Investigational drug — not FDA approved
Survodutide is investigational and not approved by the FDA for any indication. You cannot legitimately buy it from any pharmacy, telehealth provider, or online source. Do not use anything sold as “survodutide” outside a supervised clinical trial. Talk to a licensed clinician about your treatment options.
Survodutide Weight Loss Drug: Phase 3 Results, FDA Status & What to Do Now
By The RX Index Editorial Team · Last verified: — within hours of the SYNCHRONIZE-1 topline release. Next scheduled review: after the ADA Scientific Sessions, June 5–8, 2026.
Disclosure: Some links on this page are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you.
Survodutide is not available through any affiliate link on this page.The bottom line on the survodutide weight loss drug
The survodutide weight loss drug is an investigational once-weekly injection from Boehringer Ingelheim that hit up to an average of 16.6% weight loss at 76 weeks in its Phase 3 SYNCHRONIZE-1 trial — topline results released April 28, 2026. The molecule is real. The prescription path is not.
Survodutide is not FDA approved. You can't legitimately buy it from any pharmacy, telehealth provider, or “research peptide” site. Boehringer has guided toward a 2027 or 2028 launch contingent on favorable trial data — but no FDA submission, approval date, launch date, or price is confirmed today.
The part most coverage is missing:
The available approved drugs already have higher Phase 3 weight-loss numbers. Tirzepatide (Zepbound) showed about 20.9% in SURMOUNT-1. Wegovy HD (semaglutide 7.2 mg, FDA approved March 19, 2026) showed 20.7% in STEP UP. Both are on pharmacy shelves now.
Quick status — survodutide at a glance
| Question | Direct answer |
|---|---|
| Is the survodutide weight loss drug FDA approved? | No. Investigational only. |
| Can you buy it online today? | No legitimate route exists. |
| Is it a GLP-1? | Partly. It's a glucagon/GLP-1 dual agonist. |
| How much weight loss did Phase 3 show? | Up to 16.6% mean at 76 weeks vs 3.2% on placebo (efficacy estimand). |
| Average pounds lost? | Up to an average of 39.2 lb (17.8 kg). |
| Who makes it? | Boehringer Ingelheim, licensed from Zealand Pharma. |
| Realistic U.S. timeline? | Boehringer has guided toward 2027–2028 launch, contingent on data. |
| What should you do now? | Wait, find a trial, or compare available GLP-1s. |
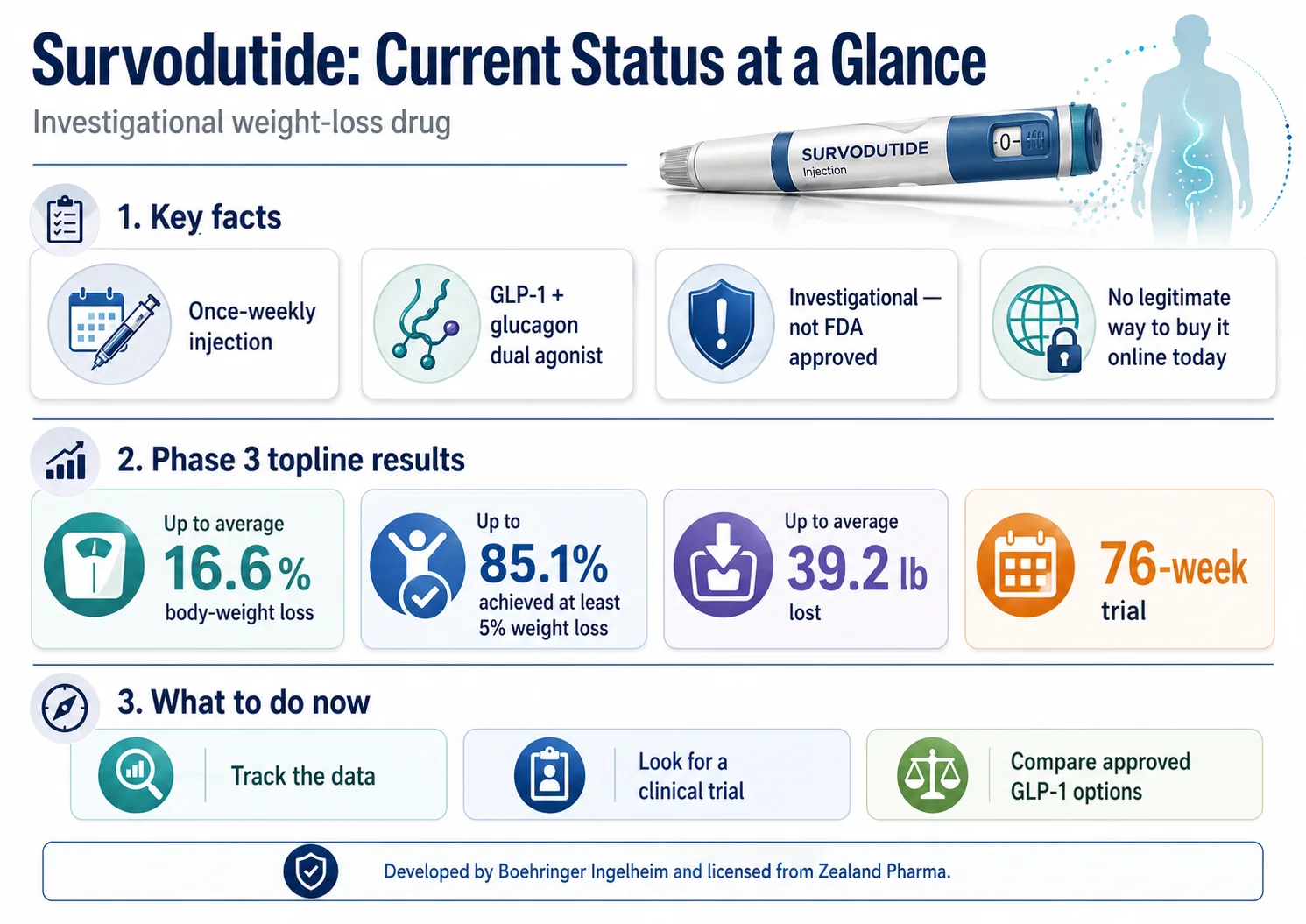
Survodutide current status at a glance — Phase 3 topline results released April 28, 2026.
