Last verified: April 22, 2026 · NEJM REDEFINE 1 & 2 data confirmed · FDA NDA status checked · Prescribing information current
Clinical Data Guide · April 2026
CagriSema Side Effects: What REDEFINE 1, 2, and 4 Actually Show (2026 Update)
By The RX Index editorial team — a pricing intelligence and comparison resource for GLP-1 telehealth providers
The bottom line on CagriSema side effects
CagriSema side effects are overwhelmingly gastrointestinal — nausea, constipation, vomiting, diarrhea, abdominal pain — and most were mild-to-moderate and transient in the REDEFINE phase 3 trials. In REDEFINE 1, 79.6% of participants on CagriSema had at least one gastrointestinal adverse event versus 39.9% on placebo. Nausea hit 55%. Constipation 30.7%. Vomiting 26.1%. About 1 in 17 people (5.9%) discontinued treatment because of side effects.
Important: CagriSema is not FDA-approved. No pharmacy can fill a CagriSema prescription today. FDA decision anticipated late 2026.
CagriSema side effects — quick answers
Every key question, sourced from REDEFINE trial data.
| Question | Short answer |
|---|---|
| Most common side effect? | Nausea (55% in REDEFINE 1) |
| How severe? | Mostly mild-to-moderate; peaks during dose escalation |
| Dropout rate due to side effects? | 5.9% (REDEFINE 1) / 8.4% (REDEFINE 2) |
| Worse than Wegovy? | Nausea rate looks higher; overall dropout rate is nearly identical |
| Worse than Zepbound 15 mg? | Nausea roughly double; overall dropout actually lower (5.9% vs 6.7%) |
| Available to buy right now? | No — investigational only |
| Expected FDA decision? | Late 2026 (NDA filed Dec 18, 2025) |
What we actually verified for this page
- ✅CagriSema FDA status and NDA timeline — Novo Nordisk press release dated December 18, 2025, plus Novo's explicit statement that CagriSema is “not approved in the US or EU.”
- ✅Every REDEFINE 1 side-effect percentage — pulled directly from Table 3 of Garvey et al., New England Journal of Medicine, 2025 (NEJMoa2502081).
- ✅REDEFINE 2 GI and discontinuation rates — Davies et al., NEJM, 2025 (NEJMoa2502082).
- ✅REDEFINE 4 headline data — Novo Nordisk company announcement dated February 23, 2026.
- ✅Wegovy comparator rates — current Wegovy FDA prescribing information, Novo Nordisk (novo-pi.com), revised 12/2025.
- ✅Zepbound comparator rates — current Zepbound FDA prescribing information, Eli Lilly (pi.lilly.com).
- ⚠️If a number isn’t in a verified public source, we don’t publish it — we say the source didn’t break it out. You’ll see that flagged in several places below.
🟢Not sure what’s approved and right for you right now? Take our free 60-second GLP-1 matching quiz. No credit card, no commitment. You’ll get a personalized action plan covering what’s FDA-approved, what your insurance is likely to cover, and which legitimate providers fit your situation.
What is CagriSema, in one paragraph
CagriSema is a once-weekly subcutaneous injection combining cagrilintide 2.4 mg (a long-acting amylin analog — it mimics amylin, a gut hormone that signals fullness) and semaglutide 2.4 mg (the GLP-1 receptor agonist in Wegovy and Ozempic). Both slow gastric emptying. Both reduce appetite. The theory behind stacking them: two different hormonal pathways should drive more weight loss than semaglutide alone. The REDEFINE 1 trial confirmed that theory — 20.4% weight loss versus 3.0% on placebo over 68 weeks. The catch, which is what most of this page is about, is that the side-effect cost of stacking two appetite-suppressing hormones is real and measurable.
For the fuller background on what CagriSema is and when it’s expected to launch, see our CagriSema overview guide. This page stays focused on one question: how rough are the side effects, actually?
