Is Compounded GLP-1 Still Available in 2026?
By The RX Index Research Team · Last verified: March 31, 2026
Sources: FDA.gov, CMS.gov, NovoCare, LillyDirect, Ro, KFF, TrumpRx.gov
The Direct Answer
Not as a routine mainstream option. The FDA declared that semaglutide and tirzepatide injection shortages are resolved, the shortage-based grace periods ended in 2025, and the FDA issued 30 warning letters to telehealth companies on March 3, 2026 after an earlier wave in September 2025.
Here’s what most people miss: the approved path is no longer $1,000+ per month. The Wegovy pill starts at $149/month, some Wegovy pen self-pay offers start at $199/month, and Zepbound self-pay options start at $299/month. If you’re currently on a compounded GLP-1, you have real options — and most of them are safer and more affordable than you think.
This page covers: what actually happened, what’s still legal, every access path and what it costs, and exactly what to do next based on your situation.
Last verified: March 31, 2026 · Sources listed below

Is Compounded GLP-1 Still Available?
For most people, not in any meaningful way — and the trend is toward less access, not more. But here’s the good news buried inside that headline: the reason most people used compounded GLP-1s was price, and the price landscape for FDA-approved options has changed dramatically.
During the GLP-1 shortage (2022–early 2025), compounding pharmacies could legally mass-produce semaglutide and tirzepatide because brand-name versions weren’t available in sufficient quantities. That window is now closed.
Novo Nordisk and Eli Lilly told the FDA they can meet national demand. The FDA agreed. The courts upheld those decisions. And in March 2026, the FDA sent 30 warning letters to telehealth companies for making misleading claims about compounded GLP-1 products — the second wave since a broader crackdown began in September 2025.
Is Compounded GLP-1 Going Away?
For routine use, yes. The FDA declared both the semaglutide and tirzepatide shortages resolved, which ended the shortage-based compounding exception. Enforcement is active and accelerating.
Limited patient-specific compounding under section 503A of the FD&C Act may still be possible when a licensed clinician documents a genuine medical need that the FDA-approved product cannot meet. But “I prefer it cheaper” is not a documented medical need under FDA rules.
Is Compounded Semaglutide Still Available?
In limited cases, but not as a routine copy of Wegovy or Ozempic. The FDA resolved the semaglutide injection shortage on February 21, 2025. The enforcement discretion period for 503B outsourcing facilities ended May 22, 2025. State-licensed pharmacies compounding under 503A may still produce semaglutide for individual patients with a documented medical need — but not as a standard, mass-marketed product.
Is Compounded Tirzepatide Still Available?
For mainstream consumer use, essentially no. The FDA removed tirzepatide from the shortage list in October 2024. By March 2025, courts denied the compounding industry’s request for an injunction. The 503B grace period ended March 19, 2025. If you still see heavily marketed compounded tirzepatide, treat that as a verification problem, not proof it’s still a normal path.
Compounded GLP-1 Ban Update: 2026 Timeline
The phaseout dates already passed. Here’s the regulatory timeline:
| Date | What Happened |
|---|---|
| March 2022 | FDA adds semaglutide to drug shortage list — broad compounding begins |
| December 2022 | Tirzepatide added to shortage list |
| October 2, 2024 | FDA removes tirzepatide from shortage list |
| December 19, 2024 | FDA sets 90-day grace period for tirzepatide compounders |
| February 21, 2025 | FDA removes semaglutide from shortage list |
| March 5, 2025 | Judge denies OFA injunction for compounded tirzepatide; 503A discretion ends |
| March 19, 2025 | 503B grace period for compounded tirzepatide expires |
| April 24, 2025 | Semaglutide preliminary injunction denied; 503A discretion ends |
| May 22, 2025 | 503B grace period for compounded semaglutide expires |
| May 2025 | Judge rules in FDA's favor on tirzepatide compounding |
| September 2025 | FDA launches crackdown on misleading GLP-1 advertising |
| February 2026 | FDA announces intent to restrict APIs for mass-marketed non-approved GLP-1s |
| March 3, 2026 | FDA issues 30 warning letters to telehealth companies over compounded GLP-1 marketing |
Timeline sourced from FDA.gov.
