Research Guide · Last verified: April 24, 2026
By The RX Index Research Team · Trial data verified against primary sources · Updated monthly and within 48 hours of major announcements · 15-min read
Triple Agonist vs Dual Agonist Weight Loss:
What the 2026 Data Actually Shows
The 100-word answer
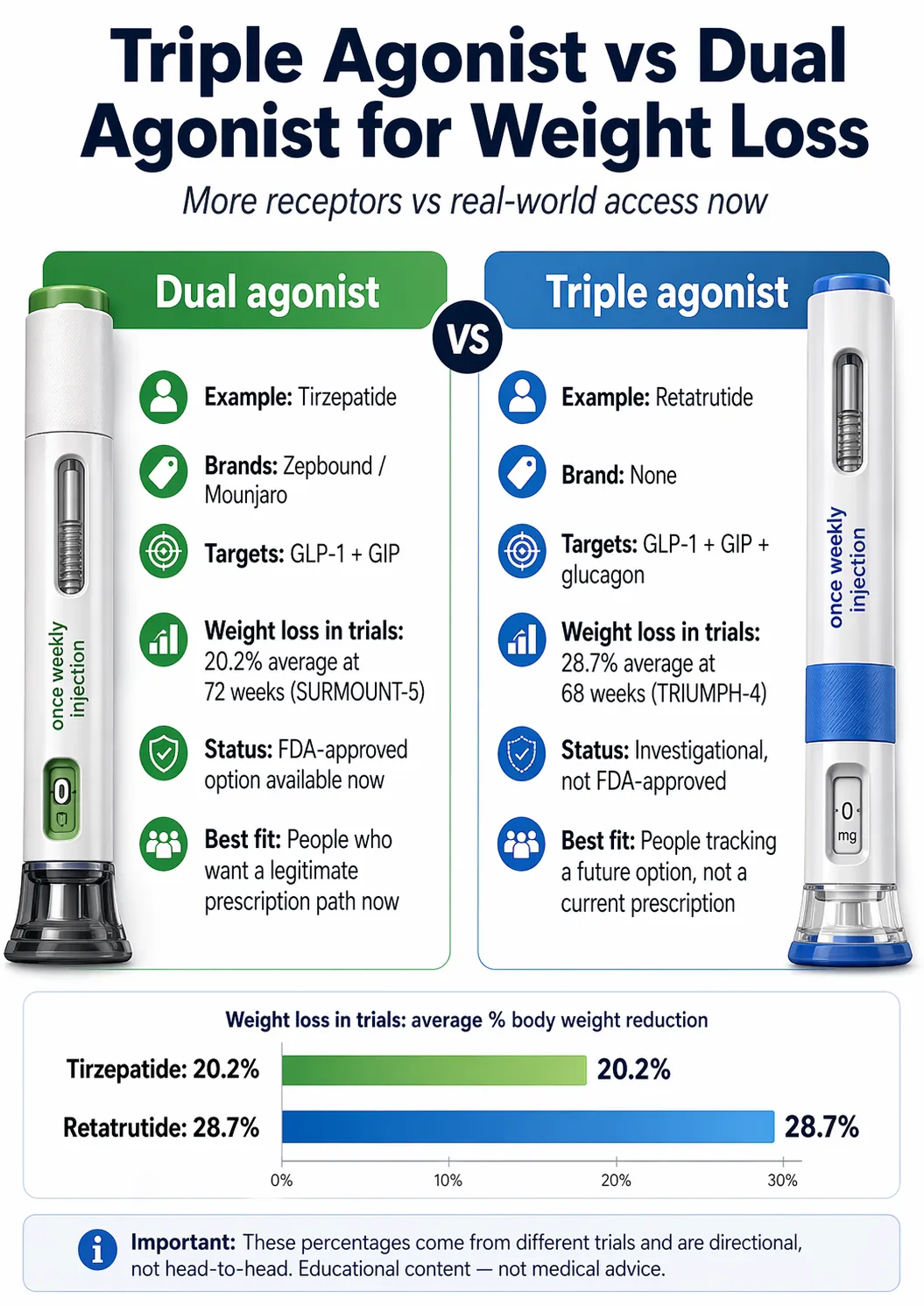
For triple agonist vs dual agonist weight loss, the 2026 answer is short. Triple agonists (one drug — investigational retatrutide) activate three hormone receptors and have produced up to 28.7% average weight loss at 68 weeks in Phase 3 (TRIUMPH-4, 12 mg). Dual agonists (FDA-approved tirzepatide / Zepbound / Mounjaro) activate two and produced up to 20.2% at 72 weeks in the head-to-head SURMOUNT-5 trial. Triple looks stronger on paper. But as of April 24, 2026, no triple agonist is FDA-approved, available by legitimate prescription, or accessible outside a clinical trial. The dual agonist is already at pharmacy.