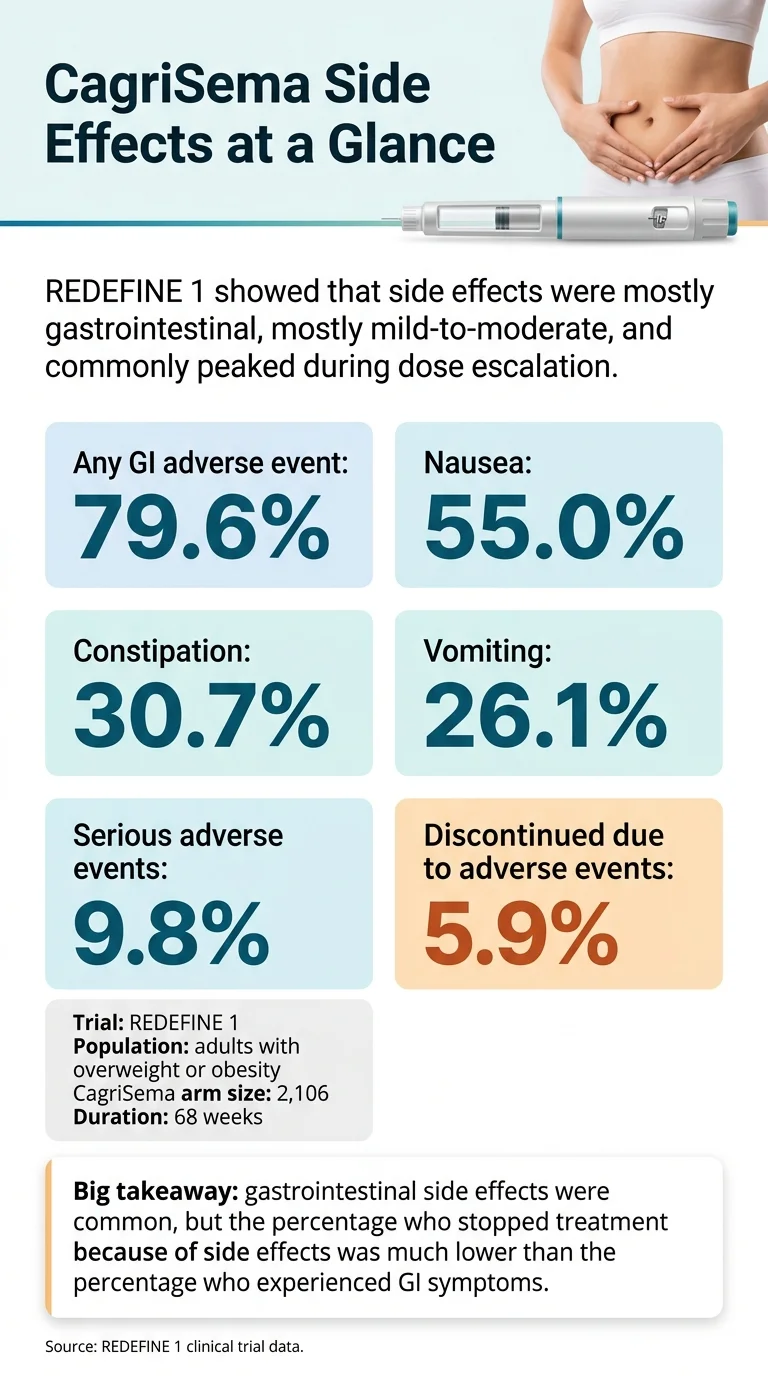
Source: REDEFINE 1 clinical trial data (Garvey et al., NEJM 2025).
Every CagriSema side effect from REDEFINE 1, with the exact numbers
Answer capsule: REDEFINE 1 was the pivotal phase 3 trial that powered CagriSema’s FDA application — 3,417 adults with overweight or obesity and no type 2 diabetes, followed for 68 weeks. 92.3% of CagriSema participants had at least one adverse event of any kind. 79.6% had a gastrointestinal adverse event. 9.8% had a serious adverse event (versus 6.1% on placebo). 5.9% discontinued treatment because of side effects. Two deaths were reported in the CagriSema arm — causes established by the external event adjudication committee were suicide and cancer of unknown primary source.
