Last verified: April 20, 2026 · FDA status checked · Pricing verified · Trial data confirmed
Comparison Guide · April 21, 2026
Cagrisema vs Wegovy (2026): Should You Wait or Start Now? The Honest Verdict
Published:
By The RX Index Research Team — a pricing intelligence and comparison resource for GLP-1 telehealth providers
The bottom line up front
Cagrisema posted stronger weight-loss numbers than Wegovy in trials — roughly 20–23% mean weight loss vs ~15% — but Cagrisema is not FDA-approved. The decision gets more interesting when you add Zepbound: in the only Phase 3 trial that put Cagrisema and Zepbound side-by-side, Cagrisema lost (23.0% vs 25.5%). The drug you were planning to wait for already has a stronger one at your pharmacy.
Wegovy is FDA-approved, available today, and ranges from $149–$399/month self-pay through NovoCare or as low as $25/month with commercial insurance. For most readers: don’t wait for Cagrisema.
If you want the strongest currently available GLP-1, Zepbound (tirzepatide) outperformed Cagrisema head-to-head in REDEFINE 4. Ro carries Zepbound, handles prior authorization, and runs a free GLP-1 insurance coverage check.
 (sponsored affiliate link, opens in a new tab)
(sponsored affiliate link, opens in a new tab)CagriSema vs Wegovy: dual-hormone combination vs semaglutide-only. CagriSema is not FDA-approved as of April 2026.
The short answer
For cagrisema vs wegovy in April 2026, here’s where things actually stand. Cagrisema (cagrilintide + semaglutide) posted stronger weight-loss numbers than Wegovy in trials — roughly 20–23% mean weight loss vs ~15% — but Cagrisema is not FDA-approved. Novo Nordisk filed the New Drug Application on December 18, 2025, and the company expects FDA review in 2026. No PDUFA date has been publicly confirmed. Realistic consumer availability is not before late 2026 at the earliest.
Wegovy is FDA-approved and available today. Self-pay pricing through NovoCare Pharmacy is form- and dose-specific: $149/month for the lower-dose pill, $199/month for the pen introductory offer, $349/month for the standard pen, $399/month for Wegovy HD 7.2 mg, and $299/month for the higher-dose pill. Commercial insurance + the Wegovy Savings Offer drops cost to as low as $25/month for eligible patients.
Here’s the piece of decision intelligence that reshapes this question: in the only Phase 3 trial that directly compared Cagrisema to tirzepatide (Zepbound), Cagrisema lost. If your goal is the strongest currently available weight-loss GLP-1, the honest answer isn’t Wegovy or a future Cagrisema launch — it’s Zepbound, and it’s available now.
For most readers: don’t wait for Cagrisema. Any website selling “compounded cagrisema” or “compounded cagrilintide” today is operating outside federal law.
What we actually verified on April 20, 2026
We built this page by checking primary sources, not paraphrasing other comparison articles.
| ✓ Verified | ✗ We refused to guess |
|---|---|
| Cagrisema FDA status (not approved) — FDA Orange Book, Drugs.com regulatory history, Novo Nordisk press releases | Cagrisema launch price — not disclosed by Novo Nordisk |
| NDA filing date (Dec 18, 2025) — Novo Nordisk investor release | Exact PDUFA date — FDA has not publicly confirmed |
| Wegovy self-pay pricing (pen and pill, all dose tiers) — novocare.com, wegovy.com | Individual insurance out-of-pocket — varies by plan |
| Ro GLP-1 program pricing structure — ro.co | Specific state Medicaid coverage — changes frequently |
| Success by Sesame subscription pricing — sesamecare.com | Any claim that “compounded cagrisema” could be safe |
| FDA position on cagrilintide compounding (prohibited) — fda.gov unapproved-GLP-1 page | Any testimonial we couldn’t attribute to a named person |
| Trial data: REDEFINE 1, 2, 4 and REIMAGINE 2 — NEJM (REDEFINE 1 and 2) + Novo Nordisk topline press releases | Whether Cagrisema will be approved (decision pending) |
Cagrisema vs Wegovy at a glance
The fast comparison. The decision framework below explains which column you should care about more.
