By The RX Index Editorial Team · Sources: STEP-1 extension, STEP-4, SURMOUNT-4, eClinicalMedicine 2026 meta-regression, BMJ 2026 meta-analysis, Cleveland Clinic 2026 cohort
Last verified: · Evidence, pricing & provider details re-checked April 29, 2026
Editorial Standards · Affiliate Disclosure · Next audit: May 29, 2026
GLP-1 STOP & MAINTENANCE — 2026 EVIDENCE GUIDE
Weight Regain After Stopping GLP-1: 2026 Data + Stop Plan
Published:
The short, honest answer
Weight regain after stopping GLP-1 medication is common — but the amount that comes back depends on which version of “stopping” you’re doing, and most people don’t know there are five.
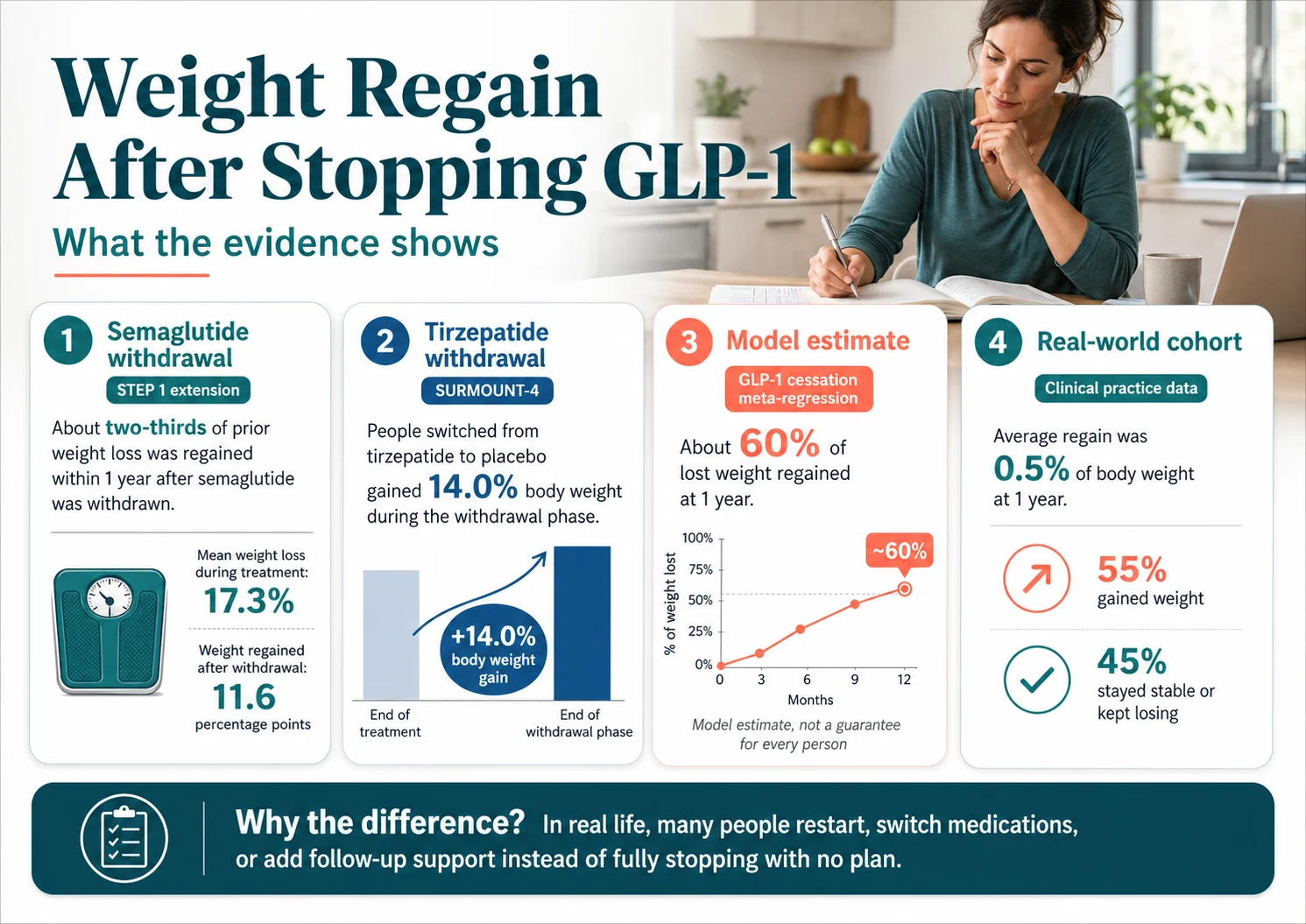
In the major withdrawal trials, participants who had semaglutide and lifestyle support discontinued regained about two-thirds of their lost weight within a year. People randomized off tirzepatide regained about 14 percentage points of body weight in the same window. A 2026 eClinicalMedicine meta-regression of GLP-1 cessation studies estimated 60% regain at week 52. Real-world cohorts look completely different: the Cleveland Clinic 2026 dataset (n=7,938) showed an average one-year regain of just 0.5%. Both are true. Both matter. The difference is what you do.
Disclosure: Some links on this page are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you.
Weight Regain After Stopping GLP-1: What Actually Happens (And What You Can Do About It)
Here’s the bottom line up front. In the major withdrawal trials, participants who had semaglutide and lifestyle support discontinued regained about two-thirds of their lost weight within a year. People randomized off tirzepatide regained about 14 percentage points of body weight in the same window. A 2026 eClinicalMedicine meta-regression of GLP-1 cessation studies estimated 60% regain at week 52 (the longer-term plateau in that model is a projection, not a directly observed outcome). And real-world cohorts look completely different: the Cleveland Clinic 2026 dataset (n=7,938) showed an average one-year regain of just 0.5%, primarily because most people who stopped switched, restarted, or layered on lifestyle support rather than fully stopping.
So the question isn’t really “will I regain weight after stopping GLP-1?” It’s which version of stopping are you doing? This page maps the five real options — continue, lower-dose maintenance, taper-with-infrastructure, switch, or hard stop — against the published evidence, and gives you the framework to decide. The goal is for you to walk into your prescriber’s office knowing exactly what to ask.
Quick answer table
| Question | Best current answer |
|---|---|
| Will I regain weight? | Common, not guaranteed. Depends on what replaces the medication. |
| How much? | Trial models estimate ~60% of loss back at 1 year with no plan; real-world averages can be much lower when people switch, restart, or maintain support. |
| How fast? | Appetite returns in weeks 2–6; weight regain typically becomes measurable months 2–6. |
| Best next step? | Use the 5-factor framework below, then bring it to your prescriber. |
Skip to: The Numbers · Decision Framework · Tapering Protocols · Take the Quiz
Will I regain weight after stopping GLP-1?
Some weight regain after stopping a GLP-1 is common, but it is not automatic and the amount varies widely by medication, by reason for stopping, and by what you do in the months that follow. The randomized withdrawal trials show substantial regain when people stop the medication and lifestyle support without replacement. The real-world cohort data show much smaller average regain, because most people don’t stop in that exact way. Both are true. Both matter.
The phrase you’ll see on most pages — “you’ll gain it all back” — is wrong, but only because it’s incomplete. The honest version: if you stop your GLP-1 with no replacement plan, no taper, no lifestyle infrastructure, and no follow-up, you should expect to regain a substantial portion of what you lost over the following 12–18 months. If you stop with a plan, your odds change.
Three things determine where you land on that spectrum:
- Whether the medication is replaced by a maintenance dose, a different medication, or a structured non-drug program
- Whether you have durable habits in place before the medication leaves your system — resistance training, protein, sleep, monitoring
- Why you stopped — voluntary cessation with a plan looks completely different from forced cessation due to cost, supply, or insurance loss
That’s the framework. The rest of this page is the evidence behind it and the practical decisions it leads to.
