Last verified: April 21, 2026 · FDA status checked · Pricing verified · Trial data confirmed
Comparison Guide · April 21, 2026
CagriSema vs Ozempic: Which One Actually Matters Right Now?
Published:
By The RX Index Editorial Team — a pricing intelligence and comparison resource for GLP-1 telehealth providers
The short answer
CagriSema looks like a stronger future GLP-1 than Ozempic on the numbers — but the “CagriSema beat Ozempic” headline isn’t quite what it sounds like. In REIMAGINE 2, CagriSema was compared against semaglutide 2.4 mg. Ozempic’s FDA-approved maximum dose is 2 mg weekly. That’s a real difference most articles skip.
The bigger real-world issue: CagriSema is not FDA-approved. You can’t fill it at any U.S. pharmacy. For anyone who needs treatment in the next 90 days, Ozempic — or a stronger option like Zepbound — is the only real answer.
- Future efficacy signal: CagriSema
- What you can actually do today: Ozempic (or Zepbound)
- The catch most pages miss: Semaglutide 2.4 mg (trial comparator) ≠ Ozempic 2 mg (real-world max)
 (sponsored affiliate link, opens in a new tab)
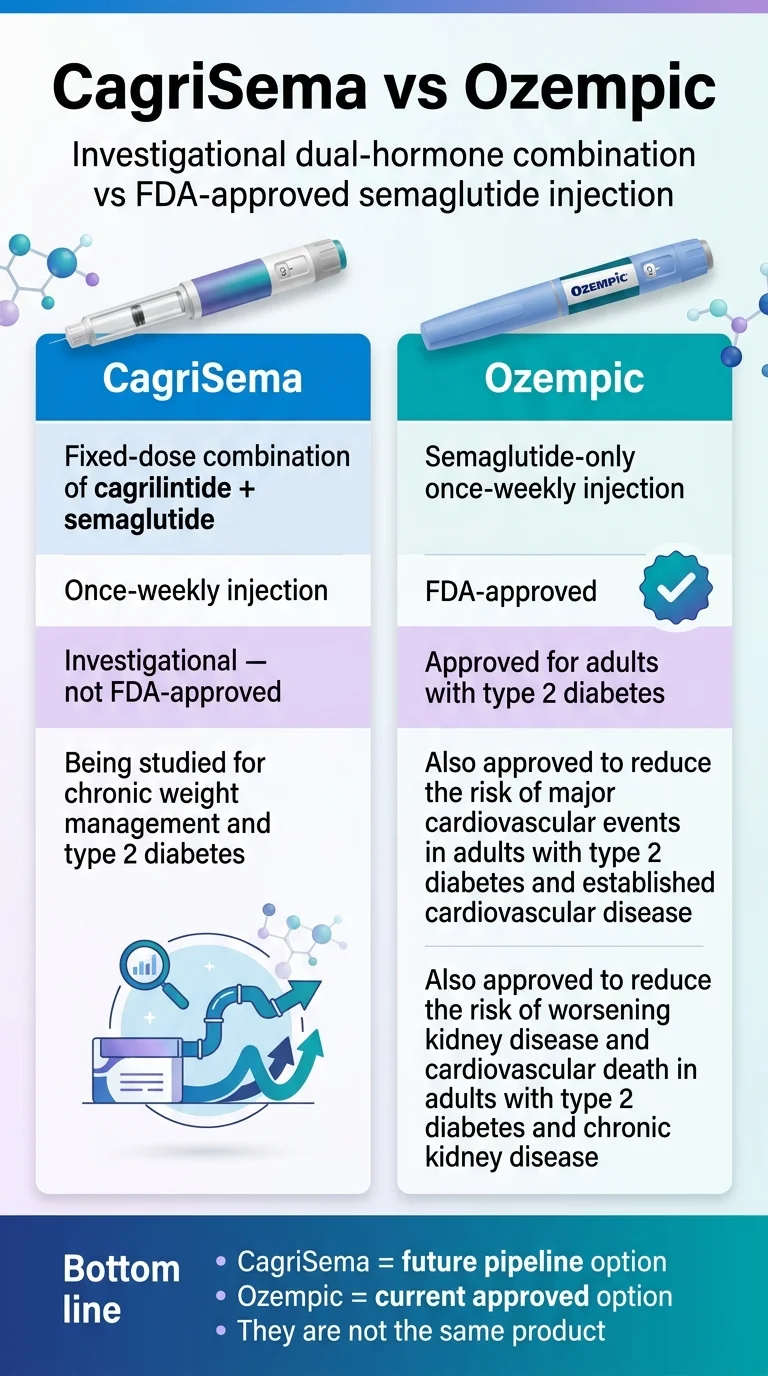
(sponsored affiliate link, opens in a new tab)CagriSema = future pipeline option. Ozempic = current approved option. They are not the same product.
