Affiliate disclosure: The RX Index earns a commission when you sign up with some of the providers mentioned on this page. It does not affect what you pay, and it never determines our rankings or which providers we cover. Read the full disclosure.
Best Tirzepatide for Diabetes (2026): Mounjaro, Cost & How to Get It
Published: · Last reviewed:
By The RX Index Editorial Team
The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We do not provide medical advice. Talk to a licensed clinician before starting or changing any diabetes medication. The RX Index may earn a commission from some of the providers linked on this page. That does not change our rankings, our regulatory accuracy standards, or our willingness to recommend a non-affiliate path when it fits better. Read our full affiliate disclosure.
This content is educational and is not medical advice. Talk to a licensed clinician before starting any diabetes medication.
Bottom line up front
The best tirzepatide for diabetes is Mounjaro. It's the only FDA-approved tirzepatide for type 2 diabetes -- approved May 13, 2022 for adults, and now for kids 10 and older. In the SURPASS-2 head-to-head trial, Mounjaro lowered A1C by 2.01% to 2.30% across the 5, 10, and 15 mg doses, beating semaglutide (Ozempic) 1 mg at 1.86%. Zepbound is the same active drug, but it's labeled for weight management and obstructive sleep apnea -- not diabetes -- so the name on the prescription matters for your coverage. Mounjaro's current list price is $1,112.16 for a 28-day supply. With commercial insurance covering Mounjaro for diabetes plus the Lilly Mounjaro Savings Card, eligible patients pay as low as $25 per month. Compounded tirzepatide is no longer the workaround it was -- the FDA ended shortage-based enforcement discretion in 2025, and on April 30, 2026 proposed excluding tirzepatide from the 503B Bulks List.
If you have a type 2 diabetes diagnosis and you want a real path to Mounjaro today, the fastest legitimate route we've verified is Sesame's Everyday Rx program. A licensed provider does a video visit, prescribes Mounjaro if it's appropriate, and helps with your prior authorization.
What we actually verified for this page
✓ Mounjaro's FDA label and pediatric indication (current label revised April 2026)
✓ SURPASS-1 through SURPASS-5 A1C and weight results, plus the published SURPASS-CVOT cardiovascular outcomes trial (December 2025)
✓ March 2026 Cleveland Clinic / JAMA Cardiology analysis on heart and kidney outcomes
✓ Mounjaro current list price: $1,112.16 per 28-day supply (pricinginfo.lilly.com)
✓ Mounjaro Savings Card terms -- including the $25 floor, the $1,950 annual cap (when covered), and the December 31, 2026 program expiration
✓ Medicare Part D and Medicaid patient-cost data reported by Lilly
✓ FDA ending 503A and 503B enforcement discretion for tirzepatide (February 18 and March 19, 2025) and the April 30, 2026 FDA proposal to exclude tirzepatide from the 503B Bulks List
✓ Sesame Care's Everyday Rx program prescribing Mounjaro for type 2 diabetes
✓ Ro's statement that it does not currently prescribe Mounjaro
Quick decision table -- find yourself, then keep reading
| If this is you | Ask your doctor about | Your best next step |
|---|---|---|
| Type 2 diabetes diagnosis + want tirzepatide | Mounjaro | Check clinical fit and start the prior authorization |
| No diabetes diagnosis + weight-loss goal | Zepbound | Use the weight-loss GLP-1 path, not this page |
| Commercial insurance + unknown coverage | Mounjaro coverage check | Run Ro's free coverage checker (sponsored affiliate link, opens in a new tab) before booking |
| Complex diabetes -- insulin, kidney/heart issues, or prior denial | Mounjaro through full-chart care | Start with your PCP or endocrinologist |
Don't know your coverage yet? Check it free in two minutes (sponsored affiliate link, opens in a new tab) before you book any visit.
Who this page is for (and who it isn't)
This page is for you if:
- You have a type 2 diabetes diagnosis (or your doctor just told you your A1C is in diabetes range) and you want the real answer on which tirzepatide is for diabetes, what it costs, and how to actually get it.
- You've heard Mounjaro, Zepbound, and tirzepatide used like they mean the same thing, and you want to know which one your prescription should say.
- You're worried about insurance denial, prior authorization, or paying $1,000+ a month out of pocket -- and you want a plan before you book the visit.
This page is NOT the right starting point if:
- Type 1 diabetes. Mounjaro isn't approved for type 1, and your care should run through your endocrinologist.
- Weight loss without a diabetes diagnosis. Zepbound is the tirzepatide brand labeled for that -- see our tirzepatide providers guide or take the quiz.
- Complex diabetes -- you're on multiple insulins, have kidney or heart complications, or have already been denied. A local endocrinologist may be a better first stop.
What is the best tirzepatide for diabetes?
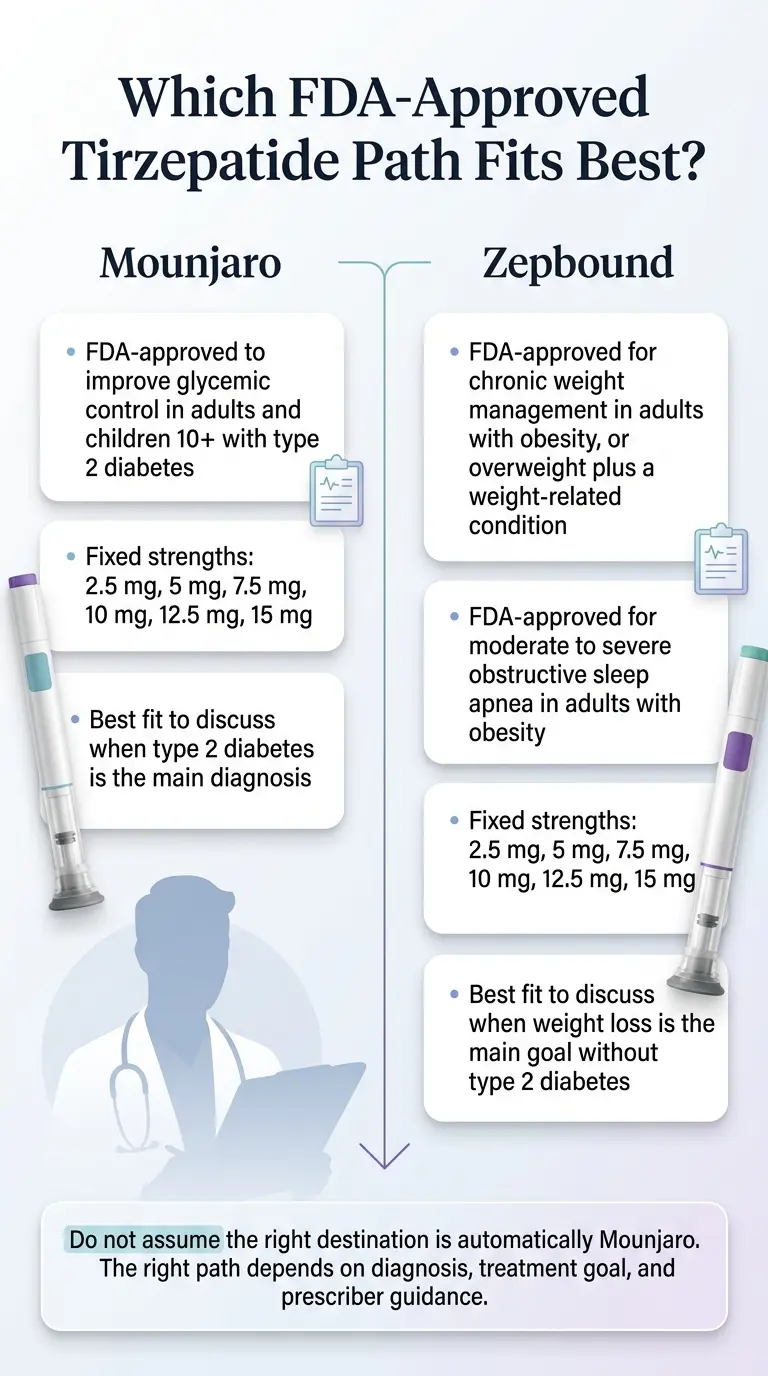
The best tirzepatide for diabetes is Mounjaro, because it's the only FDA-approved tirzepatide brand indicated to improve blood sugar control in adults -- and pediatric patients 10 and older -- with type 2 diabetes. Mounjaro carries the diabetes label. Zepbound is the same active drug but is labeled for weight management and obstructive sleep apnea. Compounded tirzepatide is not FDA-approved for any use, and as of March 2025, it lost the legal shelter that let pharmacies make it in volume.
