Last verified: April 21, 2026 · FDA status checked · Pricing verified · Phase 3 data confirmed
Comparison Guide · April 21, 2026
Retatrutide vs Ozempic: Which Makes Sense Right Now?
Published:
By The RX Index Research Team — a pricing intelligence and comparison resource for GLP-1 telehealth providers
The short answer before you scroll
Retatrutide vs Ozempic is a future-vs-now decision, not a head-to-head fight. Retatrutide has the bigger weight-loss headline — 28.7% average body-weight reduction at 68 weeks in Eli Lilly’s Phase 3 TRIUMPH-4 trial. Ozempic (semaglutide) is FDA-approved for adults with type 2 diabetes, carries labeled cardiovascular and kidney benefits, and has a real, current price you can look up. Retatrutide is investigational.
If you need treatment now, the practical answer is almost always “start an approved GLP-1.” For most people that means Ozempic or Mounjaro if the goal is type 2 diabetes, and Wegovy or Zepbound if the goal is weight loss. Retatrutide is not a treatment decision you can make today — Lilly has not announced an FDA filing date, approval date, or launch date.
 (sponsored affiliate link, opens in a new tab)
(sponsored affiliate link, opens in a new tab)Retatrutide wins on future weight-loss potential. Ozempic wins on the real-world decision today.
Snapshot: retatrutide vs Ozempic at a glance
| Factor | Retatrutide (LY3437943) | Ozempic (semaglutide) |
|---|---|---|
| Maker | Eli Lilly | Novo Nordisk |
| Mechanism | Triple agonist: GLP-1 + GIP + glucagon | Single agonist: GLP-1 |
| FDA-approved? | No. Investigational. | Yes. Approved Dec 2017. |
| Approved uses | None yet | Type 2 diabetes glycemic control; MACE reduction in T2D + CVD; kidney-risk reduction in T2D + CKD |
| Best public weight-loss result | 28.7% at 68 weeks (TRIUMPH-4, 12 mg) | 7.1–14.1 lb across A1C studies (Ozempic is not FDA-approved for weight loss; Wegovy at 2.4 mg produced ~14.9% in STEP 1) |
| Can you legally get it in 2026? | Clinical trials only | Yes, by prescription |
| Self-pay price | No legitimate public price | $199/mo first 2 months (intro, through June 30, 2026), then $349/mo (0.25/0.5/1 mg) or $499/mo (2 mg) via NovoCare |
| With commercial insurance | Not applicable | As low as $25/mo for covered patients (Novo savings offer; eligibility restrictions apply) |
Sources: FDA Ozempic label (2025 revision); Novo Nordisk cost-and-coverage page (verified April 2026); Eli Lilly TRIUMPH-4 investor release (Dec 11, 2025); Lilly retatrutide FAQ.
The quick verdict
The one-line answer: Retatrutide probably wins on raw future weight-loss potential. Ozempic wins on everything that matters to a real decision today — FDA approval, legal availability, defined dosing, official pricing, and labeled cardiovascular and kidney benefits in adults with type 2 diabetes.
Ozempic doesn’t have the biggest weight-loss headline number, and it never will. If you only care about the top-line weight-loss figure, retatrutide looks stronger on paper. But Ozempic isn’t supposed to win on that metric. Ozempic is a diabetes drug that also causes weight loss. The FDA-approved weight-loss sister of Ozempic is Wegovy — same molecule, different dose, different label.
What Ozempic wins on is decision quality. You can get it. You know the dose. You know the price. You know what the FDA says it does. Every one of those sentences is impossible to write about retatrutide in 2026.
Not sure whether to keep waiting for retatrutide, start an approved GLP-1, or move to a different approved drug entirely?
Takes about 60 seconds — no sign-up to see the match.
Is this even the right comparison?
Not perfectly. Retatrutide is being developed primarily for obesity and type 2 diabetes. Ozempic is FDA-approved for type 2 diabetes, and Novo Nordisk’s own patient site tells readers that Ozempic is not a weight-loss drug. For a cleaner obesity comparison, Wegovy (semaglutide 2.4 mg) is the benchmark most clinicians would use. People still search “retatrutide vs Ozempic” because Ozempic is the famous brand — and that’s the search we’re writing for.