CagriSema vs Ozempic: the 15-second answer
If you mean future upside, CagriSema is the more interesting drug. If you mean what should I do right now, Ozempic wins — because it’s FDA-approved, it’s available, and it has a mature label covering type 2 diabetes, major cardiovascular event reduction, and kidney disease progression. CagriSema is still investigational with no approved label yet.
Both drugs come from Novo Nordisk. Both are once-weekly injections. Both contain semaglutide. But they are not the same drug, they are not approved for the same things, and only one of them is something you can start this month.
For the broader picture on how CagriSema works and its full trial program, see our What Is CagriSema? guide. This page stays focused on the Ozempic decision.
CagriSema vs Ozempic at a glance: the reality-check table
Most comparison pages bury the actual decision data under 1,500 words of generic explainer. Every row of this table answers a specific decision question.
| Decision question | CagriSema | Ozempic | Why this matters |
|---|---|---|---|
| What is it? | Fixed-dose combination of cagrilintide (amylin analog) + semaglutide, once weekly | Semaglutide-only, once weekly | CagriSema is not “Ozempic with a new name” — it’s a two-drug combination. |
| FDA status today | Not approved. NDA filed Dec 18, 2025. Review anticipated 2026. | Approved for T2D (2017), CV risk reduction in diabetics with heart disease (2020), kidney disease + CV death in diabetics with CKD (Jan 2025) | This is the single most important difference. |
| Can you fill it at a U.S. pharmacy today? | No. | Yes. | Most “CagriSema vs Ozempic” searches are really this question in disguise. |
| What is it approved for? | Nothing yet. Filing is for chronic weight management in adults with obesity or overweight + at least one weight-related condition. | Type 2 diabetes; CV risk reduction in diabetics with heart disease; kidney disease progression and CV death reduction in diabetics with CKD. | Ozempic is not approved for weight loss. Wegovy — same molecule, higher dose — is. |
| Best obesity trial result | 22.7% mean weight loss at 68 weeks (adherent); 20.4% intention-to-treat (REDEFINE 1) | Not tested at a weight-loss dose. Wegovy (2.4 mg) produces 14.9–17.4% across STEP trials. | Ozempic maxes out at 2 mg. A clean obesity comparison is CagriSema vs Wegovy-dose semaglutide. |
| Best diabetes trial result (head-to-head) | 14.2% weight loss + 1.91-point HbA1c reduction at 68 weeks (REIMAGINE 2) | 10.2% weight loss + 1.76-point HbA1c reduction — but at semaglutide 2.4 mg, not Ozempic’s 2 mg max | The headline “CagriSema beat Ozempic” uses a dose higher than Ozempic’s FDA maximum. |
| Head-to-head vs tirzepatide (Zepbound) | 23.0% vs tirzepatide’s 25.5% in REDEFINE 4 at 84 weeks. Non-inferiority not met. | Not tested head-to-head vs tirzepatide as Ozempic. | Tirzepatide is already approved and available now. |
| GI side-effect signal | 79.6% of CagriSema patients had GI events in REDEFINE 1 (vs 39.9% placebo). ~5.9% discontinued. | Labeled: nausea, vomiting, diarrhea, abdominal pain, constipation. Boxed warning for thyroid C-cell tumors. | CagriSema trial data ≠ FDA-approved label. Final warnings will be established at approval. |
| Price you can verify today | Not announced. [Needs verification at launch] | List: $1,027.51/pen. NovoCare self-pay: $349/mo (0.25–1 mg) or $499/mo (2 mg). Intro offer $199/mo for first 2 fills through June 30, 2026. Commercially insured as low as $25/mo via Savings Card. | Ozempic pricing is published and verifiable. CagriSema’s isn’t yet. |
| Realistic U.S. availability | FDA decision anticipated in 2026. Post-approval commercial launch timing not publicly confirmed. | Available now at retail pharmacies and through NovoCare. | Even if approval lands on schedule, new drugs typically reach pharmacies only after manufacturing ramp and distribution setup. |
Sources: Novo Nordisk press releases (Dec 18, 2025; Feb 2, 2026; Feb 23, 2026); NEJM REDEFINE 1 (June 22, 2025); Ozempic prescribing information (2025 label); NovoCare Pharmacy pricing pages; STEP trial program. Last verified April 21, 2026.