“Tirzepatide” is the active drug. It's a dual GIP and GLP-1 receptor agonist -- meaning it acts on two of your body's gut hormones at once. Those two hormones do most of the work of telling your pancreas when to release insulin, slowing how fast your stomach empties, and quieting hunger signals in your brain.
Eli Lilly makes two FDA-approved brand names of tirzepatide: Mounjaro (approved for type 2 diabetes, May 13, 2022) and Zepbound (approved for chronic weight management and obstructive sleep apnea). Same active drug. Same core adult dose strengths. Different labels. Different insurance treatment.
For someone with type 2 diabetes, asking for Mounjaro by name is the cleanest path. Insurance plans usually have Mounjaro on their formulary for diabetes. Your doctor can document the diabetes diagnosis on the prior authorization form. The Lilly Mounjaro Savings Card only works when the prescription is for an FDA-approved use -- which, for tirzepatide and diabetes, means Mounjaro.
Best overall: Mounjaro
For most adults with type 2 diabetes who are candidates for a GLP-1, Mounjaro is the first-line tirzepatide answer. The clinical case is strong. The coverage case is strong. The supply is stable. And Lilly's manufacturer savings card can take the sticker shock out of the picture for most commercially insured patients.
Best online path: Sesame Everyday Rx
Among telehealth platforms we've checked, Sesame's Everyday Rx program (sponsored affiliate link, opens in a new tab) is the cleanest match for a diabetes-labeled Mounjaro prescription. It's specifically built for managing chronic conditions like type 2 diabetes -- not just weight loss. The program starts at $59/month on the annual plan, up to $99/month month-to-month. That fee covers the visit, ongoing provider messaging, and lab support. The medication is billed separately through your pharmacy and insurance.
Best free tool: Ro's GLP-1 Insurance Coverage Checker
Ro is one of the largest GLP-1 telehealth brands. On most pages on The RX Index, we recommend Ro for FDA-approved GLP-1s. But for this search -- best tirzepatide for diabetes -- Ro is not our primary recommendation, because Ro's own page says it does not currently prescribe Mounjaro (sponsored affiliate link, opens in a new tab). Ro prescribes Zepbound (the weight-loss tirzepatide) through its Body program.
Where Ro still earns its place on this page: their free GLP-1 Insurance Coverage Checker (sponsored affiliate link, opens in a new tab). Ro's team contacts your insurance company on your behalf and emails you a personalized report showing your Mounjaro coverage, your estimated copay, prior-authorization status, and supply availability. Free. No prescription required.
Best for complex diabetes: a local endocrinologist or your existing PCP
If you're using insulin, have diabetic kidney disease or heart failure, have been denied for a GLP-1 before, or have had pancreatitis or other significant GI issues, an online visit isn't the right starting point. A local endocrinologist or PCP can manage all of that in one place, with your full chart, and order labs in person. We'd rather you get the right care than the fastest one.
Ready to take the next real step?
If you have a type 2 diabetes diagnosis and you want a same-day video visit with a licensed provider who can prescribe Mounjaro and help with your prior authorization, Sesame Everyday Rx is the path we'd point you to first.
Subscription starts at $59/month on the annual plan. The Mounjaro itself is billed separately through your pharmacy and insurance. Prescription is not guaranteed -- your provider decides what's clinically right for you.
Check Mounjaro eligibility with Sesame Everyday Rx → (sponsored affiliate link, opens in a new tab)Mounjaro vs. Zepbound for diabetes -- which name should you ask for?
Mounjaro and Zepbound contain the same active drug (tirzepatide) at the same core adult dose strengths. The difference is the label. Mounjaro is FDA-approved for type 2 diabetes. Zepbound is FDA-approved for chronic weight management and obstructive sleep apnea. For a diabetes patient, asking for Mounjaro by name is almost always the right paperwork -- insurance plans cover Mounjaro for type 2 diabetes far more reliably than they cover Zepbound, and the Mounjaro Savings Card only works with an FDA-approved-use prescription.
