cardiovascular death, heart attack, and stroke — in adults with established cardiovascular disease who also have obesity or overweight. The agency gave it Priority Review, fast-tracking the decision within six months.
This is a separate indication from Wegovy's weight loss approval. It's backed by a different trial, carries different insurance implications, and changes how your doctor codes the prescription. When Wegovy is prescribed for cardiovascular risk reduction, the diagnosis code reflects your cardiovascular condition — not obesity. That distinction is the reason Medicare Part D can now cover it (more on that below).
A few things this approval does not mean:
It's not a cure for heart disease. Wegovy reduces the risk of future events, used alongside your existing medications and lifestyle changes. It doesn't replace statins, blood pressure meds, or anything your cardiologist already has you on — it's additive. And the SELECT trial only included people with existing cardiovascular disease, so the cardiovascular indication doesn't currently apply to people who are at risk but haven't had an event.
Source: FDA press announcement, March 8, 2024
What Did the SELECT Trial Actually Show?
Wegovy reduced major cardiovascular events by 20% over 3.3 years in adults with heart disease — and the heart protection may not be explained by weight loss alone.
The SELECT trial is the foundation for everything on this page. Here's what it showed in plain English.
The basics
Researchers enrolled 17,604 adults across 41 countries. Every participant had established cardiovascular disease — a prior heart attack, prior stroke, or symptomatic peripheral artery disease — plus a BMI of 27 or higher. None had diabetes. (People with diabetes were already covered by earlier Ozempic cardiovascular data.)
Half received Wegovy 2.4 mg weekly by injection. Half received placebo. Everyone continued their existing heart medications — statins, blood pressure drugs, blood thinners — throughout the study.
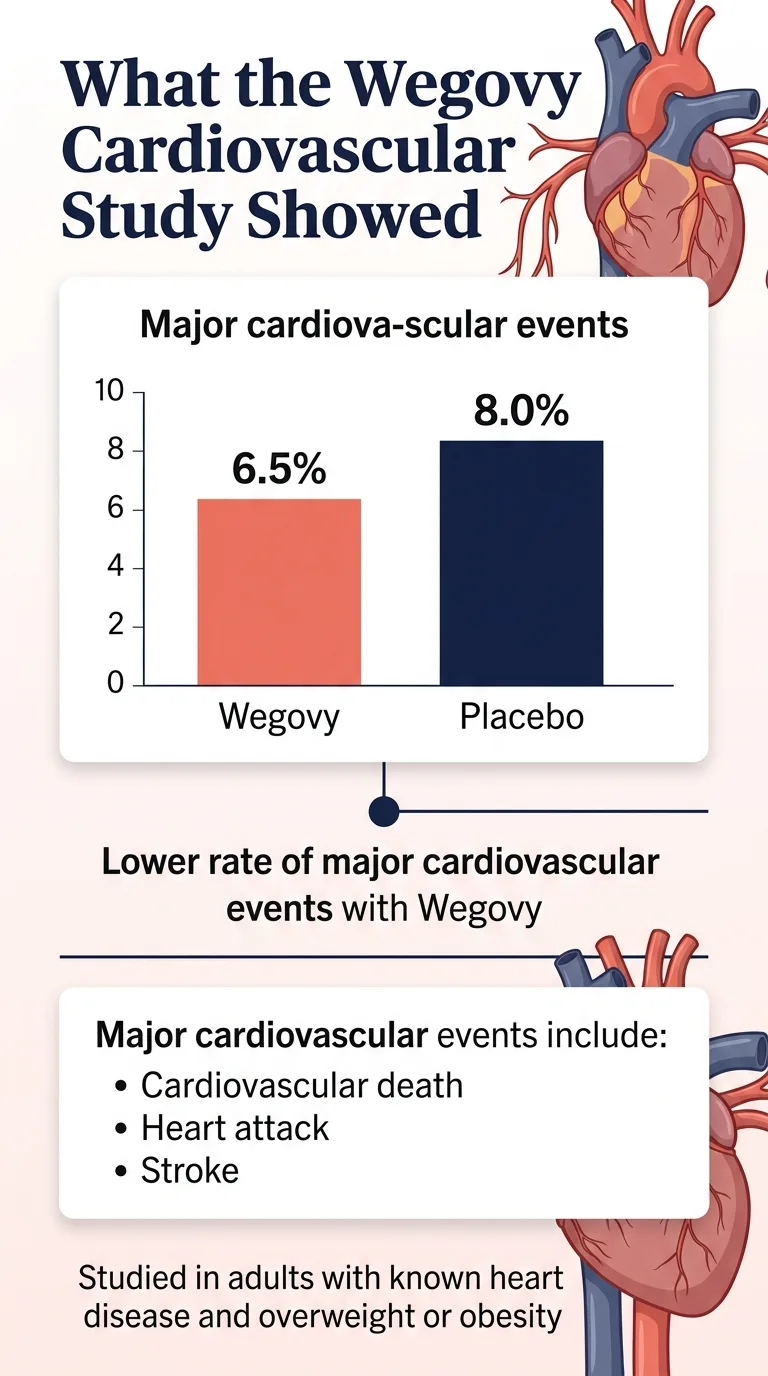
The results
Over a mean follow-up of 39.8 months:
| Outcome | Wegovy | Placebo | Reduction |
|---|---|---|---|
| Major CV events (death, MI, stroke) | 6.5% | 8.0% | 20% |
| Cardiovascular death | 2.5% | 3.0% | ~15%* |
