Affiliate disclosure: The RX Index earns a commission when you sign up with some of the providers mentioned on this page. It does not affect what you pay, and it never determines our rankings or which providers we cover. Read the full disclosure.
Verified GLP-1 Comparison ·
By The RX Index Editorial Team ·
Orforglipron vs Ozempic: Real Differences, Cost, and Who Each Fits
Published:
The orforglipron vs Ozempic decision comes down to three things: what your label actually needs, what your wallet can handle, and which daily friction you’ll stick with for months. Foundayo (orforglipron) is now FDA-approved specifically for chronic weight management and starts at $149/month self-pay. Ozempic (semaglutide) is FDA-approved for type 2 diabetes with cardiovascular and kidney risk-reduction labeling in certain adults with T2D — but it is not FDA-approved for weight loss.
One thing most comparison pages won’t tell you: there is no direct head-to-head clinical trial between Foundayo and injectable Ozempic. The strongest comparative data pit orforglipron against oral semaglutide — a lower-dose diabetes tablet, not the weekly Ozempic injection most people mean when they type this query. That matters, and we’ll explain exactly why below.
Here’s the quick comparison before we go deeper:
| Foundayo (orforglipron) | Ozempic (semaglutide) | |
|---|---|---|
| FDA-approved for | Chronic weight management | Type 2 diabetes, CV risk reduction, kidney risk reduction in certain T2D adults |
| How you take it | Once-daily pill, any time, no food/water rules | Once-weekly injection (subcutaneous) |
| Self-pay starting price | $149/mo (lowest dose) | $199/mo intro through June 30, 2026, then $349/mo |
| Best fit | Weight-loss-first readers who want an oral option | T2D readers who need diabetes/CV/kidney label breadth |
Not sure which path fits? Get your personalized GLP-1 action plan — free, 60 seconds
Find My GLP-1 Path →
Which is better for most people: orforglipron or Ozempic?
There is no universal winner. The right choice depends on whether your primary need is weight management, type 2 diabetes control, or simply removing the injection barrier — and each scenario has a different “best” answer.
This is the part that most comparison pages get wrong. They flatten orforglipron vs Ozempic into “pill vs shot” and call it a day. But the real decision has three forks, and which fork you’re in changes everything.
If your goal is chronic weight management
Foundayo is the cleaner fit. It’s the only one of these two that carries an FDA label specifically for weight loss in adults with obesity or overweight with at least one weight-related condition. Ozempic is frequently prescribed off-label for weight loss, but “off-label” means your insurance is less likely to cover it, your pharmacy may question the prescription, and you’re relying on a use the FDA hasn’t formally evaluated for that brand. Insurance formularies, prior authorization decisions, and coverage appeals all reference the FDA-approved indication.
If your goal is type 2 diabetes control
Ozempic is the stronger fit, and it’s not close. Its label covers glycemic control, major adverse cardiovascular event reduction in adults with T2D and established cardiovascular disease, and kidney risk reduction in adults with T2D and chronic kidney disease. That is the product of extensive outcome trial data baked directly into the Ozempic prescribing information. Foundayo is not currently FDA-approved for type 2 diabetes. Eli Lilly plans to seek that indication later in 2026, and the ACHIEVE-3 trial showed strong A1C results, but right now the label says weight management only.
If your real blocker is injections
If needles are the reason you haven’t started GLP-1 treatment, Foundayo removes that barrier entirely. This isn’t trivial — real people describe the experience in these exact words:
“I’ve been standing there, needle ready, and I just can’t bring myself to do it.”
— r/Ozempic
“I can’t see myself doing it forever.”
— r/glp1
A daily pill you take with breakfast (or whenever — no food rules) is a fundamentally different commitment than a weekly subcutaneous injection. For some people, that difference is the difference between starting treatment and not starting.
If your real blocker is cost or coverage
Without insurance, Foundayo’s lowest dose ($149/mo) is cheaper than Ozempic’s post-intro pricing ($349/mo at standard doses). But Foundayo’s maintenance doses reach $299–$349/mo, narrowing that gap. With commercial insurance and a manufacturer savings card, both can drop to $25/month. The deeper question is which one your plan will cover. Foundayo’s on-label weight management indication may actually improve coverage odds for obesity-first patients, because insurers are increasingly adding anti-obesity medications to formularies. Ozempic’s coverage is strongest when prescribed for its approved indication — type 2 diabetes.
Still unsure which fork you’re in? Get your personalized GLP-1 action plan
Get My Personalized Plan →Orforglipron vs Ozempic at a glance: the full verified comparison
We assembled data from FDA prescribing information, manufacturer pricing pages, and published clinical trials into a single view that doesn’t exist on any competing page in this exact form. Every cell is sourced.