The 30-second comparison
| Question | Dual agonist (tirzepatide) | Triple agonist (retatrutide) |
|---|---|---|
| Receptors targeted | GLP-1 + GIP | GLP-1 + GIP + glucagon |
| Brand name | Zepbound, Mounjaro | None — investigational |
| FDA-approved for weight loss? | Yes (Zepbound) | No |
| Best Phase 3 weight loss | 20.2% (SURMOUNT-5, 72 wk) | 28.7% (TRIUMPH-4, 68 wk) |
| Available by prescription today? | Yes | No (clinical trials only) |
| Real cash-pay price | $299–$449/mo (Zepbound self-pay) | None — no legitimate retail price |
| Best next step | Check FDA-approved access | Track legitimate trials only |
What we actually verified for this page
- Tirzepatide SURMOUNT-5 head-to-head (20.2% vs semaglutide 13.7% at 72 weeks): Aronne LJ et al., NEJM, May 2025; NCT05822830
- Zepbound labeled obesity data (up to 20.9% at 72 weeks vs 3.1% placebo): DailyMed Zepbound prescribing information
- Retatrutide TRIUMPH-4 topline (28.7% at 12 mg, 68 weeks; dysesthesia 20.9%): Eli Lilly press release, December 11, 2025
- Retatrutide TRANSCEND-T2D-1 topline (2.0% A1C, 16.8% weight loss, 40 weeks): Eli Lilly press release, March 19, 2026
- TRIUMPH-5 head-to-head retatrutide vs tirzepatide: ClinicalTrials.gov NCT06662383, ongoing
- Retatrutide FDA status: Investigational. Not in Drugs@FDA approvals as of April 24, 2026
- Foundayo (orforglipron) FDA approval: April 1, 2026; -11.1% weight change at 17.2 mg vs -2.1% placebo at 72 weeks (Trial 1)
- Zepbound self-pay pricing, Ro pricing, Medicare GLP-1 Bridge (CMS, updated April 6, 2026)
- FDA warnings on unapproved and compounded retatrutide products
We re-verify this page monthly — and within 48 hours of any major Lilly, Novo Nordisk, or FDA announcement.
What "triple agonist vs dual agonist weight loss" actually means
A dual agonist is a single weight-loss medication that activates two different gut-hormone receptors at once. A triple agonist activates three. The third receptor — glucagon — is the one driving the bigger trial numbers, because it works through a different mechanism than the first two. That's the entire concept. The rest is execution.
Why each generation of GLP-1 drug got bigger
Single agonist (one receptor — GLP-1)
Semaglutide is the GLP-1-only obesity benchmark. You know it as Wegovy (weight loss) and Ozempic (type 2 diabetes). In SURMOUNT-5 — the head-to-head Phase 3b trial published in the New England Journal of Medicine — semaglutide produced a 13.7% average weight loss at 72 weeks.
Dual agonist (two receptors — GLP-1 + GIP)
Tirzepatide adds a second receptor, GIP (glucose-dependent insulinotropic polypeptide). It's sold as Zepbound for weight loss and Mounjaro for diabetes. In SURMOUNT-5, tirzepatide produced 20.2% average weight loss at 72 weeks — beating semaglutide by 47% in relative terms. The Zepbound DailyMed label lists Study 1 results of up to 20.9% body-weight reduction at 72 weeks with the 15 mg dose.
Triple agonist (three receptors — GLP-1 + GIP + glucagon)
Retatrutide is the leading candidate. Eli Lilly's TRIUMPH-4 Phase 3 trial, released December 2025, reported 28.7% average weight loss at the 12 mg dose over 68 weeks in adults with obesity and knee osteoarthritis. That's the highest Phase 3 obesity-drug weight-loss number we verified for this comparison as of April 24, 2026.