| All-cause death | 4.3% | 5.2% | ~19%* |
| Mean weight change at ~2 years | −9.4% | −0.9% | — |
*CV death and all-cause death were secondary/exploratory endpoints, not the primary outcome. The CV death result did not reach conventional statistical significance (p = 0.07). These numbers provide useful context but should be interpreted with that caveat.
Sources: Lincoff AM et al., NEJM, November 2023; ACC SELECT trial summary
What the 20% actually means in real terms
That 20% number is everywhere. But here's what it looks like in absolute terms: major cardiovascular events happened in 6.5% of people on Wegovy versus 8.0% on placebo. That's a 1.5 percentage-point absolute reduction — which translates roughly to one major cardiac event prevented for every 67 patients treated over about 3.3 years.
That may sound modest in isolation. But this is on top of statins, blood pressure meds, and standard cardiovascular care. Adding Wegovy produced additional protection beyond what the best existing treatments already provide. Very few interventions clear that bar.
Why researchers think it's more than just weight loss
A subanalysis published in The Lancet in October 2025 found that Wegovy's cardiovascular benefit was not fully explained by the amount of weight patients lost. People who lost minimal weight still saw heart protection. The exact biological pathways are still being studied, but the benefit likely reflects a combination of weight loss and broader cardiometabolic effects — including reductions in inflammation, blood pressure, and markers of vascular stress — rather than weight loss alone.
Why this matters for you: Even if you don't hit dramatic weight loss numbers, the cardiovascular protection may still be meaningful.
Who Qualifies for Wegovy for Heart Disease?
If you've had a prior heart attack, stroke, or symptomatic PAD and your BMI is 27+, you likely qualify at the label level. But your insurer adds additional requirements — and they differ by payer.
At the FDA label level, the indication is straightforward: adults with established cardiovascular disease and either obesity (BMI ≥ 30) or overweight (BMI ≥ 27). In practice, insurance companies layer requirements on top, and understanding both levels saves you time and frustration.