The catch most pages miss: semaglutide 2.4 mg is not Ozempic 2 mg
This is the thing almost nobody explains clearly. We’re going to.

Semaglutide 2.4 mg (the trial comparator) aligns more closely with Wegovy dosing than Ozempic dosing.
REIMAGINE 2 (Novo Nordisk press release, February 2, 2026) compared multiple doses of each drug. The headline comparison was CagriSema 2.4/2.4 mg versus semaglutide 2.4 mg. Here’s where it gets tangled: semaglutide 2.4 mg is not Ozempic. It’s the Wegovy dose. Ozempic’s FDA label lists doses of 0.25 mg (start), 0.5 mg, 1 mg, or 2 mg — and 2 mg is the maximum.
So when you read “CagriSema beat Ozempic,” what actually happened is:
- CagriSema 2.4/2.4 mg produced 14.2% weight loss and a 1.91-point HbA1c reduction at 68 weeks.
- Semaglutide 2.4 mg produced 10.2% weight loss and a 1.76-point HbA1c reduction at 68 weeks.
- The trial did not test Ozempic’s actual maximum FDA-approved dose of 2 mg.
Does that invalidate the comparison? No — it’s still evidence that a higher dose of semaglutide plus cagrilintide outperforms the same higher dose of semaglutide alone. But it changes how a real person should think about it:
- For diabetes at maximum Ozempic dose (2 mg), we don’t have clean head-to-head data.
- For weight loss at Wegovy-equivalent dose (2.4 mg), CagriSema produced roughly 4 percentage points more weight loss over 68 weeks.
- For obesity in general, CagriSema’s best result (22.7% in REDEFINE 1) beats Wegovy’s STEP benchmark (14.9–17.4%) by about 5–8 percentage points.
Meaningful? Yes. Revolutionary? Not quite. And there’s a reason it feels smaller than the news suggested — Novo Nordisk had publicly pointed toward a 25% weight-loss target for CagriSema. When REDEFINE 1 came back at 22.7%, Novo’s shares fell sharply. The efficacy is real. The “next-gen Ozempic killer” framing was investor theater.
Does CagriSema actually beat Ozempic on the numbers?
On trial signal, yes — CagriSema produced more weight loss and slightly better A1C reduction than semaglutide alone in head-to-head studies. But the comparator dose was 2.4 mg of semaglutide, higher than Ozempic’s 2 mg maximum, so “beat Ozempic” slightly overstates the case. Here’s the full breakdown.
REDEFINE 1 — obesity, no diabetes (NEJM, June 2025)
3,417 adults with obesity or overweight plus at least one weight-related condition. No diabetes. 68 weeks.
- CagriSema 2.4/2.4 mg: 22.7% mean weight loss (trial-product estimand, adherent patients). 20.4% in the full intention-to-treat population.
- Semaglutide 2.4 mg alone: 16.1% weight loss.
- Cagrilintide 2.4 mg alone: 11.8% weight loss.
- Placebo: ~2.3% weight loss.
Proportions reaching thresholds (adherent patients): ≥20% weight loss in 60.2%, ≥25% in 40.4%, ≥30% in 23.1%. One important detail: approximately 57% of participants actually reached the full 2.4/2.4 mg target dose. Real-world patients who can’t tolerate the full dose will likely see something closer to 20.4% than 22.7%.
REDEFINE 2 — obesity + type 2 diabetes (Novo Nordisk, March 2025)
1,207 adults with overweight or obesity AND type 2 diabetes. 68 weeks.
- CagriSema: 15.7% weight loss vs 3.1% for placebo.
- 89.7% of CagriSema patients reached ≥5% weight loss vs 30.3% on placebo.
- Roughly 74% achieved an HbA1c of 6.5% or lower.
REIMAGINE 2 — head-to-head vs semaglutide 2.4 mg (Novo Nordisk, February 2026)
2,728 adults with type 2 diabetes inadequately controlled on metformin. 68 weeks. The headline comparison:
- CagriSema 2.4/2.4 mg: 14.2% weight loss, 1.91-point HbA1c reduction.