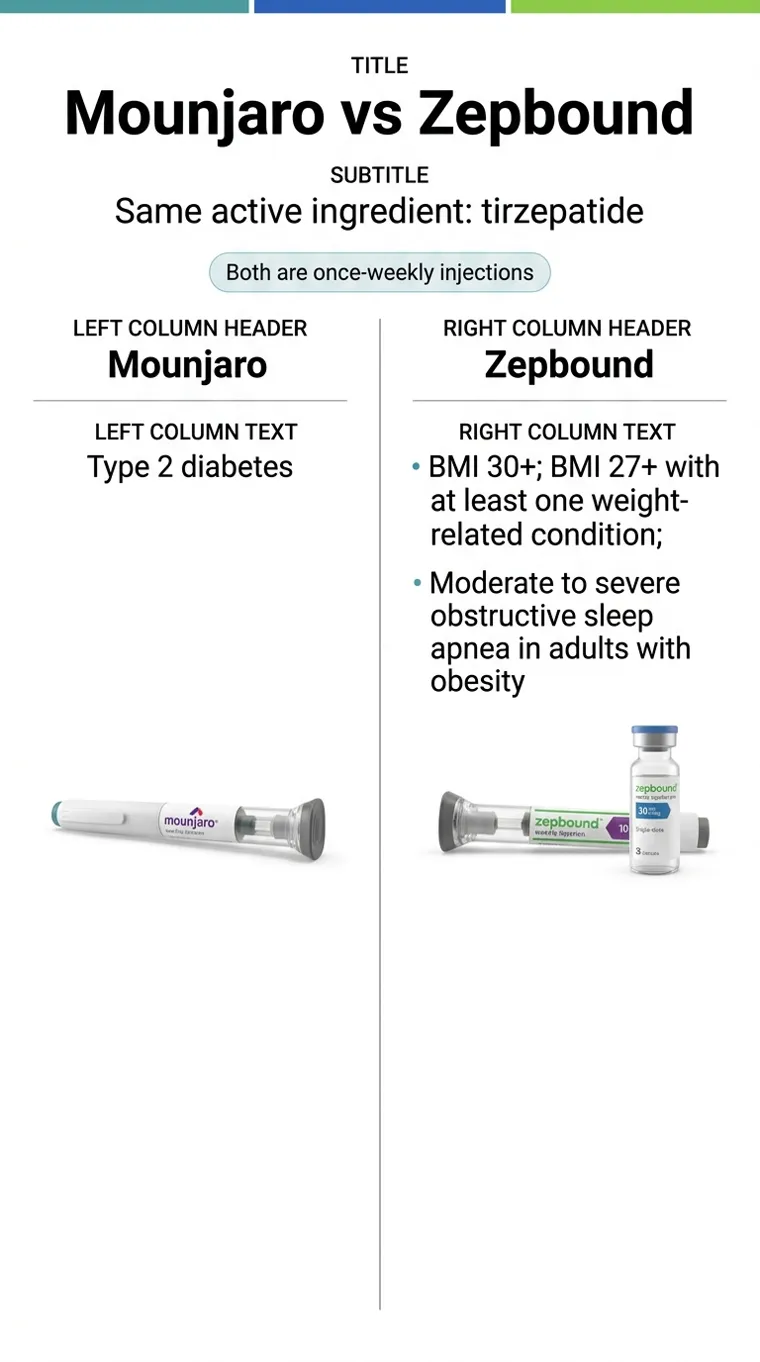
Mechanically, Mounjaro and Zepbound are the same molecule. Both are tirzepatide. The strengths overlap. Eli Lilly makes them both. If you held a Mounjaro pen and a Zepbound pen with the labels removed, you couldn't tell them apart. But the label is the whole game.
| Mounjaro | Zepbound | |
|---|---|---|
| FDA-approved for type 2 diabetes? | Yes -- adults and kids 10+ | No |
| FDA-approved for chronic weight management? | No | Yes (BMI ≥30, or ≥27 with a weight-related condition) |
| FDA-approved for obstructive sleep apnea? | No | Yes |
| Current list price (28-day supply) | $1,112.16 | Varies; vial cash-pay path via LillyDirect is lower |
| Manufacturer savings card | Mounjaro Savings Card -- for FDA-approved use (diabetes) | Zepbound Savings Card -- for FDA-approved use (weight loss / OSA) |
| Cash-pay route via LillyDirect | Limited | Self-pay vials starting at $299/month for 2.5 mg dose |
| Insurance coverage for your diagnosis | Usually covered for T2D, often with prior authorization | Often excluded for weight loss; OSA coverage is improving |
The reason this matters more than it should: most insurance plans treat diabetes coverage and obesity coverage as completely different rules. Diabetes coverage is broad and old. Obesity coverage is narrower and newer, and many plans still exclude weight-loss medication entirely.
So if you have type 2 diabetes and you've been told “ask about tirzepatide” -- ask for Mounjaro. That single word change is the difference between a clean prior authorization and a denial loop.
One honest admission, because we'd rather earn your trust than your click
Ro is one of the strongest GLP-1 telehealth brands overall, and on most pages on The RX Index we'd recommend Ro for FDA-approved GLP-1s. But for this search -- best tirzepatide for diabetes -- Ro is not our primary recommendation, because Ro's own page says it does not currently prescribe Mounjaro (sponsored affiliate link, opens in a new tab). Ro prescribes Zepbound (the weight-loss tirzepatide) through its Body program. That's a great fit for someone whose goal is weight loss. It's not the right fit for someone with a diabetes diagnosis who needs the diabetes-labeled prescription for coverage. That's why we're sending diabetes searchers to Sesame Everyday Rx instead.
Mounjaro vs. Ozempic for diabetes -- the SURPASS-2 head-to-head
In the 40-week SURPASS-2 trial of 1,879 adults with type 2 diabetes inadequately controlled on metformin, Mounjaro 5 mg, 10 mg, and 15 mg lowered A1C by 2.01%, 2.24%, and 2.30% respectively, compared with 1.86% for semaglutide (Ozempic) 1 mg. Weight loss was 7.6 kg, 9.3 kg, and 11.2 kg on Mounjaro vs. 5.7 kg on Ozempic. Ozempic currently carries an FDA-approved indication for reducing cardiovascular risk in type 2 diabetes with established heart disease. Mounjaro does not yet have that specific FDA-approved cardiovascular indication.
The SURPASS trials (SURPASS-1 through SURPASS-5) are the clinical backbone of Mounjaro's approval. Across 7,769 adults with type 2 diabetes -- used as monotherapy, added to metformin, added to sulfonylureas or SGLT2 inhibitors, and added to basal insulin -- Mounjaro lowered A1C by roughly 1.87% to 2.59% depending on the trial design and the dose.
The numbers people actually want to see
| Treatment | A1C reduction at 40 weeks | Weight loss at 40 weeks |
|---|---|---|
| Ozempic 1 mg | −1.86% | −5.7 kg (~12.6 lb) |
| Mounjaro 5 mg | −2.01% | −7.6 kg (~16.8 lb) |
| Mounjaro 10 mg | −2.24% | −9.3 kg (~20.5 lb) |
| Mounjaro 15 mg | −2.30% | −11.2 kg (~24.7 lb) |
Baseline A1C in SURPASS-2 was about 8.28%. Mean diabetes duration was 8.6 years. Source: SURPASS-2 publication, NEJM 2021.
Important caveat: Ozempic is now approved at a higher 2 mg dose that wasn't tested in SURPASS-2, so some of the gap narrows in newer real-world studies. The honest summary is that tirzepatide hits A1C and weight harder than semaglutide on average, but the margin is smaller at semaglutide's newer doses, and the choice depends on more than just A1C.
What about the heart and kidneys?
In March 2026, Cleveland Clinic researchers published an analysis in JAMA Cardiology comparing tirzepatide to dulaglutide (Trulicity) in adults with type 2 diabetes and cardiovascular disease. The composite outcome -- heart attack, stroke, coronary procedures, heart failure, kidney failure, and death from any cause -- happened in 23.7% of patients on tirzepatide vs. 27.4% on dulaglutide. That's a 16% lower risk on tirzepatide, and it was statistically significant.