What we actually verified
We checked the Boehringer Ingelheim and Zealand Pharma April 28, 2026 announcements, Zealand's pipeline page, published Phase 2 obesity and MASH papers, ClinicalTrials.gov entries for SYNCHRONIZE-1 / -2 / -CVOT and LIVERAGE, FDA approval announcements for Wegovy HD (March 2026), Wegovy MASH (August 2025), and Foundayo (April 2026), and current pricing pages for the providers we recommend.
What we couldn't verify yet: The full SYNCHRONIZE-1 safety table and granular responder breakdown (≥10%, ≥15%, ≥20%) won't be public until the ADA 2026 Scientific Sessions in June. Boehringer's NDA timing, FDA review priority for obesity, the eventual brand name, and the launch price are all unknown.
Find your GLP-1 path — 60-second matching quiz →
Take the free matching quiz →What is the survodutide weight loss drug?
If you've taken Wegovy or Ozempic, you know GLP-1 (glucagon-like peptide-1) — that's the receptor that tells your brain you're full and slows your stomach down. Survodutide does that. But it also activates a second receptor: glucagon. Most people associate glucagon with raising blood sugar in an emergency. What's less famous is its role in energy expenditure and liver fat metabolism.
That dual-mechanism design is why this drug exists and why people care. It's also why the “BI 456906” name keeps showing up in older trial papers and on Reddit. Boehringer hasn't picked a brand name yet — that comes around approval.
Who actually makes it?
Survodutide was originally invented at Zealand Pharma in Denmark and licensed to Boehringer Ingelheim, which now runs all global development and commercialization. Zealand keeps co-promotion rights in the Nordic countries and gets royalties from sales if the drug launches. The most authoritative pages are boehringer-ingelheim.com and zealandpharma.com — not the dozens of “buy survodutide online” sites flooding search results. Neither company has partnered with any U.S. pharmacy, telehealth platform, or compounding pharmacy to distribute the drug, because there's nothing to distribute.
What did Phase 3 actually show? (The numbers Boehringer released April 28, 2026)
| SYNCHRONIZE-1 detail | What it means |
|---|---|
| Trial size | 725 adults randomized and treated |
| Population | Obesity or overweight, without type 2 diabetes |
| Duration | 76 weeks |
| Doses | Weekly 3.6 mg or 6.0 mg subcutaneous, or placebo |
| Primary endpoints | (1) % body-weight change, (2) achieving ≥5% body-weight reduction |
| Result on weight loss | Up to 16.6% mean (efficacy estimand) vs 3.2% placebo (p<0.0001) |
| Responder rate (≥5%) | Up to 85.1% vs 38.8% placebo |
| Absolute weight lost | Up to 39.2 lb (17.8 kg) |
| Body composition | Predominantly fat, not muscle, per company commentary |
| Both estimands met endpoints | Yes — efficacy and treatment-regimen |
| Where to find full data | ADA Scientific Sessions, June 5–8, 2026 |
Sources: Boehringer Ingelheim press release, April 28, 2026; Zealand Pharma announcement, April 28, 2026; Reuters and BioPharma Dive coverage, April 28, 2026; The Wall Street Journal coverage of fat-vs-muscle composition, April 28, 2026.
A note on estimands (why the topline number has a footnote)
Boehringer reported results under two statistical frameworks: the efficacy estimand (treatment effect if everyone stayed on therapy as planned) and the treatment-regimen estimand (treatment effect regardless of adherence). The 16.6% headline figure used the efficacy estimand. Both estimands met both co-primary endpoints. The full breakdown will land at ADA in June.