| Cagrisema | Wegovy | |
|---|---|---|
| Active ingredients | Cagrilintide 2.4 mg + semaglutide 2.4 mg | Semaglutide (injection 0.25–2.4 mg + HD 7.2 mg; tablet 1.5, 4, 9, 25 mg) |
| Mechanism | Amylin analogue plus GLP-1 receptor agonist | GLP-1 receptor agonist |
| Delivery | Once-weekly subcutaneous injection (dual-chamber pen used in REDEFINE trials) | Once-weekly injection pen or once-daily oral tablet |
| Manufacturer | Novo Nordisk | Novo Nordisk |
| FDA-approved? | No — NDA filed Dec 18, 2025; FDA review expected 2026 | Yes — approved 2021 |
| Available to buy today? | No — clinical-trial access only | Yes — pharmacies nationwide and direct via NovoCare |
| Self-pay price (2026) | Not disclosed; not on the market | $149–$399/month through NovoCare; as low as $25/month with commercial insurance + savings offer |
| Best-case trial weight loss | 22.7% at 68 weeks (REDEFINE 1, trial-product estimand) | ~15% average at 68 weeks (STEP 1 data) |
| Same-trial comparison (REDEFINE 1) | 20.4% | 14.9% (semaglutide arm, same trial) |
| Head-to-head vs Zepbound? | Yes — REDEFINE 4: Cagrisema 23.0% vs Zepbound 25.5%, non-inferiority not met | |
| GI adverse events (REDEFINE 1) | 79.6% | 73.8% (semaglutide arm, same trial) |
| Cardiovascular risk-reduction indication | Not established (REDEFINE 3 ongoing) | Yes — reduces major cardiovascular events in adults with heart disease |
| Legally compoundable? | No — FDA explicitly prohibits cagrilintide compounding | Semaglutide is not on FDA’s shortage list; routine shortage-based compounding no longer supported |
Sources: FDA prescribing information (Wegovy DailyMed label), NEJM NEJMoa2502081 and NEJMoa2502082, Novo Nordisk press releases (Dec 18, 2025; Feb 2, 2026; Feb 23, 2026), NovoCare pricing pages, FDA unapproved-GLP-1 guidance. All verified April 20, 2026.
Cagrisema current status tracker
This is the piece of information most searchers actually came for, and it’s the thing that goes stale fastest. We update this monthly — and weekly during the FDA review window.
| Field | Status (as of April 20, 2026) |
|---|---|
| FDA-approved in the US? | No — investigational |
| NDA filed with FDA? | Yes — December 18, 2025 |
| Expected FDA review timing | Novo Nordisk guided to FDA review in 2026 (estimate — no PDUFA date has been publicly confirmed) |
| Approved anywhere in the world? | No |
| Earliest realistic US patient availability | Not before late 2026; broader access likely 2027 (estimate) |
| Only legal access path today | Clinical trial enrollment (REDEFINE / REIMAGINE programs) |
| Legally compoundable? | No — FDA has explicitly stated cagrilintide cannot be used in compounding |
| Price disclosed by Novo Nordisk? | No |
Source: Novo Nordisk press releases (Dec 18, 2025; Feb 2, 2026; Feb 23, 2026), FDA Orange Book, Drugs.com regulatory history.
If you’re tracking this approval, bookmark this page. We’ll update it the day Novo or the FDA announces anything.