- Semaglutide 2.4 mg: 10.2% weight loss, 1.76-point HbA1c reduction.
- 43% of CagriSema patients reached ≥15% weight loss; 24% reached ≥20%.
- No weight-loss plateau observed through 68 weeks on CagriSema.
The HbA1c advantage (1.91 vs 1.76) is statistically significant but clinically narrow. The weight-loss advantage (14.2 vs 10.2) is more meaningful. Both are real.
REDEFINE 4 — head-to-head vs tirzepatide (Novo Nordisk, February 23, 2026)
This is the one that should make you think twice before waiting for CagriSema. 809 adults with obesity plus at least one comorbidity. 84 weeks. CagriSema versus tirzepatide (Zepbound) 15 mg.
- CagriSema: 23.0% weight loss (all-adherent); 20.2% (treatment-regimen estimand).
- Tirzepatide 15 mg: 25.5% (all-adherent); 23.6% (treatment-regimen).
- Non-inferiority endpoint: not met.
Tirzepatide is not hypothetical. It’s Zepbound. It’s the drug your pharmacist can put on the counter today. And it produced more weight loss than CagriSema in a direct head-to-head trial, not a cross-trial inference. If maximum weight loss is your goal, the better drug is available now. See our CagriSema vs Zepbound guide for the full breakdown.
What Ozempic actually does at its approved doses
Ozempic at its approved diabetes doses (0.25–2 mg): in the 2 mg dose studied in the SUSTAIN FORTE trial, approximately 6.9 kg mean weight loss over 40 weeks. HbA1c reduction of roughly 1.4–1.9 percentage points depending on baseline and comparator. Ozempic is designed to control blood sugar, not produce maximum weight loss — which is part of why Wegovy (semaglutide at 2.4 mg) was developed as a separate product for obesity.
If that answered your main question and you want to know what you can actually do now —
No signup. No email wall. Built as a decision tool, not a lead magnet.
Is CagriSema approved or available yet? No.
December 18, 2025. Novo Nordisk submitted its NDA for CagriSema in adults with obesity or overweight plus at least one weight-related comorbidity. The submission was based on REDEFINE 1 and REDEFINE 2. Under a standard FDA review timeline, the agency has about 10 months to issue a decision from NDA acceptance. Novo Nordisk has publicly stated FDA review is expected in 2026.
1. Approval date and pharmacy launch date are not the same thing
Even after FDA approval, new drugs take time to reach retail pharmacies at meaningful scale. Manufacturing ramp, distribution contracts, and payer formulary reviews don’t happen instantly. Novo Nordisk has not publicly announced a commercial launch date for CagriSema. Anyone telling you “CagriSema will be on shelves by [specific date]” is guessing.
2. The diabetes indication is a separate filing that hasn’t happened yet
Novo Nordisk’s December 2025 filing is for weight management only. The company has said it will approach the FDA about a type 2 diabetes indication after results from REIMAGINE 1 and REDEFINE 3 (the 7,000-patient cardiovascular outcomes trial). That timeline has not been publicly stated.
3. Compounded cagrilintide doesn’t qualify under FDA compounding rules
You may see “CagriSema” or “cagrilintide” advertised by research peptide suppliers or compounding operations. To be compounded for human use under FDA rules, a drug substance generally needs to meet one of a small set of criteria — appearing on FDA’s Section 503A bulk drug substances list, being a component of an FDA-approved drug, or having a USP/NF monograph. Cagrilintide currently does not meet those criteria. Compounding pharmacies and research suppliers advertising cagrilintide for human use are operating outside FDA’s compounding framework.
Should you wait for CagriSema or act now?