Ozempic vs Wegovy: the label difference that changes the comparison
Both Ozempic and Wegovy contain semaglutide. The key difference is dose and labeled use:
- Ozempic is approved at doses up to 2 mg weekly for glycemic control in adults with type 2 diabetes, MACE reduction in T2D with established CVD, and kidney-risk reduction in T2D with chronic kidney disease.
- Wegovy is approved at doses up to 2.4 mg weekly for chronic weight management in adults and pediatric patients 12 and older with obesity, and in adults who are overweight with a weight-related condition. Wegovy also has a cardiovascular outcome indication for adults with obesity and established CVD.
This page is our brand-level real-world decision resource — Ozempic specifically, in context. Our separate retatrutide vs semaglutide guide covers the molecule-level comparison if that’s what you need.
The full retatrutide vs Ozempic comparison
Retatrutide is a Phase 3 investigational triple-receptor agonist producing substantially more weight loss than Ozempic has shown in diabetes trials — and roughly double what Wegovy produced in STEP 1. But retatrutide isn’t FDA-approved, isn’t legally sold to the public, and has a harsher side-effect profile at peak dose. Ozempic is an FDA-approved GLP-1 agonist with a mature safety profile, official pricing, and three labeled indications retatrutide’s trials haven’t matched yet.
The RX Index Reality Matrix — Retatrutide vs Ozempic (verified April 2026)
| Factor | Retatrutide (LY3437943) | Ozempic (semaglutide) |
|---|---|---|
| Class | Triple agonist (GLP-1 + GIP + glucagon) | GLP-1 receptor agonist |
| FDA status | Investigational (not approved for any indication) | Approved Dec 2017 |
| FDA-approved uses | None | Type 2 diabetes; MACE reduction in T2D + CVD; kidney-risk reduction in T2D + CKD |
| Pivotal weight-loss readout | TRIUMPH-4: 28.7% mean weight loss at 68 weeks (12 mg, obesity + knee osteoarthritis cohort) | Adults lost 7.1–14.1 lb across A1C studies; Ozempic is not FDA-approved for weight loss. Fairer comparison: Wegovy (2.4 mg), STEP 1: ~14.9% at 68 weeks. |
| Diabetes A1C data | TRANSCEND-T2D-1: A1C reduction 1.7–2.0 pts; weight loss up to 16.8% at 40 weeks | SUSTAIN program: ~1.5–2.0 pt A1C reduction at maximum approved dose |
| Cardiovascular outcomes | CV outcomes study ongoing. No labeled CV benefit. | SUSTAIN-6 → MACE reduction in T2D + CVD. SELECT trial (Wegovy 2.4 mg) → MACE reduction in obesity + CVD. Labeled. |
| Kidney outcomes | Kidney endpoints under study. No labeled kidney benefit. | FLOW trial → reduces risk of eGFR decline, ESKD, and CV death in T2D + CKD. Added to label Jan 2025. |
| Head-to-head study | NCT06260722 — Phase 3 vs semaglutide in T2D. Results not yet published. | Same trial above |
| Novel safety signal | Dysesthesia (abnormal skin sensation / tingling) in ~20.9% at the 12 mg dose in TRIUMPH-4 — not seen at meaningful rates with Ozempic | Not reported |
| Legal access (April 2026) | Clinical trial enrollment only. FDA has stated retatrutide cannot be used in compounding under federal law. | Prescription from any licensed U.S. prescriber |
| Self-pay price | No legitimate public price (not legally sold to the public) | $199/mo first 2 months (intro, through June 30, 2026); then $349/mo (0.25/0.5/1 mg) or $499/mo (2 mg) |
| Expected U.S. availability | Not announced by Lilly. Multiple Phase 3 readouts pending through 2026. | Available today nationwide |
Cross-trial caveat: Numbers come from different studies, different populations, and different protocols. The only registered head-to-head (NCT06260722) is a type 2 diabetes trial and results haven’t been published yet. Treat every cross-study comparison as directional, not definitive.