How much weight regain after stopping GLP-1 is typical?
Across the major studies, people who stop GLP-1 medications without a maintenance plan regain about 60–67% of their lost weight within 12 months on average in trial-modeled estimates. A 2026 eClinicalMedicine meta-regression projected the trajectory plateaus longer-term (roughly 75% of original loss eventually regained in the model), but that plateau is an extrapolation beyond the directly observed 52-week trial data. Real-world cohorts show much smaller average regain, primarily because most people don’t fully stop.
This is where the pages currently ranking on the topic fail you. They cite one trial, give you one number, and call it a day. The actual evidence is a range, and you need to see the full picture to know where you’ll probably land.
The full evidence matrix
| Source | Evidence type | Population | What happened after stopping | Practical translation |
|---|---|---|---|---|
| STEP-1 extension (Wilding et al., Diabetes Obes Metab 2022) | Trial extension, observed | Semaglutide 2.4 mg, n=327, withdrawn after 68 weeks of treatment + lifestyle | Mean weight loss 17.3% during treatment. Regained 11.6 percentage points (≈two-thirds of loss) within 52 weeks of withdrawal. Net retained: 5.6% below baseline. | If you lost 17% on semaglutide, expect to give back about 11–12% over the year if you stop with no replacement. |
| STEP-4 trial (Rubino et al., JAMA 2021) | Randomized withdrawal | Semaglutide 2.4 mg, switched to placebo at week 20 | Placebo-switched group regained 6.9% over 48 weeks. Continued group lost an additional 7.9%. | Continuing held the loss; switching to placebo reversed it. |
| SURMOUNT-4 (Aronne et al., JAMA 2024) | Randomized withdrawal | Tirzepatide 10/15 mg, n=670, after 36-week lead-in | Placebo-switched group gained 14.0% from week 36 to week 88. Continued group lost an additional 5.5%. About 9 in 10 continuers maintained ≥80% of weight loss. | Tirzepatide regain is steep in raw pounds because the loss was steep. |
| SURMOUNT-4 post-hoc (Horn et al., JAMA Intern Med 2025) | Post-hoc analysis | Tirzepatide withdrawal subgroup | 82% of those who stopped regained ≥25% of their initial weight loss within a year. Greater regain was associated with greater reversal of waist, blood pressure, A1c, and lipid improvements. | The scale isn’t the only thing on the line — your blood pressure, glucose, and waist often follow. |
| eClinicalMedicine meta-regression (Budini et al. 2026) | Meta-regression model | 6 RCTs, ~3,236 GLP-1 users, with longer-term projection | Modeled 60% of lost weight regained at week 52 (observed). Projected plateau around 75.3% regain longer-term — this plateau is extrapolated, not directly observed. | The first year is the danger zone with the strongest evidence. Beyond that, the trajectory softens — but the model has more uncertainty over time. |
| BMJ systematic review (West et al., BMJ 2026) | Meta-analysis | 37 studies, 9,341 participants, multiple anti-obesity meds | Average regain ≈ 0.4 kg/month after stopping across all weight-management meds. Subgroup of newer GLP-1s (semaglutide/tirzepatide): ≈ 0.8 kg/month. | About 1.8 lb/month average for semaglutide/tirzepatide users — roughly 21 lb/year. |
| Cleveland Clinic real-world cohort (March 2026) | Retrospective cohort | 7,938 adults, semaglutide/tirzepatide, 3–12 months use | Obesity-treatment group lost 8.4% before stopping, regained just 0.5% on average at one year. 27% switched medications, 20% restarted, 14% added lifestyle visits. | What people actually do after stopping shapes the outcome — many don’t fully stop. |
What this looks like in real pounds
Trial numbers are abstract. So we ran the math against the most cited estimates — the eClinicalMedicine 60% one-year figure and the STEP-1 two-thirds figure — across common starting losses. Treat this as a population-based translation, not a personal prediction.
If you lost this much on a GLP-1 and stopped with no replacement plan, here’s roughly what the trial-modeled data suggests over 12 months:
| Weight you lost | 60% regain estimate | Two-thirds regain estimate | What we’d want you to do with this number |
|---|---|---|---|
| 20 lb | 12 lb | 13 lb | Set a “call your doctor” threshold at 5–10 lb regained. |
| 30 lb | 18 lb | 20 lb | Build the maintenance plan before the last dose, not after the appetite returns. |
| 40 lb | 24 lb | 27 lb | Consider whether continuing, lower-dose maintenance, or switching is more realistic than a full stop. |
| 50 lb | 30 lb | 33 lb | Ask about labs, blood pressure, glucose, and replacement options. |
| 75 lb | 45 lb | 50 lb | Treat stopping as a medical transition, not a finish line. |
If those numbers feel scary, that’s fine. They should feel real, not abstract. They also don’t have to be your numbers — that’s what the rest of this page is about.
Honest moment from us
No page on the internet — including ours — can promise you’ll keep all your progress. If your weight gain is driven by ongoing biology, insulin resistance, sleep apnea, PCOS, or a long history of weight cycling, long-term medical treatment may simply be the more realistic maintenance path. We say that not to push medication on you, but because the worst outcome is stopping without a plan, regaining everything, and concluding you “failed.” You didn’t fail. The plan failed.
Build your stop plan.
The framework below maps your situation against the evidence above. It takes about 60 seconds and outputs a specific next step you can bring to your prescriber.
→ Take the GLP-1 path quizHow fast does weight come back after stopping GLP-1?
The medication itself takes about 5 weeks to clear your system after the last dose (5–7 weeks for Wegovy 2.4 mg per public safety language), but appetite often starts returning before then. Meaningful regain usually shows up between months 2 and 6. The first 12 months after stopping is the highest-risk window in the available trial and modeling data.
Two pharmacokinetic facts you should know going in:
- Semaglutide (Ozempic, Wegovy, Rybelsus) has a half-life of about 7 days. It typically takes about 5 weeks to clear, with Wegovy 2.4 mg labeling commonly citing 5–7 weeks for full washout.
- Tirzepatide (Mounjaro, Zepbound) has a half-life of about 5 days. It takes roughly 4–5 weeks to clear.
That doesn’t mean appetite comes back on a clean schedule. It comes back gradually — and most people notice it before the scale moves.
The realistic timeline
| Time after last dose | Drug status | What you’ll likely feel | Confidence |
|---|---|---|---|
| Days 1–7 | Drug still active | Largely the same. “Food noise” may quietly start returning by end of week. | Pharmacokinetic |
| Weeks 2–4 | Drug levels declining substantially | Appetite gradually returning. Stomach empties faster. Cravings notice. | Pharmacokinetic + clinical observation |
| Weeks 5–7 | Largely cleared (Wegovy 2.4 mg may extend) | Hunger near pre-treatment baseline for many people. Food noise back. | Pharmacokinetic + clinical observation |
| Months 2–3 | Fully cleared | Old habits start pulling. Decision fatigue around food returns. | Trial trajectory data |
| Months 6–12 | — | Highest-risk regain window. Trial-modeled estimate: ~60% of loss back by month 12 with no replacement. | Direct observation (trial extensions) |
| Months 14–18+ | — | Regain trajectory may slow according to model projections, but uncertainty grows beyond 52 weeks. | Modeled projection, not directly observed |
If you have type 2 diabetes, your blood sugar may also rise during this window — sometimes within days to weeks of clearance. That’s a separate clinical issue from the weight regain and needs to be managed by your prescriber, not the bathroom scale. We cover this in more detail in the diabetes section below.
Why does weight regain happen after GLP-1s? (The biology, plain)
GLP-1 medications work by activating a hormone receptor that reduces appetite, slows stomach emptying, and quiets “food noise.” When the medication leaves your system, those effects leave with it — and the underlying biology that made weight loss difficult in the first place tends to come back. This is not willpower failing. It’s a treatment effect ending.
