Affiliate disclosure: The RX Index earns a commission when you sign up with some of the providers mentioned on this page. It does not affect what you pay, and it never determines our rankings or which providers we cover. Read the full disclosure.
Drug Comparison ·
By The RX Index Editorial Team · · Pricing verified via LillyDirect · Foundayo data from FDA-approved prescribing information and ATTAIN-1 (NEJM, Sept 2025) · Retatrutide data from Eli Lilly TRIUMPH-4 topline results (Dec 2025, not yet peer-reviewed)
Orforglipron vs Retatrutide: Real Differences & What to Do Now (2026)
Published:
Orforglipron vs retatrutide comes down to one thing most comparison pages won’t say upfront: you can get one of these drugs today, and you can’t get the other.
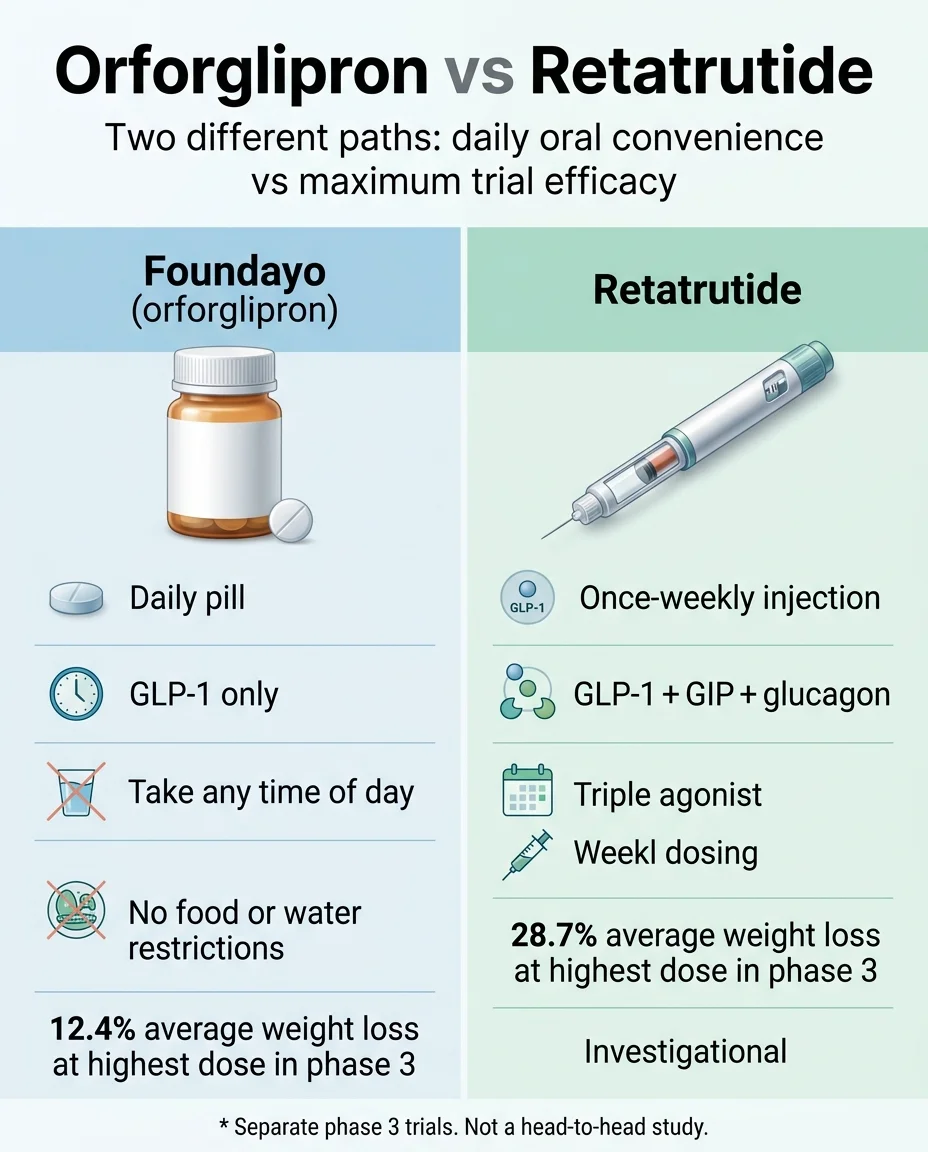
Foundayo (orforglipron) is an FDA-approved daily GLP-1 pill available right now through LillyDirect, telehealth providers, and retail pharmacies. Self-pay starts at $149/month. In Phase 3 trials, it produced 12.4% average body weight loss at the highest dose over 72 weeks — about 27 lbs for the average participant.