FDA-level criteria
- ✓Prior heart attack (myocardial infarction)
- ✓Prior stroke
- ✓Symptomatic peripheral artery disease (PAD)
- ✓BMI of 27 or higher
- ✓Age 18+
- ✓Diabetes is not required for the FDA label
What major insurers actually require
This is where most pages stop being useful. Here's what we found when we reviewed current prior-authorization criteria from three of the largest payers:
| Requirement | UnitedHealthcare | Aetna / CVS Caremark | Medicare Part D |
|---|---|---|---|
| BMI threshold | ≥ 27 | ≥ 27 | ≥ 27 |
| Qualifying CV conditions | MI, stroke, symptomatic PAD, revascularization | MI, stroke, PAD, revascularization | Established CVD (documented) |
| Age requirement | 45+ | Varies by plan | 18+ (most are 65+) |
| T2D allowed for this pathway? | Often no — routed to Ozempic | Often no — similar routing | Depends on plan |
| GDMT required? | Yes | Yes, typically | Varies |
| Prior auth needed? | Yes | Yes | Yes, typically |
| Documentation | CV event history + BMI + current meds | Diagnosis + procedure records + GDMT | Prescriber justification |
Sources: UHC PA criteria; Aetna CV criteria; CVS Caremark criteria; CMS guidance, reviewed April 2026
Key insight #1: Even if your plan excludes weight-loss drugs, many plans will still cover Wegovy when prescribed for cardiovascular risk reduction. The CV indication creates a separate coverage pathway that bypasses the weight-loss exclusion. Your doctor's prescription coding is what makes this work.
Key insight #2: If you have type 2 diabetes and established heart disease, major payers often route you toward Ozempic (which has its own CV indication for diabetic patients) rather than approving Wegovy through this pathway. That's not a dead end — it's a different door.
Quick self-check
Likely qualifies: Documented prior heart attack, stroke, or symptomatic PAD — plus BMI ≥ 27 — and your doctor will prescribe under the cardiovascular indication.
Maybe: Your chart says “coronary artery disease” or “atherosclerosis” but you haven't had a discrete event. Some payers accept revascularization procedures (stents, bypass) as qualifying events. Others are stricter. Worth checking.
Likely not through this pathway: You have cardiovascular risk factors (high cholesterol, high blood pressure, family history) but no documented cardiovascular disease. You may still qualify for Wegovy's weight management indication — that's a separate path. See our weight-loss GLP-1 guide →
What Counts as “Established Cardiovascular Disease” for Wegovy?
This is the most common sticking point — and where most people's coverage gets stuck.
Conditions that usually qualify
- ✓Prior heart attack (myocardial infarction) — documented with dates
- ✓Prior stroke — documented with imaging or hospital records
- ✓Symptomatic peripheral artery disease (PAD) — with symptoms or a procedure
- ✓Prior coronary revascularization — CABG or stent placement (PCI)
Conditions that usually do NOT qualify on their own
- ✗High cholesterol alone
- ✗High blood pressure alone
- ✗Family history of heart disease
- ✗Elevated coronary calcium score without a prior event
- ✗Obesity alone (falls under the weight management indication)
- ✗Heart failure without a qualifying atherosclerotic event
What if your chart says “heart disease” but isn't specific?
This happens constantly. If your records mention “coronary artery disease” or “cardiovascular disease” but don't document a specific qualifying event with dates, your payer may push back during prior authorization.
The fix: ask your clinician to make sure the prior-authorization request and supporting chart documentation clearly identify the qualifying cardiovascular history — the specific event, the date, and the fact that Wegovy is being prescribed for its cardiovascular indication. That documentation is the single biggest factor in whether your insurance approves or denies the claim.
Is Wegovy Safe if You Already Have Heart Disease?
Yes — Wegovy was specifically studied in 17,604 people with existing heart disease. Not only was it considered safe enough to approve, it reduced their cardiovascular events. But side effects are real, and you should know what to expect.
What the safety data showed
In SELECT, the overall rate of serious adverse events was slightly lower in the Wegovy group (33.4%) than in the placebo group (36.4%). Cardiovascular events were reduced, not increased.