The pattern is clear: enforcement is accelerating, not slowing down. And at the same time, approved access is getting cheaper and easier.
Not sure what to do next?
Take our 60-second GLP-1 matching quiz for a personalized plan.
Find My Best Option →Why Do I Still See Compounded GLP-1 Ads Everywhere?
Because the market didn’t vanish the day the shortages ended. IQVIA reported that compounded GLP-1 prescribing stayed active after the FDA declared the shortages over. Many companies kept offering “personalized” doses, alternate forms, or add-ins like B12 to argue their products weren’t simple copies.
The FDA isn’t buying it.
The “Personalized” Workaround
Under 503A, a state-licensed pharmacy can still compound a medication if a licensed clinician determines a specific patient has a medical need the commercially available product cannot meet. Some companies have used this rule to keep offering compounded semaglutide and tirzepatide — typically by adding B12, using non-standard doses, or offering oral/sublingual forms.
The FDA’s warning letters specifically targeted companies making claims that imply compounded products are equivalent to FDA-approved drugs, or that obscure who actually compounds the medication by branding it under the telehealth company’s own name.
Marketing Red Flags to Watch For
These are the exact claim types the FDA has flagged in enforcement actions:
| Claim You See | Why It’s a Red Flag |
|---|---|
| "Contains the same active ingredient as Wegovy/Ozempic" | FDA cited this exact claim type in warning letters — compounders cannot imply equivalence |
| "Clinically proven for weight loss" | FDA-approved clinical trials apply to approved products, not compounded versions |
| "FDA-approved compounded semaglutide" | No such category exists. "FDA-approved" and "compounded" are different things |
| No compounding pharmacy identified by name | Legitimate programs identify the state-licensed pharmacy producing the medication |
| Checkout before any real clinical evaluation | A licensed clinician should review your medical history before any prescription |
| Oral drops, gummies, patches, sprays | These delivery forms are not FDA-approved for semaglutide or tirzepatide |
| "Generic semaglutide" or "generic tirzepatide" | No FDA-approved generic exists in the US for either medication |

If This Is Your Situation, Here’s What to Do Next
Find your scenario. We’ve mapped the best next step for each one.
“I’m on compounded semaglutide right now”
Don’t panic, and don’t reorder blindly. Your immediate priority: know your exact current dose in milligrams, confirm which pharmacy compounds your medication, and understand why a compound was prescribed.
If the only reason was price or availability during the shortage, start planning a transition. On a medication-only basis, the Wegovy pill starts at $149–$299 per month, and the Wegovy pen starts at $199 per month for eligible new patients on starting doses.
If your clinician believes you have a genuine medical need for compounding — a documented allergy, a non-standard dose requirement — have them state the specific reason in writing.
Find Your Best Approved Match on Ro →Higher Urgency
“I’m on compounded tirzepatide right now”
This is the higher-urgency case. Tirzepatide’s shortage-based window closed earlier, the courts have ruled, and FDA enforcement is further along. If you’re still receiving compounded tirzepatide, verify with your provider how they’re justifying continued compounding under current rules.
Your practical next move: compare FDA-approved tirzepatide access. Zepbound single-dose vials are available starting at $299 per month through LillyDirect.
See Current Zepbound Pricing and Check Eligibility →“My provider just canceled my compounded GLP-1”
This is the exact moment this page was written for. The worst move right now is searching for another random compounded offer to fill the gap. The best move is comparing approved alternatives and prices before your current supply runs out.
Weight regain after GLP-1 discontinuation is well-documented — the priority is avoiding a gap in care, not finding another compounded source.