There’s a moment in almost every GLP-1 user’s experience that’s worth naming. It usually shows up around week 4 or 5 after the last dose. They walk past the kitchen and think about food in a way they hadn’t in months. The mental quiet is gone. The cravings have a voice again. Reddit users call it “food noise coming back,” and it’s the single most consistent thing we hear from people who stopped.
Three things are happening biologically:
1. Appetite signals shift back. GLP-1 medications mimic a natural gut hormone that signals fullness and slows digestion. When you stop, your body’s appetite and satiety regulation tends to drift back toward its pre-treatment pattern. The meal-to-meal sense of fullness disappears.
2. The body defends a lower weight. This is well-documented across all weight-loss approaches, not just GLP-1s. After significant loss, resting energy expenditure tends to drop and appetite signals tend to increase — the body acts like it’s trying to get back to its prior weight. The GLP-1 was effectively counteracting that defense. Without it, the defense wins.
3. Cardiometabolic improvements often reverse. This is the part most pages skip. The 2025 SURMOUNT-4 post-hoc analysis found that participants who regained ≥25% of their tirzepatide weight loss also showed measurable reversals in waist circumference, blood pressure, non-HDL cholesterol, fasting glucose, and insulin resistance — with greater regain associated with greater reversal. Those who regained less kept more of the metabolic gains. The takeaway: the scale isn’t the only thing on the line.
What this is not: classic chemical “withdrawal.” GLP-1 medications aren’t habit-forming. You’re not going through detox. You’re losing a treatment effect, the same way someone might lose blood-pressure control after stopping a blood-pressure medication. That distinction matters because the framing changes the plan.
Can you stop a GLP-1 and actually keep the weight off? (Real-world evidence)
Some people maintain most of their weight after stopping a GLP-1, and the patterns are predictable. The Cleveland Clinic 2026 cohort showed that successful maintainers often did one of three things: switched to another anti-obesity medication, restarted the original medication, or paired their stop with structured lifestyle visits.
The Cleveland Clinic 2026 cohort is the most useful counterweight to the trial data. Across 7,938 adults who stopped semaglutide or tirzepatide after 3–12 months of use, the average one-year regain in the obesity-treatment group was just 0.5% of body weight. About 55% gained weight back; 45% stayed stable or kept losing.
That’s not a contradiction of the trial data. It’s the difference between a clinical withdrawal study (placebo-controlled, no replacement, no support) and real life (where 27% switched medications, 20% restarted, and 14% added lifestyle visits per the same cohort). The trial number is what happens in a controlled withdrawal setting. The real-world number reflects what happens when people engage with the problem.
Who tends to maintain more often
Based on the published evidence and clinical observation, these factors tilt the odds — they’re not guarantees:
- The original weight gain came from a specific, time-limited trigger that’s now resolved
- Built durable habits during treatment — resistance training 2–3×/week, consistent protein intake, regular sleep, a tracking habit
- Has a clinician relationship for follow-up, not just a one-time prescription
- Self-weighs regularly without spiraling
- Has a financial and support runway to continue treatment if needed
Who tends to regain more often
- Long history of weight cycling
- Type 2 diabetes or significant insulin resistance
- Severe food noise returning quickly after the last dose
- Stopped abruptly because of cost, supply disruption, or insurance loss with no replacement plan
- No clinician follow-up
- Significant muscle loss during the active treatment phase
That last point matters more than most pages give it credit for. We’ll cover it next.
What people on Reddit are actually saying (individual experiences, not clinical evidence)
- “I went off for a month and gained 6 lbs.” — r/Ozempic
- “Stopped 4 months ago and have gained nothing back… eating healthy and tracking my food intake and gym 5 days a week.” — r/Ozempic
- “It isn’t as easy as it was on ozempic, but it’s not impossible either.” — r/Ozempic
- “I know once that food noise comes back in my head it’s game over for me.” — r/Ozempic
We include these because they reflect what we hear from readers all week. They are not typical-result claims. They are individual posts.
Body composition: what you regain vs. what you lost
A 2026 systematic review and meta-analysis in the International Journal of Obesity found that GLP-1-related weight loss is predominantly fat mass, with lean-mass reductions that were modest overall — not the extreme muscle-loss picture some headlines have suggested. But what you regain after stopping appears to come back disproportionately as fat in preclinical models, which is why protecting muscle during treatment matters even more than during loss.
This deserves a careful read because the topic is full of overclaims in both directions.
The accurate picture from the 2026 meta-analysis: Fat-mass loss predominated during GLP-1 treatment. Lean-body-mass reductions were real but modest in pooled data. Visceral fat (the dangerous kind, around organs) was preferentially reduced. That’s a more positive body-composition story than the “you’re losing all muscle” headlines suggest.
The concerning preclinical signal: A 2026 University of Pennsylvania study in mice — not yet replicated in humans — found that animals who repeatedly stopped and restarted GLP-1 medications became progressively less responsive: they lost less weight on each restart cycle, and the weight they regained between cycles was almost entirely fat. The researchers proposed a “muscle floor” mechanism. This is animal data. It’s not proven in people. But it lines up with the broader weight-cycling literature.
Why this still changes the math
Even modest muscle loss during weight loss matters more after stopping than during. If you regain weight and the regain skews toward fat (as the preclinical work suggests), your body composition can drift in the wrong direction even at the same scale weight. Your resting metabolic rate may be lower. Your future ability to respond to weight management may be harder.
This is the strongest evidence-based argument for two practical decisions:
- Resistance training during AND after GLP-1 treatment — protective of the lean mass that protects you
- Avoiding repeated stop-and-restart cycles if you can — each cycle may be biologically less efficient, based on preclinical signals
We’ll get to the practical version of both in the lifestyle section below.
Should you taper, lower-dose, switch, restart, or stay on maintenance? (Decision framework)
There is no single right answer for everyone, and there is no FDA-approved tapering schedule for any GLP-1 medication. The right path depends on five specific factors. Most readers fit into one of three patterns: continue at full dose (strongest evidence to maintain loss), continue at a lower maintenance dose (common in clinical practice, less RCT validation), or taper-with-infrastructure (the Embla 9-week protocol has the strongest published evidence for non-trial settings).
We built this framework after reading every published trial, every meta-analysis, and the clinical commentary in the obesity medicine literature through April 2026. It’s our synthesis. It’s not a substitute for your prescriber. It’s what to bring to your prescriber.
The 5 factors
Score yourself on each. Pattern recognition matters more than math.
1. Do you have a cardiometabolic indication that improved on the GLP-1?
Type 2 diabetes, prediabetes, hypertension, sleep apnea, fatty liver disease, established cardiovascular disease. If yes, you’re not just managing weight — you’re managing a chronic condition the medication is treating. Stopping reverses both. Continue or maintain at lower dose unless your clinician has a specific reason to stop.
2. What was your starting BMI and metabolic vulnerability?
A BMI in the high 20s with no comorbidities is a different stop calculus than BMI 38 with two comorbidities. The higher and more vulnerable you started, the stronger the case for ongoing treatment.
3. Have you built durable lifestyle infrastructure during treatment?
Resistance training 2–3×/week for at least 3–6 months. Consistent protein. Tracking habit. Sleep that you actually defend. This isn’t moral — it’s mechanical. The Embla cohort that showed taper success had this. People who don’t have it typically regain.
4. Are you 50+ or already showing signs of muscle loss?
Sarcopenic obesity risk rises with age. If you’re 50+ and already lost meaningful muscle on the way down, full cessation is higher-risk than for a 35-year-old who weight-trained throughout treatment. Maintenance dosing is more protective.
5. Are you stopping by choice or by force?
Voluntary cessation with a plan is one situation. Forced cessation — cost spike, insurance loss, supply disruption, compounded source change — is a different one and should trigger a replacement plan, not a hard stop.