| Category | Foundayo (orforglipron) | Ozempic (semaglutide) |
|---|---|---|
| Brand / Manufacturer | Foundayo™ / Eli Lilly | Ozempic® / Novo Nordisk |
| FDA approval date | April 1, 2026 | December 2017 (T2D); CV and kidney indications added subsequently |
| Approved indication | Chronic weight management (obesity/overweight + comorbidity) | Type 2 diabetes, CV risk reduction in T2D+CVD, kidney risk reduction in T2D+CKD |
| Approved for weight loss? | Yes — on-label | No — off-label use only |
| Drug class | GLP-1 receptor agonist (non-peptide small molecule) | GLP-1 receptor agonist (peptide) |
| Route | Once-daily oral tablet | Once-weekly subcutaneous injection |
| Food/water restrictions | None — take any time of day | N/A (injection); note: oral semaglutide requires empty stomach |
| Available doses | 0.8, 2.5, 5.5, 9, 14.5, 17.2 mg tablets | 0.25, 0.5, 1, 2 mg injection pens |
| Dose escalation | Start 0.8 mg → increase every 30+ days through 6 dose levels | Start 0.25 mg → 0.5 mg after 4 wks → 1 mg after 4 wks → optional 2 mg |
| Refrigeration | No | Yes (before first use; room temp up to 56 days after) |
| Avg weight loss (clinical trials) | 12.4% / 27.3 lbs over 72 weeks (ATTAIN-1, efficacy estimand) | Not approved for weight loss. For context: semaglutide 2.4 mg (Wegovy) showed ~15% over 68 weeks in STEP 1 |
| A1C reduction | -1.91% at 36 mg vs oral semaglutide (ACHIEVE-3, Lancet 2026) | -1.5% to -2.0% (SUSTAIN trials) |
| Cardiovascular outcome data | Not yet available (CV risk markers improved in trials) | Yes — CV risk reduction is part of the Ozempic label for certain adults with T2D and established CVD |
| GI side effects (trial rate) | ~59% reported GI symptoms (ACHIEVE-3) | ~37–45% (oral semaglutide comparator arm, same trial) |
| Discontinuation due to side effects | ~9–10% | ~4–5% (oral semaglutide in same trial) |
| Self-pay cost (monthly) | $149 (0.8 mg) → $199 (2.5 mg) → $299 (5.5/9 mg) → $299†/$349 (14.5/17.2 mg) | $199 intro‡ (first 2 fills) → $349 (≤1 mg) → $499 (2 mg) |
| With commercial insurance | As low as $25/mo (Lilly savings card) | As low as $25/mo (Novo Nordisk savings card) |
| Medicare Part D | Eligible individuals may pay ~$50/mo beginning July 1, 2026 (per Eli Lilly) | Covered for T2D (varies by plan); weight loss typically not covered |
| Where to get it | LillyDirect, Ro, Amazon Pharmacy, WW Med+, retail pharmacies | Most retail pharmacies, telehealth providers |
| Head-to-head trial vs each other? | No. No direct trial between Foundayo and injectable Ozempic exists. | Same. |
†Foundayo’s $299 price for 14.5/17.2 mg doses requires refilling within 45 days of last delivery through Lilly’s Journey Program. Miss that window and the price reverts to $349. Source: foundayo.lilly.com/coverage-savings.
‡Ozempic’s intro offer of $199 for new self-pay patients runs November 17, 2025 through June 30, 2026. After the intro period, self-pay prices are $349/mo (≤1 mg) and $499/mo (2 mg). Source: ozempic.com savings page, NovoCare.
Sources: FDA prescribing information for Foundayo and Ozempic; Eli Lilly investor press release (April 1, 2026); foundayo.lilly.com/coverage-savings; NovoCare/ozempic.com pricing pages; Lancet (ACHIEVE-3, Feb 2026); NEJM (STEP 1, ATTAIN-1).
What does the evidence actually say — and what does it not say?
The most important thing on this page: Eli Lilly states there are no ongoing or completed clinical trials comparing orforglipron directly with injectable GLP-1 medications like Ozempic or tirzepatide. The strongest direct comparative data come from ACHIEVE-3, which tested orforglipron against oral semaglutide (Rybelsus-type doses of 7 mg and 14 mg) — not the weekly Ozempic pen.
What ACHIEVE-3 actually showed
The ACHIEVE-3 trial enrolled 1,698 adults with type 2 diabetes across six countries for 52 weeks. It compared orforglipron (12 mg and 36 mg) against oral semaglutide (7 mg and 14 mg). Key results at the 36 mg orforglipron dose versus 14 mg oral semaglutide:
- A1C reduction: -1.91% vs -1.47% (orforglipron superior, p<0.001)
- Weight loss: 8.2 kg (~18 lbs) vs 5.3 kg (~11.7 lbs) (orforglipron superior)
- GI side effects: 59% vs 37–45% (orforglipron higher)
- Discontinuation due to adverse events: 9–10% vs 4–5% (orforglipron higher)
These are real results from a well-designed Phase 3 trial published in The Lancet. But they compare orforglipron to oral semaglutide doses used for diabetes — not to the weekly injectable Ozempic pen that most searchers have in mind.
Why cross-trial comparisons mislead
You’ll see other pages line up Foundayo’s 12.4% weight loss (ATTAIN-1) against Wegovy’s ~15% (STEP 1) and declare a winner. We won’t do that, because those numbers come from different trials with different patient populations, different timeframes, different baseline weights, and different protocols.
An honest reading: Foundayo produces clinically meaningful weight loss in its own trials, and injectable semaglutide at higher doses (Wegovy 2.4 mg) has generally shown higher average weight loss in its respective trials. The ~3 percentage point gap in the raw numbers is real, but it’s not a controlled comparison. Treat it as a rough directional signal, not a precise measurement.
The one honest tradeoff we think actually helps you decide
Foundayo does not have direct head-to-head data against injectable Ozempic or Wegovy, and its trial discontinuation rate due to side effects (~10%) is roughly double that of oral semaglutide (~5%) in the same study. If you need the most established evidence base for diabetes, cardiovascular, and kidney outcomes, or if tolerability is your single highest priority, injectable semaglutide is the safer known quantity.