Why Phase 3 (16.6%) is lower than Phase 2 (18.7%)
Survodutide's Phase 2 obesity trial showed up to 18.7% weight loss at 46 weeks. The Phase 3 trial is bigger, longer, and used a gentler titration — different statistical framing, larger and more diverse population, and 30 more weeks of follow-up that include people who hit a plateau. Cross-phase number comparisons aren't apples-to-apples. The 16.6% number is the one Boehringer will show the FDA.
What the topline release didn't tell us
- Responder breakdowns at ≥10%, ≥15%, ≥20%, or ≥25%
- Full safety table — actual nausea/vomiting/diarrhea rates, serious adverse event rates, discontinuation rates by dose
- Subgroup analyses (sex, age, BMI, race, baseline metabolic risk)
- Body composition detail beyond “predominantly fat”
- Cardiovascular signals (those come from SYNCHRONIZE-CVOT, which is years away)
The full SYNCHRONIZE-1 dataset will be presented at the American Diabetes Association Scientific Sessions, June 5–8, 2026. Expect updated coverage from The RX Index within 24 hours of the presentation.
How survodutide stacks up against other GLP-1s
Cross-trial comparisons are not head-to-head trials. Different populations, baselines, statistical methods, and durations make a 16.6% in one trial not directly equivalent to a 20.9% in another. With that caveat front and center:
| Drug | Mechanism | Duration | Mean weight loss | FDA status | Available now? |
|---|---|---|---|---|---|
| Survodutide | GLP-1 + glucagon | 76 wks (Phase 3) | Up to 16.6% | Investigational | No |
| Tirzepatide (Zepbound) | GLP-1 + GIP | 72 wks (SURMOUNT-1) | ~20.9% | FDA approved (Nov 2023) | Yes |
| Wegovy HD (semaglutide 7.2 mg) | GLP-1 only | 72 wks (STEP UP) | 20.7% (efficacy est.) | FDA approved (Mar 19, 2026) | Yes |
| Wegovy 2.4 mg (semaglutide) | GLP-1 only | 68 wks (STEP 1) | 14.9% | FDA approved (Jun 2021) | Yes |
| Wegovy oral pill (semaglutide 25 mg) | GLP-1 only | 64 wks (OASIS 4) | ~13.6% | FDA approved | Yes |
| Foundayo (orforglipron) | Oral GLP-1 | 72 wks (ATTAIN-1) | 12.4% (efficacy est.) | FDA approved (Apr 2026) | Yes |
| Retatrutide | GLP-1 + GIP + glucagon (triple) | 48 wks (Phase 2) | ~24.2% | Investigational | No |
Sources: Boehringer Ingelheim April 28, 2026 release; Le Roux et al., Lancet Diab Endocrinol 2024; Jastreboff et al., NEJM 2022 (SURMOUNT-1); Aronne et al., NEJM 2025 (SURMOUNT-5); Wilding et al., NEJM 2021 (STEP 1); Novo Nordisk March 19, 2026 release (STEP UP / Wegovy HD); FDA Foundayo prescribing information; Jastreboff et al., NEJM 2023 (retatrutide Phase 2).
The damaging admission we're not going to dodge:
Survodutide's Phase 3 number is good. It's not the best number in the class. Two FDA-approved drugs you can fill a script for this week already show higher mean Phase 3 weight loss: Zepbound (~20.9%) and Wegovy HD (20.7%). BioPharma Dive's analyst coverage called survodutide's headline “comparable to Wegovy” but “short of Zepbound.”
If your only metric is pounds on the scale, the available drugs already beat the unavailable one. What survodutide might offer: the glucagon arm for liver-fat reduction (the MASH program, where it already has Breakthrough Therapy designation), increased energy expenditure, and possibly a different response for people who plateau on other GLP-1s. None of that is proven for the average obesity patient yet.
Decision Resolution Point #1
If 16.6% is what brought you here, two FDA-approved drugs with higher Phase 3 numbers are available right now: Zepbound and Wegovy HD. Ro carries both, plus the insurance concierge and free coverage checker.
Ro Body membership: $39 for the first month, then as low as $74/month with annual prepay ($149/month standard ongoing). Medication billed separately. Ro matches LillyDirect / NovoCare pricing. Verified April 28, 2026.
Check eligibility on Ro for Zepbound, Wegovy HD, or Foundayo →Is the survodutide weight loss drug FDA approved?
Investigational means a drug is still being studied and has not been approved for marketing. You can take it inside a clinical trial. You cannot buy it.
Fast Track designation is a status the FDA gives drugs targeting serious conditions with unmet need — it lets the company submit data on a rolling basis. Survodutide has this for MASH.
Breakthrough Therapy designation is one step further — for drugs whose preliminary clinical data suggest substantial improvement over existing therapies. Survodutide got this for MASH in September 2024.
Neither designation is approval. And critically, the obesity NDA is a separate review from the MASH NDA — they don't share a clock.