Retatrutide is a once-weekly injectable triple agonist that produced 28.7% weight loss in Phase 3 — the highest number ever reported for any obesity drug, roughly 71 lbs on average. But it is not FDA-approved, not available at any legitimate pharmacy, and Eli Lilly has not publicly announced an FDA filing date. Analyst projections place potential approval no sooner than 2027.
Both are made by Eli Lilly. In our editorial assessment, they’re designed to serve different patients — orforglipron for accessibility, retatrutide for maximum efficacy. The rest of this page exists so you never have to open another tab.

Orforglipron vs Retatrutide: The Head-to-Head Comparison
Data assembled from ATTAIN-1 (NEJM, Sept 2025), TRIUMPH-4 topline results (Eli Lilly, Dec 2025), FDA prescribing information for Foundayo, and LillyDirect pricing. Sources cited for every row. Last verified .
| Category | Foundayo (Orforglipron) | Retatrutide (LY3437943) |
|---|---|---|
| FDA Status | ✅ Approved April 1, 2026 | ❌ Not approved — Phase 3 trials ongoing |
| Drug Type | Non-peptide oral GLP-1 receptor agonist (small molecule) | Triple agonist: GLP-1 + GIP + Glucagon (peptide) |
| Receptors Targeted | 1 (GLP-1 only) | 3 (GLP-1 + GIP + Glucagon) |
| How You Take It | Daily pill — any time of day, no food or water restrictions | Weekly subcutaneous injection |
| Phase 3 Weight Loss (highest dose, on-treatment) | 12.4% at 72 weeks / ~27.3 lbs (ATTAIN-1) | 28.7% at 68 weeks / ~71.2 lbs (TRIUMPH-4, 12mg) |
| Phase 3 Weight Loss (all participants incl. dropouts) | 11.2% / ~25.0 lbs (ATTAIN-1) | 23.7% / ~27.2 kg (TRIUMPH-4, 12mg) |
| Most Common Side Effects | Nausea, constipation, diarrhea, vomiting (FDA label) | Nausea, diarrhea, constipation, vomiting (TRIUMPH-4) |
| Dysesthesia (skin tingling) | Not reported as significant | 20.9% at 12mg · 8.8% at 9mg |
| Stopped Treatment Due to Side Effects | 10% at 17.2mg (FDA label, pooled trials) | 12.2% (9mg) · 18.2% (12mg) |
| Self-Pay Price | $149–$349/mo (dose-dependent via LillyDirect; $299/mo at highest doses via Self-Pay Journey Program) | N/A — not commercially available |
| With Commercial Insurance | As low as $25/mo with savings card | N/A |
| Availability | Now — LillyDirect, retail pharmacies, telehealth | Not available outside authorized clinical trials |
| Manufacturer | Eli Lilly | Eli Lilly |
| Biggest Advantage | Available now. Oral. No fasting. Affordable entry price. | Highest weight loss of any obesity drug ever tested. |
| Biggest Limitation | Less weight loss than injectable GLP-1s | Not approved. Timeline uncertain. Unique dysesthesia signal. |
Sources: FDA.gov approval announcement (4/1/26), Foundayo prescribing information (DailyMed), ATTAIN-1 (NEJM, Sept 2025), TRIUMPH-4 topline results (Eli Lilly press release, Dec 2025), LillyDirect pricing page (verified 4/14/26). These are separate trials with different patient populations — not a head-to-head comparison.
Separate Phase 3 trials. Not a head-to-head study.
Ready to start with the drug that’s available now?