The 2026 GLP-1 generations matrix
Trial data, FDA status, and real-world access — all in one place. Population, dose, and baseline weight differ across trials, so cross-trial percentages are directional, not direct head-to-head.
| Drug | Brand | Generation | Best reported weight loss | Trial / Duration | FDA status |
|---|---|---|---|---|---|
| Semaglutide | Wegovy / Ozempic | Single | 13.7% | SURMOUNT-5 / 72 wk | Approved |
| Semaglutide | Wegovy | Single | 14.9% (STEP 1) | STEP 1 / 68 wk | Approved |
| Liraglutide | Saxenda | Single (daily) | ~8% | SCALE / 56 wk | Approved |
| Orforglipron | Foundayo | Single (oral pill) | 11.1% at 17.2 mg | ATTAIN-1 / 72 wk | Approved April 1, 2026 |
| Tirzepatide | Zepbound / Mounjaro | Dual (GLP-1 + GIP) | 20.2% | SURMOUNT-5 / 72 wk | Approved |
| Tirzepatide | Zepbound | Dual | 20.9% (15 mg) | SURMOUNT-1 / 72 wk | Approved |
| Survodutide | None | Dual (GLP-1 + glucagon) | ~19% (Phase 2) | SYNCHRONIZE / — | Investigational |
| CagriSema | None | Dual (GLP-1 + amylin) | ~22.7% | REDEFINE 1 / 68 wk | Investigational; FDA filing Dec 2025 |
| Retatrutide | None | Triple (GLP-1 + GIP + glucagon) | 28.7% (12 mg) | TRIUMPH-4 / 68 wk | Investigational |
| Retatrutide | None | Triple | 16.8% (T2D, 12 mg) | TRANSCEND-T2D-1 / 40 wk | Investigational |
| UBT251 | None | Triple (Phase 2, China) | 19.7% | NCT06747572 / 24 wk | Investigational |
What these numbers mean in actual pounds
| Drug | Trial avg. loss | Pounds lost from 250 lb |
|---|---|---|
| Semaglutide (best) | 13.7% | ~34 lb |
| Tirzepatide (best) | 20.2% | ~50 lb |
| Retatrutide (best) | 28.7% | ~72 lb |
The retatrutide figure is close to what bariatric surgery typically produces, achieved with a weekly injection. That's the headline pulling people into this search. It's also the headline driving a wave of unsafe gray-market products — which we cover below.
Why these numbers aren't a clean apples-to-apples comparison
- No published head-to-head trial yet. TRIUMPH-5 (NCT06662383) is the direct comparison. Until it reports, the 8-point gap is suggestive, not proven.
- Different populations. TRIUMPH-4 enrolled adults with obesity + knee osteoarthritis (mean BMI 40.4). SURMOUNT-5 enrolled adults with obesity without diabetes and a comorbidity.
- Different durations. TRIUMPH-4 ran 68 weeks; SURMOUNT-5 ran 72.
- Different responder rates. In TRIUMPH-4, nearly 60% on 12 mg lost ≥25% body weight. In SURMOUNT-5, 31.6% on tirzepatide hit ≥25%. Both are remarkable.
Why a triple agonist produces more weight loss than a dual agonist
Both drug classes reduce how much you eat. Retatrutide adds a third pathway — glucagon receptor activation — that is being studied for its role in energy expenditure and fat oxidation. The simplest mental model: dual agonists work primarily on the intake side; triple agonists pull on the intake side and add an output-side mechanism.
What each receptor contributes
- GLP-1 (glucagon-like peptide-1). Slows gastric emptying. Sends "you're full" signals to your brain. This is the mechanism Ozempic and Wegovy work through.
- GIP (glucose-dependent insulinotropic polypeptide). Enhances insulin secretion when blood sugar rises. Adds appetite reduction beyond GLP-1 alone.
- Glucagon (the third receptor). Drives energy expenditure and fat oxidation. Combining it with GLP-1 + GIP is designed to balance the metabolic effects so the energy-expenditure benefit can come through.
| Claim | Evidence status |
|---|---|
| Receptor targets (which drug binds which receptor) | Verified in published structural biology and Phase 1–3 trials |
| Appetite/intake reduction | Strongly supported by clinical and mechanistic data |
| Energy-expenditure contribution from glucagon receptor activation | Mechanistic / proposed; supported in preclinical and early clinical work |
| Fat-vs-lean-mass body-composition advantage | Not settled; awaits detailed Phase 3 body-composition publication |
| Clinical superiority of retatrutide over tirzepatide | Pending TRIUMPH-5 head-to-head readout |
Can you actually get a triple agonist right now?
The legitimate path: a clinical trial
If you genuinely want to try retatrutide before approval, the only legal route is enrolling in one of Lilly's ongoing Phase 3 studies on ClinicalTrials.gov. What that actually looks like:
- You won't be guaranteed retatrutide — most trials randomize to drug or placebo.
- You must meet specific inclusion/exclusion criteria — BMI thresholds, medical history, no recent GLP-1 use.