Is retatrutide FDA-approved? Can you actually get it?
No and no — not in any legal retail sense. Ozempic is FDA-approved and available nationwide by prescription. Retatrutide is an investigational drug; Eli Lilly states publicly that legal access is only through Lilly clinical trials. The FDA has explicitly stated that retatrutide cannot be used in compounding under federal law and has issued warning letters involving compounded and unapproved retatrutide sales to U.S. consumers.
What the FDA has actually said about compounded retatrutide
FDA enforcement record — retatrutide compounding
In its September 9, 2025 warning letter to GLP1 Solution, the FDA wrote that “drug products compounded using retatrutide are not eligible for the exemptions provided by section 503A” because retatrutide is not on an applicable USP or NF monograph and is not a component of an FDA-approved drug, and not eligible for 503B exemptions because retatrutide does not appear on the 503B bulks list and is not used to compound a drug on the shortage list.
The FDA’s “Concerns with Unapproved GLP-1 Drugs” page (last updated February 2026) states that retatrutide “cannot be used in compounding under federal law” and that the agency has issued warning letters to companies distributing it.
In one sentence: If you see a telehealth site, wellness clinic, or “research peptide” vendor selling retatrutide to U.S. consumers in 2026, that product is not a legal prescription drug. The FDA has flagged such products as being of unknown quality, with potential issues including incorrect dosing, contamination, or non-sterile manufacturing.
The one legal path to retatrutide in 2026
Clinical trial enrollment. Eli Lilly’s TRIUMPH program has enrollment ongoing in multiple Phase 3 trials through 2026. Trial participation is medically supervised and uses sponsor-supplied study drug — but many trial arms include a placebo, so there’s a meaningful chance you’ll receive no active drug. Individual Patient Expanded Access (compassionate use) is extremely unlikely to be granted given that FDA-approved GLP-1s exist for the same conditions.
Why retatrutide is different from compounded semaglutide
Compounded semaglutide and tirzepatide had a legal basis during the FDA drug shortage period (2022–2024). Retatrutide has never been FDA-approved and has never been on a shortage list, so it has no shortage-based legal pathway at all. The category simply doesn’t exist for this molecule.
Is retatrutide better than Ozempic for weight loss?
By the headline numbers, yes — retatrutide produced substantially more weight loss in obesity trials than Ozempic has produced in diabetes trials, and roughly double what Wegovy produced in STEP 1. But this isn’t an apples-to-apples comparison.
The biggest public retatrutide number right now
Eli Lilly’s TRIUMPH-4 trial reported topline results on December 11, 2025. In adults with obesity and knee osteoarthritis, the 12 mg retatrutide dose produced a mean body-weight reduction of 28.7% at 68 weeks. Roughly 60% of participants lost at least 25% of their body weight. Those figures are the highest ever reported in a Phase 3 obesity trial and are in the range typically seen after bariatric surgery.
Important context: 28.7% is a company topline readout from an investor release, not yet a fully published peer-reviewed journal article. Full Phase 3 publications are pending as additional TRIUMPH readouts report out through 2026.
What a fair reader should conclude
Retatrutide looks more powerful on weight loss by a meaningful margin — not because Ozempic is weak, but because retatrutide acts on three receptors instead of one. Adding GIP amplifies insulin secretion and improves fat metabolism, and adding glucagon increases energy expenditure. That’s the mechanism reason retatrutide beats both Ozempic (GLP-1 only) and Zepbound (GLP-1 + GIP, the dual agonist).
The catch: retatrutide isn’t available to you as a prescription drug in 2026. Ozempic, Wegovy, and Zepbound are. The right question usually isn’t “which is better on the data?” It’s “which is the best thing I can actually start now?” For the CagriSema vs retatrutide angle, see our CagriSema vs retatrutide comparison.
What does Ozempic do better than retatrutide right now?
Ozempic’s advantage isn’t hype — it’s decision quality. The FDA label covers three things retatrutide doesn’t have yet: glycemic control in adults with type 2 diabetes, MACE reduction in adults with T2D and established CVD, and kidney-risk reduction in adults with T2D and chronic kidney disease. Those are labeled benefits backed by peer-reviewed outcome trials.
