Researched and written by The RX Index Editorial Team · Last verified: April 30, 2026 · Next scheduled re-verification: July 30, 2026 · Sources: FDA prescribing labels via DailyMed, JAMA Internal Medicine 2022, NIDDK, ACG 2025, Eli Lilly medical statements · Editorial standards
GLP-1 GALLBLADDER RISK — 2026 EVIDENCE GUIDE
GLP-1 Gallbladder Risk by Drug: What the FDA Labels and the JAMA Meta-Analysis Actually Say
Published:
The honest bottom line, up front.
GLP-1 medications carry a real but modest increase in gallbladder problems — roughly a 37% relative risk increase versus placebo or comparator, which works out to about 27 additional cases of gallbladder or biliary disease per 10,000 people per year in the largest pooled analysis. Risk is meaningfully higher for weight-loss-indication use (relative risk 2.29) than diabetes-indication use (relative risk 1.27), and higher with higher doses and longer treatment. The smartest moves: know the symptom triage at the top of this page, pace your weight loss, and stay in close contact with a prescriber who can respond fast.
This is the page we wished existed when readers started writing in asking which GLP-1 was “safer for the gallbladder.” The honest answer is more useful than a ranking — it's a per-drug rate table from the FDA labels, a triage card you can act on today, and a clear-eyed view of which risk factors are in your control.
Sources verified: FDA prescribing information via DailyMed (April 30, 2026); He et al., JAMA Internal Medicine 2022 meta-analysis; NIDDK gallstone guidance; Eli Lilly published medical statement on tirzepatide. Full reference list at the bottom of this page.
Disclosure: Some links on this page are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you.

When to call your prescriber, when to go to the ER
The short answer: Sudden, severe, persistent right-upper-belly pain — especially with fever, vomiting that won't stop, jaundice (yellowing of the eyes or skin), dark urine, or pale stools — is an emergency. Get evaluated today. Less severe right-upper-belly pain after fatty meals, or new pain that comes and goes, is a same-day or next-day call to your prescriber, not an “ignore it and hope” situation.
GLP-1 nausea and routine GI side effects feel different from gallbladder symptoms. The triage card below is calibrated to the difference. Save it before you start, and refer back if anything changes.
| Tier | What you might feel | What to do |
|---|---|---|
| Emergency | Severe, persistent right-upper-belly pain (often radiating to the right shoulder blade or back); fever or chills; jaundice; persistent vomiting; dark urine or pale stools. | Seek urgent medical care now. Then contact your prescriber — do not take another dose until evaluated. |
| Urgent — same day | New right-upper-belly pain lasting 30 minutes or more, especially after fatty meals; nausea or vomiting that's different from your usual GLP-1 GI side effects. | Message your prescriber today; do not escalate your dose. Document timing, location, severity, and what you ate. |
| Prompt — next visit | Intermittent mild right-side discomfort or fullness; new fat-meal intolerance; pain that comes and goes over days. | Bring it up at your next prescriber visit (within 1–2 weeks). Keep a brief food/symptom log. |
| Routine | Mild generalized bloating; usual GLP-1 nausea that improves with smaller meals. | Standard supportive care. Mention at your next routine check-in if it persists. |
One important distinction: Gallbladder pain is typically sharp or stabbing, focused under the right rib cage, often radiating to the right shoulder blade or middle back, and frequently triggered within an hour of a fatty meal. Routine GLP-1 nausea is more diffuse and central, less sharp, and usually improves with smaller, lower-fat meals. If you can't tell which one you're feeling, treat it as the higher tier and call.
Is this normal GLP-1 nausea, or is it your gallbladder?
The short answer: Routine GLP-1 GI side effects tend to be diffuse, dose-related, and improve with smaller meals and time. Gallbladder pain tends to be sharp, localized to the upper-right belly, often appears after fatty meals, and frequently radiates to the right shoulder or back. Fever, jaundice, and abnormal urine or stool color point clearly toward gallbladder or bile duct trouble — not nausea.
| Symptom pattern | More consistent with… | What to do |
|---|---|---|
| Mild nausea after a dose increase that improves with smaller meals | Standard GLP-1 GI side effect | Standard management; mention at next visit if it persists |
| Bloating, mild cramping, constipation, mild reflux | Standard GLP-1 GI side effect | Hydration, fiber, smaller meals |
| Sharp pain in the upper-right belly after fatty meals | Gallbladder attack pattern | Call prescriber today; ER if severe or with red flags |
| Pain radiating to the right shoulder or middle of the back | Gallbladder attack pattern | Same as above |
| Pain with fever or chills | Possible gallbladder inflammation or infection | Urgent medical care |
| Yellow eyes or skin (jaundice) | Possible bile duct blockage | Urgent medical care |
| Tea-colored urine or clay-colored stools | Possible bile flow problem | Urgent medical care |
| Severe abdominal pain radiating to the back with vomiting | Possible pancreatitis (separate emergency) | Urgent medical care |
| Vomiting that won't stop, can't keep fluids down | Either severe GLP-1 GI or another emergency | Call prescriber today, ER if dehydrated |
The pattern to remember: GLP-1 GI side effects most people get are predictable, central, and time-limited. Gallbladder attacks are sharp, right-sided, often meal-triggered, and pattern-repeating. They're different animals. When in doubt, lean toward calling.
GLP-1 nausea and gallbladder pain can both involve the upper belly. They feel different to clinicians, but they can feel similar to a person experiencing them for the first time. Naming the difference is what gets you to care faster — and outcomes for gallbladder problems are very good when caught early. NIDDK warns that an untreated bile duct blockage can be life-threatening, which is exactly why red flags shouldn't be dismissed.