- You commit to multiple visits over 40–80 weeks.
- You get the medication at no cost during the trial — but can't necessarily continue afterward.
The unsafe path: "research peptide" retatrutide
Search "buy retatrutide" and you'll find dozens of websites selling vials labeled "research use only" or "not for human consumption" — paired with detailed dosing protocols clearly written for human use. The FDA states that unapproved GLP-1 products do not undergo FDA review for safety, effectiveness, or quality. There's no FDA-approved retatrutide product, no labeling, no dose verification, and no medical safety net.
Why "compounded retatrutide" is different from compounded semaglutide or tirzepatide
FDA warning letters state that compounded retatrutide products fail 503A and 503B compounding conditions because retatrutide is not in an applicable USP/NF monograph, is not a component of an FDA-approved human drug, does not appear on the 503A or 503B bulks lists, and is not used to compound a drug on the shortage list. There is no legal pathway for compounded retatrutide right now. If a telehealth provider is offering "compounded retatrutide," walk away.
If you landed here because you thought retatrutide was something you could start today, don't guess on gray-market products.
Compare the FDA-approved GLP-1 paths that actually exist for your situation — most people qualify for more than they think.
Find My Legitimate GLP-1 Path (free 60-second quiz) →When will retatrutide be FDA approved?
A late-2026 obesity NDA filing has been reported as Eli Lilly's working plan. Until Lilly actually submits and the FDA accepts the application, there is no review timeline. A realistic public-availability window — assuming the remaining TRIUMPH and TRANSCEND trials read out as expected — is late 2027 or 2028. There is no guarantee.
The retatrutide trial tracker (verified April 24, 2026)
| Trial | NCT | Population | Status |
|---|---|---|---|
| TRIUMPH-4 | NCT05931367 | Obesity + knee osteoarthritis | Reported December 2025 ✓ |
| TRANSCEND-T2D-1 | — | Type 2 diabetes monotherapy | Reported March 2026 ✓ |
| TRIUMPH-1 | — | General obesity (largest trial) | Expected 2026 |
| TRIUMPH-2 | — | Type 2 diabetes + obesity/overweight | Expected 2026 |
| TRIUMPH-3 | — | Class II/III obesity + CVD | Expected 2026 |
| TRIUMPH-5 (head-to-head vs tirzepatide) | NCT06662383 | Obesity | Expected 2026 |
| TRIUMPH-6 | NCT06859268 | Maintenance of weight reduction | Expected 2026–2027 |
| TRIUMPH-7 | — | Obesity + chronic low back pain | Expected 2026–2027 |
| Lilly NDA submission (obesity) | — | — | Reported plan: late 2026 |
| Potential FDA approval | — | — | Late 2027–2028 (not guaranteed) |
Bottom line: don't plan your weight loss around retatrutide being available before 2028. If you're going to act on weight, act on what's available.
Should you wait for retatrutide or start a dual agonist now?
For most people, waiting doesn't make sense. A dual agonist started today produces an average of 15–22% weight loss over 72 weeks — real progress already banked by the time retatrutide could reach pharmacy shelves (most likely 2027 or 2028 at the earliest).
4 scenarios where starting today is right
- BMI ≥30, or BMI ≥27 with a weight-related comorbidity. Hypertension, dyslipidemia, OSA, CVD. Every month of carrying that weight has measurable health cost.
- You've never tried a GLP-1. You haven't seen what your body does on a dual agonist. You may respond at the high end (~25%+).
- You have a concrete deadline. Wedding, surgery, fertility window. Retatrutide won't land in time.
- Your insurance covers Zepbound, or you're Medicare-eligible. The cost gap between starting and waiting can be enormous.
3 scenarios where waiting might make sense
- You're already on tirzepatide and close to your goal. No urgency. Stay the course.
- You've genuinely tried tirzepatide and plateaued at an unsatisfactory weight. After optimizing dose and adherence, retatrutide tracking becomes more reasonable.
- You're medically able to wait 18–24 months with no health pressure. No comorbidity clock, no quality-of-life cost.
The damaging admission
Tirzepatide does not produce 28.7% average weight loss. If your single goal is the maximum possible weight-loss number on paper and you can afford to wait two years with zero health cost, retatrutide may eventually deliver more. But for most people — anyone with a comorbidity, anyone with a deadline, anyone who's never tried a GLP-1 — the best FDA-approved option today beats a better one that isn't here yet.