The realistic survodutide approval timeline
| Milestone | Status / Date | What it means for you |
|---|---|---|
| Phase 1 safety testing | Completed | Sufficient to continue development |
| Phase 2 obesity (Le Roux et al.) | Completed; published Lancet Diab Endocrinol, Feb 2024 | 18.7% weight loss at 46 weeks signal |
| Phase 2 MASH (Sanyal et al.) | Completed; published NEJM, June 2024 | Liver-fat improvement signal |
| FDA Fast Track (MASH) | Granted | Faster review path for MASH only |
| FDA Breakthrough Therapy (MASH) | Granted September 2024 | Even faster review for MASH only — NOT for obesity |
| EMA PRIME scheme (MASH) | November 2023 | Faster European review path |
| SYNCHRONIZE-1 last patient last visit | Q1 2026 | Trial complete |
| SYNCHRONIZE-1 topline results | April 28, 2026 | Up to 16.6% mean weight loss; both primary endpoints met |
| Full SYNCHRONIZE-1 data | ADA Scientific Sessions, June 5–8, 2026 | Detailed responder data, AE rates, subgroup analyses |
| SYNCHRONIZE-2 (obesity + T2D) topline | Expected 2026 [NEEDS VERIFICATION] | Second Phase 3 pillar |
| SYNCHRONIZE-CVOT (cardiovascular) | Ongoing — years out | Required for any CV indication |
| LIVERAGE / LIVERAGE-Cirrhosis (MASH Phase 3) | Ongoing | MASH indication |
| FDA NDA submission (obesity) | [NEEDS VERIFICATION] — not publicly committed | Clock starts on FDA review |
| Boehringer-guided launch window | 2027–2028, contingent on favorable data | Not an FDA-confirmed date |
| Brand name | Not yet announced | Boehringer assigns at approval |
What still has to happen between today and approval
- Phase 3 obesity program completion. SYNCHRONIZE-1 read out April 28, 2026. SYNCHRONIZE-2 (obesity with type 2 diabetes) is the second pillar and hasn't read out yet.
- Full data package. ADA presentation in June, journal publication, and the all-enrollees / sensitivity analyses.
- NDA filing. Boehringer hasn't publicly committed to a date.
- FDA acceptance and review. The FDA has 60 days to accept the NDA, then a standard review of ~10 months for a novel mechanism. Sometimes longer.
- Possible advisory committee. New mechanisms often get a public advisory committee meeting.
- Labeling negotiation, manufacturing inspections, REMS decisions.
- Post-approval launch. Pricing decisions, payer negotiations, distribution buildout.
If everything moves crisply, late 2027. If there's an advisory committee or a CMC follow-up, 2028. Boehringer's own public guidance has stayed inside that window.
Decision Resolution Point #2
If 18 months or more is too long for your situation, you have two honest options: an FDA-approved alternative you can start this month, or a clinical trial enrollment.
Take the 60-second GLP-1 matching quiz →Can you buy the survodutide weight loss drug today?
Why “buy survodutide online today” is a bad idea (in five reasons)
Reason 1: It is not FDA approved.
No legitimate U.S. provider can prescribe it for human use. Anyone offering to is either operating illegally or labeling the product "research only" while everyone in the transaction knows what it's for. The seller may be operating illegally, and using the product creates safety and regulatory risk for you.
Reason 2: It is protected by intellectual property.
Boehringer Ingelheim and Zealand Pharma hold rights to survodutide. U.S. compounding pharmacies are not authorized to produce a generic version of survodutide. Anything sold today as "compounded survodutide" is not a verified FDA-approved or legitimate commercial prescription product for human use.
Reason 3: It is not on the FDA shortage list.
Compounding pharmacies have a narrow legal pathway to compound versions of FDA-approved drugs that are in shortage. Survodutide has never been on that list — because it has never been an approved drug. There is no legitimate compounding lane for it.
Reason 4: There is no purity or sterility standard for human use.
"Research peptide" sellers often advertise high HPLC purity, but for an injectable peptide you also need verified sterility, endotoxin testing, and potency consistency. The FDA's own guidance warns that illegally marketed GLP-1 products may be counterfeit, contain wrong ingredients, harmful ingredients, too little or too much active ingredient, or no active ingredient at all.
Reason 5: There is no medical supervision.
In the Phase 2 trial, roughly 25% of survodutide participants discontinued during dose escalation due to GI side effects — with a clinical team monitoring them weekly. Self-titrating a peptide of unknown purity, with no clinician guidance, is the worst version of the experience.
What if you've already taken something labeled “survodutide”?
Do not take another dose until you contact a licensed clinician. Bring the vial and any documentation you have. Seek urgent care or call Poison Control (1-800-222-1222) immediately if you have severe symptoms — persistent vomiting, severe abdominal pain, signs of dehydration, or any unusual symptoms after injection.
Decision Resolution Point #3
You came here looking for survodutide. You now know it's not safely available. The fastest path forward is identifying which legitimate option matches what you wanted survodutide to do for you.
Find your real GLP-1 path →How do I find a survodutide clinical trial?
Go to ClinicalTrials.gov and search for “survodutide” or “BI 456906”. Filter by Status: “Recruiting” or “Not yet recruiting”; Country: United States; Condition: obesity, MASH, or type 2 diabetes.