What the patterns suggest
| Your pattern | Most evidence-supported path |
|---|---|
| Strong “yes” on factors 1, 2, or 4 | Continue at full or lower maintenance dose. STEP-4 and SURMOUNT-4 both showed continued treatment groups maintained loss; tirzepatide continuation produced additional loss. |
| Strong “yes” on factor 3, low on 1, 2, 4 | Tapered cessation with structured lifestyle support has the strongest non-medication evidence (Embla 9-week, ECO 2024). |
| Forced cessation (factor 5) | Replace, don’t stop. Compounded providers, cash-pay branded options, or insurance appeals are the bridge. |
| Mixed picture, unsure | Lower-dose maintenance — common clinical pattern, lower cost than full dose, lower risk than full stop. |
If you want to walk through this against your specific situation, that’s what our quiz is for.
Want a personalized stay/taper/stop plan you can bring to your prescriber?
Our free 60-second quiz maps your answers against this framework and outputs a specific next step. No signup, no provider redirect — just the plan.
→ Take the GLP-1 path quizTapering off GLP-1: the protocols with the most evidence
There is no FDA-approved tapering schedule for any GLP-1 medication. The most-cited published approaches are the Embla 9-week real-world taper (presented as posters at the 2024 European Congress on Obesity), the Oxford clinical-observation 8–12 week stepped approach, and the published switch-and-bridge strategy using older anti-obesity medications. All three are clinician-led approaches, not RCT-validated protocols.
If you and your prescriber have decided you want to taper, here’s the honest landscape of what exists.
The Embla 9-week protocol (best real-world evidence)
Presented at the European Congress on Obesity 2024 by Dr. Henrik Gudbergsen and colleagues. The broader Embla program enrolled 2,246 adults using semaglutide alongside coaching. Among the subset who reached goal weight and tapered to zero over a median of 9 weeks, 85 participants had 6-month post-taper follow-up data, and weight remained stable in that subset.
Key features:
- Tapering happened after goal weight was achieved
- The cohort’s average maximum semaglutide dose was 0.77 mg — lower than the standard 2.4 mg
- Active coaching continued throughout the taper and after
- This is conference poster real-world data, not an RCT
This is the closest thing we have to evidence for a “stop completely and keep the weight off” protocol. But notice the conditions: lower dose throughout, structured coaching, taper at goal — not at the start of frustration. Without those conditions, this isn’t the protocol you’re running.
The Oxford 8–12 week stepped approach
Published in Obesity and Endocrinology (Oxford Academic, September 2025) as clinical observation. Patients stepped from 2.4 mg → 1.0 mg → 0.5 mg → off over 8–12 weeks. The authors described more stable appetite regulation and less anxiety than abrupt cessation. They explicitly labeled this as anecdotal, hypothesis-generating, and not validated by randomized trials.
Switch-and-bridge
A 2024 analysis (Paddu et al.) found that switching from a GLP-1 to older anti-obesity medications — metformin and topiramate were among the most-used options — after 12 months helped maintain weight loss for up to 2 years in some patients. This isn’t a taper, but it’s a relevant alternative if your goal is “off injections” rather than “off all weight medication.”
The protocols, side by side
| Protocol | Source | Evidence type | Duration | What it requires | Best fit |
|---|---|---|---|---|---|
| Embla 9-week taper | Gudbergsen et al., ECO 2024 (poster) | Real-world cohort, n=85 with follow-up | ~9 weeks (median) | Reached goal + active coaching/program support | Voluntary cessation with infrastructure |
| Oxford 8–12 week stepped | Oxford Academic 2025 | Clinical observation, anecdotal | 8–12 weeks | Prescriber willing to write declining doses | Patients with stable habits |
| Switch-and-bridge | Paddu et al., 2024 | Retrospective analysis | Variable | Prescription for replacement | Want off GLP-1 but accept other pharmacotherapy |
| Cold stop | (Default if no plan) | — | 0 | Nothing | Only when forced (acute side effect, pregnancy) |
The damaging admission we owe you
Even the best tapering protocol isn’t guaranteed. The Embla data — the most encouraging real-world taper evidence — came from people inside an active digital coaching program who tapered at goal weight on lower-than-standard doses. People who taper at home, alone, after a price hike, on full doses, without support, do worse. If you’re not in a position to put real infrastructure in place, a low-dose maintenance protocol is more likely to protect your progress than a taper-and-hope approach. That’s the honest read of the evidence, not the marketing one.
A taper is a clinical conversation, not a DIY project.
If tapering is the right path for you, you need a prescriber who will write declining doses on a planned schedule and follow you through the transition. Our quiz routes you to providers known for flexible dose management.
→ See your match in the path quizGLP-1 maintenance dosing: the lower-dose option
Maintenance dosing means continuing GLP-1 medication at a lower dose after reaching goal weight, with the goal of preserving appetite regulation without continued active loss. STEP-4 and SURMOUNT-4 both showed continued treatment preserved weight loss. Lower-than-standard maintenance doses are used widely in clinical practice but are less validated by large randomized trials than full-dose continuation.
For many readers, this is the path the evidence actually supports — not “stop forever,” but “use less for less money, indefinitely.” Here’s what that looks like.
What “maintenance dose” actually means
A maintenance dose is a step-down from your weight-loss dose to the lowest dose that holds your weight stable. For semaglutide, that might mean stepping from 2.4 mg back to 1.0 mg or even 0.5 mg weekly. For tirzepatide, from 15 mg back to 7.5 mg or 5 mg. The Embla cohort effectively ran their entire program at a lower maximum dose (0.77 mg semaglutide average) and still produced strong loss and stability.
What the evidence supports — and doesn’t
- Strongly supported: Continued treatment at full dose maintains weight loss (STEP-4, SURMOUNT-4 continuation arms)
- Supported by real-world clinical observation: Lower maintenance doses can hold weight in many patients
- Not yet RCT-validated: Specific dose-reduction protocols (e.g., “drop to half dose for 6 months, then quarter dose”) — these exist in practice but lack large randomized comparisons
- Speculative: Microdosing (sub-therapeutic doses below 0.25 mg semaglutide weekly) is not supported by published RCT evidence for weight maintenance
The cost math that often favors maintenance
Here’s an underappreciated point: if the Penn Medicine “muscle floor” preclinical finding holds in humans, restarting after a stop may produce diminished weight loss compared to continuous treatment. That means the stop-then-restart-later strategy might actually cost you more — both biologically and financially — than staying on a low-dose maintenance protocol the whole time.
Maintenance dose strategies, plainly
| Strategy | Pattern | Evidence | Where it tends to fit |
|---|---|---|---|
| Full maintenance | Continue weight-loss dose | Strongest — STEP-4, SURMOUNT-4 | FDA-approved branded medication, especially with insurance coverage |
| Lower-dose maintenance | Step down to ~0.5–1.0 mg semaglutide or 5–7.5 mg tirzepatide | Clinical observation; real-world cohort data | Compounded telehealth often offers more dose flexibility for this approach |
| Microdosing | Sub-therapeutic weekly dose | Not RCT-validated for weight maintenance | Some compounded providers offer this off-label; verify with your specific provider |
| Every-other-week dosing | Standard dose, half frequency | Limited published data | Compounded providers; brand-name follows standard schedule |
If your prescriber is open to maintenance dosing, the next question is which provider supports the path. Some prescribers and programs only write standard maintenance schedules. Some compounded telehealth providers offer more dose flexibility — but compounded GLP-1 medications are not FDA-approved, and the FDA has stated that compounded drugs should generally be considered when a patient’s needs cannot be met by an FDA-approved drug.
Match yourself to a provider that fits the path you chose.
The quiz outputs a specific match based on your dose strategy, your insurance situation, and whether FDA-approved or compounded fits your needs.