In December 2025, the full SURPASS-CVOT trial was published. It met the primary noninferiority endpoint against dulaglutide -- meaning tirzepatide wasn't worse on major cardiovascular events -- but it did not demonstrate superiority. Important context: Mounjaro's current FDA label still does not include a cardiovascular risk-reduction indication. Ozempic does. So does Trulicity. If FDA-approved CV risk reduction is your priority today, those products still have the cleaner label path.
On kidneys: real-world data suggests tirzepatide has lower rates of acute kidney injury than older GLP-1s, with stable estimated GFR and reduced albuminuria. The main kidney risk in practice is dehydration from GI side effects -- drink water, especially during dose increases.
So which is “best”?
- Priority: A1C reduction and weight loss together -- Mounjaro wins on average.
- Priority: established FDA-approved cardiovascular risk reduction with known heart disease -- Ozempic or Trulicity are the more conservative choices today.
- Priority: FDA-approved kidney protection with chronic kidney disease -- Ozempic carries that specific indication.
- Can't tolerate injections -- Rybelsus (oral semaglutide) is an option for type 2 diabetes.
Talk to your clinician about which of those priorities matters most in your case. Also see our full GLP-1 for diabetes comparison.
Is compounded tirzepatide a safe path for diabetes?
For 2026, the realistic answer is no -- compounded tirzepatide is not the right path for routine type 2 diabetes management. The FDA ended enforcement discretion for 503A state-licensed pharmacies on February 18, 2025 and for 503B outsourcing facilities on March 19, 2025. On April 30, 2026, the FDA proposed excluding tirzepatide from the 503B Bulks List after finding no clinical need. Public comment runs through June 29, 2026. Eli Lilly's own testing of compounded tirzepatide samples found roughly half had potency below the labeled amount, plus bacterial contamination, high endotoxins, and chemical impurities in some samples.
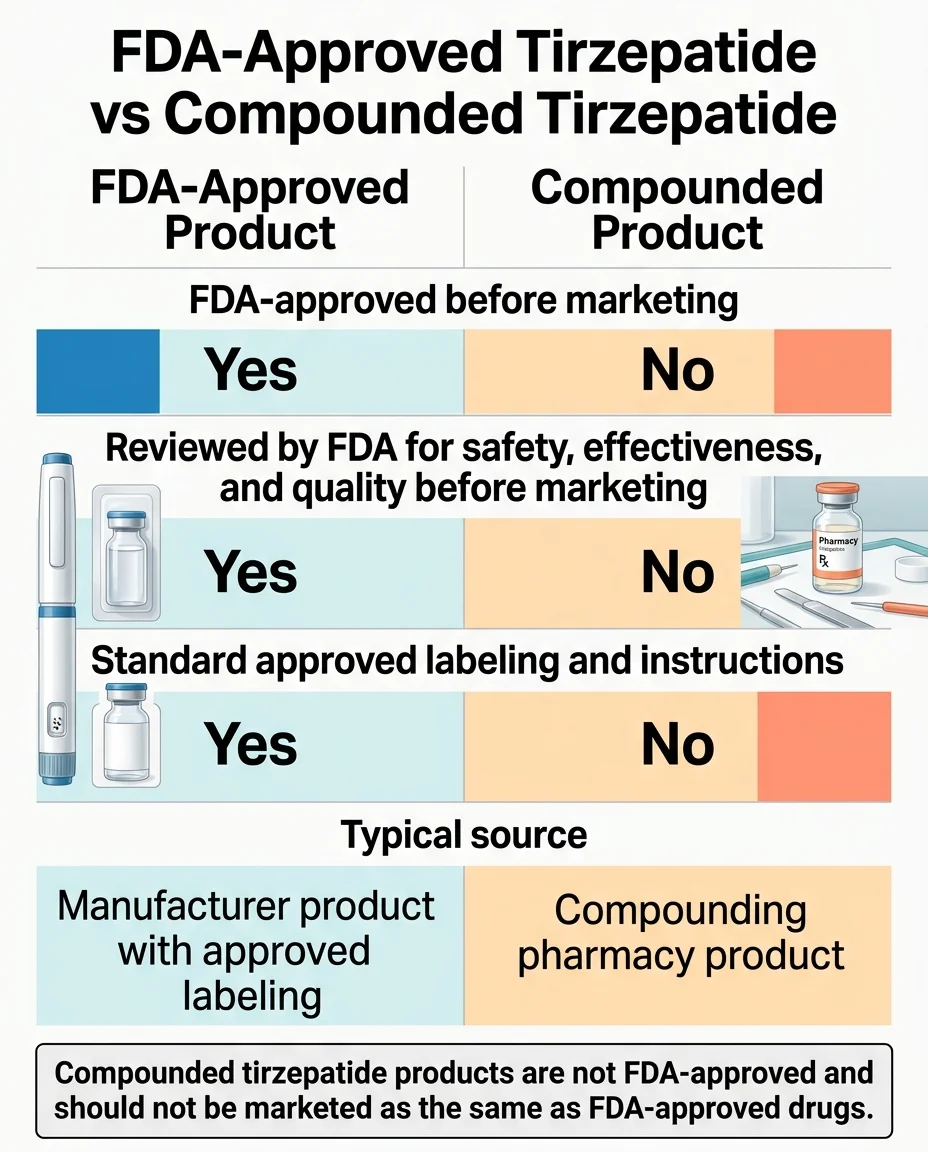
The compounded tirzepatide regulatory timeline
| Date | What happened |
|---|---|
| Oct 2, 2024 | FDA initially determined the tirzepatide shortage was resolved |
| Dec 19, 2024 | After re-evaluation, FDA confirmed the tirzepatide injection shortage was resolved |
| Feb 18, 2025 | FDA's enforcement-discretion window for 503A pharmacies compounding tirzepatide closed |
| Mar 5, 2025 | Federal court (N.D. Texas) denied the compounders' preliminary injunction in Outsourcing Facilities Association v. FDA |
| Mar 19, 2025 | FDA's enforcement-discretion window for 503B outsourcing facilities closed |
| May 7, 2025 | District court upheld the FDA's shortage determination; case on appeal to the Fifth Circuit |
| Feb 2026 | FDA published: “FDA Intends to Take Action Against Non-FDA-Approved GLP-1 Drugs” |
| Mar 12, 2026 | Eli Lilly issued an open letter flagging a novel impurity in tirzepatide samples compounded with vitamin B12, with unknown short- and long-term effects in humans |
| Apr 30, 2026 | FDA proposed excluding tirzepatide (and semaglutide and liraglutide) from the 503B Bulks List -- public comment until June 29, 2026 |
After all of that, a narrow legal pathway still exists: a 503A pharmacy can compound tirzepatide for a specific patient with a documented clinical need that an FDA-approved product cannot meet -- for example, a verified allergy to an inactive ingredient in Mounjaro. Cost or convenience alone is not a valid Section 503A justification.