Ozempic’s current FDA-labeled indications (2025 label)
- Glycemic control in type 2 diabetes (approved December 2017).
- Reducing major adverse cardiovascular events (MACE — CV death, nonfatal heart attack, nonfatal stroke) in adults with T2D and established CVD (from SUSTAIN-6).
- Reducing the risk of sustained eGFR decline, end-stage kidney disease, and CV death in adults with T2D and chronic kidney disease (from the FLOW trial; added to label January 2025).
Retatrutide’s cardiovascular and kidney outcomes studies are still in progress. For a patient with T2D, established coronary disease, and early-stage CKD — the retatrutide weight-loss headline is exciting, but the drug you should be on today is a GLP-1 with labeled cardiovascular and kidney benefits. That’s Ozempic. See our Best GLP-1 for Diabetes guide for the full decision tree by use case.
Retatrutide vs Ozempic side effects
Both drugs are GI-heavy — nausea, vomiting, diarrhea — because slowing stomach emptying is part of how they work. But the current public retatrutide data shows meaningfully higher rates across every GI category and a novel safety signal not seen with Ozempic.
| Adverse event | Retatrutide (TRIUMPH-4) | Ozempic (label, all doses) |
|---|---|---|
| Nausea | 38.1–43.2% | 15.8–20.3% |
| Diarrhea | 33.1–34.7% | 8.5–8.8% |
| Vomiting | 20.4–20.9% | 5.0–9.2% |
| Discontinuation due to adverse events | 12.2–18.2% | 3.1–3.8% |
| Dysesthesia (abnormal skin sensation) | ~20.9% at 12 mg dose | Not reported at meaningful rates |
Sources: TRIUMPH-4 Eli Lilly readout (Dec 11, 2025); FDA-approved Ozempic label adverse reactions section (2025). Different trials, different populations — treat gap as directional.
Ozempic also carries the GLP-1 class boxed warning for thyroid C-cell tumors (based on rodent studies), and labeled warnings for pancreatitis, diabetic retinopathy complications, acute kidney injury, hypoglycemia with insulin or sulfonylureas, and gallbladder events. Both drugs are contraindicated in people with a personal or family history of medullary thyroid carcinoma (MTC) or MEN 2.
Retatrutide at 12 mg is a harder medication to tolerate than Ozempic at 2 mg. If you struggled with GI side effects on Ozempic, TRIUMPH-4 numbers suggest you’d struggle more, not less.
Should you wait for retatrutide or start Ozempic now?
For almost everyone, start now. Eli Lilly has not announced an FDA filing date, approval date, or launch date for retatrutide. Multiple Phase 3 readouts are still pending through 2026. Waiting an indefinite period — or without treatment — for a drug that hasn’t been submitted to the FDA, hasn’t been priced, and has higher peak-dose side effects than the approved options is rarely the right call.
The timeline, honestly
- Lilly has seven additional Phase 3 TRIUMPH readouts scheduled throughout 2026.
- Lilly has not publicly committed to a New Drug Application (NDA) filing date.
- Approval and launch dates have not been announced.
- Even after approval, Lilly will need to scale manufacturing (they’re still catching up on Zepbound and Mounjaro) before broad commercial availability.
The honest takeaway: this isn’t happening in 2026. Beyond that, there’s no publicly committed timeline to rely on.