For most people who need treatment within the next several months, waiting is the wrong call. CagriSema is at best a year or more from pharmacy shelves, and the incremental efficacy gain over approved options (~4–7 percentage points more weight loss) rarely justifies another year of untreated metabolic disease. Here’s the framework, built profile by profile.
| Your situation | Honest answer | Why |
|---|---|---|
| Type 2 diabetes, not at A1C goal | Start an approved GLP-1 now. | Every month at elevated A1C accumulates vascular and kidney risk. CagriSema isn’t filed for diabetes yet. Ozempic already has labeled CV and kidney benefits. |
| Obesity (BMI ≥30), seeking weight loss, no current treatment | Start Wegovy or Zepbound now. | Wegovy produces 14–17% weight loss and is approved. Zepbound produces ~20–25%. Waiting a year for a 5–7 point improvement rarely beats starting treatment now. |
| BMI 27–29 with a comorbidity | You’re eligible for approved weight-loss GLP-1s today. | Wegovy and Zepbound both cover this group. CagriSema’s filing uses the same threshold. |
| Plateaued on Wegovy after 6–12 months | Talk to your prescriber about switching to Zepbound first. | A switch to tirzepatide is available now and has direct head-to-head data against CagriSema (where tirzepatide came out ahead). |
| Established cardiovascular disease + type 2 diabetes | Start Ozempic now. | Ozempic is FDA-approved for CV risk reduction in exactly this group. CagriSema has not reported CV outcomes data. This is not close. |
| Severe obesity (BMI ≥40) | Start treatment now, not later. | Time-to-treatment matters more than efficacy percentage points at this BMI. CagriSema didn’t beat Zepbound in REDEFINE 4 anyway. |
| Stable, in maintenance, just following the pipeline | Keep watching. No action needed. | You’re the one group where “waiting” genuinely makes sense — because you don’t need to act. |
Editorial framework based on current FDA labeling and published Phase 3 data. Not medical advice. Confirm any treatment decision with your clinician. Last verified April 21, 2026.
The math is simple. CagriSema’s apparent advantage in obesity trials is 5–8 percentage points of weight loss beyond Wegovy-dose semaglutide. Real-world patients who can’t tolerate the full 2.4/2.4 mg dose — and roughly 43% of REDEFINE 1 participants didn’t — will see less. And the drug that actually beat CagriSema in REDEFINE 4 — tirzepatide, sold as Zepbound — is already on pharmacy shelves today.
Still not sure which path fits you? One question, one answer, personalized to your goals, insurance situation, and what you’ve already tried.
If you have type 2 diabetes, which one matters more today?
Ozempic, clearly. It’s the only one approved for type 2 diabetes, and its label already includes three benefits CagriSema hasn’t demonstrated in published data.
Ozempic’s current FDA-labeled indications (prescribing information, 2025)
- Glycemic control in type 2 diabetes (since 2017).
- Reducing major adverse cardiovascular events (heart attack, stroke, CV death) in adults with type 2 diabetes and known cardiovascular disease (2020).
- Reducing the risk of kidney disease progression, kidney failure, and CV death in adults with type 2 diabetes and chronic kidney disease (January 2025, based on the FLOW trial).
CagriSema is approved for zero things. The U.S. filing is for chronic weight management only. It has not been filed for diabetes. Its cardiovascular outcomes trial (REDEFINE 3, 7,000 patients) is active but has not reported results.
For a patient with type 2 diabetes, the Ozempic-vs-CagriSema question isn’t “which drug is stronger.” It’s “which drug is approved for what I actually need?” Right now, for every diabetes use case, that’s Ozempic. See our Best GLP-1 for Diabetes guide for the full breakdown.
If your real goal is weight loss, is Ozempic even the right comparator?
Probably not. Ozempic is semaglutide, but its FDA label is for diabetes-related uses — not weight management. Wegovy uses the same molecule at a higher dose (2.4 mg) and is specifically approved for chronic weight management. If your real question is “CagriSema vs semaglutide for weight loss,” the cleaner comparison is against Wegovy, not Ozempic.
For a clean weight-loss comparison, here are the current benchmarks:
- Wegovy (semaglutide 2.4 mg): ~14.9% weight loss at 68 weeks in STEP 1. ~15.2% at 104 weeks in STEP 5.
- Wegovy HD (semaglutide 7.2 mg): 20.7% weight loss at 72 weeks in STEP UP. ~33% of patients reached ≥25%.
- Zepbound (tirzepatide): ~20.2% at 72 weeks in SURMOUNT-5. Up to 25.5% at 84 weeks in REDEFINE 4.
- CagriSema: 22.7% (adherent) or 20.4% (intention-to-treat) at 68 weeks in REDEFINE 1.
CagriSema lands in the top tier — but so do Wegovy HD and Zepbound, both of which are already approved and available. The gap between CagriSema and the best currently-available options is roughly 2–8 percentage points, depending on which comparator and which trial. See our CagriSema vs Wegovy guide for that specific head-to-head.