Why this matters specifically for diabetes: If a compounded batch is sub-potent, you may not know what dose you actually received. For someone managing diabetes, that uncertainty can show up as uncontrolled blood sugar before it shows up on labs. Consistent, independently tested dosing is the whole point of FDA approval.
If your reason for considering compounded was cost, we hear you -- and we want to be direct: the cost section below maps out every legitimate cheaper route to Mounjaro. There are real options. The compounded shortcut isn't one of them for diabetes in 2026.
What Mounjaro actually costs in 2026 (by your coverage situation)
Mounjaro's current list price is $1,112.16 for a 28-day supply before insurance, savings, pharmacy terms, taxes, or fees. Almost no one pays the full list price. With commercial insurance that covers Mounjaro for type 2 diabetes plus the Lilly Mounjaro Savings Card, eligible patients pay as low as $25 per month. If your commercial plan does not cover Mounjaro, the Savings Card may bring a 1-month fill as low as $499. Medicare and Medicaid patients can't legally use the Mounjaro Savings Card, but Lilly reports that about 8 out of 10 Medicare Part D patients pay $0–$50 per 28-day supply.
The Mounjaro Cost-By-Coverage Decision Tree (2026)
| Your situation | What you'll likely pay per month | Why |
|---|---|---|
| Commercial insurance that covers Mounjaro for type 2 diabetes | As low as $25 with the Lilly Savings Card | Card covers up to $150/month savings ($450 per 3-month fill), capped at $1,950 per calendar year, 13 fills max |
| Commercial insurance that does NOT cover Mounjaro | As low as $499 for a 1-month fill | Savings capped at $647 per month and $8,411 per calendar year under current Lilly terms |
| Medicare Part D with a type 2 diabetes diagnosis | ~8 out of 10 patients pay $0–$50 | Based on Lilly's Oct. 2024--Sept. 2025 IQVIA patient-cost data. Federal rules block use of manufacturer savings cards. |
| Medicare Advantage with drug coverage | Same as Part D -- plan-dependent | Same Medicare rules apply |
| Medicaid | ~$5–$11 average when covered | State-dependent. Some states have step-therapy rules requiring metformin or another GLP-1 first. |
| TRICARE / VA | Often a low copay when approved | Prior authorization with A1C documentation is the usual path |
| Uninsured | ~$1,000–$1,300 retail | Savings Card cannot be used. Mounjaro is not currently on Lilly Cares' available medications list. |
Sources: pricinginfo.lilly.com (list price); Mounjaro Savings Card terms (program through Dec 31, 2026); Mounjaro FAQ patient-cost data.
How the Mounjaro Savings Card actually works
Card requirements:
- You must have commercial (private) prescription drug insurance.
- Your prescription must be for an FDA-approved use (type 2 diabetes).
- You must enroll at mounjaro.lilly.com.
- The card is not usable if you have Medicare, Medicaid, Medigap, TRICARE, VA, DoD, or any state pharmacy assistance program.
- The program currently runs through December 31, 2026.
If your commercial plan covers Mounjaro for diabetes: You can pay as little as $25 for a 1-month fill, or as little as $25 for a 3-month fill. Maximum savings is $150 per 1-month fill, $300 per 2-month fill, or $450 per 3-month fill. Annual maximum: $1,950 per calendar year, up to 13 fills.
If your commercial plan does NOT cover Mounjaro: The card may still bring a 1-month fill as low as $499, with maximum monthly savings of $647 and a separate annual maximum of $8,411.
If you have Medicare or Medicaid: No savings card, by federal rule. Many Part D plans cover Mounjaro for diabetes. Ask about the Medicare Prescription Payment Plan (M3P), which lets you smooth your prescription costs across the year in fixed monthly payments.
What to do if you're uninsured
- Compare retail cash prices. Pharmacy cash prices vary, often $1,000–$1,300 per month. GoodRx coupons can sometimes drop the price modestly at participating pharmacies.
- Check your state Medicaid eligibility. Some patients qualify for Medicaid and don't know it.
- Consider the diabetes-clinic route. Federally Qualified Health Centers (FQHCs) and community health centers often have sliding-scale pricing and pharmacy partnerships.
- Talk to your clinician about alternatives. If Mounjaro is out of reach, semaglutide options, older GLP-1s, or non-GLP-1 diabetes medications may be more affordable starting points.
Note: Lilly Cares' available-medications list does not currently include Mounjaro. Other Lilly products like Trulicity and certain insulins are listed.
Before you book anything, find out what your plan actually covers
Most people guess at their insurance coverage and find out the real number at the pharmacy. There's a better way.
Ro's GLP-1 Insurance Coverage Checker (sponsored affiliate link, opens in a new tab) is free. No prescription required. You enter your insurance info, Ro's team contacts your insurer on your behalf, and you get a personalized report by email showing your Mounjaro coverage, your estimated copay, prior-authorization status, and supply availability. Quick honest note: Ro doesn't prescribe Mounjaro itself. The coverage checker is a free standalone tool you can use no matter where you ultimately fill your prescription.
Check what your insurance covers for Mounjaro -- free in 2 minutes → (sponsored affiliate link, opens in a new tab)How to get Mounjaro online for type 2 diabetes
Tirzepatide is not a controlled substance, so federal rules allow licensed providers to prescribe it via telehealth without a prior in-person visit. The two real paths for Mounjaro specifically are platforms that prescribe on the diabetes label -- not the weight-loss-only platforms. Sesame's Everyday Rx program (sponsored affiliate link, opens in a new tab) is built for chronic-condition management like type 2 diabetes. Program access starts at $59/month on the annual plan.
Path 1 -- Your primary care doctor or endocrinologist (the most common path)
If you already have a PCP or endocrinologist managing your diabetes, this is usually the cleanest route. They already have your A1C history, your medication list, and your chart notes.
What to bring:
- Your most recent A1C result (if you have one)
- Your current diabetes medication list (metformin, insulin, sulfonylureas, etc.)
- Any GLP-1 history (Trulicity, Ozempic, Bydureon, Byetta) -- including reasons you stopped, if applicable
- Your insurance card and pharmacy of choice
Ask your provider three things:
- “Is Mounjaro appropriate for me, given my history and contraindications?”
- “Will you submit the prior authorization for Mounjaro on the diabetes label, not Zepbound for weight loss?”
- “What's our plan if the first PA gets denied?”
Path 2 -- Sesame Everyday Rx (the cleanest online path for diabetes)
Sesame Care is a telehealth marketplace with two tirzepatide programs: Everyday Rx (for diabetes -- this is the program you want for Mounjaro) and Success by Sesame (for weight loss, prescribes Zepbound).
The Everyday Rx model:
- Answer an intake questionnaire about your diabetes history
- Browse Sesame's network -- pick your provider based on specialty, reviews, and availability
- Do a video consult (usually same-day or next-day)
- Provider reviews your history, orders labs if needed, and prescribes Mounjaro if appropriate
- Provider submits prior authorization through your insurance
- Prescription is sent to your local pharmacy
- Ongoing care includes unlimited provider messaging and monthly refill management
What it costs: Sesame's Everyday Rx subscription starts at $59/month with an annual plan, up to $99/month month-to-month. The medication is not included in the program fee -- your Mounjaro is filled at your pharmacy and runs through your insurance and the Lilly Savings Card just like it would with any other prescriber.