Here’s Table 3 from Garvey et al., reorganized so it’s actually useful:
REDEFINE 1: Adverse events across all four trial arms
| Event | CagriSema 2.4/2.4 (n=2,106) | Semaglutide 2.4 (n=302) | Cagrilintide 2.4 (n=302) | Placebo (n=705) |
|---|---|---|---|---|
| Any adverse event | 92.3% | 89.7% | 84.1% | 82.3% |
| Any GI adverse event | 79.6% | 73.8% | 54.0% | 39.9% |
| Nausea | 55.0% | — | — | 12.6% |
| Constipation | 30.7% | — | — | 11.6% |
| Vomiting | 26.1% | — | — | 4.1% |
| Injection-site reactions | 12.2% | 2.6% | 16.9% | 3.0% |
| Gallbladder-related disorders | 4.1% | 3.0% | 2.3% | 1.0% |
| Allergic reactions | 5.2% | 5.6% | 7.6% | 5.5% |
| Neoplasms (benign + malignant) | 6.4% | 6.6% | 1.7% | 4.4% |
| Pancreatitis | 0.2% | 0.3% | 0% | 0% |
| Serious adverse events | 9.8% | 5.0% | 8.9% | 6.1% |
| Discontinued due to adverse event | 5.9% | 3.6% | 2.6% | 3.5% |
| GI-specific discontinuation | 3.6% | 1.3% | 1.3% | 0.6% |
| Deaths during trial | 2 (0.1%) — 1 suicide, 1 cancer | 0 | 0 | 0 |
Source: Garvey WT, Blüher M, Osorto Contreras CK, et al. Coadministered Cagrilintide and Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2025. DOI: 10.1056/NEJMoa2502081, Table 3.
The insight nobody else is pulling out of this table
Look at the “Any GI adverse event” row across the four arms: CagriSema 79.6%, semaglutide-alone 73.8%, cagrilintide-alone 54.0%, placebo 39.9%. The gap between CagriSema and semaglutide-alone is only 5.8 percentage points. Stacking the amylin half on top of the GLP-1 half does not double the gut burden — it nudges it.
Translation: if you’re trying to decide whether CagriSema will be gentler than Wegovy, it won’t be. CagriSema literally contains semaglutide 2.4 mg — plus a smaller-but-not-zero amylin burden on top. There is no scenario, on the published data, where CagriSema is easier on the gut than Wegovy.
CagriSema side effects in people with type 2 diabetes (REDEFINE 2)
Answer capsule: REDEFINE 2 tested CagriSema in 1,206 adults with type 2 diabetes and overweight or obesity over 68 weeks (904 on CagriSema, 302 on placebo). GI side effects occurred in 72.5% of CagriSema participants versus 34.4% on placebo. Serious adverse events hit 10.4% of the CagriSema group. Discontinuation due to side effects was 8.4% on CagriSema versus 3.0% on placebo.
The REDEFINE 2 paper (Davies et al., NEJMoa2502082) does not break out nausea, vomiting, and constipation as individual percentages in the publicly available abstract or press materials. If those appear in the full supplementary appendix, we’ll update this section. We’re not going to estimate them.
What’s verified from REDEFINE 2:
- •GI adverse events: 72.5% (vs 34.4% placebo)
- •Discontinuation due to adverse events: 8.4% (vs 3.0% placebo)
- •GI-specific discontinuation: 4.8%
- •Pancreatitis: 0.3%
- •Weight loss: 13.7% (treatment-policy) / 15.7% (trial-product)
- •HbA1c reached 6.5% or below in 73.5% of CagriSema patients vs 15.9% on placebo
The tolerability pattern matches REDEFINE 1: mostly mild-to-moderate GI events, peaking during dose escalation, settling afterward. The higher overall discontinuation rate (8.4% vs 5.9% in REDEFINE 1) is worth noting without over-interpreting — population-level factors, dose-escalation practice, and concomitant oral glucose-lowering medications all likely play a role.
CagriSema vs Wegovy: Is it really worse for side effects?
Answer capsule: On the headline nausea rate, yes — CagriSema’s 55% in REDEFINE 1 runs meaningfully higher than Wegovy’s 44% per the Wegovy FDA prescribing information. Vomiting (26% vs 24%) and constipation (31% vs 24%) also tilt a few points higher. But the rate of people who actually quit because of side effects is close to identical: CagriSema 5.9% versus Wegovy 6.8%. Tolerability is directionally worse; the practical “can I actually stay on this drug” outcome lands in the same neighborhood.
| Side effect | CagriSema (REDEFINE 1) | Wegovy 2.4 mg (FDA label) |
|---|---|---|
| Nausea | 55.0% | 44% |
| Vomiting | 26.1% | 24% |
| Constipation | 30.7% | 24% |
| Diarrhea | Reported; not quantified in public source | 30% |
| Abdominal pain | Reported; not quantified in public source | 20% |
| Injection-site reactions | 12.2% | 1.4% |
| Overall discontinuation due to AE | 5.9% | 6.8% |
| GI-specific discontinuation | 3.6% | 4.3% |
Sources: Garvey et al., N Engl J Med 2025 (REDEFINE 1 Table 3); Wegovy US Prescribing Information, Novo Nordisk, revised 12/2025, Table 3.