CagriSema vs Ozempic side effects: what to expect
Ozempic has an FDA-approved label that lists specific warnings and common side effects. CagriSema has trial data showing higher GI event rates than placebo and somewhat higher rates than semaglutide alone — but it has no FDA label yet, so its final warnings and precautions are not established.
Ozempic’s current FDA-labeled warnings
- Thyroid C-cell tumors (boxed warning). In rodent studies, semaglutide caused thyroid C-cell tumors. Do not use Ozempic if you or a family member has ever had medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- Pancreatitis. Rare but serious. Stop the drug for severe, persistent stomach pain.
- Diabetic retinopathy complications. Monitored in diabetics.
- Acute kidney injury. Particularly with severe GI events.
- Hypoglycemia. Risk rises when combined with insulin or sulfonylureas.
- Gallbladder problems, including gallstones and inflammation.
- Delayed gastric emptying. Can affect absorption of other oral medications.
Common side effects per the Ozempic label: nausea, vomiting, diarrhea, abdominal pain, constipation. Source: Ozempic prescribing information, FDA DailyMed, 2025 label.
CagriSema’s trial-reported adverse events
| Event | CagriSema (REDEFINE 1) | Placebo (REDEFINE 1) |
|---|---|---|
| Any gastrointestinal adverse event | 79.6% | 39.9% |
| Discontinuation due to adverse events | ~5.9% | ~3.7% |
| Serious adverse events | ~1 in 10 patients | ~1 in 16 patients |
| Reached full 2.4/2.4 mg target dose | ~57% | — |
Source: Garvey WT et al., NEJM 2025 (REDEFINE 1 publication); Novo Nordisk public remarks on dose-reaching. A meta-analysis of four randomized CagriSema trials (4,419 patients total, ScienceDirect, February 2026) found GI adverse events occurred 32% more often on CagriSema than on semaglutide alone.
CagriSema is investigational. Its final FDA-approved label — including any boxed warnings, precautions, and listed adverse reactions — will be established at approval, not before. Post-marketing real-world safety data doesn’t exist yet. Because semaglutide is one of CagriSema’s two components, semaglutide-class warnings like the thyroid C-cell tumor signal are likely to carry over — but “likely to carry over” is not “currently on an FDA-approved label.”
How much do CagriSema and Ozempic cost?
CagriSema’s price has not been announced. Novo Nordisk typically doesn’t announce launch pricing until FDA approval. Here’s what you can actually verify for Ozempic today.
Ozempic — what you actually pay today
| Pathway | Cost | Notes |
|---|---|---|
| List price (WAC) | $1,027.51/pen | Most people don’t pay this. |
| Commercial insurance + Ozempic Savings Card | As low as $25/mo | Up to $100 max savings/mo, up to 48 months. Government beneficiaries excluded. |
| NovoCare self-pay (0.25, 0.5, or 1 mg) | $349/mo | Standard tier |
| NovoCare self-pay (2 mg) | $499/mo | Maximum approved dose tier |
| NovoCare new self-pay intro offer | $199/mo | First 2 monthly fills of 0.25 or 0.5 mg; through June 30, 2026 |
| Novo Nordisk Patient Assistance Program | Free | Uninsured patients at or below 200% of the Federal Poverty Level (~$31,200/individual in 2026) |
Source: NovoCare Pharmacy pricing pages and Ozempic.com Savings Card terms, accessed April 2026. Note: Novo Nordisk announced a list price reduction to $675 effective January 1, 2027. The self-pay channel prices ($349/$499) are explicitly unchanged by that list-price move.
CagriSema — what we don’t know yet
Novo Nordisk has not announced CagriSema pricing. Based on Novo’s pattern with Wegovy and Ozempic, a list price in the $900–$1,350 range at launch seems plausible, with self-pay pricing likely somewhere between $349 and $599. That is an educated inference, not a verified number. We’ve flagged it clearly as unverified.
The answer to “Should I wait for CagriSema to maybe be cheaper?” is: No. CagriSema will not be cheaper at launch. Waiting for a hypothetically lower price on a drug that isn’t launched yet is a bad bet.
What we actually verified for this page
- ✅ CagriSema NDA filing date (Dec 18, 2025) — Novo Nordisk press release via PR Newswire.
- ✅ REDEFINE 1 results (22.7% adherent; 20.4% intention-to-treat) — NEJM publication (June 22, 2025).