The fastest legitimate path to FDA-approved Zepbound
We compared the major branded telehealth options and Ro is our primary recommendation for FDA-approved tirzepatide weight-loss access. Ro carries Zepbound (tirzepatide for weight loss) and Foundayo (orforglipron, the new oral GLP-1 approved April 2026), matches LillyDirect / NovoCare / TrumpRx pricing, includes an insurance concierge that handles prior authorization paperwork, and offers a free GLP-1 Insurance Coverage Checker so you can see what your plan covers before you commit.
Note on what Ro carries: Ro does not currently offer Mounjaro. Mounjaro is the diabetes brand of tirzepatide; Zepbound is the weight-loss brand. If you want tirzepatide for weight loss, Zepbound through Ro is the cleaner path.
Get started for $39 the first month, then as low as $74/month with annual plan paid upfront (or $149/month standard). Medication cost is separate.
We may earn a commission from these referrals. Our recommendations are based on verified evidence and reader fit, not affiliate payout.
Triple vs dual agonist side effects compared
Both classes cause the same gastrointestinal side effects — nausea, diarrhea, vomiting, constipation — and both are dose-dependent and worst during dose escalation. Triple agonists in trials reported somewhat higher GI rates. Retatrutide also showed a side effect not seen at meaningful rates with tirzepatide: dysesthesia (abnormal skin sensations like tingling or burning) in 20.9% of participants at the highest dose.
The TRIUMPH-4 retatrutide adverse-event table
| Adverse event | Retrat. 9 mg | Retrat. 12 mg | Placebo |
|---|---|---|---|
| Nausea | 38.1% | 43.2% | 10.7% |
| Diarrhea | 34.7% | 33.1% | 13.4% |
| Constipation | 21.8% | 25.0% | 8.7% |
| Vomiting | 20.4% | 20.9% | 0.0% |
| Decreased appetite | 19.0% | 18.2% | 9.4% |
| Dysesthesia | 8.8% | 20.9% | 0.7% |
| Discontinuation due to adverse events | 12.2% | 18.2% | 4.0% |
Source: Eli Lilly TRIUMPH-4 topline data, December 2025. Full peer-reviewed dataset expected at a future medical meeting.
What dysesthesia actually is, and whether you should care
Dysesthesia is a medical term for abnormal or unpleasant sensations — tingling, burning, "pins and needles," or altered perception of touch. Three things to understand:
- Dose-dependent: 0.7% at placebo, 8.8% at 9 mg, 20.9% at 12 mg.
- Generally mild: Most cases didn't lead to people stopping the drug.
- Not reported in Phase 2: That's why it surprised analysts when it appeared in Phase 3. The FDA and Lilly will both be watching for it in TRIUMPH-1, TRIUMPH-3, and TRIUMPH-5.
Worth noting: Zepbound's DailyMed label does report dysesthesia, but at much lower rates — roughly 0.2–0.4% across doses vs 0.1% placebo, compared to 20.9% at retatrutide's highest dose.
Boxed warnings — what applies to which drugs
The FDA-approved GLP-1 drugs on this page — Zepbound and Foundayo — carry boxed warnings for thyroid C-cell tumors. Anyone with a personal or family history of medullary thyroid carcinoma (MTC) or multiple endocrine neoplasia syndrome type 2 (MEN 2) should not take these drugs. Retatrutide is investigational and has no final FDA label yet; its eventual boxed-warning language can't be stated until approval. Other class-level cautions seen across approved GLP-1s include pancreatitis, gallbladder disease, kidney injury, and possible worsening of diabetic retinopathy. Discuss your full medical history with a prescriber.
Cost comparison: what you'll actually pay
Retatrutide has no legitimate cash price because it's not a legitimate prescription product. Tirzepatide does. Here's what current dual-agonist access actually costs.
| Zepbound dose | Self-pay price | Material caveat |
|---|---|---|
| 2.5 mg vial | $299/month | Starting / titration dose |
| 5 mg vial | $399/month | Subject to Self Pay Journey Program terms |
| 7.5 mg, 10 mg, 12.5 mg, 15 mg | $449/month | Self Pay Journey Program terms; regular price applies if refill not within 45 days |
Verify current pricing on the official Zepbound savings page before purchasing — Lilly updates terms periodically. Eli Lilly Self Pay Journey Program, April 2026.
Want to know whether your insurance covers Zepbound — without paying anything to find out?
Ro's insurance concierge handles the prior authorization paperwork — which can be the single biggest barrier to actually getting the prescription approved. The coverage checker is free.
Check coverage with Ro's free GLP-1 Insurance Coverage Checker →Takes about 60 seconds. No payment required to check. If you're on Medicare, see the Bridge program details below before assuming self-pay is your only path.