A general surgeon who treats a lot of these patients put it this way: William Hinojosa, DO, of Oklahoma Surgical Associates, told Norman Regional Health System's editorial team in January 2026 that when patients on GLP-1s describe right-upper-belly pain, his recommendation is simple: “reaching out to your primary care provider, and getting evaluated.” Hold the dose conversation, call, get the imaging. That's the whole playbook.
Save the symptom checklist + prescriber script. Jump to the one-page version → Designed so nothing important gets missed at your next visit.
What the evidence actually shows
The short answer: The 2022 JAMA Internal Medicine meta-analysis pooled 76 randomized clinical trials (103,371 patients) and found GLP-1 receptor agonist use was associated with a 37% increased relative risk of gallbladder or biliary disease (RR 1.37, 95% CI 1.23–1.52) — translating to about 27 additional cases per 10,000 people per year. Real-world cohort data from the 2025 American College of Gastroenterology TriNetX study also showed elevated risk over a 3-year window. The signal is consistent. The absolute risk to any one person remains modest.
We want you to see the actual numbers, because most pages on this topic either round them up to scare you or round them down to reassure you. Here are the four numbers that matter most:
- 1.37 relative risk — the pooled hazard from the JAMA meta-analysis, versus placebo or comparator. (95% CI 1.23–1.52.)
- 27 extra cases per 10,000 people per year — the absolute-risk translation of that relative risk. Most people on GLP-1s won't develop gallbladder disease.
- RR 2.29 weight-loss indication vs RR 1.27 diabetes indication — the same drug class is roughly twice as risky when used at higher doses for faster weight loss.
- RR 1.56 high dose vs RR 0.99 low dose; RR 1.40 long use vs RR 0.79 short use — both dose and duration are modifiable levers.
The 2025 ACG real-world data point in the same direction. A 3-year retrospective cohort presented at the 2025 American College of Gastroenterology meeting compared GLP-1 users with non-users in the TriNetX research network. Cumulative gallbladder/biliary outcomes were higher among GLP-1 users at 3 years — strengthening the case that the trial signal also shows up in routine clinical care, not just in pivotal-trial conditions.
Context for the absolute risk. Roughly 10–15% of U.S. adults already have gallstones at baseline per NIDDK. Most are silent and never cause problems. So when GLP-1s push the population-level rate up, the additional cases stack onto a baseline that's already common. This is the honest framing: the risk is real, the absolute number is modest, and the controllable factors are dose, duration, and pace of weight loss.
Gallbladder rates by drug (FDA prescribing labels)
The short answer: Label-reported gallbladder event rates range from a 0.6% acute-gallbladder composite for Mounjaro to a 2.5% cholelithiasis rate for Wegovy oral tablets, with most other approved GLP-1s falling somewhere between 0.6% and 1.6% in their pivotal trials. These are not head-to-head comparisons — different drugs were studied in different populations, doses, indications, and trial durations.
This table is the original evidence asset on this page. We pulled the rates from each medication's FDA prescribing information via DailyMed rather than relying on aggregated secondary summaries. The caveats matter — we explain them below the table.