Important caveat on cross-trial comparisons
REDEFINE 1 and the STEP trials that underpin Wegovy’s label used different populations, different escalation schedules, different adverse-event reporting windows, and different definitions of “treatment-emergent.” Treat this as a decision aid, not a head-to-head verdict. Head-to-head GI data between CagriSema and Wegovy doesn’t exist in any published trial — Novo Nordisk never ran that comparison.
The damaging admission, upfront
CagriSema does NOT look like an easier, gentler, better-tolerated Wegovy. If you had a hard time on semaglutide — bad nausea, throwing up, couldn’t leave the house — CagriSema contains the exact same 2.4 mg dose of semaglutide in the exact same molecule, plus a second drug with its own gut effects. The math here doesn’t bend in your favor.
If a gentler GLP-1 is what you actually want, the honest path is either (a) a lower dose of semaglutide (1.7 mg Wegovy), (b) a switch to tirzepatide, which has meaningfully lower headline GI numbers per its FDA label, or (c) a conversation with a prescribing clinician about slower titration.
CagriSema vs Zepbound (tirzepatide): The head-to-head trial Novo lost
Answer capsule: REDEFINE 4 directly compared CagriSema 2.4/2.4 mg against tirzepatide 15 mg (Zepbound) in 809 adults over 84 weeks. CagriSema produced 23.0% weight loss versus 25.5% for tirzepatide (efficacy estimand) and did not meet its primary endpoint of demonstrating noninferiority. On side effects, Zepbound’s FDA label numbers run cleaner than CagriSema’s across every comparable metric.

Cross-trial comparison — not a head-to-head study. Sources: REDEFINE 1 data, Wegovy prescribing information, Zepbound prescribing information.
| Side effect | CagriSema (REDEFINE 1) | Zepbound FDA label (15 mg) |
|---|---|---|
| Nausea | 55.0% | 25–29% (pooled across doses) |
| Vomiting | 26.1% | 13% (15 mg) |
| Diarrhea | Reported; not quantified | 19–23% (pooled) |
| Constipation | 30.7% | 11–17% (pooled) |
| Abdominal pain | Reported; not quantified | 9–10% (pooled) |
| Any GI adverse event | 79.6% | 56% (15 mg) |
| Severe GI adverse reactions | Not broken out in public source | 3.1% (15 mg) |
| Overall discontinuation due to AE | 5.9% | 6.7% (15 mg) |
| GI-specific discontinuation | 3.6% | 4.3% (15 mg) |
| Pancreatitis | 0.2% | 0.2% (pooled) |
Sources: Garvey et al., N Engl J Med 2025; Zepbound US Prescribing Information (pi.lilly.com), current version; Novo Nordisk REDEFINE 4 company announcement, February 23, 2026.
The honest read of REDEFINE 4
Zepbound produced more weight loss and the published side-effect numbers run lower on nausea, vomiting, and severe GI events. CagriSema’s defenders will point out that the head-to-head was open-label, which can bias tolerability reporting, and that Novo is running longer, higher-dose studies (REDEFINE 11) that may shift the picture. Fair. But as of today, the publicly available peer-reviewed evidence suggests Zepbound remains the efficacy and tolerability benchmark CagriSema did not beat.
The practical consequence for a reader deciding right now: if you’re GLP-1-naive and looking for a currently available option while CagriSema is still under FDA review, Zepbound is a legitimately better candidate on the numbers we have.
When do CagriSema side effects start, and do they get better?
Answer capsule: In REDEFINE 1, nausea, vomiting, and diarrhea peaked during the 16-week dose-escalation phase and declined afterward. Constipation was the exception — it stayed relatively constant over the full 68 weeks. This timing pattern is the same one seen with semaglutide (Wegovy) and tirzepatide (Zepbound), not a CagriSema-specific quirk.
REDEFINE 1 dose escalation schedule and typical tolerability
| Weeks | Dose (each drug) | What the trial data suggests |
|---|---|---|
| 1–4 | 0.25 mg | Mild nausea possible as body adjusts |
| 5–8 | 0.5 mg | GI events begin climbing |
| 9–12 | 1.0 mg | Nausea commonly peaks |
| 13–16 | 1.7 mg | Nausea still elevated; vomiting peaks |
| 17–68 | 2.4 mg (maintenance) | Nausea and vomiting decline; constipation may persist |
Source: Garvey et al., REDEFINE 1 NEJM 2025, Figure S11B.