The honest tradeoff: Sesame is a marketplace model. You pick your provider -- that's better than being assigned one. But provider quality and prior-authorization experience can vary. The fix: read provider profiles before booking, and pick someone who explicitly mentions diabetes or endocrinology experience.
See same-day Mounjaro providers on Sesame Everyday Rx → (sponsored affiliate link, opens in a new tab)Path 3 -- Lilly Find Care (manufacturer-direct referral, not prescribing)
Lilly Find Care is a directory of independent in-person and telehealth providers that prescribe Lilly medications. It's not a prescribing service -- it's a referral tool. Useful if you want to see what's in your zip code or you specifically want a Lilly-vetted provider.
Path 4 -- Don't use telehealth (and that's okay)
We mean this seriously. Telehealth is convenient, not always optimal. Use a local clinician instead if any of these apply:
- You're on insulin or a sulfonylurea (these meds raise hypoglycemia risk when combined with tirzepatide -- close monitoring is safer in person)
- You have stage 3+ chronic kidney disease, heart failure, or known cardiovascular disease
- You've had pancreatitis, severe GI disease, or gallbladder disease
- You've already been denied Mounjaro and need an appeal
- Your A1C is uncontrolled despite multiple meds and you need a real care plan, not just a prescription
If you have type 2 diabetes and you're ready for the next step
Sesame Everyday Rx is purpose-built for the diabetes path -- not weight loss. You pick your provider (filter for diabetes or endocrinology experience), do a same-day or next-day video visit, and the provider handles the prior authorization with your insurance. Subscription starts at $59/month on the annual plan. The Mounjaro itself is billed separately.
See same-day Mounjaro providers on Sesame Everyday Rx → (sponsored affiliate link, opens in a new tab)Will insurance cover Mounjaro for type 2 diabetes?
Most commercial insurance plans cover Mounjaro for type 2 diabetes, but injectable diabetes medications like Mounjaro typically require prior authorization (PA) before they'll pay. PA is paperwork your prescriber files to prove medical necessity. The strongest PA submissions include a confirmed type 2 diabetes diagnosis (ICD-10 code E11.x), current and historical A1C results, your trial history with other diabetes medications (especially metformin), and a statement of why Mounjaro is the appropriate next step.

What insurance is actually checking when they review your PA
- Is there a confirmed type 2 diabetes diagnosis on file? (ICD-10 code E11.x)
- What is the patient's current A1C? (Plans usually want documentation, sometimes ≥7.0%)
- Has the patient tried metformin? For how long? At what dose? Was it tolerated?
- Has the patient tried another GLP-1 (Trulicity, Ozempic, Bydureon) -- and either failed it or had a documented intolerance?
- Are there contraindications that would rule out Mounjaro?
- Is the patient using insulin or a sulfonylurea? (This affects dose-adjustment planning)
- Does the plan have a preferred GLP-1 (often Ozempic or Trulicity)?
The “my A1C improved -- will I lose coverage?” fear
Plans don't have a single rule on this, and it varies. What you can do: ask your prescriber how they document continuation of therapy on annual reauthorizations. Make sure your prior A1C history (the high numbers before Mounjaro) is in the chart and referenced on the reauthorization form. If denied at reauthorization, appeal with a letter of medical necessity stating that Mounjaro is preventing diabetic complications and that discontinuation risks A1C rebound.
The Mounjaro denial triage -- what to do if your first PA gets rejected
| Why you were denied | What to do |
|---|---|
| Diagnosis not documented | Confirm with your prescriber's office that the ICD-10 code for type 2 diabetes (E11.x) was submitted, and that an A1C result is on file |
| Step therapy required | Ask which medication the plan wants you to try first. If you've already tried it, ask the prescriber to document that and resubmit |
| Not on formulary | Ask the prescriber's office to file a formulary exception with a letter of medical necessity |
| Quantity limits exceeded | Confirm the dose is within the FDA label; if so, the prescriber can document medical necessity for the prescribed dose |
| Need cardiovascular history / step from Ozempic | Ask whether Ozempic was tried and not tolerated, or whether a specific clinical reason supports Mounjaro |
The 12-question Mounjaro provider checklist
Before you book any online provider for a Mounjaro evaluation, ask:
- Do you specifically evaluate for Mounjaro for type 2 diabetes (not just Zepbound for weight loss)?
- Is the medication included in your fee, or billed separately through my pharmacy?
- Do you submit prior authorization on my behalf?
- Do you help with appeals if the first PA is denied?
- Do you order or review labs?
- Which pharmacies can fill the prescription?
- Can I use my commercial insurance and the Lilly Savings Card?
- What happens if my insurance denies Mounjaro?
- How are refills handled?
- How do you handle dose increases or reductions?
- What does cancellation look like?
- What happens if I have a side effect or a concern between visits?
Don't pay anyone who can't answer all twelve clearly.
Red flags to walk away from
- “No prescription required”
- “Research peptide” or “research use only”
- No licensed clinician named or verifiable
- No pharmacy disclosure
- “Guaranteed approval”
- Compounded product presented as if it's the same thing as FDA-approved Mounjaro
Mounjaro dosing for type 2 diabetes
Mounjaro is a once-weekly subcutaneous injection. The FDA label starts at 2.5 mg once weekly for the first 4 weeks -- a non-therapeutic “build-up” dose used to improve gastrointestinal tolerability. After 4 weeks, the dose increases to 5 mg once weekly. Based on your glycemic response and tolerability, your prescriber may increase the dose by 2.5 mg every 4 weeks. For adults, the maximum labeled dose is 15 mg once weekly. For pediatric patients ages 10 and older, the maximum labeled dose is 10 mg once weekly.
The standard adult titration schedule
| Weeks | Dose |
|---|---|
| 1–4 | 2.5 mg once weekly (non-therapeutic -- for tolerability only) |
| 5–8 | 5 mg once weekly |
| 9–12 | 7.5 mg once weekly (if needed) |
| 13–16 | 10 mg once weekly (if needed) |
| 17–20 | 12.5 mg once weekly (if needed -- adults only) |
| 21+ | 15 mg once weekly (maximum -- adults only) |
Source: Mounjaro FDA prescribing information, current label revised April 2026. Pediatric patients (10 and older) titrate by the same schedule, with a maximum of 10 mg once weekly.
Higher dose does not automatically mean better. Many patients reach excellent A1C control on 5 mg or 7.5 mg and never need to go higher. The right maintenance dose is the lowest dose that gets you to your A1C goal with side effects you can live with.
Injection basics
- Inject subcutaneously (just under the skin) in the abdomen, thigh, or upper arm.
- Rotate injection sites to avoid skin irritation.