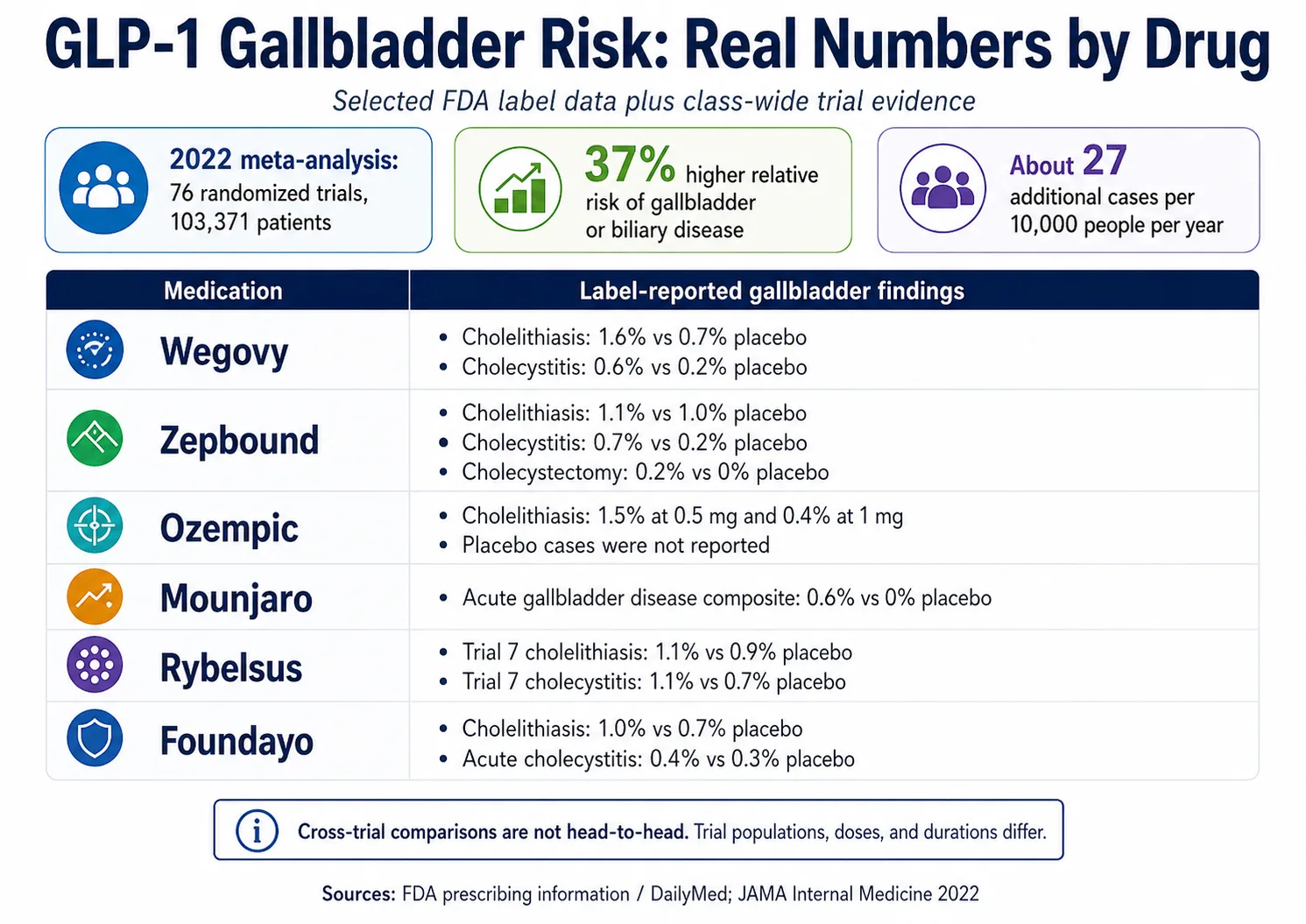
| Medication | Active ingredient / class | Approved indication | Cholelithiasis | Cholecystitis | Cholecystectomy | Source |
|---|---|---|---|---|---|---|
| Wegovy® injection | Semaglutide / GLP-1 RA | Chronic weight management | 1.6% Wegovy vs 0.7% placebo | 0.6% Wegovy vs 0.2% placebo | Not separately reported in cited section | DailyMed: Wegovy PI |
| Wegovy® oral tablets | Semaglutide / GLP-1 RA | Chronic weight management | 2.5% Wegovy vs 1.0% placebo | Reported in label | Not separately reported in cited section | DailyMed: Wegovy PI |
| Zepbound® | Tirzepatide / dual GIP + GLP-1 RA | Chronic weight management | 1.1% Zepbound vs 1.0% placebo | 0.7% Zepbound vs 0.2% placebo | 0.2% Zepbound vs 0% placebo | DailyMed: Zepbound PI |
| Mounjaro® | Tirzepatide / dual GIP + GLP-1 RA | Type 2 diabetes | 0.6% Mounjaro vs 0% placebo for an acute gallbladder disease composite (cholelithiasis + biliary colic + cholecystectomy) in SURPASS-1 and SURPASS-5 | DailyMed: Mounjaro PI; Eli Lilly medical statement | ||
| Ozempic® | Semaglutide / GLP-1 RA | Type 2 diabetes / CV risk | 1.5% on 0.5 mg and 0.4% on 1 mg vs 0% placebo (glycemic-control trials) | Acute gallbladder events reported in trials and postmarketing; placebo-controlled cholecystitis rate not given in cited section | Same | DailyMed: Ozempic PI |
| Rybelsus® | Oral semaglutide / GLP-1 RA | Type 2 diabetes | 1.0% on 7 mg in glycemic trials; in Trial 7 (4-yr CV outcomes) 1.1% on 14 mg vs 0.9% placebo | 1.1% on 14 mg vs 0.7% placebo (Trial 7) | Class warning per label | DailyMed: Rybelsus PI |
| Foundayo™ | Orforglipron / oral non-peptide GLP-1 RA | Chronic weight management (FDA-approved April 2026) | 1.0% Foundayo vs 0.7% placebo (pooled Trials 1 & 2) | 0.4% Foundayo vs 0.3% placebo | Not separately reported in cited section | DailyMed: Foundayo PI |
| Saxenda® | Liraglutide 3.0 mg / GLP-1 RA | Chronic weight management | 2.2% Saxenda vs 0.8% placebo | 0.8% Saxenda vs 0.4% placebo | Label notes the majority of Saxenda-treated patients with cholelithiasis or cholecystitis adverse events required cholecystectomy | DailyMed: Saxenda PI |
| Victoza® | Liraglutide 1.8 mg / GLP-1 RA | Type 2 diabetes | 0.3% vs 0.3% placebo (pooled glycemic trials); LEADER (CV outcomes): 1.5% Victoza vs 1.1% placebo | 1.1% Victoza vs 0.7% placebo (LEADER) | Label notes the majority of events required hospitalization or cholecystectomy | DailyMed: Victoza PI |
| Trulicity® | Dulaglutide / GLP-1 RA | Type 2 diabetes / CV risk | CV outcomes trial: 0.62 per 100 patient-years on Trulicity vs 0.56 placebo (adjusted for prior cholecystectomy) | 0.5% Trulicity vs 0.3% placebo for serious acute cholecystitis | Reported in label | DailyMed: Trulicity PI |
How to read this table without misreading it
A few honest caveats — because the table is more useful when you understand its limits:
This is not a “safest GLP-1” ranking. Trial populations differ. Wegovy, Zepbound, Saxenda, and Foundayo were studied in people taking the drug for weight loss. Ozempic, Mounjaro, Rybelsus, Victoza, and Trulicity were studied primarily in people with type 2 diabetes. Doses, durations, follow-up windows, and baseline risk profiles all differ. A lower number in a diabetes trial doesn't necessarily mean the drug is “safer for your gallbladder” if you're taking it for weight loss.