But here’s the thing: if your real problem is that you haven’t started GLP-1 treatment because injections are a genuine barrier — or you want an on-label oral option for weight management at a lower cash price — that is exactly where Foundayo fits. A medication you’ll actually take consistently outperforms one you avoid, reduce, or skip.
How much do orforglipron and Ozempic cost right now?
Without insurance, Foundayo starts cheaper but the gap narrows at higher doses. With insurance and a savings card, both can hit $25/month. The real pricing story is more complex than the “$149/month” headline — here’s what we verified.
Foundayo official self-pay pricing (verified April 14, 2026)
| Dose | Monthly cost | Notes |
|---|---|---|
| 0.8 mg (starting) | $149 | Titration dose — most patients move up within 30 days |
| 2.5 mg | $199 | Second titration step |
| 5.5 mg | $299 | |
| 9 mg | $299 | |
| 14.5 mg | $299 with Journey Program / $349 without | Must refill within 45 days to keep $299 price |
| 17.2 mg (highest) | $299 with Journey Program / $349 without | Same refill timing requirement |
Source: foundayo.lilly.com/coverage-savings, verified April 14, 2026.
Ozempic official self-pay pricing (verified April 14, 2026)
| Dose | Monthly cost | Notes |
|---|---|---|
| 0.25 mg (starting) | $199 (intro offer) | Intro price for first 2 monthly fills; new patients only through June 30, 2026 |
| 0.5 mg | $199 (intro offer) | Same intro window |
| 0.25–1 mg (ongoing) | $349 | After intro period expires |
| 2 mg | $499 | Highest dose |
Source: ozempic.com and NovoCare pricing pages, verified April 14, 2026.
What you’d actually pay over 12 months
This projection accounts for the titration period and reflects what a real patient pays across a full year, not just the lowest advertised number.
| Scenario | Foundayo 12-mo total | Ozempic 12-mo total | Difference |
|---|---|---|---|
| Self-pay, Journey pricing maintained | ~$3,338 | ~$3,888 | Foundayo saves ~$550 |
| Self-pay, Journey pricing NOT maintained | ~$3,738 | ~$3,888 | Foundayo saves ~$150 |
| Self-pay at highest dose (17.2 mg vs 2 mg) | ~$3,338–$3,738 | ~$5,238 | Foundayo saves $1,500–$1,900 |
| Commercial insurance + savings card | ~$300 ($25/mo) | ~$300 ($25/mo) | Essentially equal |
The $149 headline is real but temporary. Most patients start at 0.8 mg and titrate upward. By month 3, you’re likely paying $299/month at a maintenance dose of 5.5 mg or higher. That’s still meaningfully cheaper than Ozempic’s $349–$499 without insurance, but it’s not “$149 forever.”
When Ro makes sense for access
Ro is a confirmed launch partner for Foundayo as of April 9, 2026, offering it at the same self-pay pricing ($149/mo starting dose) plus a Ro membership fee. Get started for $39, then as low as $74/month with annual plan paid upfront. Ro also carries Ozempic and other FDA-approved GLP-1 options with insurance support and prior authorization help. Ro makes the most sense if you want a single platform that can prescribe, check insurance, and coordinate access.
Check Foundayo or Ozempic pricing on Ro — $39 first month → (sponsored affiliate link, opens in a new tab)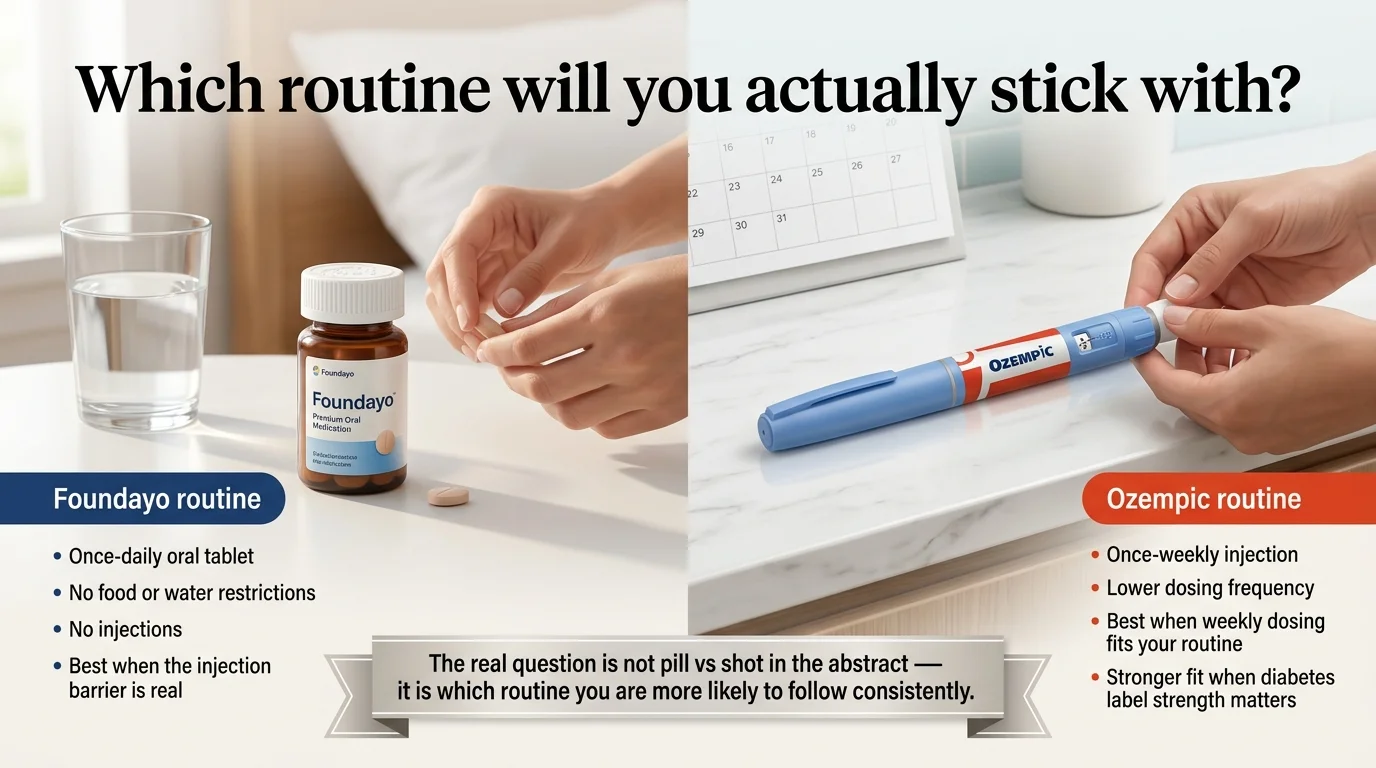
Side effects and safety: the honest comparison
Both medications cause GI side effects — nausea, vomiting, diarrhea, constipation — because that’s how the GLP-1 class works. But Foundayo’s rates ran higher in the one head-to-head trial, and its label includes practical warnings many comparison pages skip entirely.
GI side effects by the numbers (ACHIEVE-3 trial)
| Side effect | Orforglipron 36 mg | Oral semaglutide 14 mg |
|---|---|---|
| Any GI event | ~59% | ~37–45% |
| Nausea | Most common, mild-to-moderate | Most common, mild-to-moderate |