- Inject on the same day each week, at any time of day, with or without food.
- If you miss a dose: within 4 days, take it as soon as you remember. After 4 days, skip the missed dose and resume on the next regular weekly date.
- Store unopened pens or vials in the refrigerator. Once in use, single-dose pens can be at room temperature for up to 21 days.
Side effects and serious warnings -- the honest version
The most common Mounjaro side effects in the adult placebo-controlled type 2 diabetes trial pool were gastrointestinal: nausea (12–18%), diarrhea (12–17%), decreased appetite (5–11%), vomiting (5–9%), constipation (6–7%), dyspepsia (5–8%), and abdominal pain (5–6%). Most are mild to moderate and happen during dose increases. Serious-but-rare risks include pancreatitis, gallbladder problems, severe gastroparesis, acute kidney injury (usually from dehydration), and severe hypersensitivity reactions. Mounjaro carries a boxed warning for thyroid C-cell tumors including medullary thyroid carcinoma (MTC) based on rodent studies. Mounjaro should not be used by patients with a personal or family history of MTC or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Common side effects (adult placebo-controlled T2D trial pool, FDA label)
- Nausea -- 12% to 18% depending on dose
- Diarrhea -- 12% to 17%
- Decreased appetite -- 5% to 11%
- Vomiting -- 5% to 9%
- Constipation -- 6% to 7%
- Dyspepsia (indigestion) -- 5% to 8%
- Abdominal pain -- 5% to 6%
- Injection-site reactions -- lower single-digit percentages
These are worst during dose increases. Most people find they fade within a few weeks of staying at a steady dose. Eating smaller meals, staying hydrated, and avoiding very fatty or fried foods helps. See our guide to GLP-1 providers with the best side-effect support for practical nausea management strategies.
The boxed warning -- explained without panic
Mounjaro carries an FDA boxed warning about thyroid C-cell tumors, including medullary thyroid carcinoma (MTC). The warning is based on rodent studies -- the human risk hasn't been established. What it means in practice:
- Mounjaro is contraindicated if you have a personal or family history of medullary thyroid carcinoma.
- Mounjaro is contraindicated if you have Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- If you have a thyroid nodule or unexplained neck mass, your provider should evaluate it before starting Mounjaro.
- This is the same warning class that Ozempic, Wegovy, Trulicity, and the rest of the GLP-1 class carry.
Dehydration and kidney health
The most common reason Mounjaro causes acute kidney injury is dehydration from GI side effects. The fix is unglamorous: drink water, especially during dose increases. Tell your provider right away if you can't keep fluids down.
Hypoglycemia risk in diabetes
Mounjaro alone rarely causes low blood sugar. But when combined with insulin or a sulfonylurea, the hypoglycemia risk goes up. Your prescriber will usually reduce those doses when you start Mounjaro.
Other warnings worth knowing
- Pancreatitis -- rare but serious. Stop Mounjaro and seek care for severe abdominal pain that doesn't go away, especially if it radiates to the back.
- Gallbladder disease -- slightly increased risk. Watch for upper-right belly pain, fever, jaundice.
- Diabetic retinopathy -- rapid A1C improvements can temporarily worsen retinopathy. Your provider should know your retinopathy status before starting.
- Severe gastrointestinal disease -- caution with gastroparesis or significant GI motility issues.
- Pregnancy and breastfeeding -- Mounjaro is not recommended; discontinue at least 2 months before a planned pregnancy.
- Pediatric use -- approved for kids 10+ with type 2 diabetes; pediatric prescribing should be done by a pediatric endocrinologist, with a 10 mg maximum dose.
Who should NOT use Mounjaro for diabetes
Mounjaro is not appropriate for type 1 diabetes, for anyone with a personal or family history of medullary thyroid carcinoma or MEN 2, or for anyone with a severe hypersensitivity to tirzepatide. Use caution if you have a history of pancreatitis, severe gastroparesis, advanced diabetic retinopathy, severe kidney disease, are pregnant or planning pregnancy, or are breastfeeding.
If anything below applies to you, the right next step is NOT an online provider visit -- it's a conversation with your endocrinologist or PCP.
- Type 1 diabetes (use your endocrinologist)
- A personal or family history of medullary thyroid carcinoma or MEN 2 (Mounjaro is contraindicated)
- A history of pancreatitis (talk to your specialist first)
- Advanced diabetic retinopathy (needs ophthalmology input before starting)
- Severe stage 4–5 chronic kidney disease
- Active pregnancy or planning pregnancy within 2 months
- An active eating disorder (GLP-1s are not appropriate)
If that's you and you want help mapping where to start, our quiz routes people in your situation toward the right alternative rather than pushing you toward a prescription that isn't a fit.
Real reviews -- what patients actually say (and what to take with a grain of salt)
Service reviews can tell you a lot about the experience of using a telehealth platform -- wait times, provider responsiveness, billing transparency -- but they cannot tell you whether Mounjaro will work for you medically. We use attributed quotes about service experience only.
Patient experience matters. Here's what people say about Sesame, which is our primary recommendation on this page. These are real service-experience reviews published on Sesame's homepage -- not medical efficacy claims.
“I have a busy schedule and high deductible insurance, the affordability of this service is amazing and my doctor was great! I'll book again and again.”
“This is a life changing service... The appointment and meds cost me less than a typical appointment alone. Mind blown.”
“I love this service. It was extremely convenient. I was able to get an appointment within an hour! My doctor was fantastic, super kind and able to prescribe the medication I needed right away.”
The honest counterweight: Sesame, like every major telehealth platform, also has complaints -- billing disputes and expectations mismatches. The most common pattern in negative reviews is expectation misalignment: a patient books expecting a guaranteed prescription, the provider determines the medication isn't right for that patient. The prescription is never guaranteed on any legitimate telehealth platform. A platform that would guarantee a prescription regardless of clinical fit is the one to be worried about.
Individual results vary. The reviews above are service-experience reviews and do not imply typical medical results for any reader. Discuss whether Mounjaro is appropriate for your situation with a licensed clinician.
How we ranked the best tirzepatide for diabetes
We rank tirzepatide options for diabetes by FDA-approved indication first, by clinical evidence from peer-reviewed trials second, by realistic access and affordability third, and by editorial fit for the specific search intent of this page fourth. We do not rank by affiliate payout. We do not present compounded tirzepatide as equivalent to FDA-approved Mounjaro. We do not include fake reviews, fabricated medical reviewers, or schema that doesn't match visible page content.
- 30% -- FDA-approved indication for type 2 diabetes. Non-negotiable. Mounjaro has it. Zepbound does not. Compounded tirzepatide does not.
- 25% -- Clinical evidence. SURPASS-1 through SURPASS-5 published trials, SURPASS-CVOT (December 2025), the March 2026 Cleveland Clinic / JAMA Cardiology analysis, and FDA-approved prescribing information.
- 20% -- Access path quality. Can the reader actually get the prescription? Does the prescriber handle prior authorization?