The most common side effects were gastrointestinal:
- Nausea (most common — usually peaks during dose titration and improves over 4–8 weeks)
- Diarrhea
- Vomiting
- Constipation
- Abdominal pain
The honest dropout numbers
About 16% of patients on Wegovy stopped treatment due to side effects, compared to 8% on placebo. Most discontinuations were from GI issues, not cardiovascular problems.
That means roughly 5 out of 6 patients tolerated treatment well enough to continue and received the cardiovascular benefit. The 1 in 6 who couldn't tolerate it typically experienced persistent nausea or vomiting — and in most cases, a slower titration schedule or switching between pill and injection can help.
Formal contraindications (who should NOT take Wegovy)
- ✗Personal or family history of medullary thyroid carcinoma (MTC)
- ✗Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- ✗Prior serious allergic reaction to semaglutide or any ingredient in Wegovy
Important warnings and precautions
- Pancreatitis: Report severe abdominal pain immediately
- Gallbladder disease: Rapid weight loss can increase gallstone risk
- Kidney injury: Dehydration from nausea/vomiting/diarrhea can affect kidney function — stay hydrated
- Heart rate: Wegovy can cause a modest increase (typically 2–4 bpm); in SELECT, this did not translate into more cardiovascular events
- Gastroparesis: Not recommended if you have severe gastroparesis
- Pregnancy: Discontinue Wegovy when pregnancy is recognized
Can you take Wegovy with your existing heart medications?
Wegovy was used on top of standard cardiovascular therapy throughout the SELECT trial — statins, blood pressure medications, blood thinners, aspirin. Semaglutide studies did not show clinically relevant interactions with the specific oral medications that were tested. However, because Wegovy delays gastric emptying, it can affect the absorption timing of some oral medications. Your doctor may want to monitor levels of any medication that requires precise dosing — particularly Warfarin, where your INR should be checked after starting.
Source: FDA prescribing information for Wegovy
Does Medicare Cover Wegovy for Heart Disease?
Medicare Part D may cover Wegovy when it's prescribed for the FDA-approved cardiovascular indication — subject to your plan's formulary and prior-authorization rules.
How the cardiovascular pathway works (available now)
Federal law prohibits Medicare from covering weight-loss medications. But the FDA's cardiovascular indication for Wegovy is not a weight-loss use — it's a cardiovascular risk reduction use. That means it falls outside the statutory weight-loss exclusion and is potentially coverable under Part D.
- Your doctor must prescribe Wegovy for cardiovascular risk reduction, not for weight loss
- The primary diagnosis must reflect your cardiovascular condition
- Your Part D plan will almost certainly require prior authorization with documentation of your qualifying CV history
- Individual plans control their own formularies — not all Part D plans include Wegovy yet
The key number for 2026: $2,100. That's the maximum you'll spend out of pocket on all covered Part D drugs this year. Once you reach it, Medicare pays 100% for the rest of the year. And under the Medicare Prescription Payment Plan (MPPP), you can spread those costs into predictable monthly installments.
What about the Medicare GLP-1 Bridge?
The Medicare GLP-1 Bridge launches July 1, 2026 and covers certain weight-management uses outside the basic Part D benefit. It does not apply when Wegovy is prescribed for its cardiovascular indication, because that use is already potentially coverable under Part D directly.
What to do right now if you're on Medicare
- 1. Call your Part D plan — ask whether Wegovy is on the formulary for the cardiovascular risk reduction indication
- 2. Have your doctor prescribe under the CV indication with documentation of your qualifying cardiac event
- 3. Be prepared for prior authorization — your cardiologist's office handles the paperwork
- 4. If denied, appeal — many first-round denials are overturned with proper cardiovascular documentation
- 5. Calculate your costs — remember the $2,100 annual out-of-pocket cap and the MPPP installment option
For the full walkthrough of every Medicare pathway, see our complete Medicare Wegovy guide →
Source: CMS Medicare prescription drug coverage guidance
Can Commercial Insurance Cover Wegovy Even If Weight-Loss Drugs Are Excluded?