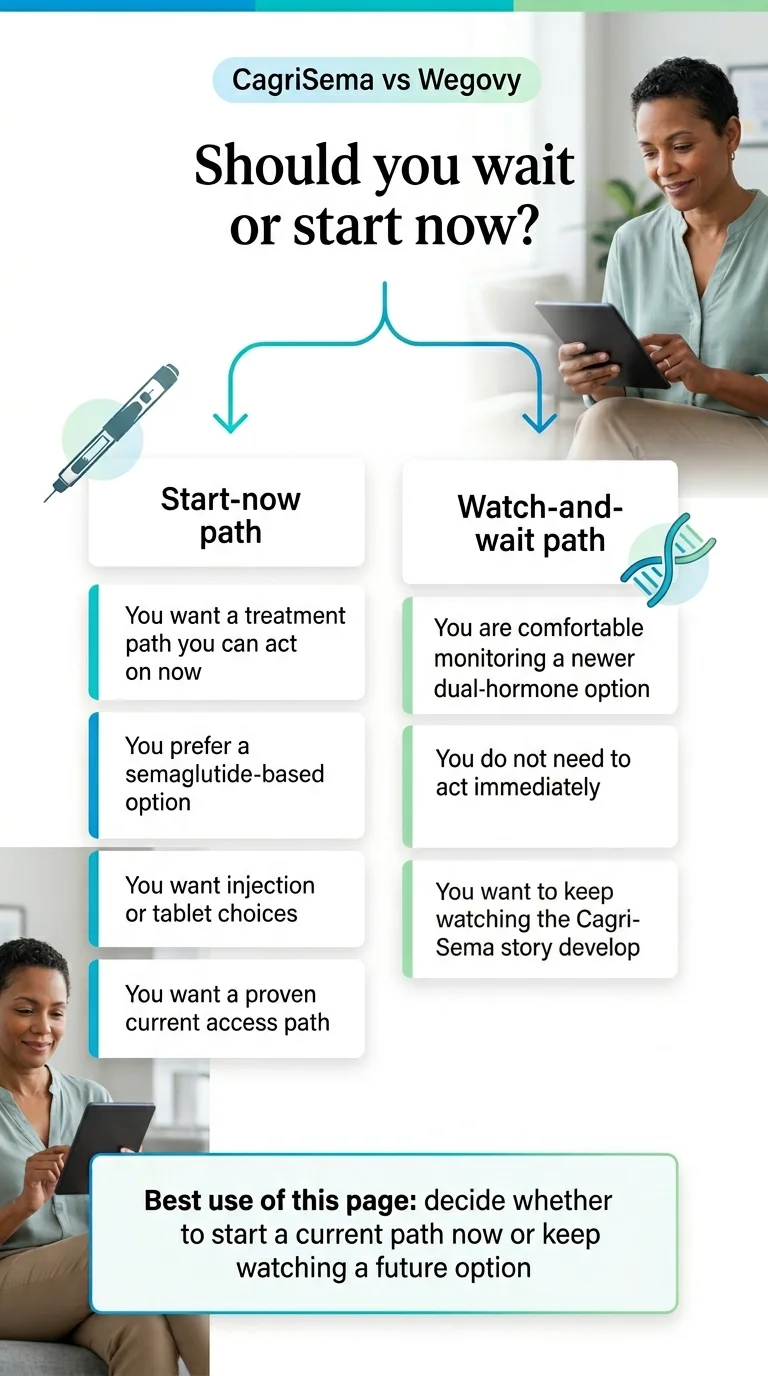
If your situation is X, here’s the better answer
Before the deep dive, here’s the quick-scan version. If one of these fits cleanly, you already have your answer.
- →You want treatment this month — start now. Don’t wait. Wegovy or Zepbound. Details in the decision framework below.
- →You’re already on Wegovy and it’s working — keep going. Don’t switch based on data about a drug you can’t access.
- →You’ve plateaued on Wegovy for 3+ months — talk to your prescriber about Zepbound. Zepbound already produced more weight loss than Cagrisema in head-to-head testing.
- →You have type 2 diabetes and obesity — start now, watch Cagrisema. This is Cagrisema’s strongest data population, but “later” isn’t a treatment plan.
- →You’ve never taken a GLP-1 and want the strongest available option — Zepbound. More below.
- →You’re uninsured and price-sensitive — start Wegovy via NovoCare or Success by Sesame. Cash-pay infrastructure exists today. Cagrisema will not have one for 12+ months post-approval.
- →You’re on Medicare or Medicaid — your path is different. Government-plan readers have separate rules. See our Wegovy Medicare coverage guide and our HSA/FSA GLP-1 guide.
- →You’re genuinely willing to wait 12–18 months — watch the status tracker. Waiting is a real choice. Just make it on purpose.
Is Cagrisema FDA-approved yet?
No. As of April 20, 2026, Cagrisema is investigational. Novo Nordisk submitted the New Drug Application on December 18, 2025. The company has guided to FDA review in 2026, but no PDUFA date has been publicly confirmed and no commercial launch date has been set.
“Submission is not approval.” A filing is a starting gun, not a finish line.
Timeline so far
- June 22, 2025REDEFINE 1 results published in the New England Journal of Medicine. Cagrisema produced 22.7% weight loss at 68 weeks (trial-product estimand) vs 16.1% for semaglutide alone.
- December 18, 2025Novo Nordisk filed the NDA with the FDA for Cagrisema for adults with obesity or overweight plus at least one weight-related condition.
- February 2, 2026Novo Nordisk announced topline REIMAGINE 2 results. In adults with type 2 diabetes, Cagrisema beat Wegovy head-to-head on both weight loss and blood sugar. (Company-announced; not yet peer-reviewed.)