The class shows a signal, and current labels reflect it. Multiple labels include explicit gallbladder warnings and report rates higher than placebo. Whether your trial reported 0.4% or 2.5% matters less than your personal risk factors and your symptom awareness.
The labels broadly agree on what to do. They say some version of: if cholelithiasis or cholecystitis is suspected, gallbladder diagnostic studies and clinical follow-up are indicated. They don't tell you to stop the medication at the first twinge. They tell you to investigate.
Why GLP-1s affect the gallbladder — the two mechanisms
The short answer: GLP-1 medications raise gallbladder risk through two simultaneous biological pathways. They slow how often the gallbladder empties, partly by suppressing cholecystokinin (CCK, the hormone that triggers gallbladder contraction). And they cause significant weight loss, which itself shifts bile chemistry. Slow emptying plus saturated bile is the classic recipe for stones.
Knowing the mechanism matters because it tells you which lever you can actually pull.
Mechanism 1 — slower gallbladder emptying. Your gallbladder is a small storage pouch under your liver that holds bile and squeezes it into the small intestine when you eat fat. The squeeze signal comes mostly from a hormone called cholecystokinin (CCK). GLP-1s appear to dampen this cycle: the gallbladder empties less often and less completely. Bile sits longer.
Mechanism 2 — rapid weight loss changes bile chemistry. When you lose body fat fast, your liver dumps extra cholesterol into bile. NIDDK identifies rapid weight loss as a recognized gallstone risk factor for any cause — surgery, very-low-calorie diets, intense exercise, or GLP-1s. At a certain saturation point, cholesterol can crystallize. Crystallized cholesterol is, mechanically, a tiny stone.
The two effects compound. This is why the JAMA meta-analysis showed weight-loss-indication GLP-1 use roughly doubled the risk versus diabetes-indication use. The drug class is the same; the difference is the dose and the speed of the weight loss.
The practical implication: pacing the weight loss is one of the biggest controllable levers. We'll get to that.
Are you higher or lower risk? A personal risk modifier
The short answer: Your personal gallbladder risk on a GLP-1 isn't the population average. It's the average modified by factors you bring with you. Specific things raise it: weight-loss use (vs. diabetes), higher doses, longer use, female sex, age 40+, BMI 30+, prior gallstones or family history, and rapid weight loss. Specific things lower it: lowest effective dose, shorter use, diabetes-only indication, and slow steady weight loss.
The “27 extra cases per 10,000 per year” number is an average across a huge mixed population. Your number is different. Here's a self-stratification framework you can use to get a rough sense of where you fall — built directly from the JAMA Internal Medicine subgroup data plus standard gallbladder epidemiology.
| Factor | Direction | Why |
|---|---|---|
| Using a GLP-1 for weight loss rather than diabetes | ↑ raises | JAMA: RR 2.29 weight loss vs 1.27 diabetes |
| Higher dose of your GLP-1 | ↑ raises | JAMA: RR 1.56 high dose vs 0.99 low dose |
| More than 26 weeks on the medication | ↑ raises | JAMA: RR 1.40 long vs 0.79 short |
| Losing weight rapidly | ↑ raises | NIDDK: rapid weight loss is a recognized gallstone risk factor |
| Female | ↑ raises | Background gallbladder epidemiology |
| Age 40 or older | ↑ raises | Background epidemiology |
| BMI 30 or higher at baseline | ↑ raises | Background epidemiology |
| Prior gallstones or close family history | ↑ raises | Established risk factor |
| Other rapid-weight-loss interventions stacked on top (very-low-calorie diet, bariatric prep) | ↑ raises | Compounds the bile chemistry effect |
| Taking the lowest effective dose that works for you | ↓ lowers | JAMA dose-response |
| Diabetes-only indication, slow weight loss | ↓ lowers | JAMA indication subgroup |
| Already had your gallbladder removed | ⚪ separate | Not a contraindication for GLP-1s — see dedicated section below |
How to use this list — as a prescriber-conversation guide, not a probability calculator:
- 0–2 risk factors stacked: baseline awareness; mention any new symptoms early.
- 3–5 stacked: worth a focused conversation with your prescriber before dose escalation — ask about pace and what they want you to report.
- 6+ stacked: ask about a slower titration and a written symptom-response plan.
This is a framework for a better prescriber conversation, not a diagnosis or validated probability. Higher-risk results don't mean you shouldn't take a GLP-1. They mean you and your prescriber should pace the weight loss, watch for symptoms, and consider a slower titration. The dose and duration findings in the meta-analysis are good news — they're levers you can pull.
Not sure which GLP-1 program fits your risk profile? Our 60-second matching quiz factors in budget, insurance status, and medication preference, then routes you to programs that fit — including options with stronger clinical wraparound for readers who want it.
Take the matching quiz →How to lower your risk — what's actually in your control
The short answer: Pacing the weight loss is the single most controllable lever — NIDDK's general guidance favors slow, gradual loss over rapid loss because rapid loss is a known gallstone risk factor. Other measurable steps: stay well-hydrated, don't stack a GLP-1 on top of an extreme low-calorie diet, ask your prescriber about slower dose titration if you have multiple risk factors, and report any right-side pain early. Ursodiol may have a role in specific clinician-supervised contexts.