- 15% -- Cost transparency. Are list prices, savings program terms, and out-of-pocket cost estimates clearly available?
- 10% -- Follow-up care and ongoing support. Refills, dose adjustments, side effect management.
Why Sesame and not Ro for this page
Our usual recommendation for FDA-approved GLP-1s is Ro. For this specific page -- the best tirzepatide for diabetes -- Ro does not currently prescribe Mounjaro according to Ro's own page. Sesame's Everyday Rx program does. Evidence overrode default priority. Also see our complete GLP-1 for diabetes comparison and best online tirzepatide providers.
Frequently asked questions
Is Mounjaro the same as tirzepatide?
Yes. Mounjaro is the brand name. Tirzepatide is the generic name of the active drug. Eli Lilly makes two FDA-approved brand-name formulations of tirzepatide: Mounjaro (labeled for type 2 diabetes) and Zepbound (labeled for chronic weight management and obstructive sleep apnea).
Is Mounjaro better than Ozempic for diabetes?
In the SURPASS-2 head-to-head trial, all three doses of Mounjaro (5, 10, and 15 mg) lowered A1C and body weight more than semaglutide (Ozempic) 1 mg over 40 weeks. However, Ozempic carries an FDA-approved indication for reducing cardiovascular risk in type 2 diabetes with established heart disease, which Mounjaro does not yet have. The right choice depends on your specific health priorities -- talk to your clinician.
Is Zepbound the same as Mounjaro for diabetes?
Zepbound and Mounjaro contain the same active drug (tirzepatide) at the same core adult dose strengths, but they are FDA-approved for different conditions. Zepbound is approved for chronic weight management and obstructive sleep apnea. Mounjaro is approved for type 2 diabetes. For a diabetes patient, asking for Mounjaro by name is almost always the right paperwork -- insurance is more likely to cover it, and the Mounjaro Savings Card only applies to FDA-approved-use prescriptions.
Can I get Mounjaro online for type 2 diabetes?
Yes. Tirzepatide is not a controlled substance, so federal law allows licensed providers to prescribe it via telehealth without a prior in-person visit. State telehealth rules still apply. Sesame's Everyday Rx program is one telehealth path specifically built for diabetes-labeled Mounjaro prescriptions. The prescription is not guaranteed -- your provider determines clinical appropriateness.
How much does Mounjaro cost without insurance in 2026?
Mounjaro's current list price is $1,112.16 for a 28-day supply. Retail cash prices typically run from about $1,000 to $1,300 per month depending on pharmacy and location. The Lilly Mounjaro Savings Card can reduce cost meaningfully even when insurance does not cover Mounjaro, but uninsured patients cannot use the card at all.
Will Medicare cover Mounjaro for type 2 diabetes?
Medicare Part D plans generally cover Mounjaro when prescribed for FDA-approved type 2 diabetes use, subject to your specific plan's formulary tier and prior-authorization requirements. Lilly reports that about 8 out of 10 Part D patients pay $0-$50 per 28-day supply, with the remaining patients averaging about $262. Medicare beneficiaries cannot legally use the Mounjaro Savings Card. Ask about the Medicare Prescription Payment Plan (M3P) for monthly cost smoothing.
Is compounded tirzepatide legal in 2026?
Mass compounding of tirzepatide by 503B outsourcing facilities became legally exposed on March 19, 2025, after the FDA's enforcement-discretion window closed. 503A state-licensed pharmacy compounding lost the same shelter on February 18, 2025. On April 30, 2026, the FDA proposed excluding tirzepatide from the 503B Bulks List; public comment is open until June 29, 2026. A narrow 503A pathway still exists for documented individual clinical needs that an FDA-approved product cannot meet. For routine type 2 diabetes management, the FDA-approved brand (Mounjaro) is the realistic legal path in 2026.
What dose of Mounjaro is used for type 2 diabetes?
The FDA label starts Mounjaro at 2.5 mg once weekly for four weeks (a non-therapeutic build-up dose), then increases to 5 mg once weekly. Your provider may increase by 2.5 mg every 4 weeks based on your A1C response and tolerability. For adults, the maximum dose is 15 mg once weekly. For pediatric patients ages 10 and older, the maximum dose is 10 mg once weekly.
How much does Mounjaro lower A1C?
In the SURPASS clinical program (SURPASS-1 through SURPASS-5), Mounjaro reduced A1C by approximately 1.87% to 2.59% across the 5, 10, and 15 mg doses, depending on trial design and background therapy. In SURPASS-2 head-to-head against semaglutide 1 mg, reductions were 2.01% (5 mg), 2.24% (10 mg), and 2.30% (15 mg). Individual results vary based on starting A1C, background therapy, adherence, and lifestyle.
What if my insurance denies Mounjaro?
Ask why it was denied, then ask your prescriber whether a letter of medical necessity, a formulary exception, or a formal appeal is the right next step. The strongest appeals include confirmed type 2 diabetes diagnosis (ICD-10 E11.x), current and historical A1C, prior medication trials (especially metformin and any GLP-1 history), any contraindications to alternative GLP-1s, and a clinical rationale for Mounjaro specifically.
Can kids with type 2 diabetes use Mounjaro?
Yes -- per the current FDA label, Mounjaro is approved for pediatric patients 10 years and older with type 2 diabetes, as an adjunct to diet and exercise. The maximum pediatric dose is 10 mg once weekly. Pediatric prescribing should be handled by a pediatric endocrinologist.
Is Mounjaro a cure for diabetes?
No. Mounjaro helps control blood sugar by improving insulin response, slowing gastric emptying, and reducing appetite -- but it does not cure type 2 diabetes. Discontinuing the medication typically causes A1C to rise again. Treatment is generally long-term.
Still not sure which GLP-1 program is right for you?
If you're not sure whether tirzepatide is the right GLP-1 for your situation -- maybe you're not sure if your A1C qualifies you, or whether your goal is really diabetes management or weight management or both -- take our free 60-second matching quiz.
Take The RX Index's free 60-second matching quiz →Sources and primary references
FDA
Eli Lilly
- Mounjaro list price reference (pricinginfo.lilly.com)
- Mounjaro Savings Card terms (current 2026 program through Dec 31, 2026)
- Mounjaro patient-cost FAQ (Medicare and Medicaid figures from IQVIA Oct 2024--Sept 2025)
- Lilly Cares available medications
Peer-reviewed clinical evidence
About this guide
This guide was researched and written by The RX Index Editorial Team. The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We track FDA labels, provider offerings, pricing changes, and regulatory developments across the GLP-1 category and update our guides on a quarterly cadence -- more often for time-sensitive pages like this one.
We earn commissions from some of the providers linked on this page when readers sign up. Affiliate payout never influences our editorial ranking or our willingness to recommend a non-affiliate alternative when the evidence calls for it. Read our full methodology and affiliate disclosure.
This page is not medical advice. Talk to a licensed clinician before starting, changing, or stopping any diabetes medication.