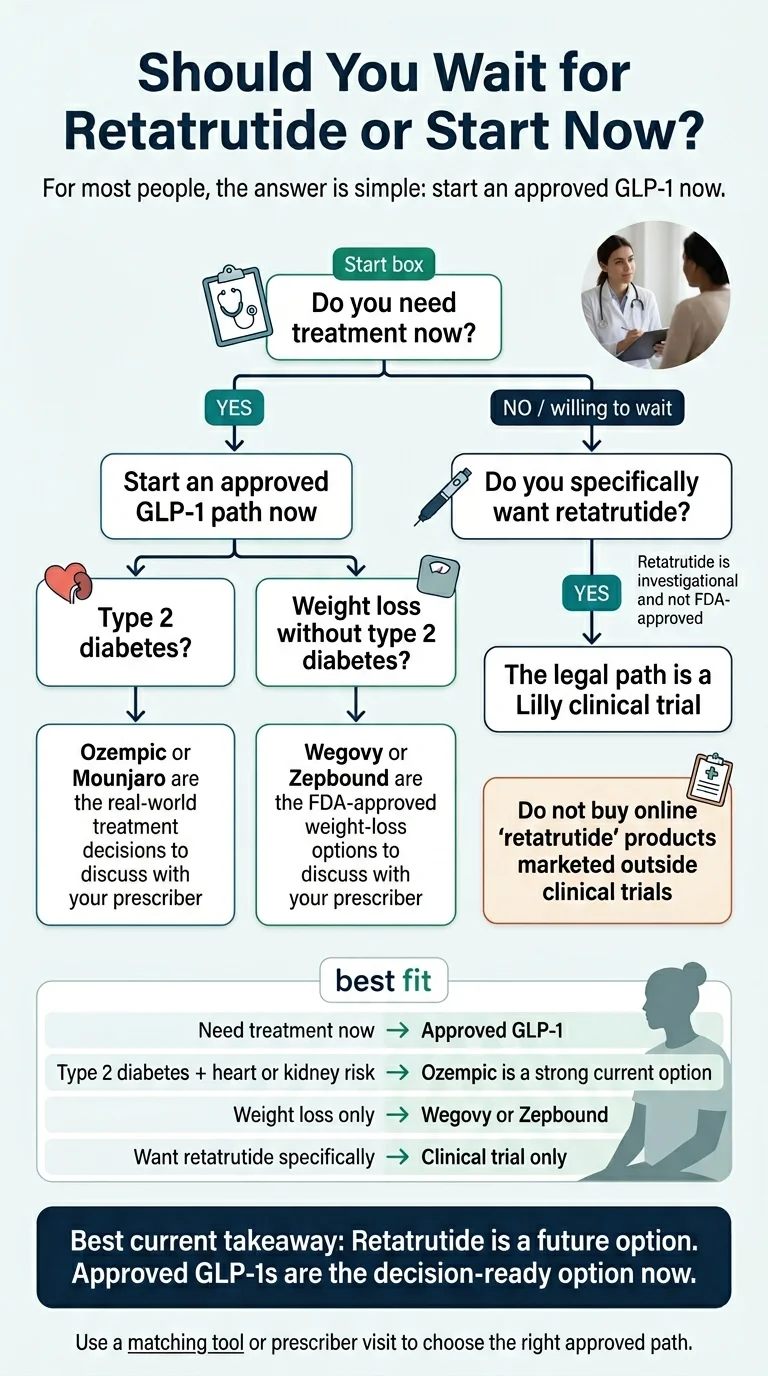
For most people, the answer is simple: start an approved GLP-1 now.
If you’re on Ozempic and plateauing
Don’t wait for an illegal product. Talk to your prescriber about switching to tirzepatide (Zepbound for weight loss or Mounjaro for T2D). Tirzepatide is a dual agonist (GLP-1 + GIP), mechanism-closer to retatrutide, and in the SURMOUNT-5 head-to-head trial it produced greater weight loss than semaglutide at the highest doses. See our retatrutide vs tirzepatide guide for how they compare.
Decision resolution point: the approved path closest to retatrutide
If what you actually want is a drug that goes beyond single-receptor GLP-1 and you can start today, Zepbound (tirzepatide) is the FDA-approved dual agonist — one receptor short of retatrutide’s triple mechanism. Clinical-trial weight loss sits between Ozempic and retatrutide. It’s available today through a prescriber or qualified telehealth provider.
Check your eligibility and see Zepbound and Wegovy pricing through Ro
Ro carries Wegovy (pill and pen), Zepbound (pen and KwikPen), and the newly approved Foundayo (orforglipron). Ro Body membership is $39 for your first month, or as low as $74/month with annual prepay. GLP-1 medication cost is billed separately — cash-pay Wegovy pill starts at $149/month through Ro’s LillyDirect and NovoCare integration, and Ro’s insurance concierge will check coverage for you first.