→ Take the 60-second match quizWhat to do BEFORE your last GLP-1 dose (10-point checklist)
The biggest mistake people make is waiting until appetite has fully returned and weight has started climbing before they call their prescriber. By that point, you’ve lost the window where the smallest interventions work. Build the plan before the last dose.
If you’re considering stopping in the next 1–6 months, run this checklist now.

- Tell your prescriber why you want or need to stop. Cost, side effects, goal reached, pregnancy, surgery, or curiosity — the answer changes the plan.
- Record your current weight, waist, and most recent labs. A1c, fasting glucose, blood pressure, lipids. These are your “before” baseline for the off-ramp.
- Decide which path you’re on: continue, lower dose, switch, taper, or stop with monitoring. Don’t leave that ambiguous.
- Set a regain threshold that triggers action. We use 5%, 10%, and 25% of lost weight regained — see the next section.
- Build a protein and fiber plan you can actually follow. Not a perfect one. A repeatable one. Discuss specific targets with your clinician or a registered dietitian rather than copying numbers from the internet.
- Start resistance training 2+ days per week before stopping, not after. Muscle preservation is the strongest practical lever you control. CDC’s adult activity guidance is the floor: 150 minutes/week moderate activity plus 2 days of muscle-strengthening activity.
- Schedule a 4–8 week post-stop follow-up. Not “we’ll see how it goes.” A booked appointment.
- Plan in advance what to do when food noise returns. It’s not “if.” Have a written response — extra protein, an emergency snack list, a 10-minute walk rule, a text-your-coach protocol.
- Confirm diabetes medication changes if applicable. Stopping a GLP-1 can require insulin or sulfonylurea adjustment. Don’t manage this alone.
- Write your “restart or switch” threshold and put a date on it. “If I’m at +10 lb regained at month 4, I will call my prescriber and discuss restarting.” Decide it now, in calm. Not later, in panic.
How to actually lower your regain risk after stopping (the real lifestyle pieces)
The published research and clinical observation point to four levers that consistently separate maintainers from regainers: structured resistance/strength activity continued after stopping, protein adequacy, ongoing monitoring, and behavioral support. The “eat protein and fiber, walk more” advice on most pages isn’t wrong — it’s just incomplete without the structural piece.
These aren’t sexy. They’re the things that work.
Resistance training (the strongest practical lever you control)
The strongest practical intervention against post-GLP-1 regain is regular resistance training. It protects the lean mass you have. It increases insulin sensitivity. It raises resting energy expenditure. And given the preclinical signal that regained weight tends to be predominantly fat, protecting muscle now is the bet that pays out later.
Practical floor: 2 days per week, full-body, progressive load. CDC’s adult guidance backs this minimum. More is better if you can sustain it. You don’t need a gym. You need consistency.
Protein and dietary structure
Protein at every meal supports satiety (delayed gastric emptying mimics what the GLP-1 was doing) and protects muscle. Specific gram targets vary by body weight, age, and clinical context — discuss yours with a clinician or registered dietitian rather than copying a number from a fitness blog. Spread protein across meals rather than loading dinner. Fiber matters for satiety too. Whole grains, beans, vegetables.
Self-monitoring without spiraling
The NIDDK recommends regular weighing as one of the most consistent maintenance behaviors in long-term studies — but it’s a tool, not a verdict. Pair it with waist measurement (more sensitive than weight to body composition shifts) and basic food logging if it works for your psychology. If self-weighing makes your mental health worse, talk to your clinician about alternatives.
Behavioral support (the boring habit that actually works)
The Embla cohort that maintained weight after tapering had active coaching. People who maintained weight in published research had structured support. People who quietly regained weight at home didn’t. This isn’t optional — it’s the structural piece.
Options:
- A registered dietitian (RDN) with obesity medicine experience
- An obesity medicine clinician (OMA-affiliated providers)
- A health coach
- A digital weight-management program with real human accountability
- A therapist or eating-disorder specialist if disordered eating is part of the picture
Things that probably matter less than the marketing suggests
- Most “metabolism-boosting” supplements
- Specific timing of meals (intermittent fasting may help some people, but it’s not a regain-prevention magic bullet)
- “Detox” protocols
- Most cleanses
The lifestyle you sustain is the lifestyle that protects you. Pick the smallest set of habits you can actually do for 12 months and keep doing them.
What if you’ve stopped and weight is already coming back? (The 5/10/25% framework)
If regain has already started, the most important thing is to respond early. We use a 5/10/25% threshold framework: 5% of your lost weight regained is the early-warning signal, 10% is the “call your prescriber” line, and 25% is the “treatment-plan reassessment” line — drawn from the SURMOUNT-4 post-hoc analysis where ≥25% regain was the category associated with greater reversal of cardiometabolic improvements. The earlier you respond, the more options you have.
Convert your regain into a percentage of your lost weight, not a raw number. A 4 lb regain after 40 lb of loss is 10%. After 80 lb of loss, it’s 5%. The percentage is what tells you how the trajectory is bending.
| Threshold | If you lost 40 lb, this is | What we’d suggest |
|---|---|---|
| 5% of loss regained | 2 lb back | Watch the trend. Pull the original plan back into focus. Track weight weekly, not daily. |
| 10% of loss regained | 4 lb back | Call your prescriber. Discuss whether to add infrastructure, switch, or restart at a low dose. |
| 25% of loss regained | 10 lb back | Schedule the appointment. This is the regain category SURMOUNT-4 post-hoc associated with meaningful cardiometabolic reversal. |
| 50% of loss regained | 20 lb back | Reassess the full plan. Lifestyle alone is rarely enough to reverse this without medical support. |
| 75%+ of loss regained | 30+ lb back | Treat as relapse of a chronic condition, not personal failure. Restart options, switch options, and escalated medical management are on the table. |
What about restarting? (And what dose?)
Restarting a GLP-1 after a stop is a real option. It’s not failure. It’s pattern recognition.
A few practical points:
- Don’t restart at your previous high dose without prescriber guidance. Your tolerance has likely reset during the stop period. Restarting at the top dose typically causes more side effects than re-titrating.
- Most prescribers re-titrate from a lower starting dose (e.g., semaglutide 0.25 mg or 0.5 mg, tirzepatide 2.5 mg or 5 mg) and step back up over weeks.
- The Penn Medicine 2026 preclinical work raised the possibility that repeated stop-restart cycles may be less effective in animal models — this hasn’t been replicated in humans yet, but argues for minimizing unnecessary cycling when possible.
- If you stopped because of side effects, talk to your prescriber about whether a different GLP-1 (or a different dose escalation pattern) might work better — semaglutide and tirzepatide have different tolerability profiles for the same person.
What if you have type 2 diabetes? (The glucose question after stopping)
If you started a GLP-1 for type 2 diabetes management, stopping affects your glucose control as much as your weight. Blood sugar can rise within days to weeks of clearance. This is a coordinated-care conversation with your prescribing clinician — not a decision to make based on a weight-management article.
Here’s what changes when diabetes is part of the picture:
- The GLP-1 is doing two jobs. It’s lowering your weight AND lowering your blood sugar. Stopping reverses both.
- Other diabetes medications may need adjustment. If you’re on insulin or a sulfonylurea, those doses may have been reduced when the GLP-1 was added. They may need to go back up — or you may need to monitor carefully for hypoglycemia in the transition.
- A1c will likely rise if no replacement diabetes management is in place. Your prescriber should be checking A1c at 3 and 6 months post-stop at minimum.
- The cardiovascular and kidney protection that some GLP-1s provide for people with diabetes (the FDA-approved cardiovascular and chronic kidney disease indications for semaglutide, for example) ends when the medication does.
This is one of the strongest arguments for not stopping a GLP-1 if you have diabetes unless there’s a clear reason to. If cost is the issue, exhaust the affordability options (insurance, prior auth, manufacturer programs, lower-cost compounded alternatives) before stopping the medication that’s actively managing your blood sugar.