Find Your Best Approved GLP-1 — 60-Second Quiz →“I pay cash and brand-name prices scare me”
We understand. For years, the only number anyone saw was $1,000+ per month. That narrative is outdated. Here’s what FDA-approved GLP-1 access actually costs right now.
| Access Path | Medication Price | Additional Fees | Notes |
|---|---|---|---|
| NovoCare — Wegovy Pill | $149/mo (1.5 mg, 4 mg); $299/mo (9 mg, 25 mg) | None | 4 mg stays at $149 until Aug. 31, 2026, then $199. Direct from Novo Nordisk. |
| NovoCare — Wegovy Pen | $199 first 2 fills (0.25/0.5 mg new patients through June 30, 2026); $349/mo standard | None | Introductory offer for eligible new patients on starting doses only. |
| Ro — Wegovy Pill | $149–$299/mo (matches NovoCare) | Ro Body membership: $45 first month, $145/mo ongoing | Includes provider access, insurance concierge, coaching, dose management. |
| Ro — Wegovy Pen | $199–$349/mo (matches NovoCare) | Ro Body membership | Same medication pricing as NovoCare + clinical support. |
| LillyDirect — Zepbound | $299 / $399 / $449 depending on dose | None | Eligible commercially insured pen patients may pay as low as $25. |
| TrumpRx | Wegovy pill $149; Wegovy pen $199; Zepbound $299 | None | Government pricing program. Visit TrumpRx.gov for current listings. |
Medication prices verified from NovoCare, Ro, LillyDirect/Zepbound, and TrumpRx.gov on March 31, 2026.
A year ago, the choice was “$200 compounded or $1,300 brand-name.” That gap has nearly closed. The Wegovy pill at $149 per month through NovoCare — no membership, no middleman — is less than what many compounded programs were charging. You can now get FDA-approved, manufacturer-quality GLP-1 medication for the same money (or less).
Check Current Pricing and Eligibility on Ro →“I want a pill, not a shot”
That option now exists. The Wegovy pill (oral semaglutide) was FDA-approved in December 2025 and became available in January 2026. It’s the first oral GLP-1 approved specifically for weight loss.
You take it daily on an empty stomach, 30 minutes before eating or drinking anything else. In clinical trials, the highest dose produced an average weight loss of about 13.6% of body weight over 64 weeks — comparable to the injection. Pricing starts at $149 per month for the starting dose through NovoCare or Ro.
There’s more on the horizon: Eli Lilly has submitted orforglipron — an oral GLP-1 pill — to the FDA, with a potential decision expected in 2026. See our orforglipron overview for what the trial data shows.
See If the Wegovy Pill Is Right for You →“I have insurance but don’t know if it covers GLP-1s”
Check before you assume. Coverage has expanded: KFF’s 2025 survey found that 19% of firms with 200+ workers covered GLP-1s for weight loss, rising to 43% among firms with 5,000+ employees. If you work for a larger employer, odds are better than you think.
With qualifying commercial insurance, the Wegovy Savings Offer can bring costs to as low as $25 per month. Ro offers a free insurance checker and a dedicated insurance concierge team that handles prior authorization for you.
A coverage denial isn’t always the final answer. See our guide on getting tirzepatide covered by insurance and GLP-1 cost without insurance for more.
Check Your GLP-1 Insurance Coverage for Free on Ro →
Are Compounded GLP-1s Legal, Safe, and FDA-Approved?
Are Compounded GLP-1s Legal?
Compounding itself is a lawful practice with a long history. Under section 503A of the FD&C Act, a state-licensed pharmacy can still compound a medication when a clinician prescribes it for a specific patient whose medical needs can’t be met by the commercially available product.
What’s no longer legal in most situations is mass-producing compounded copies of semaglutide or tirzepatide based on the (now-resolved) drug shortage. Price or convenience alone doesn’t qualify as a medical need.
Are Compounded GLP-1s FDA-Approved?
No. FDA-approved medications go through years of clinical trials and rigorous review for safety, effectiveness, and manufacturing quality. Compounded medications do not. They are not FDA-approved products. This is a critical distinction — any program claiming “FDA-approved compounded semaglutide” is making a factually impossible claim.
Are Compounded GLP-1s Safe?