Foundayo (orforglipron) is FDA-approved, available through telehealth, and starts at $149/month. Ro membership starts at $39 for the first month (medication billed separately at LillyDirect rates starting at $149/month).
Check Foundayo Eligibility on Ro → (sponsored affiliate link, opens in a new tab)How Do Orforglipron and Retatrutide Actually Work?
Orforglipron activates one metabolic receptor — GLP-1 — as a once-daily pill. Retatrutide activates three receptors simultaneously — GLP-1, GIP, and glucagon — as a once-weekly injection. That third receptor is the whole story.
GLP-1 receptor activation
Suppresses appetite and slows gastric emptying. This is what every GLP-1 drug on the market does — semaglutide, tirzepatide, and now orforglipron. It works reliably.
GIP receptor activation (retatrutide adds this)
Enhances insulin sensitivity and contributes to fat metabolism. Tirzepatide (Zepbound/Mounjaro) adds this second receptor, which is partly why it outperforms single-agonist GLP-1s.
Glucagon receptor activation (unique to retatrutide)
Increases energy expenditure. Your body burns more calories, particularly through hepatic lipid oxidation — your liver processes more fat. This third pathway is likely why retatrutide’s weight loss numbers are so much higher than anything else in the pipeline. But it also means more metabolic activity — and more side effects, including a unique dysesthesia signal.
What This Means in Plain Language
| Factor | Foundayo (Orforglipron) | Retatrutide |
|---|---|---|
| Route | Pill — any time of day | Weekly injection |
| Timing restrictions | None | N/A (injection) |
| Storage | Room temperature, protect from light | Expected: refrigeration (injection) |
| Titration time to max dose | ~5 months (30-day steps) | ~4 months (4-week steps in trials) |
| FDA-approved today? | Yes | No |
| Legal access right now | Prescription via any licensed provider | Authorized clinical trials only |
| Biggest unknown | No cardiovascular outcomes data yet | Long-term safety profile, dysesthesia persistence |
How Much Weight Will You Actually Lose? The Honest Numbers
This is the section where we need to slow down, because the headline comparison — 12.4% vs 28.7% — is real but misleading without context.
Foundayo (Orforglipron): ATTAIN-1 Trial
Published in the New England Journal of Medicine, September 2025. Enrolled 3,127 adults with obesity or overweight (without diabetes). Over 72 weeks at the highest dose (17.2mg marketed dose):
- On-treatment (efficacy estimand): 12.4% body weight lost — about 27.3 lbs for the average participant
- All participants including dropouts (treatment-regimen estimand): 11.2% — about 25.0 lbs
- 59% of patients on the highest dose lost at least 10% of their body weight
- 18% lost 20% or more
- Up to 91% of participants with prediabetes at baseline achieved near-normal blood sugar levels
For real-world context: if you weigh 250 lbs, the on-treatment average translates to roughly 31 lbs lost. That moves you from a BMI of 40 to about 35.
Retatrutide: TRIUMPH-4 Trial
Eli Lilly topline press release, December 11, 2025. Phase 3 results from 445 adults with obesity/overweight AND knee osteoarthritis. Over 68 weeks:
At 12mg (highest tested dose)
- On-treatment: 28.7% — about 71.2 lbs average
- All participants incl. dropouts: 23.7%
- WOMAC knee pain reduction: 74.3%
- 12.0% became completely pain-free
At 9mg
- On-treatment weight loss: 26.4%
- WOMAC knee pain reduction: 75.8%
- 14.1% became completely pain-free
Mean baseline weight was 248.5 lbs (BMI 40.4). For a 250-lb person: the 12mg on-treatment average translates to roughly 72 lbs lost — a BMI drop from 40 to about 29.
Why You Can’t Simply Compare These Numbers
These numbers come from different trials with different populations. ATTAIN-1 enrolled adults with obesity but without diabetes. TRIUMPH-4 enrolled adults with obesity AND knee osteoarthritis — a population with higher baseline BMI (40.4) that tends to lose more absolute weight. There is no head-to-head trial comparing these two drugs directly.
That said, the gap is large enough that the directional conclusion is clear: retatrutide produces substantially more weight loss than orforglipron. The question is whether that additional weight loss is worth the trade-offs — availability, route of administration, side effects, and cost.