What if cost or insurance is forcing you off? (The access pathway)
Cost-driven stopping is the single most preventable kind of GLP-1 cessation. Before you quit a medication that’s still working, run through the access options: insurance with prior authorization support, manufacturer/cash-pay branded pricing, transparent pharmacy programs, or compounded telehealth alternatives. If access is the only reason you’re stopping, you may not need to stop at all.
We are an affiliate site. We will earn a commission if you click through to a provider from this section. We disclose that openly. We also believe — based on every reader who has come through our site after stopping for cost — that working through these options is almost always cheaper and less harmful than stopping cold.
First, identify which access problem you actually have
| Problem | Best next step |
|---|---|
| Insurance denied my GLP-1 | Check insurance coverage with a prior-authorization-capable provider |
| Insurance covered it, then dropped it | Same — many people get it back through PA appeals |
| I’m paying retail brand price out of pocket and it’s unsustainable | Check transparent cash-pay pharmacy programs OR compounded telehealth |
| My compounded source disappeared (FDA enforcement, supply, telehealth shutdown) | Switch to a different verified compounded provider OR transition to FDA-approved brand |
| I’m on Medicare/Medicaid | Most retail telehealth programs cannot coordinate government coverage; speak with your prescriber about alternatives |
If insurance or prior authorization is the issue: Ro
For readers whose problem is “I want my insurance to cover this,” Ro is currently the strongest fit on our site. Ro publicly offers a free GLP-1 Insurance Coverage Checker, a dedicated insurance concierge that handles prior-authorization paperwork, and matches LillyDirect / NovoCare / TrumpRx pricing on FDA-approved branded medication. Ro carries Zepbound® (tirzepatide) and Foundayo™ (orforglipron), among other FDA-approved GLP-1 options.
Ro pricing (verify current at ro.co before enrolling): Ro Body membership is $39 for the first month, then $149/month — or as low as $74/month with annual plan paid upfront. Medication costs are separate unless covered by your insurance or available through a manufacturer cash-pay price Ro matches.
One material limitation: Ro states it cannot coordinate coverage for government-funded plans, with the exception of Federal Employee Health Benefits (FEHB) plans. If you have Medicare or Medicaid, this isn’t your path — talk to your prescriber about direct manufacturer programs or other options.
Free, takes about 90 seconds.
→ Check your GLP-1 insurance coverage on RoIf you want self-pay continuity: Eden, MEDVi, and Amazon One Medical
For readers whose insurance won’t cover GLP-1s and the manufacturer brand price is unworkable, three paths are worth knowing about.
Eden — FDA-approved branded, no membership fee
Eden carries FDA-approved Wegovy and Zepbound with no membership fees and same pricing at every dose. As of recent verification, Eden’s public pricing for Wegovy is $1,695/month and Zepbound is $1,399/month — these are mainstream cash-pay brand prices, not low-cost prices. Eden’s HSA/FSA-eligible language and no-membership-fee structure make it a clean option if FDA-approved branded medication is what you want and price isn’t the blocker. (Confirm current pricing at tryeden.com.)
MEDVi — compounded GLP-1, lower price point
MEDVi offers compounded GLP-1 options (compounded medications are not FDA-approved) at lower price points than retail brand. As of public-page verification, MEDVi’s program starts at $179 for the first month and $299/month for refills, with no contract and medication included.
About MEDVi specifically: We feature MEDVi for cash-pay continuity because the pricing structure works for many readers stopping for cost reasons. You should also know that the FDA issued a warning letter to MEDVi in February 2026 concerning marketing claims about compounded semaglutide and tirzepatide products — specifically, claims that misleadingly characterized the relationship between compounded products and FDA-approved versions. This is a labeling and marketing concern, not a stated safety recall. We mention it because a reasonable reader making a $300/month decision deserves to know. Verify current MEDVi marketing claims, product information, and your state’s compounded GLP-1 regulations before enrolling.
Amazon One Medical — transparent-pricing newcomer
Amazon One Medical (launched April 21, 2026) integrates Amazon Pharmacy with One Medical telehealth visits and offers transparent pricing: oral GLP-1 options starting at $149/month and injectable options starting at $299/month, with same-day delivery in nearly 3,000 cities. We’re not affiliated with Amazon, but we mention it because for readers stopping purely because of cost, this is a credible new access path that didn’t exist a year ago. (Read Amazon’s announcement.)
Compliance note
We never describe compounded GLP-1 medications as having the “same active ingredient” as FDA-approved versions, and we don’t claim compounded medications are “clinically proven.” Compounded and FDA-approved medications are regulated differently, and the FDA has stated that compounded drugs should generally be considered only when a patient’s needs cannot be met by an FDA-approved drug. The FDA has also issued public concerns about unapproved GLP-1 products including dosing errors, salt-form issues, and storage concerns. Verify the prescribed medication, the pharmacy, the source disclosure, your state’s availability, and the cancellation terms with any compounded provider before enrolling.
What we actually verified for this section (as of April 29, 2026)
- Ro’s Insurance Coverage Checker URL, pricing structure, and government-plan limitation: verified at ro.co
- Ro’s product list (Zepbound, Foundayo): verified at ro.co
- MEDVi pricing structure ($179 first month / $299 refill, no contract): verified at glp.medvi.org
- MEDVi FDA warning letter (February 2026): verified at fda.gov
- Eden’s brand pricing structure ($1,695 Wegovy / $1,399 Zepbound, same price at every dose): verified at tryeden.com
- Amazon One Medical GLP-1 Management Program ($149/$299 starting prices, April 21, 2026 launch): verified via Amazon’s announcement
- We did NOT verify each medication’s specific real-time price on each provider’s checkout flow today. Confirm at the provider’s site before enrolling.
If cost is the only reason you’re stopping, check your options before your last dose.
Stopping a medication that’s working — when an affordable alternative exists — is the most preventable kind of regain.
What if you’re stopping because of side effects?
If side effects are why you want to stop, the next step is your prescriber, not a provider comparison. Dose, titration speed, medication choice, hydration, and supportive medications can all change the risk-benefit calculus before you decide to fully stop.
Common tolerability issues that may not require stopping:
- Nausea (especially during dose escalation)
- Constipation or diarrhea
- Reflux
- Fatigue
- Appetite that’s gotten too low
- Hair shedding (often from the rate of weight loss, not the medication itself)
Things that often help without stopping:
- Slower titration (longer at each dose level)
- Holding the current dose for an additional 4–8 weeks before stepping up
- Anti-nausea medications (e.g., ondansetron) prescribed alongside
- Hydration and fiber for constipation
- Switching from semaglutide to tirzepatide or vice versa (different tolerability profiles)
- Working with your prescriber on appropriate dose adjustments using the prescribed dose strengths — not splitting FDA-approved fixed-dose pens or KwikPens. Lilly specifically states that splitting Zepbound KwikPen doses into smaller doses is not recommended.
When you should NOT just push through
These are clinician-immediately situations, not “tolerate it” situations:
- Severe abdominal pain (possible pancreatitis or gallbladder issue)
- Persistent vomiting and dehydration
- Symptoms of gallbladder disease
- Hypoglycemia if you’re on insulin or a sulfonylurea
- Pregnancy (planned or suspected)
- New or growing thyroid nodule
A note on suicidal thoughts and FDA labeling (updated 2026)
For a period after the early GLP-1 weight-loss launches, FDA monitored a potential signal for suicidal ideation and behavior. In 2026, after a comprehensive review, the FDA requested removal of the suicidal behavior and ideation warning language from Saxenda, Wegovy, and Zepbound labeling, having found no increased risk linked to these medications.
This does not mean mental health concerns should be ignored. New or worsening depression, suicidal thoughts, or significant mood changes during any major weight loss require immediate attention from your clinician or an urgent mental health resource. But the specific FDA warning language has been removed from these GLP-1 weight-loss labels, and pages still listing it as a current FDA warning are out of date.