Often, yes — and this is one of the biggest gaps in existing content on this topic.
Most commercial insurance plans exclude weight-loss medications. When people hear that, they assume Wegovy is off the table. But the cardiovascular indication creates a separate coverage pathway that many plans approve through a different prior-authorization process.
How to improve your prior-authorization odds
- 1. Make sure the prescription is coded correctly. The primary diagnosis must be cardiovascular — not obesity. Your clinician should document the qualifying event, not just "overweight."
- 2. Include supporting documentation. Prior auth for the CV indication typically requires chart notes confirming your qualifying event, current BMI, current medications, and evidence of guideline-directed medical therapy (GDMT).
- 3. Don't assume a denial is final. First-round denials are common and frequently overturned on appeal when proper documentation is resubmitted.
- 4. Check for the Wegovy Savings Offer. If your commercial plan covers Wegovy, the manufacturer savings card can bring your copay down to as low as $25/month (maximum savings $100/month). Commercially insured patients only.
If your plan does cover Wegovy and you activate the savings card, you're looking at roughly $25/month for a medication that reduced major cardiac events by 20% in the largest trial of its kind. That's less than most gym memberships.
Free coverage check · Prior-authorization support · FDA-approved Wegovy (pill or injection) · Eligibility typically determined within days
How Much Does Wegovy Cost for Heart Disease Patients?
The list price of $1,349/month is what almost nobody actually pays. Here's what real access looks like in each situation.
| Your Situation | Best Access Path | Est. Monthly Cost | Notes |
|---|---|---|---|
| Commercial insurance covers Wegovy | Pharmacy + Savings Card | As low as $25/mo | Apply at WegovyTerms.com |
| Commercial insurance, not covered | Self-pay via NovoCare or Ro | $149–$299/mo (pill) | Lower doses start at $149; 25 mg is $299/mo |
| Medicare + documented heart disease | Part D under CV indication | Varies; capped at $2,100/yr | Spread via MPPP; $0 after cap |
| No insurance, want the pill | Wegovy pill via NovoCare or pharmacy | $149/mo (lower doses) or $299/mo | 4 mg at $149 through 8/31/2026 |
| No insurance, want the injection | Self-pay via NovoCare or Ro | $199/mo (intro) then $349/mo | Lower-dose intro pricing; limited windows |
| No insurance, lowest long-term cost | 12-month subscription via Ro | $249/mo (injection) | Ro also charges $145/mo membership |
All prices verified April 2026 via Novo Nordisk/Wegovy.com, NovoCare, and Ro. Prices and promotional windows change — verify directly before purchasing.
Why “cheapest option” isn't always your first question: If you have heart disease and may qualify for the cardiovascular indication, check your insurance first before defaulting to self-pay. Even plans that exclude weight-loss drugs may cover Wegovy under the CV pathway — and insured pricing with the savings card ($25/month) is dramatically cheaper than any self-pay route.
Ro checks your specific plan and tells you what to expect before you commit
Wegovy Pill vs. Injection: Which Is Better for Heart Disease?
Both carry the FDA cardiovascular indication. The injection has the direct SELECT trial data behind it. The pill is newer, often cheaper for self-pay, and eliminates needles.

| Feature | Wegovy Injection | Wegovy Pill |
|---|---|---|
| Dose | 2.4 mg once weekly | 25 mg once daily (maintenance) |
| Titration | ~20 weeks to full dose | ~12 weeks to full dose |
| CV trial data | SELECT trial (direct) | Same molecule; OASIS 4 trial |
| Self-pay cost | $199–$349/mo | $149–$299/mo |
| Insured cost w/ savings card | As low as $25/mo | As low as $25/mo |
| How you take it | Weekly injection (thin needle, under skin) | Daily pill — empty stomach, ≤ 4 oz water, 30 min before food |
| Refrigeration? | Yes | No |
| Available since | 2021 (weight); 2024 (CV) | January 2026 |
Practical guidance for heart patients
If you're already comfortable with injections — many heart patients are, from blood thinners or insulin — the weekly injection is convenient and carries the most direct cardiovascular evidence.