Here’s what most comparison pages won’t say
If raw weight loss is your only metric, Foundayo doesn’t compete with retatrutide. It also produces less weight loss than currently available injectables — Zepbound produces about 20.9%, injectable Wegovy 15–17%, and Foundayo 12.4%. Foundayo’s value isn’t in being the most powerful GLP-1. It’s in being the most accessible: a pill you can take any time, no needles, no fasting, no refrigeration, starting at $149/month. The drug you actually take every day beats the drug you skip.
Does a convenient daily pill that delivers meaningful weight loss sound like what you need right now?
Ro membership starts at $39 for the first month (then as low as $74/month with annual plan). Foundayo medication is billed separately starting at $149/month through LillyDirect pricing.
Check Foundayo Availability and Pricing on Ro → (sponsored affiliate link, opens in a new tab)Can You Actually Get These Drugs Right Now?
Foundayo: ✅ Available now
FDA-approved April 1, 2026. Shipping started April 6 through LillyDirect. Available through telehealth providers (including Ro), retail pharmacies, and LillyDirect direct-to-consumer. You need a prescription from a licensed healthcare provider.
Retatrutide: ❌ Not yet available
Investigational drug in Phase 3 clinical trials. Eli Lilly has not publicly announced an NDA filing date. The TRIUMPH Phase 3 program includes eight trials, with seven expected to read out in 2026.
Retatrutide Approval Timeline (Projections Only)
| Period | Expected Milestone | Source / Status |
|---|---|---|
| 2026 | Remaining TRIUMPH trial results accumulate | Eli Lilly investor materials |
| Late 2026 / Early 2027 | NDA filing expected | Analyst projection — NOT confirmed by Lilly |
| 2027 | Potential FDA review and approval | Lilly 2025 investor materials indicated launch by end of 2027 |
| Late 2027 / 2028 | Commercial launch possible | Analyst projection — NOT confirmed by Lilly |
These are projections based on analyst reports (Citi, BMO, UBS, Jefferies) and Lilly investor materials — not confirmed regulatory timelines. Lilly’s official position: retatrutide is investigational and legally available only to participants in authorized clinical trials.
Want to start a GLP-1 you can access today? Check eligibility on Ro → (sponsored affiliate link, opens in a new tab)The grey-market warning you need to read
Some websites sell products labeled “retatrutide” as “research peptides” or “for research use only.” The FDA has warned companies illegally selling unapproved GLP-1 products — including retatrutide — directly to consumers. These products are of unknown quality, not legally available for human use outside authorized clinical trials, and used without medical supervision. If you want retatrutide specifically, the only legitimate path today is enrolling in a TRIUMPH clinical trial through ClinicalTrials.gov.
Side Effects Head-to-Head: The Dysesthesia Difference
Both drugs cause the gastrointestinal side effects common to all GLP-1 medications — nausea, diarrhea, constipation, vomiting. These are typically worst during dose escalation and improve over time. But retatrutide introduces one side effect that doesn’t appear with any approved GLP-1: dysesthesia.
| Side Effect | Foundayo 17.2mg (FDA Label, Pooled Trials) | Retatrutide 9mg (TRIUMPH-4) | Retatrutide 12mg (TRIUMPH-4) |
|---|---|---|---|
| GI adverse events (overall) | 69% | — | — |
| Nausea | Most common (≥5%); listed first in label | 38% | 43% |
| Diarrhea | Most common (≥5%) | 35% | 33% |
| Constipation | Most common (≥5%) | 22% | 25% |
| Vomiting | Most common (≥5%) | 20% | 21% |
| Dysesthesia (skin tingling) | Not reported as significant | 8.8% | 20.9% ⚠️ |
| Stopped treatment due to AEs | 10% | 12.2% | 18.2% |
Sources: Foundayo prescribing information (DailyMed, April 2026) — pooled data from ATTAIN-1 and ATTAIN-2; TRIUMPH-4 topline results (Eli Lilly, Dec 2025). Different trials, different populations.