| Discontinued due to side effects | ~9–10% | ~4–5% |
| Timing of side effects | Primarily during dose escalation | Primarily during dose escalation |
| Resolution | Most resolved within 4–8 weeks | Most resolved within 4–8 weeks |
Important context: these rates come from a trial comparing orforglipron to oral semaglutide in type 2 diabetes patients. Side effect profiles in an obesity-only population may differ.
Foundayo-specific label warnings most pages miss
Oral contraceptives
The Foundayo prescribing information states that the effect on oral contraceptive absorption has not been evaluated in a clinical trial. Because Foundayo delays gastric emptying, the label advises patients using oral hormonal contraceptives to switch to a non-oral contraceptive method or add a barrier method for 30 days after starting Foundayo and for 30 days after each dose increase. Non-oral hormonal contraceptives (patch, ring, IUD, implant, injection) should not be affected.
Simvastatin interaction
The label advises against taking more than 20 mg of simvastatin daily while on Foundayo, because orforglipron increases the active metabolite of simvastatin.
Concomitant GLP-1 use
The Foundayo label states that concomitant use with another GLP-1 receptor agonist is not recommended. You would not take Foundayo and Ozempic at the same time.
Boxed warnings — similar but not identical
Both medications carry boxed warnings related to thyroid C-cell tumors. However, the label language differs in an important way:
- Foundayo’s boxed warning notes that orforglipron was not pharmacologically active in rats or mice and did not produce tumors in rodents. The warning is included because the GLP-1 receptor agonist class as a whole has shown this risk in rodent studies.
- Ozempic’s boxed warning states that semaglutide caused thyroid C-cell tumors (adenomas and carcinomas) in rodents at clinically relevant exposures.
Both drugs share the same contraindication: they should not be used by patients with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
For a more detailed look at Foundayo’s side effect profile, see our orforglipron side effects breakdown with trial data →
Important safety information and contraindications
This is not a substitute for reading the full prescribing information for either medication or talking with your prescriber. But if you’re comparing these two drugs, you should know the major caution flags from both labels in one place.
| Warning category | Foundayo label | Ozempic label |
|---|---|---|
| Acute pancreatitis | Yes — discontinue if suspected | Yes — discontinue if suspected |
| Acute kidney injury | Yes — related to dehydration from GI side effects | Yes — related to dehydration from GI side effects |
| Hypoglycemia | Yes — risk increases with concomitant insulin or secretagogues | Yes — risk increases with concomitant insulin or secretagogues |
| Serious hypersensitivity | Yes — discontinue if reaction occurs | Yes — discontinue if reaction occurs |
| Acute gallbladder disease | Yes | Yes |
| Pulmonary aspiration | Yes — consider for patients undergoing anesthesia/sedation | Yes — consider for patients undergoing anesthesia/sedation |
| Diabetic retinopathy | Yes — monitor in T2D patients | Yes — monitor in T2D patients |
| Pregnancy | May cause fetal harm; discontinue when pregnancy recognized | May cause fetal harm; not recommended during pregnancy |
| Severe hepatic impairment | Not recommended (Foundayo-specific) | Not listed as a specific restriction |
| Heart rate increase | Not specifically listed | Listed in Ozempic warnings |
This table is a summary, not a complete safety disclosure. Read the full prescribing information for Foundayo and Ozempic before starting either medication.