- NCT06066528 (SYNCHRONIZE-2) — obesity with type 2 diabetes
- NCT06745284 — head-to-head survodutide vs semaglutide for energy expenditure (Phase 1)
- LIVERAGE / LIVERAGE-Cirrhosis — MASH and MASH cirrhosis (~1,800 participants combined)
- SYNCHRONIZE-CVOT — cardiovascular outcomes in obesity with elevated CV risk
Questions to ask before enrolling
- Will I receive placebo? What's the randomization ratio?
- How long is the trial, and what happens after? (Some trials offer extension access; many don't.)
- What are the visit and lab requirements?
- Are medication, visits, labs, and travel covered?
- What side effects should trigger urgent contact, and who do I call?
- Can I switch to approved treatment if I'm not satisfied?
- What's the trial site's track record?
If you're trial-curious, talk to your primary care clinician or an obesity-medicine specialist before you enroll.
Survodutide vs Wegovy, Wegovy HD, Zepbound, Mounjaro & retatrutide
The cleanest comparison today is by status and mechanism, not headline percentage. There are no head-to-head trials proving survodutide is better than any of them.
Survodutide vs Wegovy (semaglutide 2.4 mg)
Mechanism: Survodutide is GLP-1 + glucagon. Wegovy 2.4 mg is GLP-1 only. Phase 3 weight loss: Survodutide 16.6% vs Wegovy 14.9% (STEP 1) — survodutide edges standard-dose Wegovy. FDA status: Survodutide is investigational. Wegovy 2.4 mg was approved June 2021 for chronic weight management; in August 2025 it also became the first GLP-1 approved for MASH with moderate-to-advanced fibrosis, based on the ESSENCE trial showing 62.9% vs 34.3% placebo MASH resolution at 72 weeks. Cardiovascular evidence: Wegovy has the SELECT trial (significant reduction in major adverse cardiovascular events). Survodutide has SYNCHRONIZE-CVOT in progress.
Survodutide vs Wegovy HD (semaglutide 7.2 mg) — the comparison most coverage is missing
Mechanism: Wegovy HD is the same molecule as standard Wegovy (semaglutide), at a higher 7.2 mg weekly dose. Phase 3 weight loss: Survodutide 16.6% vs Wegovy HD 20.7% (efficacy estimand) / 18.8% (treatment-regimen estimand) in STEP UP. About 31.2% of Wegovy HD participants achieved at least 25% weight loss. FDA status: Wegovy HD was FDA approved March 19, 2026. It's the natural step-up for people already on standard Wegovy who need more.
Survodutide vs Zepbound (tirzepatide)
Mechanism: Both are dual agonists, but on different second receptors. Survodutide hits GLP-1 + glucagon. Zepbound hits GLP-1 + GIP. Phase 3 weight loss: Survodutide 16.6% vs Zepbound ~20.9% (SURMOUNT-1) and 20.2% (SURMOUNT-5 head-to-head vs semaglutide). Zepbound currently shows the highest mean Phase 3 weight loss in an approved drug. Survodutide's edge — if it materializes — will be the liver/MASH application and possibly a different response for tirzepatide plateaus.
Survodutide vs retatrutide
Mechanism: Retatrutide is a triple receptor agonist — GLP-1 + GIP + glucagon. Phase 2 weight loss: Up to 24.2% at 48 weeks. Phase 3 results not yet published. If you want the absolute highest-efficacy investigational drug, retatrutide is the headliner. If you want the next available next-generation drug, survodutide is likely to reach the U.S. market first.
Survodutide vs Foundayo (orforglipron) and oral Wegovy
If injection avoidance is your priority, you have two FDA-approved oral GLP-1 options as of April 2026: Foundayo (orforglipron) — a daily small-molecule oral GLP-1 from Eli Lilly, approved April 1, 2026, with 12.4% mean weight loss in ATTAIN-1, taken any time of day with or without food. Oral Wegovy (semaglutide 25 mg) — a daily oral version of semaglutide with ~13.6% mean weight loss in OASIS 4. Both are lower mean weight loss than the injectables, but for many people the convenience of a daily pill outweighs the gap.
Decision Resolution Point #4
If you want the highest available injectable weight loss right now, Zepbound and Wegovy HD are both approved and on shelves. If you want oral and FDA approved, Foundayo and oral Wegovy are both options. Ro carries Zepbound, Wegovy (including HD when available), and Foundayo, with insurance support.
See current pricing on Ro for Zepbound, Wegovy HD & Foundayo →How does the survodutide weight loss drug actually work?
What GLP-1 does (the part you've heard of)
GLP-1 is a gut hormone. When you eat, your gut releases it. It travels to your brain, signals fullness, and tells your stomach to empty more slowly. Drugs that mimic GLP-1 — semaglutide (Wegovy/Ozempic), tirzepatide (Zepbound/Mounjaro), liraglutide (Saxenda), orforglipron (Foundayo) — all use this pathway. Survodutide is a synthetic 29-amino-acid peptide modified with a fatty acid chain to keep it active for about a week, which is why it's a once-weekly injection.