If you prefer no needles, want a lower self-pay entry point, or travel frequently (no refrigeration), the pill is strong. The trade-off: you take it daily on an empty stomach, and the 30-minute wait before eating or taking other oral medications requires planning, especially if you take morning heart meds.
Both deliver semaglutide at therapeutic doses. Your doctor can help you decide, and switching between formats is possible if one doesn't work for you.
For a deeper dive on the pill specifically, see our Wegovy Pill vs. Injection comparison →
Source: FDA prescribing information, Wegovy tablets
Is Wegovy Better Than Zepbound or Ozempic for Heart Disease?
For cardiovascular risk reduction specifically, Wegovy has the strongest label today. But the right answer depends on whether you also have type 2 diabetes.
| Drug | Active Ingredient | FDA CV Indication? | Best Fit For |
|---|---|---|---|
| Wegovy | Semaglutide (2.4 mg injection or 25 mg pill) | Yes | Heart disease patients with or without diabetes who want proven CV protection |
| Ozempic | Semaglutide (0.5–2 mg injection) | Yes — for T2D + CVD | Heart disease + type 2 diabetes patients |
| Zepbound | Tirzepatide (injection) | No — CV trial ongoing | Weight loss or sleep apnea — not heart disease yet |
If your primary goal is cardiovascular protection and you don't have type 2 diabetes, Wegovy is the only GLP-1 with a directly matching label.
If you do have type 2 diabetes and established heart disease, Ozempic may be the more straightforward route. See our Ozempic vs. Wegovy comparison →
Zepbound may eventually show cardiovascular benefit — the SURMOUNT-MMO trial is ongoing — but it doesn't have a CV indication today.
Should Your Cardiologist Prescribe Wegovy, or Can You Use Telehealth?
If your cardiologist has your records and is willing to prescribe, start there. If not — and many cardiologists still refer out — telehealth fills the gap.
When to start with your cardiologist
Your cardiologist already has your cardiac history, knows your medication list, and can prescribe directly under the CV indication. If your plan covers Wegovy and your cardiologist will write the script, this is typically the fastest and most affordable route.
What to say at your appointment: “I'd like to discuss Wegovy for cardiovascular risk reduction based on the SELECT trial. I have [your specific condition] and my BMI is [X]. Can you prescribe it under the cardiovascular indication?”
When telehealth adds value
A platform like Ro prescribes FDA-approved Wegovy (pill and injection) and adds value in three situations:
- 1. Your cardiologist doesn't prescribe it — Ro's providers can prescribe with your existing medical records.
- 2. You need prior-authorization help — Ro has a dedicated insurance team that handles PA paperwork.
- 3. You want self-pay — Ro offers self-pay pricing with direct home delivery.
Important: If you're on Medicare, telehealth platforms generally cannot coordinate government insurance coverage for GLP-1 medications. For the Medicare Part D cardiovascular pathway, work directly with your doctor and your Part D plan.
What telehealth cannot do
Telehealth cannot create a qualifying cardiovascular diagnosis that doesn't already exist in your medical records. If you need the CV indication for insurance coverage, you need documented evidence of your heart condition from a clinician who examined you.
Upload your cardiac records · Licensed physician reviews your case · FDA-approved Wegovy (pill or injection) · Insurance check or self-pay delivery
What to Do Next If You Likely Qualify
You've read the evidence. You've checked the eligibility criteria. Here's the action checklist.
Step 1: Gather your documentation
- Cardiac history: Records showing your qualifying event — including dates
- Current BMI: Your doctor's office can calculate this
- Current medication list: Everything you take, including dosages
- Insurance card: You'll need your plan details for the coverage check
Step 2: Choose your route
Option A — Your cardiologist: Call their office and request an appointment to discuss Wegovy's cardiovascular indication. Mention the SELECT trial. Bring your BMI.