Is orforglipron just Ozempic in pill form?
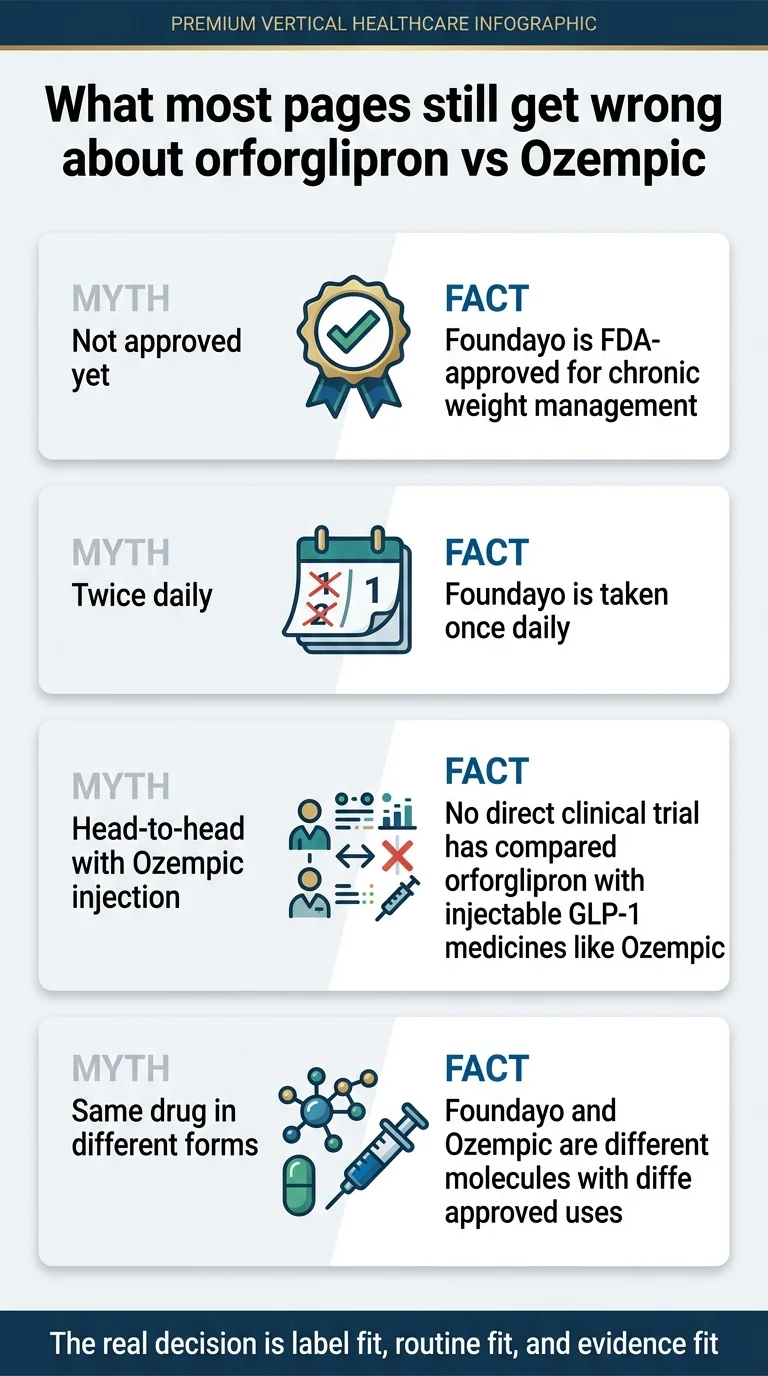
No. Foundayo and Ozempic are different molecules made by different companies with different FDA-approved uses. This is the single most common misconception we see in forums and on competing pages.
Orforglipron is a non-peptide small molecule discovered by Chugai Pharmaceutical and licensed by Eli Lilly. Semaglutide is a peptide-based molecule developed by Novo Nordisk. They both activate GLP-1 receptors, but they do it with fundamentally different molecular structures. This distinction has practical consequences:
- Absorption: Orforglipron’s small-molecule structure survives digestion easily, which is why Foundayo has no food or water restrictions. Peptide-based oral semaglutide requires an empty stomach and restricted water intake.
- Manufacturing: Small molecules are cheaper and simpler to produce at scale than peptides, which contributes to Foundayo’s lower pricing.
- Storage: Foundayo doesn’t require refrigeration. Ozempic pens do (before first use).
One freshness note for 2026
The “pill vs shot” framing is actually incomplete now. As of 2026, there are multiple oral semaglutide products: Rybelsus (T2D), Ozempic tablets (T2D), and the Wegovy pill (weight loss). Most searchers typing “orforglipron vs Ozempic” still mean the weekly Ozempic injection pen, and that’s what this comparison focuses on.
| Foundayo | Ozempic pen (injection) | Oral semaglutide (Rybelsus / Ozempic tablets / Wegovy pill) | |
|---|---|---|---|
| Molecule | Orforglipron (non-peptide) | Semaglutide (peptide) | Semaglutide (peptide) |
| Frequency | Once daily | Once weekly | Once daily |
| Food restrictions | None | N/A (injection) | Empty stomach, ≤4 oz water, wait 30 min |
| Approved for weight loss | Yes (Foundayo) | No | Wegovy pill: Yes. Rybelsus / Ozempic tablets: No |
Should you switch from Ozempic to orforglipron — or stay?