Compounded GLP-1s carry additional uncertainty because the FDA does not review them like approved finished drugs. The FDA states that compounded versions should only be used when a patient’s medical needs cannot be met by an FDA-approved drug, and the agency has received multiple dosing-error reports, some requiring hospitalization. As of July 31, 2025, the FDA had received 605 adverse-event reports for compounded semaglutide and 545 for compounded tirzepatide.
Approved products come in manufacturer-labeled packaging with defined dosing instructions. Compounded versions typically come in multi-dose vials requiring you to measure and draw your own injection — where many dosing errors originate.
How to Verify a Compounded GLP-1 Program
If you believe you have a legitimate medical need for compounded GLP-1, use this checklist:
✓ Green Flags
- The prescribing clinician states a specific medical reason the FDA-approved product won’t work for you
- The compounding pharmacy is identified by name and is state-licensed
- Your dose and formulation are documented in a patient-specific prescription
- The program clearly discloses the medication is not FDA-approved
✗ Red Flags
- Claims the product is “the same as” Wegovy, Ozempic, or Zepbound
- No compounding pharmacy is identified
- You can check out before any clinical review
- Advertised as “clinically proven” or “FDA-approved”
- Product comes as oral drops, gummies, patches, or sprays
Not sure if what you’re seeing is legitimate?
Get a Clinical Evaluation Through Ro →Compounded vs. FDA-Approved GLP-1: What Actually Changes
This isn’t about scaring you away from compounded medications. It’s about the math.
| Factor | FDA-Approved | Compounded (503A) |
|---|---|---|
| FDA-reviewed for safety and effectiveness? | Yes — full clinical trial program | No |
| Manufacturing oversight | FDA cGMP inspections | State pharmacy board oversight |
| Dosing format | Manufacturer-labeled tablets, pens, or single-use formats | Multi-dose vials requiring self-measurement |
| Dosing error risk | Very low | Higher — FDA reports errors requiring hospitalization |
| Legal status in 2026 | Fully legal with prescription | Legal only in narrow, documented circumstances |
| Insurance eligible | Yes (many commercial and some government plans) | No |
| Manufacturer savings programs | Yes — as low as $25/mo with qualifying insurance | No |
The entire case for compounded GLP-1s was always about one thing: price. Both of those conditions have changed. Supply has stabilized. Self-pay starting prices have dropped to $149/mo. When the price advantage disappears, what remains is less oversight, more variability, higher dosing error risk, no insurance eligibility, and an increasingly uncertain legal environment.
If you’re among the small number who genuinely need a compounded formulation for a documented medical reason, that path still exists. For everyone else, the move to approved options is overdue — and far easier than you think.
Looking for an affordable approved alternative?
MEDVi offers FDA-approved GLP-1 weight loss treatment with licensed clinician support.
Check Eligibility on MEDVi →How to Switch From Compounded to FDA-Approved Without Losing Progress
If you’re worried about weight regain during a transition, this is for you. Weight regain after GLP-1 discontinuation is well-documented — so the priority is avoiding a gap in treatment, not finding another compounded source. Transitioning between equivalent formulations with a clinician mapping the dose is very different from stopping cold turkey.