One practical note worth reading twice
The REDEFINE 1 protocol allowed investigators to pause dose escalation or step the dose down when a patient was struggling. Only 57.4% of CagriSema participants were on the full 2.4/2.4 mg dose at week 68 — 74.7% reached it at some point, but about a third didn’t stay there. Significant weight loss still happened at submaximum doses. For someone worried about dose-related tolerability, that’s the most encouraging finding in the trial: you don’t have to power through to the top dose to see real results.
Community anecdotes (not medical evidence)
Trial participants are real people, and some of them post on Reddit. Treat the following as emotional context, not safety data:
- •A user in r/CagriSema on a higher dose described significant nausea and needing more sleep than usual.
- •Another described starting low specifically to avoid the nausea people had been talking about, and reported side effects staying relatively mild as a result.
Both line up with what the trial data shows: the slower the titration, the more tolerable the early weeks tend to be. These are not evidence of anything. They’re directional only.
What serious or less-common CagriSema risks have shown up?
Answer capsule: Serious adverse events occurred in 9.8% of REDEFINE 1 CagriSema participants (versus 6.1% on placebo) and in 10.4% of REDEFINE 2 participants. Hepatobiliary and gastrointestinal disorders were the most common serious events. Pancreatitis occurred in 0.2%. Gallbladder-related disorders in 4.1%, versus 1.0% on placebo.
🫁 Gallbladder disease (4.1%)
4.1% of CagriSema participants had a gallbladder-related adverse event, versus 1.0% of placebo. This is a known GLP-1 class effect — semaglutide and tirzepatide trials have both reported elevated gallbladder disease rates, and rapid weight loss itself (regardless of drug) is an independent risk factor for gallstones. Steady pain in your upper-right abdomen, especially after eating, warrants a same-day call to your clinician.
🔴 Pancreatitis (0.2%)
0.2% in CagriSema vs 0% in placebo. Four cases total across 2,106 participants. Consistent with low rates seen across the GLP-1 class. Severe abdominal pain that radiates to your back is the classic warning sign — same-day medical attention, every time.
💉 Injection-site reactions (12.2%)
12.2% in CagriSema (versus 3.0% in placebo, and just 2.6% in the semaglutide-alone arm). This is meaningfully higher than Wegovy’s label rate of 1.4% — the cagrilintide component appears to drive most of it, since cagrilintide-alone also ran 16.9%. Usually mild (redness, itching, induration at the injection site) and typically self-limiting.
😴 Fatigue, dizziness, and hair loss
All occurred more frequently with CagriSema than placebo in REDEFINE 1. Exact percentages weren’t broken out in the public source we verified. Hair loss on any rapid weight-loss regimen is a well-documented phenomenon called telogen effluvium — it’s linked to the metabolic stress of fast weight loss more than to any specific drug, and it’s typically reversible once weight stabilizes.
🧠 Mental health
REDEFINE 1 measured suicidal ideation (Columbia–Suicide Severity Rating Scale) and depression (PHQ-9) across all four arms. The scores were similar across groups, and no important changes emerged. The one completed suicide in the CagriSema arm is tragic and not to be minimized; causation for individual deaths is determined by external adjudication committees. On the published data, there is no trial-level signal that CagriSema worsens mental health.
❤️ Heart rate
CagriSema raised resting heart rate by an average of 0.94 beats per minute. Semaglutide alone raised it by 1.17 bpm. Cagrilintide alone actually dropped it by 3.31 bpm. Translation: the amylin half of CagriSema partially offsets whatever modest heart-rate elevation GLP-1 drugs produce.
⚠️ Thyroid warnings
Wegovy and Zepbound both carry boxed warnings about thyroid C-cell tumors seen in rat studies. CagriSema does not yet have an FDA-approved label — the final warning language will be set at approval. We don’t predict labels. If the final CagriSema label includes similar language, we’ll update this section the day it becomes public.
One more honest statement. The REDEFINE trials lasted 68 weeks (1, 2) and 84 weeks (4). That’s long enough to catch most common side effects. It is not long enough to catch rare, delayed, or truly long-term effects that only appear once tens of thousands of people have taken the drug for years. Post-market surveillance, once CagriSema is actually approved and being used outside a trial, will reveal things the trials didn’t. That’s a real unknown — and it’s the same unknown every newly approved drug has.
Known vs. unknown: what the REDEFINE program has and hasn’t answered
This is the honest grid. Most pages on CagriSema side-step the right column.