Can Medicare cover Zepbound, Wegovy, or Foundayo for weight loss in 2026?
Yes — for the first time, through a temporary CMS program. The Medicare GLP-1 Bridge begins July 1, 2026 and provides eligible Medicare Part D beneficiaries with access to certain weight-loss GLP-1s at a flat $50/month copay, extended through December 31, 2027.
| Drug | Covered under Bridge? |
|---|---|
| All formulations of Foundayo (orforglipron) | Yes |
| All formulations of Wegovy (semaglutide) | Yes |
| Zepbound KwikPen formulation | Yes |
| Zepbound single-dose vial and single-dose pen | No |
| Mounjaro, Ozempic, Saxenda, Rybelsus | No |
| Any compounded GLP-1 | No |
Copay: flat $50/month. Your Bridge copay does not count toward your Part D True Out-of-Pocket (TrOOP) costs or the $2,100 annual out-of-pocket cap — it runs alongside your existing Part D coverage, not through it. Prior authorization is required through a CMS central processor. If you're 65+ on Medicare and have been priced out of GLP-1s for years, this is the door opening.
Can you switch from a dual agonist to a triple agonist later?
Not yet — no triple agonist is FDA-approved. There is no established switching protocol or dose conversion from tirzepatide to retatrutide because retatrutide has no FDA label. Once retatrutide is approved, switching guidance will come from the FDA label and your prescriber.
This section exists because it's the question that quietly stops people from starting tirzepatide today. The fear is "if I start now, am I locked in?"
You're not. Every pound you lose on tirzepatide is a pound that's already gone — and a pound your starting weight on retatrutide doesn't have to deal with.
| Item | Status |
|---|---|
| Retatrutide is investigational | Known |
| TRIUMPH trial titration starts at 2 mg once weekly, escalating every 4 weeks | Known (trial design only) |
| FDA-approved starting dose after approval | Not known yet |
| Standard switching protocol from tirzepatide to retatrutide | Not known yet |
| Dose conversion between tirzepatide and retatrutide | Not known yet |
What about CagriSema, Foundayo, and the other next-gen drugs?
Foundayo (orforglipron, Eli Lilly) — FDA-approved April 1, 2026
Lilly's once-daily oral GLP-1 receptor agonist for chronic weight management in eligible adults. In Trial 1 (without diabetes), the 17.2 mg dose produced -11.1% mean body-weight change at 72 weeks vs -2.1% placebo. In Trial 2 (with type 2 diabetes), the 17.2 mg dose produced -9.6% vs -2.5% placebo. Foundayo can be taken at any time of day without food or water restrictions. If you're needle-averse, this changes the dual-vs-triple math significantly. Ro carries Foundayo.
CagriSema (Novo Nordisk) — Investigational
A dual co-formulation of semaglutide + cagrilintide (an amylin analog — not GIP or glucagon). REDEFINE 1 Phase 3 reported approximately 22.7% weight loss at 68 weeks — between tirzepatide and retatrutide on the data table. Novo Nordisk filed for FDA approval in December 2025; FDA review is expected in 2026.
Survodutide (Boehringer Ingelheim / Zealand Pharma) — Investigational
A dual GLP-1/glucagon agonist — different from tirzepatide's GLP-1/GIP combination. Phase 3. Notable for early signals of strong liver-fat reduction, which matters for MASLD/NASH patients.
By 2028, the FDA-approved obesity menu may include semaglutide (Wegovy), tirzepatide (Zepbound), orforglipron (Foundayo), retatrutide, CagriSema, and possibly survodutide. The "best" drug for you will depend less on the topline weight-loss number and more on how you tolerate it, how it fits your life (oral vs injection), and what your insurance covers.
What real searchers are actually trying to figure out
"should I use Reta or Tirzepatide… reason to go with Tirzepatide, since it is already being used and approved."
"Tirz does seem to have stronger appetite suppression while reta is giving a significant boost in energy…"
"Reta is kind of Tirz plus additional benefits?"
Public forum comments are included to show the questions real people ask. They are not clinical evidence, not medical advice, and not proof of typical results. Individual experiences vary. Anything from a "research peptide" community involves products that are not the same as what's in legitimate clinical trials.
Your path forward today
Path 1: You want to start on something that exists today
The strongest legitimate option is an FDA-approved dual agonist (tirzepatide / Zepbound) or oral option (Foundayo) through a telehealth provider that handles prescription, insurance, and pharmacy logistics. Our primary recommendation is Ro — see CTA above. Sesame Care is the secondary option for maximum prescriber choice.