If Ozempic is working, your insurance covers it, and the injection isn’t a real barrier, the honest answer is: don’t switch. The case for switching is weaker than many pages suggest because you’d be moving from a medication with years of real-world data and established coverage to one that’s been available for less than two weeks.
When switching makes sense
- You’re using Ozempic off-label for weight loss and want an option that’s FDA-approved for that use — Foundayo gives you an on-label path that may improve insurance coverage odds
- Injection fatigue is real and getting worse — adherence matters more than theoretical efficacy, and if you’re skipping doses because you dread the needle, a daily pill you actually take beats a weekly shot you avoid
- You’re self-paying and the cost difference matters — at higher doses, Foundayo can save $1,500+/year compared to Ozempic self-pay pricing
- You travel frequently and the refrigeration/sharps disposal logistics are a genuine burden
When staying on Ozempic makes sense
- Your diabetes is well-controlled and your prescriber is satisfied with your A1C, CV risk markers, and kidney function
- Insurance covers it — switching means restarting the coverage and prior authorization process
- You value the evidence depth — Ozempic (initial U.S. approval: 2017) has years of real-world outcome data; Foundayo has strong clinical trial data but minimal real-world experience so far
- Side effect tolerance is good — if you’ve already passed the GI adjustment period on Ozempic, there’s no guarantee you won’t restart that process on a different medication
What Lilly’s own switch data shows
Lilly tested what happens when patients switch from injectable GLP-1s to Foundayo in the ATTAIN-MAINTAIN trial. The finding: patients who switched from injectable semaglutide (Wegovy) to orforglipron regained an average of about 2 pounds — broadly maintaining their prior weight loss. That’s reassuring, but it’s one trial, and “maintenance” isn’t the same as “continued improvement.” Switching requires a new prescription and medical supervision. These are different molecules — this is not like switching from brand to generic.
Not sure whether to start, switch, or stay? Get your personalized recommendation
Get My Recommendation →Will insurance cover Foundayo or Ozempic?
Coverage depends almost entirely on why you’re prescribed the medication. Foundayo’s weight-loss label and Ozempic’s diabetes label put them on different formulary tracks, and that distinction can mean the difference between $25/month and $349/month.
Foundayo insurance reality
Foundayo is FDA-approved for weight management — a category many insurers have historically excluded. That is changing as more plans add anti-obesity medication coverage, but it’s still inconsistent. If your commercial plan covers Foundayo, Lilly’s savings card can bring your cost to $25/month. The on-label weight management indication may actually help during prior authorization — you’re asking your insurer to cover a drug for exactly what it’s approved for.
Ozempic insurance reality
Ozempic’s diabetes indication means most commercial plans and Medicare Part D cover it when prescribed for type 2 diabetes. Coverage for off-label weight loss use is far less consistent. If your plan covers Ozempic for T2D, Novo Nordisk’s savings card can bring your cost to $25/month.
What to do if you’re denied
- Ask your prescriber to submit a prior authorization with clinical documentation
- If denied, file a formal appeal — many denials are overturned on first appeal
- Check whether the manufacturer’s patient assistance program applies to your situation
For a deeper walkthrough, see our guide to GLP-1 providers that help with prior authorization →
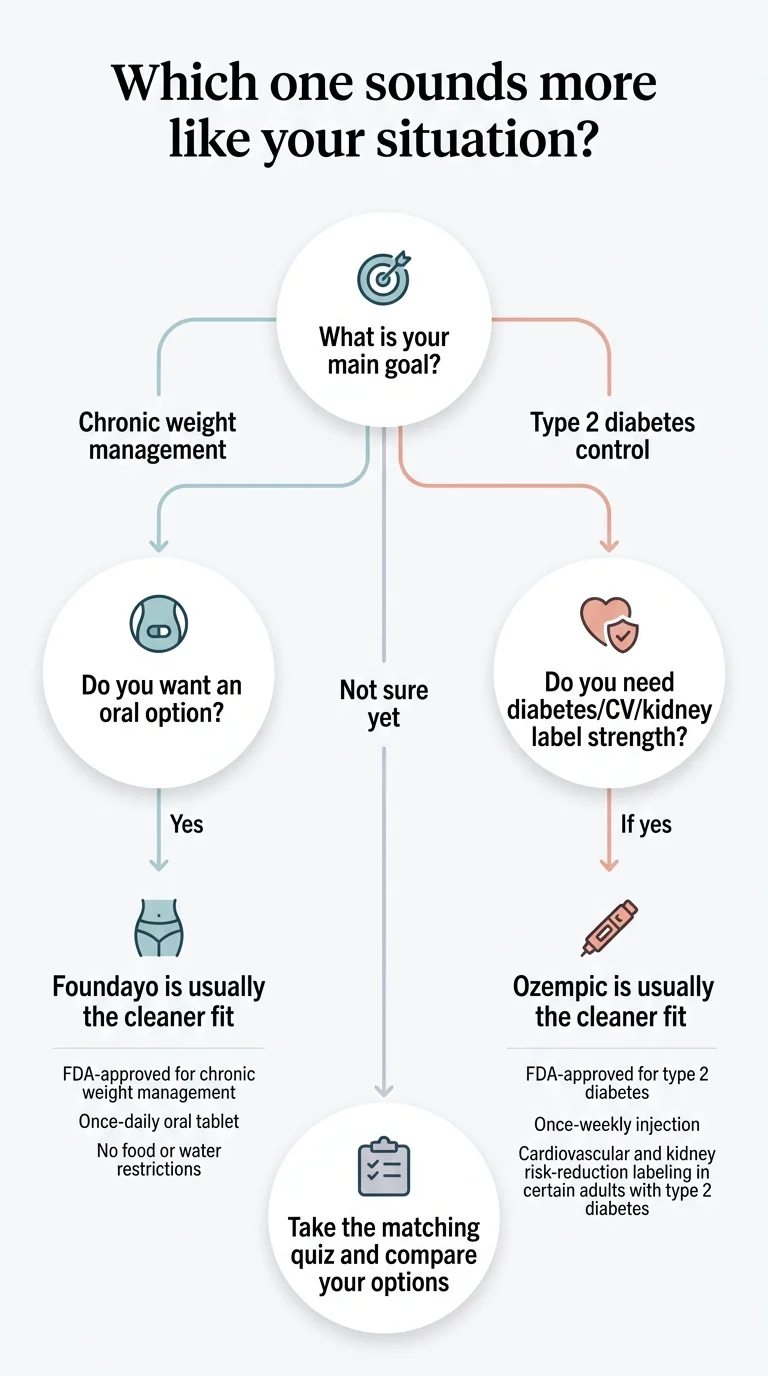
What the web still gets wrong about orforglipron vs Ozempic
We built this block because the search results for this query are riddled with stale claims. Every item below was verified against primary sources on .