- ✅ REDEFINE 2 results (15.7% weight loss) — Novo Nordisk announcement (March 10, 2025).
- ✅ REIMAGINE 2 head-to-head (14.2% vs 10.2% weight; 1.91 vs 1.76 HbA1c) — Novo Nordisk press release (February 2, 2026).
- ✅ REDEFINE 4 results (23.0% vs tirzepatide 25.5%; non-inferiority not met) — Novo Nordisk announcement via GlobeNewswire (February 23, 2026).
- ✅ Ozempic FDA indications (T2D 2017, CV 2020, CKD January 2025) — Ozempic prescribing information (2025 label) and Novo Nordisk CKD approval announcement (January 28, 2025).
- ✅ Ozempic list price ($1,027.51) and self-pay tiers — NovoCare Pharmacy pricing pages and Ozempic.com Savings Card terms (accessed April 2026).
- ✅ Semaglutide injection shortage resolved Feb 21, 2025 — FDA Drug Shortage database declaratory order.
- ⚠️ CagriSema pricing — not announced; flagged as unverified.
- ⚠️ Exact CagriSema PDUFA date / commercial launch date — not publicly confirmed; FDA review anticipated in 2026 per Novo Nordisk.
Frequently asked questions
Is CagriSema FDA-approved yet?
No. As of April 21, 2026, CagriSema is not FDA-approved. Novo Nordisk filed its New Drug Application for chronic weight management on December 18, 2025. FDA review is anticipated in 2026.
Is CagriSema actually better than Ozempic?
On trial signal, CagriSema produced more weight loss and a narrowly better HbA1c reduction than semaglutide alone in head-to-head studies. But the comparator was semaglutide 2.4 mg — higher than Ozempic’s FDA-approved maximum dose of 2 mg weekly. The “better than Ozempic” framing is directionally reasonable but not a label-to-label comparison. CagriSema is closer to “better than Wegovy-dose semaglutide” than to “better than Ozempic.”
Can you get CagriSema now?
No. It is not FDA-approved and is not available through U.S. retail pharmacies. Public, documented access is through authorized clinical research. Cagrilintide does not currently meet the FDA’s criteria that would permit legal compounding for human use.
When will CagriSema be available?
The FDA decision is anticipated in 2026 per Novo Nordisk. Commercial launch timing has not been publicly confirmed and typically follows approval by some months. Anyone giving you a specific launch date is guessing.
Why do articles say CagriSema beat Ozempic if the trial used semaglutide 2.4 mg?
Because "CagriSema beat Ozempic" is a cleaner headline than "CagriSema beat semaglutide at a dose higher than Ozempic's FDA maximum." Ozempic and Wegovy both contain semaglutide — same molecule, different doses, different approved indications. The efficacy comparison is real; the label-to-label interpretation is blurred.
Has CagriSema been filed for diabetes too?
Not yet. The December 2025 U.S. filing covers chronic weight management only. Novo Nordisk has stated it will approach the FDA about a type 2 diabetes indication after the results of REIMAGINE 1 and REDEFINE 3 (the 7,000-patient cardiovascular outcomes trial). That timeline has not been publicly stated.
Is Ozempic approved for weight loss?
No. Ozempic is FDA-approved for type 2 diabetes, cardiovascular risk reduction in diabetics with heart disease, and kidney disease progression and CV death reduction in diabetics with chronic kidney disease. Wegovy — same molecule, higher dose (2.4 mg) — is the FDA-approved semaglutide product for chronic weight management.
How much does Ozempic cost without insurance?
Self-pay patients pay $349/month for 0.25, 0.5, or 1 mg and $499/month for the 2 mg dose through NovoCare Pharmacy. New self-pay patients qualify for $199/month for their first two fills of 0.25 or 0.5 mg doses through June 30, 2026. The list price is $1,027.51 per pen. Uninsured patients at or below 200% of the Federal Poverty Level may qualify for free medication through Novo Nordisk’s Patient Assistance Program.
Is Ozempic still hard to find?
The FDA resolved the national semaglutide injection shortage on February 21, 2025. Limited localized supply disruptions can still occur, but the national shortage designation is resolved.
Can you take CagriSema and Ozempic together?
No. CagriSema already contains semaglutide — the same active drug in Ozempic. Taking both would increase semaglutide exposure beyond established dosing without safety data or labeling support.