If you're needle-averse, consider Foundayo — the FDA-approved oral GLP-1 pill (also available through Ro). Lower peak weight loss than Zepbound (~11% vs ~20%), but it's a pill taken any time of day. If you're on Medicare, see the GLP-1 Bridge section above — $50/month copay starting July 1, 2026.
Path 2: You specifically want retatrutide and you're willing to wait
The legitimate path is enrollment in a TRIUMPH or TRANSCEND clinical trial. Search "retatrutide" on ClinicalTrials.gov to find currently recruiting studies. Be realistic about randomization risk — you may be assigned to placebo or to a different dose than you expect. Whatever you do, don't buy "research retatrutide" online.
Path 3: You're not sure and want a personalized recommendation
Our matching quiz takes about a minute, is free, and requires no payment information. It matches based on your weight, goals, insurance, and timeline.
Still not sure which GLP-1 program is right for you?
Get a personalized action plan based on your weight, your goals, your insurance situation, and your timeline. Free, 60 seconds, no payment required.
Take our free 60-second matching quiz →Frequently asked questions
Is a triple agonist better than a dual agonist for weight loss?
In Phase 3 trials, the leading triple agonist (retatrutide) produced about 6–9 percentage points more average weight loss than the leading dual agonist (tirzepatide) — 28.7% vs 20.2%. But better depends on more than the topline number. FDA approval status, safety data, tolerability, cost, and access all matter. Today, the dual agonist is approved and the triple agonist is not.
Is tirzepatide a triple agonist or a dual agonist?
Tirzepatide is a dual agonist. It activates two hormone receptors — GLP-1 and GIP. It is sold as Zepbound for weight loss and Mounjaro for type 2 diabetes.
Is there an FDA-approved triple agonist for weight loss?
No. As of April 2026, no triple agonist is FDA-approved for any indication. Retatrutide is investigational and in Phase 3 trials. Realistic public availability is late 2027 or 2028, with no guarantee.
Is TRIUMPH-2 or TRIUMPH-5 the head-to-head retatrutide vs tirzepatide trial?
TRIUMPH-5 is the head-to-head trial directly comparing retatrutide vs tirzepatide in adults with obesity (NCT06662383). TRIUMPH-2 is a separate placebo-controlled retatrutide trial in people with type 2 diabetes and obesity or overweight. Both are ongoing in 2026.
How much weight did people lose on retatrutide in trials?
In Eli Lilly's TRIUMPH-4 Phase 3 trial, participants on the 12 mg retatrutide dose lost an average of 28.7% of their body weight at 68 weeks. The 9 mg dose produced 26.4% weight loss. Placebo produced 2.1%. Nearly 60% on the 12 mg dose lost ≥25% of their body weight.
How much weight did people lose on tirzepatide in trials?
In the head-to-head SURMOUNT-5 Phase 3b trial, tirzepatide produced 20.2% average weight loss at 72 weeks vs 13.7% for semaglutide. The Zepbound prescribing information lists Study 1 weight loss of up to 20.9% at the 15 mg dose at 72 weeks vs 3.1% for placebo.
Can my doctor prescribe retatrutide right now?
No. Retatrutide is not FDA-approved and cannot be prescribed as a normal weight-loss medication. It is legally available only to participants in active Lilly clinical trials.
Can I buy retatrutide online?
Not legally as a weight-loss medication. Websites selling retatrutide — typically labeled 'research use only' or 'not for human consumption' — are selling unapproved products that have not undergone FDA review for safety, effectiveness, or quality.
Can Medicare cover Zepbound, Wegovy, or Foundayo for weight loss in 2026?
Yes — through the new Medicare GLP-1 Bridge, starting July 1, 2026 and extended through December 31, 2027. Eligible Medicare Part D beneficiaries can access all formulations of Foundayo, all formulations of Wegovy, and the Zepbound KwikPen at a flat $50/month copay. The Zepbound single-dose vial, Mounjaro, Ozempic, and Saxenda are not covered under the Bridge.
Does Ro carry Mounjaro?
No. Ro does not currently offer Mounjaro. Ro carries Zepbound (tirzepatide for weight loss) and Foundayo. If you specifically need Mounjaro for type 2 diabetes, you'll want a primary care provider rather than a Ro Body subscription.
When will retatrutide be FDA approved?
Eli Lilly has reportedly planned a New Drug Application filing for late 2026 after the remaining Phase 3 readouts. A realistic public availability window is late 2027 or 2028, assuming the trials complete as planned and the FDA review proceeds without a hold. There is no guarantee.