✅ What’s known right now
- ✓Short-to-medium-term GI burden. Heavy, mostly mild-to-moderate, peaks during dose escalation.
- ✓Overall serious adverse event rate. 9.8% in REDEFINE 1 (vs 6.1% placebo); 10.4% in REDEFINE 2.
- ✓Class-typical signals. Gallbladder disease, pancreatitis, injection-site reactions at rates consistent with or slightly above the broader GLP-1 class.
- ✓Tolerability vs semaglutide alone. Modestly worse — adding cagrilintide costs about 6 percentage points of additional GI AE risk.
- ✓Tolerability vs tirzepatide. Looks worse on nausea and vomiting in indirect comparison.
❌ What’s unknown right now
- ✗Final FDA label. Contraindications, warnings, and precautions are set at approval. Not yet available.
- ✗Symptom-by-symptom breakdown from REDEFINE 2. Individual percentages not in public sources.
- ✗Full adverse-event profile from REDEFINE 4. Topline only — peer-reviewed publication pending.
- ✗Long-term safety beyond 84 weeks. REDEFINE 3 (cardiovascular outcomes, 7,000 adults) still running.
- ✗Real-world US post-market data. Zero — no one outside a clinical trial has legally used it yet.
- ✗Dose-response above 2.4/2.4 mg. Higher-dose trial planned; results aren't in.
If anyone tells you they know CagriSema’s long-term safety picture, they’re ahead of the data.
Should you wait for CagriSema, or start an approved GLP-1 now?
Answer capsule: If you already tolerate Wegovy or Ozempic well and are losing weight steadily, there’s no urgent reason to switch — CagriSema’s expected incremental weight loss over semaglutide was about 5 percentage points in REDEFINE 1, and comes with a modestly worse side-effect profile. If you’re GLP-1-naive and want something you can start now, Wegovy and Zepbound both have years of real-world safety data that CagriSema will not have at launch. If you had a rough time on semaglutide specifically, CagriSema contains the same semaglutide dose and is unlikely to feel easier.
Four-quadrant decision guide
| Your situation | What the data suggests |
|---|---|
| Doing well on Wegovy or Ozempic. Weight is dropping, side effects manageable. | Stay the course. CagriSema's expected incremental benefit is modest. The drug hasn't earned a switch. Bookmark this page for the FDA decision. |
| GLP-1-naive, want to start soon. | A currently approved option (Wegovy, Zepbound) makes more sense than waiting 6–12+ months. Real-world safety data matters. |
| Poor tolerance on semaglutide. Couldn't handle the nausea, or had to come off. | CagriSema contains the same 2.4 mg semaglutide dose. It probably won't be easier. Tirzepatide has meaningfully lower headline GI rates per its FDA label — that's a better conversation with your clinician. |
| Patient, not on therapy yet, open to waiting. | Waiting is reasonable. Add yourself to the FDA decision watch list — we'll email you when the ruling lands. |
Two concrete next steps from here
🟢If you want the personalized version of the answer above, take the matcher. We’ll ask about your current medication, tolerability, timeline, and insurance, and return a personalized action plan in under a minute. No credit card, no commitment.
🔵Interested in Zepbound (tirzepatide) through a legitimate telehealth route? Ro carries Zepbound® and Foundayo™ (the newly FDA-approved oral GLP-1 from Eli Lilly). Ro runs a dedicated insurance concierge that handles prior-authorization paperwork and offers a free GLP-1 insurance coverage checker. Ro Body membership is $39 for the first month, then $149/month or as low as $74/month with annual prepay — medication is charged separately and Ro matches LillyDirect / NovoCare pricing on the medication itself.
If Zepbound isn’t your fit, the matching quiz above will route you to what does fit.
Can I get CagriSema from a compounding pharmacy?
No — and this is a firm legal answer.
Cagrilintide is an investigational drug and is not FDA-approved. The FDA has specifically warned that unapproved GLP-1 medications cannot legally be used in compounding for human use outside of registered clinical trials. Any pharmacy, clinic, or online seller offering “CagriSema,” “cagrilintide,” or “cagrilintide-semaglutide” compounded for human use is operating outside US federal law.
This isn’t a CagriSema-specific rule. It applied to semaglutide before Wegovy was approved, and it applies to retatrutide right now. Compounding law permits pharmacies to prepare custom medications under specific conditions — but an investigational drug whose active ingredient is not FDA-approved falls outside those conditions.