The Dysesthesia Signal — Unique to Retatrutide
Dysesthesia is an abnormal touch sensation — tingling, tenderness, “pins and needles,” skin sensitivity, or discomfort when touched normally. In TRIUMPH-4:
- 20.9% of patients on the 12mg dose experienced dysesthesia
- 8.8% on the 9mg dose
- 0.7% on placebo
This side effect does not appear at meaningful rates with semaglutide, tirzepatide, or orforglipron. Researchers believe it’s related to glucagon receptor activation — the component unique to retatrutide’s triple mechanism.
What this means practically
The 9mg dose may offer a better risk-reward balance — 26.4% weight loss with only 8.8% dysesthesia. If retatrutide reaches the market, many patients and clinicians may prefer 9mg over 12mg for this reason. Dysesthesia events were generally classified as mild and rarely led to treatment discontinuation — but we don’t yet know whether they resolve over time or persist.
Foundayo Safety: What the Label Says
Foundayo carries a boxed warning for thyroid C-cell tumors (standard for the GLP-1 class). From the FDA-approved prescribing information:
| Safety Category | Details |
|---|---|
| Contraindications | Personal or family history of medullary thyroid carcinoma (MTC) or MEN 2; known serious hypersensitivity to orforglipron |
| Not recommended | Severe gastroparesis; pregnancy (may cause fetal harm); breastfeeding (not studied) |
| Drug interactions | Strong CYP3A4 inhibitors cap max dose at 9mg. Simvastatin capped at 20mg daily. Oral contraceptive effectiveness may be reduced — backup contraception required for 30 days after starting and after each dose increase. |
| Other warnings | Acute pancreatitis, acute kidney injury (from dehydration due to GI side effects), hypoglycemia risk when combined with insulin or sulfonylureas, acute gallbladder disease, pulmonary aspiration risk during general anesthesia |
| No cardiovascular outcomes data yet | Unlike injectable semaglutide (Wegovy), Foundayo has not yet demonstrated cardiovascular risk reduction in a completed outcomes trial. |
What Will You Actually Pay?
Foundayo Pricing — Verified via LillyDirect,
| Dose | Self-Pay (LillyDirect) | Notes |
|---|---|---|
| 0.8mg (starter) | $149/mo | — |
| 2.5mg | $199/mo | — |
| 5.5mg | $299/mo | — |
| 9mg | $299/mo | — |
| 14.5mg | $299/mo* | *Self-Pay Journey Program price. Standard price is $349/mo. |
| 17.2mg (max) | $299/mo* | *Self-Pay Journey Program price. Standard price is $349/mo. |
- With commercial insurance: As low as $25/month using the Foundayo Savings Card (eligible commercially insured patients; governmental beneficiaries excluded)
- Medicare Part D: Eligible individuals may be able to get Foundayo for approximately $50/month beginning as soon as July 1, 2026, per Eli Lilly
How Foundayo Compares to Other GLP-1 Options
| Option | Route | Approx. Monthly Self-Pay |
|---|---|---|
| Foundayo (orforglipron) | Daily pill | $149–$349 (dose-dependent) |
| Wegovy pill (oral semaglutide) | Daily pill (morning, empty stomach) | $149–$299 |
| Zepbound (tirzepatide) | Weekly injection | $299–$549 (dose-dependent via LillyDirect) |
| Injectable Wegovy (semaglutide) | Weekly injection | ~$1,349 list (insurance typically required) |
| Compounded semaglutide or tirzepatide | Varies by provider | ~$199–$399 (varies widely) |
| Retatrutide | Weekly injection (investigational) | Unknown — not yet approved |
Note on compounded options: Compounded medications are not FDA-approved versions of brand-name drugs. They are produced by compounding pharmacies under different regulatory oversight.
Foundayo starts at $149/month — no insurance needed, no injections, no fasting rules.
See current pricing and check eligibility on Ro. Ro membership is $39 for the first month, then as low as $74/month with annual plan (medication priced separately at LillyDirect rates).
See Current Pricing and Check Eligibility on Ro → (sponsored affiliate link, opens in a new tab)Should You Start Foundayo Now or Wait for Retatrutide?