What is dysesthesia, and should I be worried about it?
Dysesthesia is an abnormal skin sensation — tingling, burning, 'pins and needles,' or altered touch. In TRIUMPH-4, it occurred in 20.9% of participants on the 12 mg retatrutide dose vs 0.7% on placebo. It was generally mild and rarely caused people to stop the drug. The Zepbound label also reports dysesthesia, but at much lower rates (roughly 0.2–0.4% across doses vs 0.1% placebo).
Should I wait for retatrutide or start Zepbound now?
For most people with a comorbidity, a deadline, or no prior GLP-1 experience, starting an FDA-approved dual agonist today is the stronger call. Waiting two years means carrying that weight through that period. If you are already on tirzepatide and doing well, there is no urgency to switch. If you have genuinely failed on tirzepatide, retatrutide tracking becomes more reasonable.
Is Wegovy a triple agonist?
No. Wegovy is semaglutide — a single GLP-1 agonist. It activates only one hormone receptor.
Are there any other triple agonists besides retatrutide?
Novo Nordisk's UBT251 is in Phase 2 — it reported 19.7% weight loss at 24 weeks in a Chinese trial. Other triple agonists, including HM15211 and SAR441255, are in earlier development. None are close to FDA review.
What's the difference between CagriSema and a triple agonist?
CagriSema is a dual co-formulation of semaglutide (GLP-1) and cagrilintide (an amylin analog) — not a triple agonist. It targets two receptor pathways: GLP-1 and amylin. Retatrutide targets three: GLP-1, GIP, and glucagon. CagriSema's reported Phase 3 weight loss is approximately 22.7% — between tirzepatide and retatrutide. Novo filed for FDA approval in December 2025.
Is Zepbound a dual agonist?
Yes. Zepbound is the brand name for tirzepatide, a dual agonist that targets GLP-1 and GIP receptors. Mounjaro is the same molecule, branded for type 2 diabetes.
Sources
- Aronne LJ et al. Tirzepatide as Compared with Semaglutide for the Treatment of Obesity. NEJM 2025; SURMOUNT-5 (NCT05822830).
- Eli Lilly press release: "Lilly's triple agonist, retatrutide, delivered weight loss of up to an average of 71.2 lbs along with substantial relief from osteoarthritis pain in first successful Phase 3 trial." December 11, 2025.
- Eli Lilly press release: "Lilly's triple agonist, retatrutide, demonstrated significant reductions in A1C and weight in first Phase 3 trial for treatment of type 2 diabetes." March 19, 2026 (TRANSCEND-T2D-1).
- Jastreboff AM, Kaplan LM et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. NEJM 2023;389:514-526.
- DailyMed Zepbound (tirzepatide injection) prescribing information.
- ClinicalTrials.gov: TRIUMPH-4 (NCT05931367), TRIUMPH-5 (NCT06662383), TRIUMPH-6 (NCT06859268).
- Giblin J et al. Retatrutide for the treatment of obesity, obstructive sleep apnea and knee osteoarthritis: Rationale and design of the TRIUMPH registrational clinical trials. Diabetes, Obesity and Metabolism 2026;28(1):83-93.
- Eli Lilly press release: "FDA approves Lilly's Foundayo (orforglipron)." April 1, 2026.
- FDA Drugs@FDA database (verified April 24, 2026).
- FDA consumer alerts on unapproved GLP-1 products; FDA Warning Letter: GenLabMeds, September 9, 2025 (compounded retatrutide).
- Novo Nordisk press release: "Novo Nordisk files for FDA approval of CagriSema." December 2025.
- Novo Nordisk announcement on UBT251 Phase 2 results, February 26, 2026.
- CMS — Medicare GLP-1 Bridge program page (cms.gov), updated April 6, 2026.
- Eli Lilly Zepbound Self Pay Journey Program — official savings page.
About this page
The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We don't manufacture, prescribe, or sell medications. We compare what's currently available, verify pricing and access details against primary sources, and update high-impact pages monthly — or within 48 hours of any major FDA or manufacturer announcement. This page was researched and written by The RX Index Research Team. It is educational, not medical advice. Prescription decisions belong with a licensed clinician.
Last verified: April 24, 2026 by The RX Index Research Team. Trial data, FDA status, Medicare coverage, and provider pricing change. We re-check this page monthly and within 48 hours of any major announcement.
This page is for educational purposes only. It is not medical, legal, or insurance advice. Prescription decisions belong with a licensed clinician.