The legal route to access cagrilintide in the US today is participation in a legitimate, registered clinical trial. You can find active REDEFINE studies at ClinicalTrials.gov by searching “cagrisema” or “cagrilintide.” Enrollment criteria vary by study. ClinicalTrials.gov is the authoritative source.
When will CagriSema actually be available?
Answer capsule: Novo Nordisk filed the New Drug Application for CagriSema with the FDA on December 18, 2025. An FDA decision is anticipated by late 2026. No public PDUFA target date has been confirmed. Even after approval, several more months typically pass before a new drug is widely stocked at retail pharmacies.
Novo has flagged several additional trials still running: REDEFINE 3 (cardiovascular outcomes in 7,000 adults), REDEFINE 11 (longer duration, different titration), and early-stage higher-dose CagriSema studies. None of these gate the initial approval — which is based on REDEFINE 1 and 2 — but they’ll shape the final label and any future indication expansions.
What we will not do: pretend to know the exact launch date. We’ll update this page within 5 business days of any meaningful FDA action.
🔔Email me when the FDA rules on CagriSema. One email when the ruling lands, with what it actually means for your situation — not just that it happened. No spam.
Frequently asked questions about CagriSema side effects
- Is nausea the most common CagriSema side effect?
- Yes. In REDEFINE 1, 55% of CagriSema participants reported nausea, compared with 12.6% on placebo. It was the most frequent adverse event, peaked during dose escalation, and declined after participants reached the maintenance dose of 2.4 mg of each drug.
- Are CagriSema's side effects worse than Wegovy's?
- On headline GI rates, yes — CagriSema's nausea (55%), vomiting (26.1%), and constipation (30.7%) in REDEFINE 1 run higher than Wegovy's 44% nausea, 24% vomiting, and 24% constipation per Wegovy's current FDA prescribing information. But the rate of people who quit because of side effects is close to identical (CagriSema 5.9% vs Wegovy 6.8%). Tolerability is directionally worse; the practical dropout rate is a wash.
- Does CagriSema cause constipation?
- Yes. 30.7% of CagriSema participants in REDEFINE 1 reported constipation, versus 11.6% on placebo. Unlike nausea and vomiting — which peak during dose escalation and then fade — constipation rates stayed relatively constant throughout the trial. Hydration, fiber, and dietary adjustment are the usual first-line management; persistent constipation belongs in a conversation with your clinician.
- Do CagriSema side effects get better over time?
- For most people in REDEFINE 1, yes. Nausea, vomiting, and diarrhea peaked during the 16-week dose-escalation period and declined afterward. Constipation was the exception. Injection-site reactions generally resolve on their own. Rare serious events such as pancreatitis and gallbladder problems don't follow a predictable arc and require clinical attention.
- Has pancreatitis been reported in CagriSema trials?
- Yes. In REDEFINE 1, pancreatitis occurred in 0.2% of CagriSema participants (4 of 2,106), 0.3% of semaglutide-alone participants (1 of 302), and 0% of the cagrilintide-alone and placebo groups. REDEFINE 2 reported pancreatitis at 0.3%. These rates are consistent with the broader GLP-1 drug class. Severe abdominal pain that radiates to the back is the classic warning sign and warrants immediate medical attention.
- Have gallbladder problems been reported in CagriSema trials?
- Yes. Gallbladder-related disorders occurred in 4.1% of CagriSema participants in REDEFINE 1, versus 1.0% on placebo. This is a known GLP-1 class effect, amplified by rapid weight loss, which is itself an independent risk factor for gallstones.
- Will CagriSema cause hair loss?
- REDEFINE 1 reported alopecia (hair loss) as more common in the CagriSema group than placebo, but the exact percentage wasn't broken out in the public source we verified. For context, Zepbound's FDA label reports hair loss in 4–5% of users overall, and hair shedding on any rapid weight-loss regimen is generally understood as a side effect of fast weight loss (telogen effluvium) rather than a direct drug effect. Adequate protein intake and slower titration are the usual mitigation strategies; hair typically regrows once weight stabilizes.
- How many people quit CagriSema because of side effects?
- 5.9% of REDEFINE 1 CagriSema participants and 8.4% of REDEFINE 2 participants discontinued because of adverse events. For comparison: Wegovy's FDA label reports 6.8% overall discontinuation, and Zepbound 15 mg reports 6.7%. So in round numbers, roughly 1 in 15 to 1 in 17 people can't stay on any of these drugs because of side effects — including CagriSema.