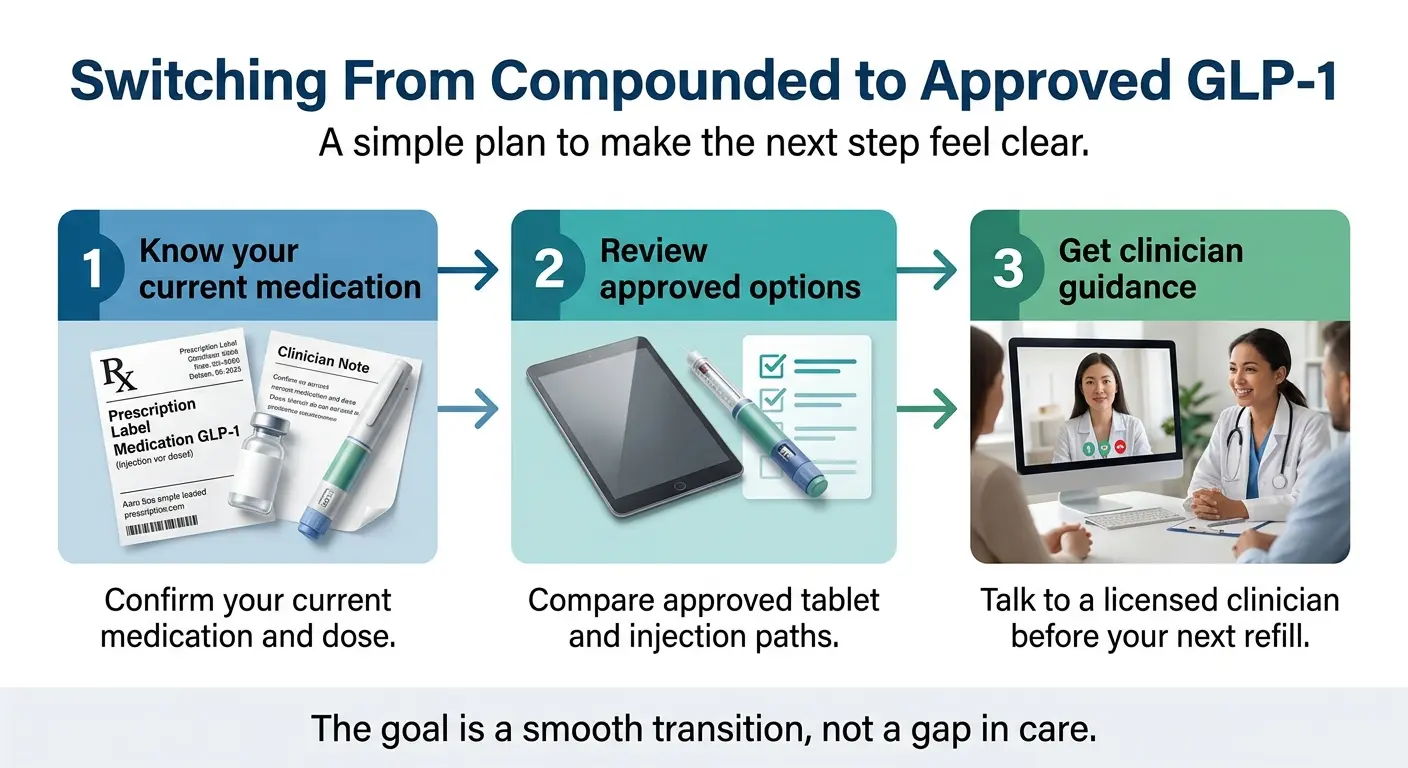
Your Transition Plan
Know your current dose in milligrams
Check your vial label or prescription records. Get the exact number — not just the brand name or frequency.
Start the conversation early
Don’t wait until your supply runs out. Telehealth evaluations can happen within days, and medication ships directly to you.
Find the FDA-approved equivalent
Work with a provider. Standard Wegovy pen doses range from 0.25 mg to 2.4 mg. Zepbound doses range from 2.5 mg to 15 mg. In most cases, there’s a direct match or a close one.
Expect a brief adjustment period
Minor side-effect differences between formulations are normal and usually mild. Stay in contact with your clinician during the first few weeks.
What to Ask Your Clinician
Five questions that matter most right now:
- 1.What exact product and dose am I currently taking? (Get this in milligrams.)
- 2.Why was I prescribed a compound? Was it price, shortage-era availability, or a specific medical need?
- 3.What FDA-approved product is the closest match?
- 4.If you’re recommending I stay on compounded, what is the documented medical reason? A vague answer is a signal to explore approved alternatives.
- 5.Can you help me navigate insurance coverage or manufacturer savings programs?
You don’t have to figure this out alone. That’s what licensed providers are for.
Start Your Transition Plan on Ro →What’s Coming Next for GLP-1 Access
Two developments worth watching:
Medicare Coverage
Medicare coverage starting July 2026
CMS’s Medicare GLP-1 Bridge will provide eligible Medicare Part D beneficiaries access to Wegovy and Zepbound for weight reduction at $50/month from July through December 2026. The BALANCE Model takes over in Part D starting January 2027 for participating plans.