Here are five levers, in rough order of impact:
1. Pace the weight loss. NIDDK supports slow, gradual weight loss with dietary changes — and explicitly warns that rapid weight loss raises gallstone risk. This is the part of GLP-1 therapy patients most often want to ignore because the rapid loss is the part that feels like winning. It's also the part that quietly drives most of the gallbladder risk. Your prescriber can help you set a realistic weekly target.
2. Stay hydrated. Standard supportive care during any weight-loss period. It also helps with the routine GI side effects of GLP-1s, which is its own win.
3. Don't stack extreme caloric restriction on top of a GLP-1. GLP-1s already shrink your appetite. Combining that with a very-low-calorie diet (under ~800 kcal/day) compounds the rapid-weight-loss biology. NIDDK identifies very-low-calorie dieting as a separate gallstone risk factor on its own. If you're working with a clinician on a structured medical weight-loss program, that's different. Solo crash dieting on top of Wegovy or Zepbound is not the move.
4. Eat regular meals — don't skip them. Long fasting periods between meals can be associated with sluggish gallbladder emptying. Consistent meal timing is the more practical version of “keep things moving.” If fatty meals reliably trigger pain, bring that pattern to your prescriber.
5. Ask about titration speed if you have stacked risk factors. If you scored high on the personal risk modifier (female, 40+, BMI 30+, weight-loss indication), a slower dose escalation is a perfectly legitimate ask. Most prescribers will accommodate. The clinical trial titration schedules are designed for tolerability and convenience — not personalized risk.
Optional — bring this up only if it applies to you: NIDDK states that ursodeoxycholic acid (ursodiol) can help prevent gallstones in people losing weight rapidly through very-low-calorie diets or weight-loss surgery. If you're losing weight rapidly on a GLP-1, ask your clinician whether that context applies to you. This is a prescriber decision, not something to self-source.
The honest disclosure: None of these eliminates the risk. They reduce it. But the JAMA data is clear that dose and duration are two big modifiable drivers, and the speed of your weight loss is the third. You can move all three.
Can you take a GLP-1 if you already have gallstones?
The short answer: Often yes — but it depends on whether your stones are silent (no symptoms) or symptomatic (causing attacks), and your prescriber makes the call. NIDDK notes that silent gallstones usually don't need treatment, while symptomatic stones often do. A history of gallstones is not an automatic disqualifier for GLP-1 therapy, but it does change the conversation.
This is one of the most common questions readers arrive at this page asking, and it has three different answers depending on your situation.
If you have silent gallstones (found incidentally on imaging, no attacks): NIDDK says silent gallstones often don't require treatment. Many people with silent stones take GLP-1s safely. The conversation with your prescriber should cover: how you'd recognize a first attack, what symptoms warrant calling, and whether they want any baseline labs before you start or escalate the dose.
If you've had gallstone attacks before but never had your gallbladder removed: This deserves a more careful pre-start conversation. Active or recurrent symptomatic gallstones often lead to a surgical referral. Some clinicians will defer GLP-1 initiation until the gallbladder issue is resolved; others will proceed with closer monitoring. The right answer depends on attack frequency, severity, your overall medical picture, and your prescriber's judgment.
If you have a known family history of gallstones but no personal stones yet: Mention it. It modestly raises your baseline risk and is worth flagging. It generally doesn't change whether you can start a GLP-1, but it might change your prescriber's monitoring posture.
What you should ask before starting if you have any gallstone history:
- Given my gallstone history, do you want me to get a baseline ultrasound before starting?
- What's your titration plan for someone with my history?
- What symptoms should make me call you immediately versus mention at the next visit?
- If I do have an attack, what's the protocol — hold the dose, ER, or evaluate first?
The headline: don't write yourself off because you've had stones before. People with gallstone histories take GLP-1s every day. The conversation just looks slightly different than it does for someone with a clean history.
Should you stop your GLP-1 if you have gallbladder pain?
The short answer: If your symptoms match the EMERGENCY row of the triage card — severe pain, fever, jaundice, persistent vomiting, dark urine, or pale stools — seek urgent medical care and contact your prescriber before taking another dose. The FDA labels for Wegovy, Zepbound, Foundayo, and other GLP-1s say that if cholelithiasis or cholecystitis is suspected, gallbladder diagnostic studies and clinical follow-up are indicated. They don't tell you to stop the medication unilaterally — they tell you to investigate.
The most common mistake we see in patient discussions: people quietly stopping their medication for a week, feeling better, then restarting at the same dose without telling anyone. That's the worst of both worlds. You lose the benefit and you lose the diagnostic information your prescriber needs to figure out what's actually happening.
The right play if you suspect gallbladder trouble:
- Hold the dose conversation. Don't escalate. Don't restart. Don't decide alone.
- Contact your prescriber today if symptoms are mild but new. Seek urgent care if they match the emergency row.
- Document the timing, location, severity, what triggered it, what made it better or worse.
- Let the clinician confirm what's happening and tell you when and how to resume.
Confirmed gallstones don't automatically end your GLP-1 therapy. Asymptomatic stones found incidentally on ultrasound often don't require stopping the medication at all — that's a clinical judgment call your prescriber makes with you.
The labels on Wegovy, Zepbound, and Foundayo are surprisingly readable on this. All three say some version of: if cholelithiasis or cholecystitis is suspected, gallbladder diagnostic studies and clinical follow-up are indicated. Investigate first. Decide second.
You're allowed to advocate for yourself here. If you're worried about gallbladder symptoms, you don't need to wait for a routine appointment — most telehealth and in-person prescribers have same-day or next-day messaging for symptom concerns. Use it.