What’s the difference between CagriSema and Wegovy?
Wegovy is semaglutide 2.4 mg alone. CagriSema is semaglutide 2.4 mg plus cagrilintide 2.4 mg — a fixed-dose combination of two hormones. CagriSema shows more weight loss in trials (22.7% vs Wegovy’s 14.9–17.4%) but also higher gastrointestinal event rates. Wegovy is FDA-approved and available. CagriSema is not.
Should I switch from Ozempic to CagriSema when it’s available?
No approved switching recommendations or post-approval switching data exist yet. Any future switch decision would depend on CagriSema’s final label, your coverage, your goals, and how well Ozempic is working. Patients most likely to benefit are probably those who plateau on semaglutide despite reaching the full dose.
The bottom line
CagriSema is genuinely the most impressive next-generation GLP-1 in the Novo Nordisk pipeline. The REDEFINE 1 weight-loss numbers are real. The REIMAGINE 2 diabetes advantage over semaglutide alone is real. The dual-hormone approach is a meaningful innovation.
And none of that changes the fact that Ozempic is approved today, available today, and already has a deeper FDA label than CagriSema will have at its launch. For type 2 diabetes, Ozempic’s CV and kidney indications are clinical benefits CagriSema hasn’t demonstrated in reported data yet. For weight loss, Ozempic isn’t the right comparator anyway — Wegovy is, and even there the gap is more modest than headlines suggest.
The one thing almost nobody should do is freeze in research mode waiting for a drug that might arrive later, might underperform its hype the way REDEFINE 1 slightly underperformed Novo Nordisk’s 25% target, and will almost certainly cost more than what you can start today.
Your next step
Still not sure which GLP-1 path fits you?
One question, one personalized answer. No signup. No email wall. Built to help you decide, not to sell you.
If you already know you want to start an FDA-approved GLP-1 today:
Ro carries the broadest current menu of FDA-approved GLP-1 options — including Wegovy pen and pill, Zepbound pen and KwikPen, and Foundayo (orforglipron). Ro matches LillyDirect and NovoCare medication pricing, runs an insurance concierge who handles prior-authorization paperwork for you, and provides a free GLP-1 Insurance Coverage Checker. Get started for $39, then as low as $74/month with annual plan paid upfront.
Note: Ro currently does not coordinate GLP-1 coverage for government insurance plans (Medicare, Medicaid, VA, TRICARE).
If Ozempic specifically is what you’re weighing, see our CagriSema vs Wegovy comparison — the same molecule at Wegovy-dose is the cleaner side-by-side with CagriSema’s trial comparator.
Sources
- Novo Nordisk. “Novo Nordisk files for FDA approval of CagriSema.” PR Newswire, December 18, 2025.
- Novo Nordisk. REIMAGINE 2 topline results press release. GlobeNewswire, February 2, 2026.
- Novo Nordisk. REDEFINE 4 topline results press release. GlobeNewswire, February 23, 2026.
- Novo Nordisk. REDEFINE 1 publication announcement. PR Newswire, June 22, 2025.
- Garvey WT, et al. REDEFINE 1 trial publication. New England Journal of Medicine, June 22, 2025.
- U.S. Food and Drug Administration. Ozempic® (semaglutide) prescribing information, current label (2025).
- FDA. “FDA approves Ozempic® to reduce the risk of kidney disease worsening, kidney failure, and death due to cardiovascular disease in adults with type 2 diabetes and CKD.” January 28, 2025.
- NovoCare Pharmacy. Ozempic® pricing and self-pay offer pages (accessed April 2026).
- FDA. Declaratory Order: Resolution of Shortages of Semaglutide Injection Products. February 21, 2025.
- ClinicalTrials.gov. REDEFINE 3 (NCT05669755) trial status listing.
- ScienceDirect. Meta-analysis: “CagriSema Versus Semaglutide Monotherapy or Placebo for Obesity.” February 2026.
- STEP clinical trial program — STEP 1, 3, 4, 5, 8, STEP UP.
- SURMOUNT-5 trial — Eli Lilly head-to-head data, December 2024.
This page is informational, not medical advice. Talk to your prescriber before starting, stopping, or changing any GLP-1 medication.
The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We receive affiliate compensation when readers start with some of the providers we reference; that compensation does not influence our clinical coverage, our pricing verification, or what we recommend — evidence does.
Last verified: April 21, 2026.