What the glucagon arm adds (the part that's actually new)
Activating glucagon receptors at sustained, controlled levels has three effects relevant to weight and metabolism:
- Increases energy expenditure. The body uses more calories at rest.
- Mobilizes fat from the liver. Glucagon receptors are concentrated in liver tissue, and activation tells the liver to break down stored fat.
- Reduces hepatic steatosis. This is why survodutide has Breakthrough Therapy designation for MASH.
The catch: glucagon also raises blood sugar. The engineering challenge of survodutide was tuning the GLP-1-to-glucagon ratio so the GLP-1 effect (which lowers blood sugar via insulin) offsets the glucagon effect. Phase 2 and Phase 3 trials suggest the balance works.
Survodutide side effects: what we know and what we're still waiting on
What Phase 2 actually showed
- Nausea (most common, especially during dose escalation)
- Vomiting
- Diarrhea
- Constipation
- Abdominal pain
- Headache, fatigue, dizziness in some participants
The discontinuation rate due to side effects was ~25% in survodutide groups vs ~4% on placebo. The vast majority of those discontinuations happened during the rapid 20-week dose escalation, not during the maintenance phase. Among participants who tolerated escalation, ongoing side effects were typically mild. Serious adverse events were actually slightly lower on survodutide (4.2%) than on placebo (6.5%) in the same trial.
Class-level cautions worth knowing
These are common cautions on approved GLP-1 drug labels. Survodutide's final label is unknown because it isn't approved.
- Pancreatitis — rare but reported with GLP-1s. Persistent severe abdominal pain is a warning sign. See our GLP-1 pancreatitis risk guide.
- Gallbladder issues — rapid weight loss of any kind increases gallstone risk. Reported with multiple GLP-1s.
- Theoretical thyroid C-cell tumor risk — observed in rodent studies for the GLP-1 class. Approved GLP-1s are contraindicated in people with personal or family history of medullary thyroid carcinoma or MEN 2.
- Hypoglycemia — low risk on its own but elevated when combined with insulin or sulfonylureas.
What we don't know yet about survodutide safety
- Long-term cardiovascular outcomes (SYNCHRONIZE-CVOT, years away)
- Long-term cancer risk (no signal yet, but long-term human follow-up doesn't exist)
- Drug-drug interactions in real-world populations
- Pregnancy and lactation safety
What about MASH and fatty liver disease?
The Phase 2 survodutide MASH trial (Sanyal et al., NEJM 2024) randomized 293 adults with biopsy-confirmed MASH and fibrosis stages F1–F3 to weekly survodutide or placebo for 48 weeks. The primary endpoint — improvement in MASH without worsening fibrosis — was met dose-dependently. Phase 3 LIVERAGE and LIVERAGE-Cirrhosis (~1,800 participants combined) are underway.
If MASH/fatty liver is your real concern, here are the legitimate paths today
| If your situation is… | Best next step | Why |
|---|---|---|
| Diagnosed MASH with F2-F3 fibrosis, no cirrhosis | Talk to a hepatologist about Wegovy or Rezdiffra | Both are FDA-approved for this indication |
| MASH with cirrhosis | Talk to a hepatologist; consider LIVERAGE-Cirrhosis enrollment | No FDA-approved drug for MASH cirrhosis yet |
| NAFLD/MASLD (fatty liver) but no biopsy or staging | Get evaluated — imaging, FibroScan, or biopsy as directed | Treatment paths depend on stage |
| You want survodutide because of the liver angle | Track ADA June 2026 + LIVERAGE readouts; talk to a hepatologist about trial eligibility | Survodutide MASH approval is years away |
What can you do today? (The decision matrix)
The right next step depends on what attracted you to survodutide in the first place. Match yourself to the row below.

Match your reason for wanting survodutide to the best approved option available today.
| If what attracted you to survodutide is… | Best available option today | Why this matches |
|---|---|---|
| The high % weight loss (16.6% Phase 3) | Zepbound (tirzepatide) or Wegovy HD (semaglutide 7.2 mg) | Zepbound shows ~20.9% in SURMOUNT-1; Wegovy HD shows 20.7% in STEP UP — both higher than survodutide's Phase 3, both available now |
| Step-up after standard Wegovy plateau | Wegovy HD (semaglutide 7.2 mg) | Approved March 2026 specifically for adults who've tolerated 2.4 mg for ≥4 weeks and need more |
| The dual mechanism (more than just GLP-1) | Tirzepatide (GLP-1 + GIP) | Different second receptor than survodutide's glucagon arm, but the only dual agonist available today |
| The liver / MASH benefit | Wegovy (FDA-approved for MASH F2-F3) or Rezdiffra; consider LIVERAGE trial | Wegovy got FDA MASH approval in Aug 2025. Survodutide's liver advantage is still trial-only. |
| Avoiding injections | Foundayo (orforglipron) or oral Wegovy (semaglutide 25 mg) | Both FDA-approved oral GLP-1s as of early 2026 |
| Insurance / HSA coverage | Wegovy or Zepbound through an insurance concierge | Both have established prior-auth pathways |
| You'd rather wait for survodutide itself | Bookmark this page; track the milestones | Boehringer-guided launch window is 2027–2028, contingent on data |
Three specific provider routes
For FDA-approved branded GLP-1s with insurance support → Ro
Ro is our primary recommendation for FDA-approved, insurance-sensitive, or branded medication. They carry Zepbound (tirzepatide), Foundayo (orforglipron), Wegovy pen, Wegovy pill, and Zepbound KwikPen. They match LillyDirect, NovoCare, and TrumpRx pricing, and include an insurance concierge that handles prior authorization paperwork, plus a free GLP-1 Insurance Coverage Checker.