- Is CagriSema FDA-approved yet?
- No. Novo Nordisk filed the NDA on December 18, 2025. An FDA decision is anticipated by late 2026. Until then, CagriSema is investigational — available only through active REDEFINE clinical trials. No pharmacy, telehealth provider, or compounding pharmacy can legally dispense it in the US.
- Should I wait for CagriSema if I already had a bad time on semaglutide?
- The published evidence suggests probably not. CagriSema contains the same 2.4 mg dose of semaglutide that's in Wegovy, plus a second drug (cagrilintide) that also has GI effects. If Wegovy made you miserable, CagriSema is very unlikely to be easier. A conversation with your clinician about a different mechanism — such as tirzepatide — is a more promising path.
How we verified this page
The trust claim on a YMYL page is only as good as the sources behind it. Here are ours.
Primary peer-reviewed sources
- Garvey WT, Blüher M, Osorto Contreras CK, et al. Coadministered Cagrilintide and Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2025. DOI: 10.1056/NEJMoa2502081. (REDEFINE 1; Table 3 for every adverse event percentage on this page.)
- Davies MJ, Bajaj HS, Broholm C, et al. Cagrilintide–Semaglutide in Adults with Overweight or Obesity and Type 2 Diabetes. N Engl J Med. 2025. DOI: 10.1056/NEJMoa2502082. (REDEFINE 2.)
- Wharton S, Calanna S, Davies M, et al. Gastrointestinal tolerability of once-weekly semaglutide 2.4 mg in adults with overweight or obesity. Diabetes Obes Metab. 2022;24(1):94–105. (Pooled Wegovy STEP data; used for historical context only.)
Manufacturer sources
- Novo Nordisk. Novo Nordisk files for FDA approval of CagriSema. Press release, December 18, 2025.
- Novo Nordisk. CagriSema 2.4 mg / 2.4 mg demonstrated 22.7% mean weight reduction. Press release, June 22, 2025.
- Novo Nordisk. CagriSema demonstrated 23% weight loss in REDEFINE 4. Company announcement, February 23, 2026.
Regulatory sources
- Wegovy® (semaglutide) US Prescribing Information, Novo Nordisk, revised 12/2025. Available at novo-pi.com.
- Zepbound® (tirzepatide) US Prescribing Information, Eli Lilly and Company. Available at pi.lilly.com.
- FDA: Postmarket Drug Safety Information — FDA’s Concerns with Unapproved GLP-1 Drugs Used for Weight Loss.
- ClinicalTrials.gov registrations: NCT05567796 (REDEFINE 1), NCT05394519 (REDEFINE 2), NCT07011667, NCT06131437.
How this page was built. Our editorial team pulled every CagriSema adverse-event number directly from NEJM REDEFINE 1 and 2 publications or from Novo Nordisk’s dated press releases. Comparator data (Wegovy, Zepbound) came from FDA-approved prescribing information. Where a figure was not available in a verifiable public source, we marked that gap plainly. Nothing here is paraphrased from other side-effect articles; every number traces to an original source.
What we are not. We are not clinicians. This page summarizes published clinical trial data for an investigational medication and is for educational purposes only. Nothing here is medical advice. If you are weighing any GLP-1 treatment decision, talk with a qualified healthcare provider who can see your full medical history.
Refresh schedule. Monthly during the CagriSema FDA review window, within 5 business days of any meaningful FDA action, quarterly thereafter.
Still not sure which GLP-1 program is right for you?
CagriSema isn’t available to anyone outside a clinical trial, and likely won’t be for several more months. In the meantime, there are FDA-approved GLP-1 medications that are available, have years of real-world data, and work — if you match the right one to your situation, your insurance, and your tolerability.
🟢Take our free 60-second matching quiz. You’ll get a personalized action plan covering what’s FDA-approved, what’s likely to be covered by your plan, and the most legitimate current providers for your situation.
🔔Or: email me when the FDA rules on CagriSema. One email when the ruling lands, with what it actually means for your situation. No spam.
Related guides
Medical disclaimer. This page summarizes published clinical trial data and is for educational purposes only. Nothing here constitutes medical advice, diagnosis, or treatment recommendations. Always consult a qualified healthcare provider before starting, stopping, or changing any medication.
Last verified: · Page updated:
Affiliate disclosure: some links on this page use affiliate tracking URLs. This does not affect the price you pay or our editorial independence.