Option B — Telehealth (Ro): Complete an online health assessment, upload your cardiac records, and a licensed physician reviews your case. Start here →
Step 3: Handle insurance
Your doctor or Ro's insurance team submits the prior authorization under cardiovascular diagnosis codes. If you have commercial insurance that covers Wegovy, apply the Wegovy Savings Card for copays as low as $25/month. If denied, appeal — proper CV documentation frequently overturns first-round denials.
Step 4: Start treatment
Wegovy uses a gradual dose-titration schedule to minimize side effects:
Injection: 0.25 mg weekly for 4 weeks → 0.5 mg → 1.0 mg → 1.7 mg → 2.4 mg (therapeutic dose around month 5)
Pill: 1.5 mg daily for 30 days → 4 mg → 9 mg → 25 mg daily from day 91 onward
Step 5: Monitor and follow up
Continue all existing heart medications. Follow up with your cardiologist at 3 and 6 months to check blood pressure, weight, kidney function, and how you're tolerating the medication. In SELECT, cardiovascular protection was observed within the first several months of treatment.
Free coverage check · Pill or injection · Delivered to your door
Real Patient Experiences With Wegovy for Heart Disease
Clinical trial numbers tell you what happens on average. Real patients tell you what it feels like.
“I want other people out there, especially people my age, to know that there's help. It's not easy taking that first step, and you shouldn't be embarrassed to ask for help.”
— Terry, Wegovy patient since 2023 (Wegovy Ambassador; compensated by Novo Nordisk)
“My palpitations are really under control, and my blood pressure is way down.”
— Verified patient review, Drugs.com (cardiovascular risk reduction)
“Losing the weight is very important for my heart, and my numbers have improved. But feeling crappy all the time just may not be worth it — still figuring that out.”
— Verified patient review, Drugs.com (cardiovascular risk reduction)
Individual results vary. These experiences are not a guarantee of outcomes.
That last review is honest — and it's why we included it. Not everyone has a smooth ride, especially during the first weeks of dose titration. But the question isn't whether you'll feel some discomfort. It's whether the trade-off is worth it. For 5 out of 6 patients in SELECT, the answer was yes.
What About Wegovy for Heart Failure?
Not FDA-approved for heart failure — but the clinical data is promising, especially for a specific type of heart failure linked to excess weight.
Heart failure with preserved ejection fraction (HFpEF) is a condition where the heart pumps normally but doesn't fill properly, leading to fatigue, shortness of breath, and exercise intolerance. Obesity is a major driver of HFpEF, and treatment options have been limited.
The STEP-HFpEF trials showed that Wegovy significantly improved symptoms, exercise capacity, and quality of life in people with HFpEF and obesity. The current Wegovy label includes heart-failure hospitalization data from SELECT as an exploratory endpoint, but the FDA has not established an effect on heart failure as an approved indication. If you have HFpEF, this is worth discussing with your cardiologist.
What If You Don't Qualify for the Heart Disease Indication?
You still have options — they're just different pathways.
If you have type 2 diabetes + established heart disease: Ozempic has its own CV indication for this population. See our Ozempic vs. Wegovy comparison →
If you want Wegovy for weight loss only: Self-pay pill starts at $149/month for lower doses. See our GLP-1 provider comparison →
If you're on Medicare without heart disease: The Medicare GLP-1 Bridge launching July 2026 covers certain weight-management uses at $50/month. See our Medicare Wegovy guide →
How We Verified This Page
We built this guide from primary sources — not by summarizing other websites.
- FDA approval announcement — March 8, 2024
- FDA prescribing information — Wegovy injection — current label
- FDA prescribing information — Wegovy tablets — current label
- SELECT trial — Lincoff AM et al., NEJM, November 2023
- Lancet subanalysis — weight-independent CV effects, October 2025
- CMS Medicare guidance — GLP-1 Bridge and Part D coverage
Editorial independence: The RX Index earns affiliate commissions from some providers linked on this page. This never influences our clinical recommendations or the data in our tables. If we stripped every affiliate link from this page, it would still be the most complete Wegovy cardiovascular guide online.