FDA-approved only — we’re not blurring approved and compounded on a branded comparison page.
Or take the 60-second matching quiz to see every legitimate FDA-approved option — and, as a separate cash-pay lane, legitimate compounded semaglutide providers — matched to your BMI, state, insurance, and goals.
What if your real goal is weight loss, not diabetes control?
If your goal is pure weight management and you don’t have type 2 diabetes, Ozempic is usually the wrong comparison drug. The FDA-approved weight-loss semaglutide is Wegovy, and the FDA-approved dual agonist is Zepbound. Both are closer to what retatrutide is trying to be than Ozempic is.
- Wegovy is semaglutide (same molecule as Ozempic) at the higher dose approved for chronic weight management.
- Zepbound is tirzepatide, a dual GLP-1 + GIP agonist approved for chronic weight management. Higher peak efficacy than Wegovy in head-to-head trials. Mechanism-closest to retatrutide.
- Foundayo (orforglipron) is Lilly’s once-daily oral GLP-1 that received FDA approval on April 1, 2026, for chronic weight management. First oral GLP-1 pill that can be taken any time of day without food or water restrictions. See our orforglipron vs retatrutide comparison.
How much does Ozempic cost today — and what does retatrutide cost?
Ozempic has a real, current self-pay price. Retatrutide doesn’t — because it isn’t legally sold to the public. If a website is advertising a “retatrutide price” in the $199–$499/month range in 2026, that’s a red flag — no such legal price exists.
Ozempic self-pay, as of April 2026
| Pathway | Cost | Notes |
|---|---|---|
| Intro offer (months 1–2, 0.25 or 0.5 mg) | $199/mo | Through June 30, 2026; first-time users only |
| NovoCare self-pay (0.25, 0.5, or 1 mg ongoing) | $349/mo | Standard tier |
| NovoCare self-pay (2 mg ongoing) | $499/mo | Maximum dose tier |
| Commercial insurance + Ozempic Savings Card | As low as $25/mo | Eligibility restrictions apply; Medicare/Medicaid excluded |
| Retatrutide | No legitimate public price | Not legally sold to the public |
Source: Novo Nordisk cost-and-coverage page, verified April 21, 2026.
Who should choose what? (Decision by use case)
The worst decision is treating retatrutide vs Ozempic as a single winner-vs-loser question. Here’s how to map yourself to the right choice today.
Type 2 diabetes + established CVD or CKD
Ozempic has the strongest case. The FDA label covers glycemic control, MACE reduction, and kidney-risk reduction — three labeled benefits retatrutide doesn’t have. Don’t wait on retatrutide here.
Type 2 diabetes without CVD or CKD
Ozempic or Mounjaro are both appropriate. Mounjaro tends to produce more weight loss and comparable or better A1C; Ozempic has the broader cardiovascular and kidney label. Discuss with your prescriber.
Goal is weight loss without diabetes
Wegovy or Zepbound. Zepbound is mechanism-closer to retatrutide. Wegovy has CV outcome data in people with obesity and established CVD. Don’t default to Ozempic — it’s a diabetes drug.
Plateaued on Ozempic
Don’t wait for retatrutide. Talk to your prescriber about switching to tirzepatide (Zepbound for weight loss, Mounjaro for T2D). SURMOUNT-5 showed tirzepatide outperformed semaglutide head-to-head.
Specifically want retatrutide and willing to wait
Search actively enrolling TRIUMPH trials at ClinicalTrials.gov. That’s the only legal path. Placebo randomization is part of most arms.
You bought compounded retatrutide online
Stop using it and talk to your prescriber. The FDA has warned these products are of unknown quality. “Prescribed by a licensed telehealth provider” does not make it legal — retatrutide is not eligible for compounding under federal law regardless of the prescription.
If none of the above exactly describes your situation, the fastest way to get a clear answer is our matching quiz. It accounts for your BMI, insurance status, state, prior GLP-1 experience, and goals.
A real patient perspective
“With Ozempic, I lowered my A1C and saw noticeable weight loss.”