| What other pages claim | What we verified | Why it matters |
|---|---|---|
| Orforglipron is "pending FDA approval" or "expected in 2026" | Foundayo was FDA-approved April 1, 2026. Available via LillyDirect since April 6, Ro and retail pharmacies since April 9. | If the page you're reading still says "not yet approved," everything else on it is suspect. |
| Foundayo is "twice daily" | Current FDA labeling is once daily. | Dosing errors on a health page are disqualifying. |
| Orforglipron has been tested head-to-head against Ozempic injection | Lilly states there are no trials comparing orforglipron with injectable GLP-1s. ACHIEVE-3 compared it to oral semaglutide (14 mg) only. | This is the biggest honesty test for any comparison page. |
| "Foundayo costs $149/month" (full stop) | $149 is the lowest titration dose (0.8 mg). Maintenance doses are $299/month with Journey pricing, or $349 without. | Headlines that stop at $149 create price disappointment. |
| "Pill vs shot" is the whole comparison | The real comparison is label fit (weight loss vs T2D/CV/kidney). In 2026, Ozempic also has tablet prescribing information. | The old framing is incomplete and misleading. |
| Foundayo and Ozempic are "the same drug in different forms" | Different molecules (orforglipron vs semaglutide), different manufacturers (Lilly vs Novo Nordisk), different approved uses. | Getting this wrong undermines everything else. |
How to access Foundayo or Ozempic today
Both medications require a prescription. Here’s where to get each one as of April 2026.
| LillyDirect (Foundayo) | Ro | NovoCare (Ozempic) | Retail pharmacy | |
|---|---|---|---|---|
| Medications | Foundayo only | Foundayo, Ozempic, other GLP-1s | Ozempic only | Both |
| Membership/consultation fee | None | $39 first month, then as low as $74/mo (annual plan) | None | None (but need existing Rx) |
| Medication pricing | Self-pay per dose table above | Same manufacturer pricing (separate from membership) | Self-pay per dose table above | Varies; insurance or GoodRx |
| Delivery | Free home shipping | Foundayo: home delivery. Ozempic: pharmacy pickup. | Pharmacy pickup with savings card | In-store pickup |
| Insurance check | Auto-checks coverage routinely | Yes, with PA support | Savings card for insured patients | Through your plan |
| Best for | Self-pay Foundayo patients | Patients wanting one platform for consultation + Rx + insurance support | Insured Ozempic patients | Patients with existing prescriptions |
LillyDirect — Foundayo direct
No membership fee. Free home shipping. Automatic insurance check. Best for self-pay Foundayo patients going direct to Lilly.
Start with LillyDirect → (sponsored affiliate link, opens in a new tab)Ro — one platform for both
$39 first month. Covers Foundayo and Ozempic. Insurance support and prior authorization help included. Best for patients wanting a single platform.
Check eligibility on Ro — $39 to start → (sponsored affiliate link, opens in a new tab)Who should NOT choose each one
The fastest way to earn your trust is to tell you who each option is wrong for.