We don’t have a provider CTA in this section. This is a medical conversation, not a shopping decision.
What if you’re stopping for pregnancy or surgery?
Pregnancy and surgery are separate medical situations, and a generic weight-regain article isn’t your stop instructions. Wegovy’s prescribing information directs patients to stop at least 2 months before planned pregnancy because of its long half-life. The October 2024 multi-society anesthesia guidance updated previous “stop the week before surgery” advice — most patients can now continue GLP-1s before elective surgery, with higher-risk patients requiring individualized steps. Talk to the relevant clinical team, not the internet.
Pregnancy planning
GLP-1 weight-loss medications are not used during pregnancy. Wegovy’s prescribing information specifically advises stopping at least 2 months before planned pregnancy because of semaglutide’s long half-life. For other GLP-1 medications (tirzepatide, liraglutide, oral semaglutide), check the current prescribing information and follow your prescriber’s specific guidance — pregnancy stop timing differs by medication.
If you’re trying to conceive, are pregnant, or suspect you might be, contact your prescriber immediately — both for the medication discussion and for diabetes management if applicable.
Surgery and anesthesia
The October 2024 multi-society anesthesia guidance (American Society of Anesthesiologists, AGA, and others) walked back the previous “stop the week before surgery” advice. Current consensus is more nuanced: most patients can continue GLP-1s before elective surgery with modifications (clear liquid diet for longer, possible delay if delayed gastric emptying is suspected). Higher-risk patients may need specific protocols.
Tell your surgeon and anesthesiologist you’re on a GLP-1. They will give you the protocol that fits your specific procedure. Don’t follow old internet advice. The guidance has changed.
Diabetes-specific caution for either situation
If you started the GLP-1 for type 2 diabetes management, stopping (whether for pregnancy or surgery) requires a glucose-management plan. Don’t navigate this alone. This is a coordinated-care moment.
A quick word on the “gut reset” and other emerging research
A clinical research team announced preliminary results in April 2026 from a procedure called duodenal mucosal resurfacing, suggesting it may help people regain less weight after stopping GLP-1s. The full data are scheduled for presentation at Digestive Disease Week 2026, and the larger pivotal REMAIN-1 trial is ongoing. This is genuinely interesting research, but it remains investigational, not widely available, and not a substitute for a current maintenance plan.
In April 2026, researchers shared early findings showing that duodenal mucosal resurfacing — a minimally invasive procedure that renews the lining of the upper small intestine — appeared to help a subset of participants regain less weight after discontinuing tirzepatide. The pivotal REMAIN-1 trial is fully enrolled, with topline 6-month data expected in late 2026.
What this means for you, today:
- Status: investigational. Not approved. Not widely available.
- It’s being studied as a post-GLP-1 maintenance option, not a replacement.
- If approved in the future, it would expand the toolkit. It doesn’t change your current decision.
If you see breathless headlines about “the procedure that ends GLP-1 dependency,” that’s what they’re referring to. The actual data are early-stage and worth tracking, not pivoting on.
What we actually verified for this page
We pride transparency on YMYL pages. Here’s the audit trail.
Clinical evidence reviewed and cited
- STEP-1 extension (Wilding et al., Diabetes, Obesity and Metabolism 2022)
- STEP-4 trial (Rubino et al., JAMA 2021)
- SURMOUNT-4 trial (Aronne et al., JAMA 2024)
- SURMOUNT-4 post-hoc cardiometabolic analysis (Horn et al., JAMA Internal Medicine November 2025)
- eClinicalMedicine meta-regression (Budini et al., 2026)
- BMJ systematic review/meta-analysis (West et al., BMJ January 2026)
- Cleveland Clinic real-world cohort (March 2026)
- 2026 systematic review and meta-analysis on GLP-1 body composition (International Journal of Obesity)
- Embla 9-week tapering data (Gudbergsen et al., ECO 2024 posters)
- Oxford Academic clinical observation (Obesity and Endocrinology September 2025)
- Paddu et al. 2024 weight maintenance after GLP-1 with cost-effective anti-obesity medications
- Penn Medicine preclinical stop-restart study (2026, animal model)
Regulatory sources reviewed
- FDA prescribing information for Wegovy, Ozempic, Zepbound, Mounjaro
- FDA 2026 communication requesting removal of suicidal behavior/ideation warning from Saxenda, Wegovy, and Zepbound labeling
- FDA February 2026 warning letter to MEDVi
- FDA public communications on unapproved/compounded GLP-1 products
- October 2024 multi-society anesthesia guidance on GLP-1s and surgery
- CDC adult physical activity guidelines
- NIDDK weight management guidance
Provider information verification status (as of April 29, 2026)
- Ro pricing structure (membership $39 first month / $149 ongoing / $74 with annual prepay) and Insurance Coverage Checker URL: verified at ro.co
- Ro government-plan limitation (no Medicare/Medicaid coordination, FEHB excepted): verified at ro.co
- Ro product list (Zepbound, Foundayo): verified at ro.co
- MEDVi pricing ($179 first month / $299 refill, no contract): verified at glp.medvi.org
- MEDVi FDA warning letter: verified at fda.gov
- Eden pricing (Wegovy $1,695, Zepbound $1,399, no membership fee, same price every dose): verified at tryeden.com
- Amazon One Medical GLP-1 Management Program details: verified via Amazon announcement
- Specific real-time provider checkout pricing was NOT verified individually today; confirm at provider site before enrolling.
Affiliate disclosure: The RX Index earns commissions when readers enroll with some providers mentioned on this page. Compensation does not influence which clinical claims we make, which trials we cite, or which medications we describe as FDA-approved versus compounded. We disclose verified regulatory actions involving providers we cover.
This page is not medical advice. It is educational content that synthesizes published evidence. Do not start, stop, or change a prescription medication without your prescriber.
The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers.
Frequently asked questions
Will I regain all the weight back if I stop GLP-1?
Probably not all of it. Trial-modeled estimates suggest most people regain about 60–67% of their lost weight within 12 months of stopping with no maintenance plan. The 2026 eClinicalMedicine meta-regression projected the trajectory plateaus longer-term, with about 25% of the original loss typically retained — though that plateau is a model projection, not directly observed long-term data. Real-world cohorts (Cleveland Clinic 2026) show much smaller average regain because most people don't fully stop — they switch, restart, or maintain habits.
How long after stopping Ozempic does weight come back?
Semaglutide (Ozempic, Wegovy) takes about 5 weeks to clear your system due to its 7-day half-life, with Wegovy 2.4 mg labeling commonly citing 5–7 weeks for full washout. Appetite typically returns within 2–6 weeks of the last dose. Measurable weight regain usually appears between months 2 and 6.
How quickly will I gain weight after stopping Wegovy?
The BMJ January 2026 meta-analysis estimated average regain of about 0.4 kg/month across all weight-management medications, with the semaglutide/tirzepatide subgroup at roughly 0.8 kg/month — about 1.8 lb/month, or roughly 21 lb/year on average. Individual results vary significantly.
Can you take GLP-1 forever?
Some people may need long-term GLP-1 treatment to maintain weight loss and cardiometabolic improvements; others may not. The FDA-approved indications for medications like Wegovy and Zepbound are framed around chronic weight management — meaning the labels assume long-term use. Whether you personally need lifelong treatment is a clinical decision that depends on your starting BMI, comorbidities, and how your body responds.
Is "Ozempic rebound" real?
"Ozempic rebound" is the popular term for weight regain after stopping semaglutide. The medical framing is that appetite-regulation pressure returns when the medication leaves the system. It is not chemical addiction or withdrawal in the traditional sense. It's a treatment effect ending.
Should I taper off GLP-1 or stop cold turkey?