Disclosure: Michael is a paid Novo Nordisk spokesperson. This quote is presented as a patient perspective, not as proof of comparative efficacy. Individual results vary. Ozempic is FDA-approved for adults with type 2 diabetes; it is not approved for weight management. There are no legitimate public patient testimonials for retatrutide in 2026 — it’s available only in blinded clinical trials.
What we actually verified for this page
Every comparison page claims to be “thoroughly researched.” Here’s the actual list.
| Claim | Source | Verified on |
|---|---|---|
| Retatrutide 28.7% mean weight loss at 68 weeks (12 mg, TRIUMPH-4) | Eli Lilly investor press release, Dec 11, 2025 | Apr 21, 2026 |
| Retatrutide Phase 3 T2D A1C reduction 1.7–2.0 pts; weight loss up to 16.8% at 40 weeks | Eli Lilly TRANSCEND-T2D-1 investor release | Apr 21, 2026 |
| TRIUMPH-4 side effects (nausea 38.1–43.2%, discontinuation 12.2–18.2%) | Eli Lilly TRIUMPH-4 readout | Apr 21, 2026 |
| Retatrutide legally available only through Eli Lilly trials | Lilly retatrutide FAQ (lilly.com) | Apr 21, 2026 |
| FDA: retatrutide cannot be used in compounding under federal law | FDA warning letter to GLP1 Solution (Sept 9, 2025); FDA “Concerns with Unapproved GLP-1 Drugs” (Feb 2026) | Apr 21, 2026 |
| Registered head-to-head retatrutide vs semaglutide Phase 3 T2D trial | ClinicalTrials.gov NCT06260722 | Apr 21, 2026 |
| Ozempic FDA indications (T2D; MACE in T2D + CVD; kidney risk in T2D + CKD) | FDA-approved Ozempic label (2025 revision) | Apr 21, 2026 |
| Ozempic self-pay pricing ($199/$349/$499) | Novo Nordisk cost-and-coverage page | Apr 21, 2026 |
| Foundayo (orforglipron) FDA approval, April 1, 2026 | FDA press announcement; Lilly press release | Apr 21, 2026 |
Frequently asked questions
Is retatrutide better than Ozempic for weight loss?
On trial data, yes — retatrutide produced 28.7% mean weight loss at 68 weeks in TRIUMPH-4 (12 mg, obesity + knee OA), while Wegovy (the weight-loss-approved form of Ozempic’s semaglutide) produced ~14.9% in STEP 1 at 68 weeks. But retatrutide is investigational, not FDA-approved, and has higher peak-dose side effects than approved options.
Is retatrutide FDA-approved in 2026?
No. As of April 2026, retatrutide is in Eli Lilly’s Phase 3 TRIUMPH program and has not been submitted to the FDA. Lilly has not announced an NDA filing date, approval date, or launch date. Multiple Phase 3 readouts remain pending through 2026.
Can you buy retatrutide online?
There is no legal way to buy retatrutide for human use in the U.S. in 2026. Eli Lilly says it is available only through Lilly clinical trials. The FDA has stated retatrutide cannot be used in compounding under federal law and has issued warning letters involving compounded and unapproved retatrutide sales.
Is Ozempic approved for weight loss?
No. Ozempic is FDA-approved for adults with type 2 diabetes — for glycemic control, cardiovascular risk reduction in T2D with established CVD, and kidney-risk reduction in T2D with chronic kidney disease. The FDA-approved weight-loss form of semaglutide is Wegovy.
Should I wait for retatrutide?
Usually no. Eli Lilly has not announced an FDA filing date, approval date, or launch date for retatrutide, and Phase 3 readouts remain pending. The defensible reasons to wait are narrow: you qualify for and want to enroll in a legitimate TRIUMPH clinical trial, or you’ve already failed on both semaglutide and tirzepatide with no approved next step.
What if Ozempic stopped working for me?
Plateaus are common on any GLP-1. The legitimate next steps are to reassess your dose and adherence with your prescriber, and discuss switching to tirzepatide (Zepbound for weight loss or Mounjaro for T2D). In SURMOUNT-5, tirzepatide produced more weight loss than semaglutide head-to-head. Chasing compounded retatrutide isn’t a reasonable plateau solution.