Pricing: $39 for the first month, then as low as $74/month with annual prepay ($149/month standard ongoing). Membership and medication billed separately. Verified April 28, 2026.
Check eligibility on Ro for Zepbound, Wegovy or Foundayo →For maximum FDA-approved provider choice → Sesame Care
If you want to choose your own clinician across the broadest FDA-approved formulary (Wegovy, Zepbound, Ozempic, Mounjaro, Foundayo, Saxenda), Sesame is the secondary FDA-approved option. Particularly strong if you want Costco-member pricing on Wegovy or Ozempic and the ability to pick your provider yourself.
See Sesame Care GLP-1 options →For flat-rate cash-pay → Eden
If insurance isn't your path and you want predictable cash pricing for FDA-approved or compounded medications, Eden uses a flat-rate model with no membership fees and accepts HSA/FSA at checkout.
Compliance note: Compounded medications are not FDA-approved and are not the same as branded Wegovy, Ozempic, Zepbound, or Mounjaro. They are individually compounded under applicable 503A/503B rules where a documented clinical need exists. Not interchangeable from a regulatory standpoint.
Check Eden's flat-rate GLP-1 pricing →If none of those three feel right, the matching quiz routes you through a few more questions and matches you to the option that fits your specific situation.
Take the 60-second matching quiz →Frequently asked questions about the survodutide weight loss drug
Is survodutide a GLP-1?
Yes, but not only a GLP-1. Survodutide activates both the GLP-1 receptor and the glucagon receptor. That’s why it’s called a dual agonist rather than a standard GLP-1 drug.
Is survodutide the same as Ozempic, Wegovy, or Mounjaro?
No. Ozempic and Wegovy are both semaglutide (a GLP-1-only drug). Mounjaro and Zepbound are both tirzepatide (a GLP-1 + GIP dual agonist). Survodutide is a different molecule — GLP-1 + glucagon — made by Boehringer Ingelheim, licensed from Zealand Pharma.
Is survodutide FDA approved?
No. As of April 28, 2026, survodutide is investigational and not approved for any indication. It has FDA Breakthrough Therapy and Fast Track designations for MASH (a liver disease), but those are not approvals.
Can you buy survodutide online?
No. There is no legitimate consumer purchase route for survodutide today. Websites currently selling it are selling unapproved, non-verified material that should not be used by humans. The FDA’s own consumer guidance warns that unsafe online pharmacies sell drugs that can be unapproved, counterfeit, contaminated, or otherwise unsafe.
When will survodutide be available in the U.S.?
There is no confirmed availability date. Boehringer Ingelheim has guided publicly toward a 2027 or 2028 launch contingent on favorable trial data. The earliest meaningful update is the full SYNCHRONIZE-1 dataset at the ADA Scientific Sessions, June 5–8, 2026.
How much weight do you lose on survodutide?
In the SYNCHRONIZE-1 Phase 3 trial released April 28, 2026, adults with obesity or overweight without type 2 diabetes lost up to an average of 16.6% of body weight at 76 weeks (up to 39.2 lb / 17.8 kg average) under the efficacy estimand. Up to 85.1% of survodutide participants achieved at least 5% body weight reduction.
Is survodutide better than Wegovy?
Survodutide’s Phase 3 number (16.6%) edges standard Wegovy 2.4 mg’s STEP 1 number (14.9%). But it’s lower than the new Wegovy HD 7.2 mg (20.7% in STEP UP, FDA approved March 19, 2026). There are no head-to-head trials. Wegovy is approved and on shelves; survodutide is not.
Is survodutide better than Wegovy HD?
On the topline weight-loss numbers in their respective Phase 3 trials, Wegovy HD (20.7% efficacy estimand in STEP UP) edges survodutide (16.6% in SYNCHRONIZE-1). No head-to-head trial exists.
Is survodutide better than Zepbound?
On topline weight loss, no — Zepbound (tirzepatide) showed about 20.9% in SURMOUNT-1, which is higher than survodutide’s 16.6%. Survodutide may have advantages on liver fat and energy expenditure due to the glucagon arm, but those are not yet proven head-to-head, and Zepbound is on pharmacy shelves now.
Is survodutide a pill or an injection?
Injection — once weekly, subcutaneous (under the skin). Survodutide itself is not an oral formulation.
What is BI 456906?
BI 456906 is the development code for survodutide. The two names refer to the same molecule. You’ll see “BI 456906” in older clinical trial papers and on ClinicalTrials.gov; “survodutide” is the generic name Boehringer is now using publicly.