There is no FDA-approved tapering schedule, but tapering with active lifestyle support has the strongest non-medication evidence (Embla 9-week protocol, ECO 2024 posters). Stopping with no replacement plan has the largest documented regain in the trials. Tapering should be a clinician-led conversation, not a DIY decision.
Can you restart GLP-1 after stopping?
Yes, restarting is a real option and is not a sign of failure. Don't restart at your previous high dose without prescriber guidance — re-titration is often necessary to manage side effects. A 2026 preclinical study from Penn Medicine raised the possibility that repeated stop-restart cycles may produce diminished weight loss in animal models; this hasn't been replicated in humans yet, but argues for minimizing unnecessary cycling when possible.
What's the lowest effective maintenance dose of semaglutide?
Clinical observation and the Embla real-world cohort suggest many people maintain weight on doses well below the 2.4 mg weight-loss target — often 0.5–1.0 mg weekly. The Embla cohort's average maximum dose was 0.77 mg. The "right" maintenance dose is the lowest dose that holds your weight stable, determined by working with your prescriber.
Does insurance cover GLP-1 maintenance dose?
Coverage varies dramatically by insurer, by indication (diabetes vs. weight management), and by medication. Many insurers cover GLP-1s for type 2 diabetes; coverage for weight management without diabetes is far more restrictive. Tools like Ro's free GLP-1 Insurance Coverage Checker can flag whether your specific plan covers a specific medication for your indication. Note: Ro doesn't coordinate coverage for Medicare or Medicaid plans (FEHB excepted).
Will I have withdrawal symptoms from stopping Ozempic?
GLP-1 medications are not habit-forming and do not cause classic chemical withdrawal. The physiological changes you'll experience after stopping — increased appetite, food noise returning, faster gastric emptying, possible glucose changes if you have type 2 diabetes — are the loss of treatment effect, not withdrawal. They're real, but they're a different category than what the word "withdrawal" usually describes.
What if I'm stopping because I can't afford it anymore?
Don't stop without exploring options. Insurance prior authorization with provider support, manufacturer cash-pay programs (Wegovy, Zepbound), transparent pharmacy programs like Amazon Pharmacy, compounded telehealth providers, and lower-dose maintenance protocols can all dramatically lower the monthly cost. Stopping a medication that's working when an affordable alternative exists is the most preventable kind of regain.
Does stopping GLP-1 affect my blood sugar if I have diabetes?
Yes. If you started a GLP-1 for type 2 diabetes management, stopping affects your glucose control as much as your weight. A1c will likely rise without replacement diabetes management, and other diabetes medications (insulin, sulfonylureas) may need adjustment. This is a coordinated-care conversation with your prescriber, not a DIY decision.
Is microdosing GLP-1 a real maintenance option?
Some clinicians use very low doses (sometimes called microdosing) for maintenance, but this approach is not validated by large randomized trials for weight maintenance specifically. It exists in clinical practice; it's not standard. Compounded providers offer the most dose flexibility if you and your prescriber want to explore it, but compounded GLP-1 medications are not FDA-approved.
Still not sure which path is right for you?
Your situation depends on factors that need to be weighed together — your cardiometabolic profile, your starting weight, the habits you’ve built, your age, and whether you’re stopping by choice or by force. We built a free 60-second quiz that maps your answers against the evidence on this page and gives you a specific, defensible plan you can bring to your prescriber.
It costs nothing. It doesn’t sign you up for anything. It outputs a personalized next step you can act on this week.
Get your personalized stay/taper/stop plan in under a minute.
→ Take the free 60-second GLP-1 path quizSources
- Wilding JPH, Batterham RL, Davies M, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes Obes Metab. 2022;24(8):1553-1564.
- Rubino D, Abrahamsson N, Davies M, et al. Effect of Continued Weekly Subcutaneous Semaglutide vs Placebo on Weight Loss Maintenance: The STEP 4 Randomized Clinical Trial. JAMA. 2021;325(14):1414-1425.
- Aronne LJ, Sattar N, Horn DB, et al. Continued Treatment With Tirzepatide for Maintenance of Weight Reduction in Adults With Obesity: The SURMOUNT-4 Randomized Clinical Trial. JAMA. 2024;331(1):38-48.
- Horn DB, Linetzky B, Davies MJ, et al. Cardiometabolic Parameter Change by Weight Regain on Tirzepatide Withdrawal in Adults With Obesity: A Post Hoc Analysis of the SURMOUNT-4 Trial. JAMA Intern Med. Published online November 24, 2025.
- Budini A, et al. Long-term weight regain trajectories after GLP-1 receptor agonist discontinuation: a meta-regression of randomized controlled trials. eClinicalMedicine. 2026.
- West S, et al. Weight regain and cardiometabolic effects after stopping weight management medications: systematic review and meta-analysis. BMJ. January 2026.
- Cleveland Clinic. What Happens When Patients Stop Taking GLP-1 Drugs? Real-World Insights from a 7,938-Patient Cohort. Cleveland Clinic Newsroom, March 2026.
- GLP-1 agonists and changes in body mass and composition in adults with overweight or obesity with or without type 2 diabetes: systematic review and meta-analysis. International Journal of Obesity. 2026.
- Gudbergsen H, et al. Slow tapering of semaglutide preserves weight loss: real-world data from 2,246 participants. Posters presented at the European Congress on Obesity (ECO), Venice, Italy, May 2024.
- Semaglutide in a real-world outpatient setting: discontinuation patterns and weight maintenance. Obesity and Endocrinology (Oxford Academic), September 2025.
- Paddu N, et al. Weight maintenance on cost-effective anti-obesity medications after GLP-1RA therapy. 2024.
- University of Pennsylvania Perelman School of Medicine. Stopping and restarting GLP-1s may make it less effective. Penn Medicine, 2026 (preclinical, animal model).
- FDA. FDA Requests Removal of Suicidal Behavior and Ideation Warning from Saxenda, Wegovy, and Zepbound Labeling. Drug Safety Communication, 2026.
- FDA. FDA’s Concerns with Unapproved GLP-1 Drugs Used for Weight Loss. fda.gov.
- FDA. Warning Letter to MEDVi, LLC. February 20, 2026, fda.gov.
- American Society of Anesthesiologists, AGA, et al. Multi-Society Guidance on GLP-1 Receptor Agonists and Elective Surgery. October 2024.
- CDC. Physical Activity Basics for Adults. cdc.gov.
- NIDDK. Eating & Physical Activity to Lose or Maintain Weight. niddk.nih.gov.
- Wegovy Prescribing Information. Novo Nordisk. wegovy.com.
- Ro. GLP-1 Insurance Coverage Checker and Pricing. ro.co.
- Eli Lilly Medical. Zepbound KwikPen Dose-Splitting Guidance. medical.lilly.com.
- Amazon. Amazon One Medical Introduces Weight Management Program. April 21, 2026, aboutamazon.com.
- Digestive Disease Week 2026. Duodenal mucosal resurfacing prevents weight regain after tirzepatide withdrawal: REMAIN-1 multicenter trial midpoint cohort results. Sullivan S, et al. Scheduled May 2026 (preliminary findings reported April 2026).
The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We earn commissions when readers enroll with some providers mentioned. We do not accept payment to alter clinical claims, FDA status, or safety information. To flag an inaccuracy: editorial@therxindex.com.
Last verified: . Next scheduled update: .
Related guides
- GLP-1 Absolute Disqualifiers: The FDA-Labeled Hard Contraindications →
- GLP-1 Cost Without Insurance: What You’ll Actually Pay in 2026 →
- Cheapest GLP-1 Without Insurance: Real Prices in 2026 →
- Best GLP-1 Providers That Accept Insurance →
- Best GLP-1 Providers That Help With Prior Authorization →
- GLP-1 Superbill Guide: When It Works, What Insurers Need →
- Find My GLP-1 Path: 60-Second Match Quiz →