Is there a head-to-head retatrutide vs Ozempic trial?
A registered Phase 3 head-to-head comparing retatrutide to semaglutide in adults with type 2 diabetes is listed on ClinicalTrials.gov as NCT06260722. It’s sponsored by Eli Lilly. Results haven’t been published. Once those results are public, the cross-trial caveat in this page’s comparison will partially resolve.
What are the side effects of retatrutide vs Ozempic?
Both drugs cause GI side effects — nausea, vomiting, diarrhea, constipation. Public Phase 3 data show higher rates on retatrutide at peak dose than Ozempic at its maximum approved dose (nausea 38.1–43.2% vs 15.8–20.3%; discontinuation 12.2–18.2% vs 3.1–3.8%). Retatrutide also shows a novel signal — dysesthesia, abnormal skin sensation — in about 20.9% of patients at 12 mg.
How much does Ozempic cost without insurance?
Novo Nordisk’s self-pay pricing is $199/month for the first two months on the 0.25 or 0.5 mg starter doses, then $349/month for ongoing 0.25/0.5/1 mg doses or $499/month for the 2 mg dose. The intro savings offer is time-bound through June 30, 2026. Commercial insurance savings can bring covered patients as low as $25/month. Medicare and Medicaid patients are excluded from the savings offer.
Who is Ozempic actually best for?
Ozempic is best for adults with type 2 diabetes — especially those who also have established cardiovascular disease or chronic kidney disease, because of the drug’s labeled MACE and kidney-risk reductions. The FDA-approved semaglutide for weight loss is Wegovy, and the mechanism-closer FDA-approved option to retatrutide is Zepbound.
Can you take Ozempic and retatrutide together?
No. GLP-1-class medications should not be self-combined. Retatrutide also isn’t legally prescribable outside a clinical trial in the U.S. If you’re enrolled in a TRIUMPH trial, the protocol will require you to wash out of any other GLP-1 before participating.
What’s the closest FDA-approved drug to retatrutide?
Zepbound (tirzepatide) is the closest FDA-approved option by mechanism. Retatrutide is a triple agonist (GLP-1 + GIP + glucagon); Zepbound is a dual agonist (GLP-1 + GIP). In SURMOUNT-1 trial data, Zepbound produced about 20.9% mean weight loss at 72 weeks — between Wegovy’s 14.9% and retatrutide’s 28.7%. It’s FDA-approved for chronic weight management and available today.
Still not sure which GLP-1 program is right for you?
You’ve read the Phase 3 data, you know the FDA status, you know the real Ozempic prices, and you know the three legitimate paths forward. If you still aren’t sure which fits your situation, that’s what our matching tool is for.
Take our free 60-second GLP-1 matching quiz
It asks about your BMI, state, insurance status, prior GLP-1 experience, and goals — then matches you to the legitimate FDA-approved option that fits, with verified pricing and no sales pressure. If cash-pay compounded semaglutide is a better fit for your cost situation, the quiz routes to vetted options there too, in a separate lane. If the right answer for you is “wait for retatrutide and enroll in a TRIUMPH trial,” the quiz will tell you that too.
One last note on retatrutide
If retatrutide is the specific drug you want, the only legal path in 2026 is a clinical trial. Search actively enrolling retatrutide trials on ClinicalTrials.gov using “retatrutide” or “LY3437943.” Trial participation is medically supervised and uses sponsor-supplied study drug from Eli Lilly. Placebo randomization is part of most arms — read the informed consent carefully before enrolling.
Any other path you see advertised in 2026 is one the FDA has already warned about. Protect yourself.
Related comparisons
About this page: The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. This page was researched and written using primary sources — FDA-approved labels, manufacturer investor releases, FDA enforcement records, ClinicalTrials.gov, and current provider pricing pages. We re-verify every data point at least monthly.
Nothing on this page is medical advice. Talk to a licensed prescriber before starting, stopping, or switching any GLP-1 medication.
Last verified: April 21, 2026.