Take the prescriber question scripts to your visit. Jump to the three scripts → One short page so you don't forget anything important under stress.
Can you take a GLP-1 after gallbladder removal?
The short answer: Cholecystectomy (gallbladder removal) is generally not a barrier to GLP-1 therapy. Eli Lilly's published medical statement says prior cholecystectomy is not a contraindication for tirzepatide (Mounjaro / Zepbound), and the same is true for many other GLP-1s based on their labels and standard clinical practice. The decision belongs to your prescriber and depends on your specific situation.
Cholecystectomy is one of the most common abdominal surgeries done in the U.S. NIDDK says people can live normally without a gallbladder — bile just flows continuously from the liver into the small intestine instead of being stored.
What changes for GLP-1 use after cholecystectomy:
- Generally not a contraindication. Lilly's published clinical Q&A on tirzepatide explicitly states it's not contraindicated in patients who have had a cholecystectomy, and that prescribing decisions should be made by the treating clinician.
- Possible GI overlap. Some people post-cholecystectomy already have looser stools, fat sensitivity, or post-meal urgency. GLP-1s slow gastric emptying — which can either smooth those symptoms or amplify them. Titration may go more slowly.
- Pancreatitis remains a separate concern. Severe upper-belly pain radiating to the back with vomiting can signal pancreatitis, which is a separate emergency from anything gallbladder-related and stays on the watch list whether or not you still have a gallbladder.
What to ask your prescriber if you're post-cholecystectomy and considering a GLP-1:
- Does my surgical history change your starting dose or titration speed?
- Should I expect any change in my post-cholecystectomy GI symptoms when I start?
- What signs of pancreatitis should I watch for, separate from gallbladder concerns?
The headline: don't write yourself off. People without gallbladders take GLP-1s every day.
What tests will doctors run if gallbladder trouble is suspected?
The short answer: Workup typically starts with a focused history and physical exam, blood tests (liver enzymes, bilirubin, lipase to rule out pancreatitis), and an abdominal ultrasound — which NIDDK identifies as the best initial imaging test for gallstones. Depending on what those show, they may add a HIDA scan, MRI/MRCP, or ERCP to look at gallbladder function or the bile ducts.
You'll be a better patient if you walk in knowing the test menu.
| Test | What it's looking for | What it can change about your GLP-1 plan |
|---|---|---|
| Blood tests | Liver enzymes (ALT, AST), bilirubin (jaundice marker), lipase/amylase (pancreatitis markers), CBC (infection signs) | Elevated lipase shifts the conversation toward pancreatitis; elevated bilirubin/liver enzymes shift toward bile duct evaluation |
| Abdominal ultrasound | Gallstones, gallbladder wall thickening (sign of inflammation), bile duct dilation | Confirms or rules out stones; usually decides whether you go to surgical consult or watchful waiting |
| HIDA scan (cholescintigraphy) | How well your gallbladder empties (gallbladder ejection fraction) | Helpful when symptoms are classic but the ultrasound is normal; can identify functional gallbladder problems |
| CT scan | Complications, alternative causes; CT can miss many gallstones | Used for atypical presentations or to rule out other abdominal pathology |
| MRI / MRCP | Detailed look at bile ducts and pancreatic ducts | Used when bile duct stones are suspected; informs whether ERCP is needed |
| ERCP | Diagnose and remove stones in the common bile duct | A treatment as much as a test; usually done when imaging suggests duct involvement |
What to bring to the appointment:
- The exact name and dose of your GLP-1
- Date you started and date of your last dose increase
- How much weight you've lost and over what timeframe
- Pain location, timing relative to meals, what makes it better or worse
- Any history of gallstones or family history of gallbladder disease
- Any fever, chills, jaundice, urine or stool color changes, or persistent vomiting
A 5-minute prep document gets you a 10x better visit. The prescriber-question scripts in the next section are built for exactly that.
Are compounded GLP-1 medications different for gallbladder risk?
The short answer: We don't have FDA-label-quality gallbladder-event data for compounded GLP-1 products, so the FDA-approved drug rates in our table above shouldn't be extrapolated to compounded products. What we can say is that GLP-1 receptor activity slows gallbladder emptying and rapid weight loss changes bile chemistry — those mechanisms are well-established at the receptor level. The biggest practical safety differences in the compounded conversation are dose precision, prescriber follow-up, and pharmacy legitimacy.
This is where we have to be careful with language, because the FDA is.
Compounded GLP-1 medications are not FDA-approved and are not interchangeable with FDA-approved branded drugs. That's the FDA's own positioning, and it's important on a safety page. Compounded drugs don't undergo FDA premarket review for safety, effectiveness, or quality.
Where the gallbladder conversation intersects with the compounded conversation:
Don't extrapolate the rate table. The percentages in the FDA-approved drug table above came from the pivotal trials that supported each drug's approval. Those trials were not conducted on compounded products. We're not telling you compounded GLP-1s have higher or lower gallbladder rates than branded — we're telling you we don't have the data to tell you, and you shouldn't infer from one to the other.
The biology of receptor activity and rapid weight loss is well-established. GLP-1 receptor agonism slows gallbladder emptying. Rapid weight loss saturates bile with cholesterol. These are mechanisms that operate at the level of receptors and bile chemistry. The actionable point for compounded users is the same as for branded users: pace the weight loss, watch for symptoms, and have a prescriber you can reach.