Frequently Asked Questions
Is Wegovy FDA-approved for heart disease?▼
Yes. Since March 2024, Wegovy is approved to reduce the risk of cardiovascular death, heart attack, and stroke in adults with established cardiovascular disease and obesity or overweight. This is a separate indication from its weight management approval.
Who qualifies for Wegovy for heart disease?▼
Adults with a documented history of heart attack, stroke, or symptomatic peripheral artery disease and a BMI of 27 or higher. Diabetes is not required at the label level, though major payers may route patients with type 2 diabetes toward Ozempic's cardiovascular indication instead.
What counts as established cardiovascular disease for Wegovy?▼
Payers typically require a prior heart attack, prior stroke, symptomatic peripheral artery disease, or prior coronary revascularization. High cholesterol, high blood pressure, or family history alone usually do not qualify.
Does Medicare cover Wegovy for heart disease?▼
Medicare Part D may cover Wegovy when prescribed for cardiovascular risk reduction, subject to formulary inclusion and prior authorization. The 2026 Part D out-of-pocket maximum is $2,100 for all covered drugs. The separate Medicare GLP-1 Bridge launching July 2026 covers weight-management uses but does not apply to the cardiovascular indication.
Can commercial insurance cover Wegovy if weight-loss drugs are excluded?▼
Often yes. The cardiovascular indication creates a separate coverage pathway. Major payers including UnitedHealthcare, Aetna, and CVS Caremark have documented prior-authorization criteria for Wegovy's cardiovascular use even though they typically exclude weight-loss medications.
Can you take the Wegovy pill for heart disease?▼
Yes. Both the Wegovy injection and Wegovy pill carry the FDA cardiovascular risk reduction indication. The pill is taken daily on an empty stomach. Self-pay pricing starts at $149/month for lower doses; the 25 mg maintenance dose is $299/month.
Do you need diabetes to qualify for Wegovy's heart disease indication?▼
No. The SELECT trial studied patients without diabetes. However, if you have type 2 diabetes with established heart disease, major payers may route you toward Ozempic, which has its own cardiovascular indication for diabetic patients.
Is Wegovy better than Zepbound for heart disease?▼
For cardiovascular risk reduction today, yes. Wegovy is the only GLP-1 with an FDA-approved cardiovascular indication for non-diabetic patients. Zepbound does not currently have a cardiovascular label. Its cardiovascular outcomes trial is ongoing.
What if insurance denies Wegovy for heart disease?▼
Appeal. First-round denials are common and frequently overturned when proper cardiovascular documentation is resubmitted. Ensure the prescription is coded under a cardiovascular diagnosis with your qualifying event clearly documented.
Is Wegovy approved for heart failure?▼
Not at this time. Clinical trials showed promising results for heart failure with preserved ejection fraction, but the FDA has not established an approved heart failure indication.
Related guides
- Ozempic vs Wegovy: 9 Real Differences in 2026
- Wegovy Pill vs Injection: Which to Choose (2026)
- Does Medicare Cover Wegovy? 2026 Coverage Guide
- Does Insurance Cover Wegovy for Weight Loss? (April 2026)
- Cheapest Wegovy Without Insurance in 2026
- How to Appeal a Wegovy Denial — 7 Real Steps (2026)
- Best Online Wegovy Provider: 7 Legit Options (2026)
- Best GLP-1 for Weight Loss: Every Option Compared (2026)
- Zepbound for Sleep Apnea: 7 Real Answers on Cost, Coverage & Access (2026)
Last updated: April 4, 2026. Published by The RX Index editorial team.
The RX Index is an independent health information resource. We may earn commissions through affiliate partnerships, but editorial content is never influenced by compensation. See our editorial policy for details.