Who makes survodutide?
Boehringer Ingelheim, the German pharmaceutical company. The molecule was originally invented at Zealand Pharma in Denmark and licensed to Boehringer, which now runs all global development and commercialization.
Does survodutide help fatty liver disease (MASH)?
Phase 2 data showed liver-fat reduction and a MASH-resolution signal. Survodutide has FDA Breakthrough Therapy designation specifically for non-cirrhotic MASH with moderate-to-advanced fibrosis. Phase 3 LIVERAGE and LIVERAGE-Cirrhosis trials are ongoing. As of August 2025, Wegovy became the first FDA-approved GLP-1 for MASH with moderate-to-advanced fibrosis.
What’s the difference between survodutide and retatrutide?
Survodutide is a dual agonist (GLP-1 + glucagon). Retatrutide is a triple agonist (GLP-1 + GIP + glucagon). Both are investigational. Retatrutide showed higher Phase 2 weight loss (~24.2%) but is on a slightly later development timeline.
How much could survodutide cost if approved?
No price is verified because survodutide is not approved or commercially available. Approved GLP-1 list prices range around $1,000–$1,300/month before discounts. Survodutide’s eventual price will depend on Boehringer’s commercial strategy, payer negotiations, and the competitive landscape at launch. Do not trust any page that quotes a price for survodutide today.
Can my doctor prescribe survodutide off-label?
No. Off-label prescribing requires the drug to first be FDA approved for some indication. Survodutide is not yet approved for any indication, so off-label prescribing isn’t possible.
Should I wait for survodutide or start another GLP-1 now?
If you need treatment now and qualify for an FDA-approved option, talk to a clinician about starting one. Wegovy HD, Zepbound, and Foundayo all became available in 2026 and offer paths your insurance may already cover. If you’re tracking pipeline drugs out of curiosity or have a specific reason to prefer survodutide’s mechanism, bookmark this page and check back after the ADA presentation in June 2026.
Related guides on The RX Index
How we built this page (and how to spot-check it)
Source hierarchy we used
- FDA and regulatory sources — drugs.fda.gov, FDA press announcements, FDA consumer guidance, ClinicalTrials.gov
- Company primary announcements — Boehringer Ingelheim and Zealand Pharma April 28, 2026 press releases; Novo Nordisk Wegovy HD and MASH announcements; Eli Lilly Foundayo announcement
- Peer-reviewed clinical literature — Lancet Diabetes Endocrinology (Le Roux et al., 2024 Phase 2 obesity); NEJM (Sanyal et al., 2024 Phase 2 MASH; Jastreboff et al., 2022 SURMOUNT-1; Aronne et al., 2025 SURMOUNT-5; Wilding et al., 2021 STEP 1; Jastreboff et al., 2023 retatrutide Phase 2); STEP UP and ESSENCE trial data
- Reputable medical and trade media — Reuters, The Wall Street Journal, BioPharma Dive, BioSpace
- Provider pricing pages — Ro, Sesame, Eden — verified
What we couldn't verify yet
- Full SYNCHRONIZE-1 safety and granular responder data (coming at ADA June 2026)
- Boehringer's exact NDA submission date for obesity
- Whether Boehringer will request priority review for the obesity submission
- SYNCHRONIZE-2 readout timing
- Future commercial pricing or insurance coverage
- The eventual brand name
Correction policy: If you find a factual error or a source that supersedes one we cited, write to us — we update pages and preserve the “Last verified” timestamp. We re-verify the timeline monthly and immediately on any Boehringer Ingelheim or FDA announcement.
So — wait, switch, or start?
If you want survodutide specifically — for the dual mechanism, the liver angle, or because the headline got your attention — bookmark this page. We update it. The next big inflection is the ADA Scientific Sessions in June 2026, when full SYNCHRONIZE-1 data drops. If you have specific MASH concerns, ask your hepatologist about the LIVERAGE Phase 3 trial.
If you want survodutide-level results you can actually start this month — Zepbound (tirzepatide, ~20.9% in SURMOUNT-1) and Wegovy HD (semaglutide 7.2 mg, 20.7% in STEP UP) are both FDA-approved and available now, with higher Phase 3 weight loss than survodutide. Through Ro, you can get started for $39 the first month membership (medication billed separately). If you want the broadest FDA-approved provider choice, Sesame Care has Wegovy, Zepbound, Ozempic, Mounjaro, Foundayo, and Saxenda.
If you came here to buy “survodutide” online — please don't. Whatever's in the vial is not a verified FDA-approved commercial product, and the legal and safety risks are stacked against you.
If you're not sure which path is yours — the quiz takes about 60 seconds and routes you based on your goal weight, insurance status, state, tolerance for waiting, and preference between injection and oral.
Still not sure which GLP-1 program is right for you?
Take our free 60-second matching quiz — it routes Medicare, Medicaid, commercial, and uninsured scenarios to the right next step based on your situation.
Take the free 60-second GLP-1 matching quiz →