This is the question this page exists to answer. We’ve given you the data. Here’s the decision framework.
Start a GLP-1 now if:
- ✓You need to lose weight and you’re ready. Every month you wait is a month of potential health improvements you don’t get — reductions in blood pressure, cholesterol, blood sugar, and joint pain
- ✓Needles are a dealbreaker. Foundayo is the only GLP-1 pill you can take any time of day without food or water restrictions
- ✓You want an affordable cash-pay option. $149/month to start
- ✓You’re already on an injectable. The ATTAIN-MAINTAIN trial showed patients switching from Wegovy to Foundayo regained only about 0.9 kg on average — oral maintenance after injectable weight loss is clinically viable
Consider waiting for retatrutide if:
- →You need to lose 25%+ of your body weight and want the most powerful pharmacological option ever studied
- →You’re comfortable with weekly injections and the potential for higher GI side effects and dysesthesia
- →You can genuinely wait an uncertain timeline. Lilly has not confirmed when retatrutide will be available. (But honestly ask yourself: could you start something available now and switch later?)
- →You accept a less-characterized safety profile. Retatrutide’s full side effect picture is still being defined
The smartest play for most people: start now.
Published switching data already exists for moving between Lilly’s medications. While there is no published data yet on switching from Foundayo to retatrutide specifically, the clinical expectation — based on how these drugs work on related receptor pathways — is that a transition path will be available if and when retatrutide is approved.
Waiting means delaying every pound you’d lose in the next year-plus. It means delaying the cardiometabolic improvements that ATTAIN-1 documented — reductions in non-HDL cholesterol, triglycerides, waist circumference, and systolic blood pressure. And it means delaying the quality-of-life changes that come with losing even 10% of body weight.
Don’t let the perfect be the enemy of the good. The drug that’s available and that you take consistently will always outperform the drug that doesn’t exist yet.
Check Eligibility on Ro — Get Started for $39 → (sponsored affiliate link, opens in a new tab)Who Should NOT Take Foundayo
Foundayo is not for everyone. The FDA-approved prescribing information specifies:
Do not take Foundayo if you:
- ✗ Have a personal or family history of medullary thyroid carcinoma (MTC)
- ✗ Have Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- ✗ Have a known serious allergy to orforglipron or any ingredient in Foundayo
Foundayo is not recommended if you:
- ⚠️ Have severe gastroparesis (severely delayed stomach emptying)
- ⚠️ Are pregnant or planning to become pregnant — may cause fetal harm
- ⚠️ Are breastfeeding (not studied)
Talk to your doctor before starting if you:
- → Have a history of pancreatitis or kidney problems
- → Take oral birth control pills (backup contraception required for 30 days after starting and after each dose increase)
- → Take simvastatin (dose must be capped at 20mg daily) or strong CYP3A4 inhibitors (Foundayo max dose reduced to 9mg)
- → Have an upcoming surgery requiring general anesthesia (increases aspiration risk)
If any of these apply to you, that doesn’t mean you’re out of options. Injectable GLP-1s like Zepbound or Wegovy may still be appropriate depending on your specific situation. Take our matching quiz or talk with your healthcare provider.
What If You Want More Weight Loss Than Foundayo Delivers?
Fair question. If Foundayo’s 12.4% isn’t enough for your goals and you don’t want to wait for retatrutide, here’s what’s FDA-approved and available today:
Zepbound (tirzepatide)
Lilly’s injectable dual agonist. ~20.9% weight loss in trials. Self-pay starts at $299/month for the lowest dose through LillyDirect, with higher doses ranging up to $549/month. Weekly injection. This is the most powerful FDA-approved weight loss drug available right now.
Injectable Wegovy (semaglutide)
15–17% weight loss. Weekly injection. Often requires insurance (list price is ~$1,349/month). Also has proven cardiovascular risk reduction data.
Compounded GLP-1 options
For readers exploring more affordable options, some telehealth providers offer compounded GLP-1 medications. These are NOT FDA-approved versions of brand-name drugs — they are produced by compounding pharmacies under different regulatory oversight. That said, compounded options can be significantly more affordable for cash-pay patients.