Foundayo is not the best fit if:
- ✗Your primary medical need is type 2 diabetes management
- ✗You need cardiovascular or kidney risk-reduction evidence
- ✗You cannot reliably take a daily medication
- ✗You’re on oral hormonal contraceptives and cannot easily switch methods
- ✗You need the most established long-term GLP-1 evidence base
Ozempic is not the best fit if:
- ✗Your primary goal is weight loss without type 2 diabetes — you’d be using it off-label
- ✗Injections are a genuine barrier to starting or continuing treatment
- ✗You’re self-paying and cost is the deciding factor at maintenance doses
- ✗You want an on-label oral GLP-1 specifically
What we actually verified — April 14, 2026
| Claim on this page | Source verified | Date checked |
|---|---|---|
| Foundayo FDA approval for weight management | FDA.gov press announcement + Eli Lilly investor press release | April 14, 2026 |
| Foundayo self-pay pricing by dose | foundayo.lilly.com/coverage-savings (official Lilly pricing page) | April 14, 2026 |
| Foundayo Journey Program refill timing ($299 vs $349) | foundayo.lilly.com/coverage-savings terms and conditions | April 14, 2026 |
| Ozempic self-pay pricing + intro offer window | ozempic.com savings page + NovoCare (offer valid through June 30, 2026) | April 14, 2026 |
| ACHIEVE-3 trial results (orforglipron vs oral semaglutide) | Lancet publication, February 2026 | April 14, 2026 |
| ATTAIN-1 trial results (Foundayo weight loss data) | Eli Lilly press release + FDA label | April 14, 2026 |
| ATTAIN-MAINTAIN trial (switch/maintenance data) | Eli Lilly press release, December 2025 | April 14, 2026 |
| No direct trial between Foundayo and injectable Ozempic | Eli Lilly clinical trial disclosures | April 14, 2026 |
| Ro availability of Foundayo ($149/mo + membership) | Ro press release (April 9, 2026) + ro.co/weight-loss | April 14, 2026 |
| Ozempic approved indications (T2D, CV, kidney) | FDA prescribing information for Ozempic | April 14, 2026 |
| Foundayo oral contraceptive / gastric emptying warning | Foundayo FDA prescribing information, Section 7.3 and 8.3 | April 14, 2026 |
| Foundayo boxed warning language | Foundayo FDA prescribing information, boxed warning | April 14, 2026 |
Frequently asked questions
Is orforglipron better than Ozempic for weight loss?
For weight-loss-first readers without type 2 diabetes, Foundayo (orforglipron) is usually the better fit because it is FDA-approved specifically for chronic weight management and costs less without insurance ($149–$349/mo depending on dose vs $349–$499/mo for Ozempic). Ozempic is not FDA-approved for weight loss. In their respective clinical trials, injectable semaglutide at weight-loss doses (Wegovy 2.4 mg) showed somewhat higher average weight loss (~15%) than orforglipron (~12%), but those numbers come from different trials and are not directly comparable.
Is orforglipron the same as Ozempic in pill form?
No. Orforglipron (Foundayo) and semaglutide (Ozempic) are different molecules made by different companies. They both activate GLP-1 receptors but have different molecular structures, different approved uses, and different manufacturers. The actual pill versions of semaglutide are Rybelsus (for T2D), Ozempic tablets (for T2D), and the Wegovy pill (for weight loss).
Is Ozempic better if I have type 2 diabetes?
Generally yes. Ozempic is FDA-approved for type 2 diabetes with additional indications for cardiovascular and kidney risk reduction in certain adults with T2D. Foundayo is currently approved only for weight management and does not yet have a diabetes indication — though Eli Lilly plans to seek one later in 2026.
Is there a direct head-to-head trial of orforglipron vs Ozempic?
No. Eli Lilly states there are no clinical trials comparing orforglipron directly with injectable GLP-1 medications like Ozempic. The ACHIEVE-3 trial compared orforglipron to oral semaglutide tablets (7 mg and 14 mg doses) — not the weekly Ozempic injection pen. Cross-trial comparisons provide rough directional guidance but are not controlled evidence.
Which is cheaper without insurance?
Foundayo starts at $149/month for the lowest dose (0.8 mg) and reaches $299/month at maintenance doses with Journey pricing, or $349 without. Ozempic starts at $199/month (intro offer through June 30, 2026) and reaches $349/month (≤1 mg) or $499/month (2 mg). Over 12 months targeting maintenance dose, Foundayo typically costs $550–$1,900 less than Ozempic depending on dose.
Can you switch from Ozempic to orforglipron?
Yes, but only under medical supervision with a new prescription. These are different molecules, not different forms of the same drug. Lilly's ATTAIN-MAINTAIN trial showed patients who switched from injectable semaglutide to orforglipron broadly maintained their prior weight loss (average regain ~2 lbs). Concomitant use with another GLP-1 receptor agonist is not recommended per the Foundayo label.
Are side effects worse with the pill?
In the ACHIEVE-3 trial, orforglipron had higher rates of GI side effects (~59% vs ~37–45%) and higher discontinuation due to adverse events (~9–10% vs ~4–5%) compared to oral semaglutide. Side effects were primarily gastrointestinal, occurred mainly during dose escalation, and were mostly mild to moderate. The Foundayo label also includes a specific warning about potential impact on oral contraceptive absorption that Ozempic does not share.
What if I want semaglutide but don't want injections?
Oral semaglutide options in 2026 include the Wegovy pill (FDA-approved for weight loss), Rybelsus (for T2D), and Ozempic tablets (for T2D). All require empty-stomach dosing with restricted water intake. Foundayo is a different molecule (orforglipron) with no food or water restrictions — but it is not semaglutide.
Still not sure which GLP-1 program is right for you?
Take our free 60-second matching quiz and get a personalized recommendation based on your goals, insurance status, and preferences.
Take the Free 60-Second Quiz →Related guides
- Orforglipron side effects — trial data and what to expect
- Orforglipron vs Wegovy pill — comparing two oral weight loss options
- Foundayo prior authorization — how to get approved
- Best GLP-1 providers that help with prior authorization
- Best brand-name GLP-1 telehealth providers compared
- Compounded GLP-1 alternatives — what to know in 2026
- Foundayo providers that accept HSA and FSA