The FDA has issued direct safety warnings about compounded GLP-1 products — separate from gallbladder risk. These include alerts about dosing errors (vials labeled in confusing ways, “units” vs. mL confusion leading to overdoses and hospitalizations), counterfeit and illegally marketed products, and salt-form concerns. None of these is a gallbladder issue per se. All of them are reasons to verify your dose and your pharmacy.
Compounded-specific safety checklist (FDA-supported items):
| Check | Why it matters |
|---|---|
| Is the pharmacy state-licensed and reputable? | FDA warns about counterfeit and illegally marketed GLP-1 products sold online |
| Is the concentration clearly labeled (mg/mL)? | Concentration confusion is a leading dosing-error cause |
| Are dosing instructions in mg, mL, and units? | “Units” vs. “mL” mix-ups have led to FDA-reported hospitalizations |
| What salt form is used? | FDA has flagged salt-form concerns; ask before assuming equivalence |
| Do you have a prescriber you can reach with symptoms? | This is the clinical-wraparound question that matters most for any safety conversation |
If the program selling you a compounded vial doesn't have a clear answer to all five questions, that's the program to leave.
What to ask your prescriber
The short answer: Bring four questions to any GLP-1 visit if gallbladder safety is on your mind: (1) Given my personal risk factors, what dose escalation pace do you recommend? (2) What symptoms should I report immediately, and how? (3) How fast can I reach you in an emergency, and through what channel? (4) Do you screen for prior gallstones or family history before starting? A program that can answer all four directly is the program you want.
We've split this into three short scripts depending on where you are.
If you haven't started yet
“Do I have any gallbladder risk factors that change your plan? Should I avoid rapid weight loss? What symptoms should make me call you? What should I do if I develop upper-right abdominal pain? Do you recommend any baseline labs based on my history?”
If you're already on a GLP-1 and you have new symptoms
“I'm taking [medication] at [dose]. I started/increased it on [date]. I've had [pain location] for [duration], with [associated symptoms]. I've lost [amount] in [time period]. Do I need labs, an ultrasound, a dose hold, a slower titration, or urgent evaluation?”
If you're using a compounded medication
“What exact dose am I taking in mg? How many units or mL should I inject? What is the concentration? Which pharmacy compounded it, and is it state-licensed? What should I do if I have right-upper-belly pain?”
Two telehealth models worth knowing about
If you're having red-flag symptoms, skip this section and seek urgent medical care first. The information below is for readers who aren't currently symptomatic and want a program with stronger clinical wraparound.
If you've gotten this far and decided you want a program with structured clinical oversight — which is exactly what a safety-conscious reader should want — there are two telehealth models worth knowing about. We're naming them because their structure fits the question this page is about. (Disclosure: we earn affiliate commissions from some of the providers we cover. This never changes our medical claims or which drugs we describe. Affiliate disclosure linked in the footer.)
| Ro | Sesame Care | |
|---|---|---|
| Best for | Readers who want a structured program with FDA-approved meds, an organized clinical workflow, and insurance handled | Readers who want to choose their own clinician and pay cash for branded medications |
| GLP-1 medications offered | FDA-approved GLP-1 options including Wegovy® pill, Wegovy® pen, Foundayo™ pill, Zepbound® KwikPen, Zepbound® pen, and Ozempic® | Wegovy® pill, Wegovy® pen, Zepbound® KwikPen, Zepbound® vial, Foundayo™, and Ozempic® |
| Pricing structure | Ro Body membership: $39 first month, then as low as $74/month with annual prepay (or $149/month month-to-month). GLP-1 medication cost is billed separately. | Per-visit clinician fees plus medication cost. Sesame says Costco members with Wegovy or Ozempic prescriptions can access Costco Pharmacy self-pay pricing; Wegovy pill currently available for as little as $149/month per Sesame. |
| Why it fits this page's reader | Ro Body is structured around ongoing prescriber follow-up; Ro publishes a free GLP-1 Insurance Coverage Checker and an insurance concierge that handles prior authorization. That's exactly the kind of clinical and administrative wraparound that matters when you're managing a known side effect like gallbladder risk. | Direct clinician choice + the broadest publicly listed branded formulary on a single platform; works well if you want a specific clinician you can build a relationship with. |
| Honest limitation | Ro doesn't carry compounded GLP-1s. If your priority is the lowest possible price and you've decided compounded is right for you, Ro isn't the fit — our matching quiz will route you to programs that are. | Costs more than telehealth-only flat-fee programs; you're paying for clinician choice and visit flexibility. |
| Check Ro eligibility & current GLP-1 options → | See Wegovy & Zepbound pricing on Sesame → |
The damaging admission, used as designed: Neither of these is the cheapest GLP-1 option you'll find online. If price is your dominant concern and you have lower personal risk factors, our matching quiz routes to the full price spectrum — including compounded programs that can be dramatically cheaper. We're naming Ro and Sesame here specifically because the question this page is about is clinical infrastructure for someone managing a known side effect. The right program depends on what you're solving for.
Frequently asked questions about GLP-1 gallbladder risk
Can GLP-1 medications cause gallbladder problems?▾
Which GLP-1 has the lowest gallbladder risk?▾
What does GLP-1 gallbladder pain feel like?▾
How long after starting a GLP-1 do gallbladder problems usually appear?▾
Should I get an ultrasound before starting a GLP-1?▾
Can I take Ozempic or Wegovy if I already have gallstones?▾
Does Mounjaro cause more gallbladder problems than Ozempic?▾
Can I take Wegovy or Zepbound after gallbladder removal?▾
Does Foundayo (orforglipron) carry the same gallbladder risk?▾
Should I stop my GLP-1 right now just to be safe?▾
Are compounded GLP-1s FDA-approved?▾
What doctor should I call for GLP-1 gallbladder symptoms?▾
What we actually verified for this article
We made a deliberate choice on a YMYL safety topic to be transparent about our sources. Here's the bench.