Not sure which GLP-1 path fits your situation?
Take our free 60-second matching quiz to get a personalized recommendation based on your goals, budget, and preferences.
Take the Free 60-Second GLP-1 Matching Quiz →Can You Switch From Foundayo to Retatrutide Later?
There is no published data on switching from orforglipron to retatrutide. That specific transition has not been studied. What we do have:
- ATTAIN-MAINTAIN trial (Eli Lilly, December 2025): Patients switching from injectable Wegovy to oral Foundayo maintained their weight loss, regaining an average of only 0.9 kg (about 2 lbs). Patients switching from Zepbound to Foundayo regained about 5.0 kg.
- Mechanism compatibility: Retatrutide activates all three receptors that orforglipron (GLP-1) and Zepbound (GLP-1 + GIP) individually target. A step-up from Foundayo to retatrutide would mean adding two additional receptor pathways, not switching mechanisms entirely.
The bottom line: starting Foundayo now does not lock you out of retatrutide later. No published data suggests prior GLP-1 use would reduce the effectiveness of a future triple-agonist medication, and any weight you lose in the interim gives you a better starting point.
How All of This Fits Into the Broader GLP-1 Landscape
Here’s where each option sits as of :
| Drug | Type | Route | Avg Weight Loss | FDA Status | Self-Pay / Month |
|---|---|---|---|---|---|
| Foundayo (orforglipron) | GLP-1 agonist | Daily pill | 12.4% | ✅ Approved | $149–$349 |
| Wegovy pill (oral semaglutide) | GLP-1 agonist | Daily pill | ~14% | ✅ Approved | $149–$299 |
| Zepbound (tirzepatide) | Dual GLP-1/GIP | Weekly injection | ~20.9% | ✅ Approved | $299–$549 |
| Wegovy (injectable semaglutide) | GLP-1 agonist | Weekly injection | 15–17% | ✅ Approved | ~$1,349 list |
| Retatrutide | Triple GLP-1/GIP/Glucagon | Weekly injection | 28.7% | ❌ Investigational | Unknown |
Weight loss figures from separate Phase 3 trials — cross-trial comparisons are directional, not definitive.
What Should You Do While Waiting for Retatrutide?
If you’ve decided retatrutide is what you ultimately want, here’s a practical plan for right now:
Start an available GLP-1 now
Every month of delay is a month of potential weight loss and health improvement you don't get back. Published data supports transitions between GLP-1 medications.
Monitor the TRIUMPH program
Seven Phase 3 trials are expected to read out throughout 2026, covering obesity, type 2 diabetes, obstructive sleep apnea, metabolic dysfunction-associated steatotic liver disease, cardiovascular and renal outcomes, and more. We'll update this page as results publish. Bookmark it.
Consider a TRIUMPH clinical trial
If you're eligible, trial participation gives you access to pharmaceutical-grade retatrutide under medical supervision at no cost.
Search enrolling retatrutide trials on ClinicalTrials.gov →Do NOT buy 'research' retatrutide online
The FDA has warned companies selling unapproved GLP-1 products, including retatrutide. Lilly's position is clear: retatrutide is legally available only to participants in authorized clinical trials. Products sold online are of unknown quality and may be harmful.
Build the lifestyle foundation
Every obesity treatment works better with consistent nutrition and movement habits. The time between now and retatrutide's potential availability is time you can use to build those habits.
What We Actually Verified
How this page was produced: We assembled this comparison from the following primary sources, verified as of :
- Foundayo FDA approval: FDA.gov press announcement, April 1, 2026
- Foundayo clinical data: ATTAIN-1 trial, published in the New England Journal of Medicine, September 2025
- Foundayo prescribing information: FDA-approved label via DailyMed, April 2026
- Foundayo pricing: LillyDirect pricing page and Lilly press releases, verified
- Retatrutide clinical data: TRIUMPH-4 topline results, Eli Lilly press release, December 11, 2025 (not yet peer-reviewed)
- Retatrutide regulatory status: FDA.gov, ClinicalTrials.gov, Eli Lilly investor communications
- Retatrutide timeline context: Lilly 2025 investor materials, analyst reports (Citi, BMO, UBS, Jefferies)
- FDA safety communications: FDA warnings on unapproved GLP-1 drugs sold to consumers
- Switching data: ATTAIN-MAINTAIN (Eli Lilly, December 2025) — studied Wegovy/Zepbound → Foundayo transitions
Where we cite cross-trial comparisons, we say so explicitly. Where data comes from company press releases rather than peer-reviewed publications, we note that. We update this page as new TRIUMPH results publish.