Pipeline Drug
Orforglipron (oral GLP-1 from Eli Lilly)
Not yet FDA-approved. Lilly submitted it for obesity and has stated self-pay pricing would start at $149 for the lowest dose if approved. A potential FDA action for obesity could come in 2026. See our orforglipron overview and side effects guide for what the trial data shows.
The trend is unmistakable: GLP-1 access is getting cheaper, easier, and more diverse. If price was keeping you on a compounded path, that barrier is eroding fast.
Frequently Asked Questions
Is compounded GLP-1 still available?
For most people, not as a routine option. The FDA declared semaglutide and tirzepatide shortages resolved in 2025, ending the shortage-based compounding exception. Limited patient-specific compounding remains possible under section 503A when a clinician documents a genuine medical need. Plan around FDA-approved options first.
Did the FDA ban compounded semaglutide?
Not as a blanket ban, but the practical effect is similar for most consumers. The shortage-based mass-compounding window ended in 2025, and the FDA resumed enforcement. On March 3, 2026, the FDA issued 30 warning letters to telehealth companies over misleading compounded GLP-1 marketing.
Is compounded tirzepatide still available?
Routine compounded tirzepatide is no longer a normal consumer path. The shortage was declared resolved in October 2024, courts denied industry injunctions, and enforcement is active.
Are compounded GLP-1s FDA-approved?
No. FDA-approved GLP-1 medications (Wegovy, Ozempic, Zepbound, Mounjaro) go through rigorous clinical trial review. Compounded versions do not and are not FDA-approved.
How much does Wegovy cost without insurance in 2026?
The Wegovy pill starts at $149/mo for the starting dose through NovoCare. The Wegovy pen starts at $199/mo for eligible new patients on starting doses (through June 30, 2026), then $349/mo standard. With qualifying commercial insurance and the Wegovy Savings Offer, eligible patients may pay as low as $25/mo.
Can I switch from compounded to brand-name GLP-1 without gaining weight?
Weight regain is primarily associated with stopping GLP-1 treatment entirely. If you're transitioning to an equivalent dose with clinician guidance, the priority is maintaining continuity of care — not the brand on the label. Work with a provider to map the transition.
What is TrumpRx?
TrumpRx is a government direct-to-consumer pricing platform at TrumpRx.gov that launched in February 2026. It offers medication-specific pricing: Wegovy pill from $149/mo, Wegovy pen from $199/mo, Zepbound from $299/mo. Designed primarily for cash-paying patients.
Is there a GLP-1 pill for weight loss?
Yes. The Wegovy pill (oral semaglutide) was FDA-approved in December 2025 and is available starting at $149/mo. Eli Lilly's orforglipron is under FDA review and not yet approved.
Will compounded GLP-1 come back if there's another shortage?
The FDA can reinstate shortage-based compounding permissions if supply issues return. But basing current treatment decisions on that possibility isn't recommended. Plan around what's available and legal today.
How do I know if a GLP-1 program online is legitimate?
Look for: prescriptions of FDA-approved medications, a clearly identified licensed clinician, medical evaluation before any prescription, no claims that compounded products are 'equivalent' to approved drugs, and a state-licensed pharmacy. The FDA's BeSafeRx campaign offers additional resources.
How We Verified This Page
Regulatory information: FDA timeline dates, enforcement actions, and policy statements sourced from FDA.gov. Warning letter details from the March 3, 2026 FDA announcement.
Pricing data: Verified from NovoCare, Ro, LillyDirect/Zepbound, and TrumpRx.gov on March 31, 2026.
Insurance/coverage data: Employer coverage from KFF 2025 Employer Health Benefits Survey. Medicare coverage from CMS Medicare GLP-1 Bridge and BALANCE Model.
Safety data: FDA’s concerns about unapproved GLP-1 drugs. Adverse-event totals as of July 31, 2025.
Ready to find your approved GLP-1 path?
FDA-approved options now start at $149/mo. A licensed clinician can help you map the transition — with no gap in care.