For this article, we verified:
- Cholelithiasis, cholecystitis, and cholecystectomy rates from each medication's FDA prescribing label, accessed via DailyMed on April 30, 2026
- Pooled relative-risk and absolute-risk numbers from the 2022 JAMA Internal Medicine meta-analysis (He et al.)
- 3-year real-world cohort data from the 2025 American College of Gastroenterology TriNetX cohort study
- Mechanism description against the 2025 mechanistic review in Therapeutic Advances in Endocrinology
- Manufacturer guidance on post-cholecystectomy use from Eli Lilly's published medical Q&A on tirzepatide
- Symptom triage against NIDDK gallstone guidance and FDA labels (Wegovy, Zepbound, Foundayo, Ozempic, Saxenda, Victoza, Trulicity)
- FDA safety communications on compounded GLP-1 products (dosing errors, counterfeit products, salt-form concerns)
- Baseline U.S. gallstone prevalence (10–15% of adults) from NIDDK
- Ro pricing and current FDA-approved GLP-1 options from Ro's public pricing page
- Sesame pricing and current GLP-1 program options from Sesame's public weight-loss program page
- Public clinical commentary from William Hinojosa, DO, via Norman Regional Health System (January 2026)
What we did not do: We did not perform original clinical research, conduct a meta-analysis, or examine patients. The editorial conclusions on this page are conclusions about how to interpret published evidence, not original medical findings.
Last verified:
Next scheduled re-verification: (or sooner if any FDA label revision affects the table)
Sources
FDA prescribing information (DailyMed / accessdata.fda.gov):
- Wegovy® (semaglutide injection and oral tablets) — DailyMed
- Ozempic® (semaglutide) — DailyMed
- Rybelsus® (oral semaglutide) — DailyMed
- Zepbound® (tirzepatide) — DailyMed
- Mounjaro® (tirzepatide) — DailyMed
- Foundayo™ (orforglipron) — DailyMed (FDA-approved April 2026)
- Saxenda® (liraglutide 3.0 mg) — DailyMed
- Victoza® (liraglutide 1.8 mg) — DailyMed
- Trulicity® (dulaglutide) — DailyMed
Peer-reviewed literature:
- He L, Wang J, Ping F, et al. Association of Glucagon-Like Peptide-1 Receptor Agonist Use With Risk of Gallbladder and Biliary Diseases: A Systematic Review and Meta-analysis of Randomized Clinical Trials. JAMA Internal Medicine. 2022;182(5):513-519. doi:10.1001/jamainternmed.2022.0338
- Ramírez-Mejía MM, Ponciano-Rodriguez G, Eslam M, Méndez-Sánchez N. GLP-1 receptor agonists and gallbladder disease risk: insights into molecular mechanisms and clinical implications. Therapeutic Advances in Endocrinology and Metabolism. 2025
- Tao C, et al. Glucagon-like peptide-1 receptor agonist-induced cholecystitis and cholelithiasis: a real-world pharmacovigilance analysis using the FAERS database. Frontiers in Pharmacology. 2025;16:1557691
- ACG 2025 Annual Meeting: Increased Risk of Gallbladder and Biliary Diseases With GLP-1 Receptor Agonists: A United States Collaborative Network Study. American Journal of Gastroenterology. 2025;120(10S2):S40
Government health guidance:
- National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK): Definition & Facts for Gallstones; Symptoms & Causes; Diagnosis; Treatment; Dieting & Gallstones
- FDA: Concerns with Unapproved GLP-1 Drugs Used for Weight Loss
- FDA: Alerts on Dosing Errors Associated with Compounded Injectable Semaglutide
Manufacturer medical statements:
- Eli Lilly Medical Information: Use of tirzepatide in patients with prior cholecystectomy
Clinical perspective:
- William Hinojosa, DO. Gallbladder Risks with GLP-1 Medications. Norman Regional Health System. January 2026
The bottom line, again
GLP-1 medications are real medicine with real benefits and a real, modest, manageable gallbladder side-effect signal. The honest version of this story is the one most worth knowing: pace the weight loss, know the symptoms, work with a prescriber who can respond fast, and the absolute risk to most people stays small. If something feels off, trust the triage card at the top of this page.
We built this guide because the existing pages on this topic either over-reassure you (“don't worry, it's rare”) or scare you (“Ozempic causes gallstones”) without giving you the actual numbers, the actual symptom playbook, or the actual next action. You deserve all three. That's the page we tried to make.
Still not sure which GLP-1 program is right for you?
Take our free 60-second matching quiz. We'll factor in your medical history, budget, insurance status, and medication preferences, then route you to programs that fit — across both branded FDA-approved and compounded options, with the kind of clinical wraparound that fits your situation.
Take the matching quiz →The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We earn affiliate commissions when readers connect with some of the providers we cover; this never changes our editorial recommendations or our medical descriptions. Read our Affiliate Disclosure. The information on this page is for educational purposes and is not medical advice. If you have symptoms that worry you, contact your prescriber or seek urgent care — don't rely on a webpage to make medical decisions for you.
Author: The RX Index Editorial Team · Last verified: · More GLP-1 guides →