Frequently Asked Questions
Is Foundayo the same as orforglipron?
Yes. Foundayo is the FDA-approved brand name for orforglipron, manufactured by Eli Lilly. It was approved on April 1, 2026 for chronic weight management in adults with obesity, or overweight with at least one weight-related medical condition.
Are orforglipron and retatrutide made by the same company?
Yes. Both are developed by Eli Lilly and Company. In our editorial view, Lilly positions orforglipron for oral convenience and broad accessibility, and retatrutide for maximum efficacy.
Which drug produces more weight loss, orforglipron or retatrutide?
Based on Phase 3 trial data, retatrutide produces substantially more weight loss: 28.7% at 68 weeks versus Foundayo's 12.4% at 72 weeks. These are from separate trials with different patient populations — not a head-to-head comparison — but the gap is large enough that the directional conclusion holds.
When will retatrutide be available?
Eli Lilly has not publicly announced a regulatory filing date. Lilly's 2025 investor materials indicated a planned launch by end of 2027. Seven remaining Phase 3 trials are expected to read out in 2026, after which Lilly is expected to file for FDA review. These are projections, not confirmed timelines.
Can I take orforglipron and retatrutide together?
No clinical data exists on combining these drugs. Foundayo's FDA label states it should not be used with other GLP-1 receptor agonists.
Is it safe to buy retatrutide online?
No. The FDA has warned companies illegally selling unapproved GLP-1 products to consumers, including retatrutide. These products are of unknown quality and may be harmful. Lilly states retatrutide is legally available only to participants in authorized clinical trials.
Will retatrutide be a pill?
As of April 2026, retatrutide is being studied exclusively as a once-weekly subcutaneous injection. Eli Lilly has not announced plans for an oral formulation.
How long do I need to take Foundayo?
Foundayo's FDA label approves it for long-term use in maintaining weight reduction. Clinical evidence across the GLP-1 drug class consistently shows that stopping treatment leads to weight regain.
What if I can't afford Foundayo?
If $149–$349/month is outside your budget, consider: compounded GLP-1 options through telehealth providers (some start lower), checking whether your commercial insurance covers Foundayo ($25/month with the Foundayo Savings Card), or Medicare Part D coverage, which Lilly says eligible beneficiaries may access beginning as soon as July 2026.
Does retatrutide cause skin tingling?
Yes. In the TRIUMPH-4 Phase 3 trial, 20.9% of patients on the 12mg dose reported dysesthesia — an abnormal touch sensation described as tingling, tenderness, or skin sensitivity. At the lower 9mg dose, the rate was 8.8%. On placebo, it was 0.7%. These events were generally mild and rarely led to discontinuation. This side effect does not appear with semaglutide, tirzepatide, or orforglipron.
Still Not Sure Which GLP-1 Program Is Right for You?
Answer a few questions about your goals, preferences, and budget, and we’ll recommend the specific path — and specific provider — that fits your situation. Whether that’s Foundayo, an injectable, a compounded option, or something else entirely. No spam, no pressure, just a personalized action plan.
Related guides
- Orforglipron vs Ozempic — full comparison with pricing and data
- Orforglipron vs Zepbound — pill vs injection head-to-head
- Orforglipron vs Wegovy pill — two oral GLP-1s compared
- Orforglipron (Foundayo) side effects — what clinical data shows
- Best GLP-1 telehealth providers compared for 2026
- Cheapest GLP-1 options without insurance — full pricing breakdown
- Best compounded GLP-1 providers in 2026
- Ro Body reviews — FDA-approved GLP-1 with insurance support
- Eden Health GLP-1 reviews — cost, complaints, and verdict