- February 23, 2026Novo Nordisk announced topline REDEFINE 4 results. Cagrisema was tested directly against Zepbound (tirzepatide 15 mg) and missed its non-inferiority goal. (Company-announced.)
When will Cagrisema actually be available?
If the FDA approves it in late 2026, Novo Nordisk still needs to complete launch preparation. Pharmacy availability typically follows 1–3 months after approval for a commercial launch. Insurance coverage typically follows another 6–12 months as payers negotiate pricing.
Realistic timeline for a typical US consumer: not before late 2026 at the earliest; broader access likely 2027. That’s why “I’ll just wait” is a bigger time commitment than many searchers realize. Twelve to eighteen months of no progress is a long time in a body that’s ready to change.
Start-now vs watch-and-wait: the decision paths for Cagrisema vs Wegovy in 2026.
Cagrisema vs Wegovy: the actual head-to-head trial data
The short answer: Cagrisema beat Wegovy in every comparison that’s been run. It also lost — clearly — to Zepbound in the only trial against tirzepatide. That second fact is the one most comparison pages skip, and it changes the decision.
| Trial | Population | Duration | Cagrisema | Comparator | Difference |
|---|---|---|---|---|---|
| REDEFINE 1 (NEJM) | 3,417 adults with obesity, no diabetes | 68 weeks | −22.7% (trial-product) | Semaglutide: −16.1% / Cagrilintide: −11.8% / Placebo: −2.3% | +6.6 pp vs Wegovy |
| REDEFINE 1 (treatment-policy) | Same | 68 weeks | −20.4% | Semaglutide: −14.9% / Placebo: −3.0% | +5.5 pp vs Wegovy |
| REDEFINE 2 (NEJM) | 1,206 adults with T2D | 68 weeks | −13.7% to −15.7% | Placebo: −3.1% to −3.4% | +10.6 to +12.6 pp vs placebo |
| REIMAGINE 2 (topline) | ~2,728 adults with T2D | 68 weeks | −14.2%, HbA1c −1.91 pp | Wegovy: −10.2%, HbA1c −1.76 pp | +4.0 pp vs Wegovy |
| REDEFINE 4 (topline) | 809 adults with obesity | 84 weeks | −23.0% (trial-product) / −20.2% (treatment-policy) | Tirzepatide: −25.5% / −23.6% | −2.5 pp / −3.4 pp — non-inferiority NOT met |
Sources: Garvey WT et al., NEJM 2025 (NEJMoa2502081) and Davies MJ et al., NEJM 2025 (NEJMoa2502082) for REDEFINE 1 and 2; Novo Nordisk press releases Feb 2 and Feb 23, 2026 for REIMAGINE 2 and REDEFINE 4.
The cleaner apples-to-apples number
A lot of articles throw around “22.7% vs 15%” like it’s a clean comparison. It isn’t — those numbers come from different trials, different populations, different analysis methods. The cleaner comparison lives inside REDEFINE 1, where Cagrisema and semaglutide were tested in the same trial, same population, same conditions.
Using the treatment-policy estimand — which counts everyone enrolled, the way real life does — Cagrisema produced 20.4% weight loss vs 14.9% for semaglutide alone. That’s a 5.5 percentage point advantage. Translated into pounds for a 230-pound starting weight: roughly 47 lbs on Cagrisema vs 34 lbs on Wegovy. Real difference.
REIMAGINE 2 is the direct head-to-head between Cagrisema and Wegovy in adults with type 2 diabetes. Topline results: Cagrisema produced 14.2% weight loss vs 10.2% for Wegovy, plus stronger blood sugar reduction (HbA1c −1.91 vs −1.76 percentage points). If you have T2D and obesity, Cagrisema is a more potent drug in this setting. The problem is it’s still not a drug you can get.
Is Zepbound actually better than both Cagrisema and Wegovy?
Here’s the honest thing we have to put plainly: if you’re searching “cagrisema vs wegovy” because you want the strongest weight-loss drug available to you, the honest answer isn’t Wegovy — it’s Zepbound.
Zepbound is FDA-approved. It’s available now. In the only Phase 3 trial that’s put Cagrisema and Zepbound side-by-side, Zepbound produced more weight loss than Cagrisema did. We’d be doing you a disservice to point you anywhere else if maximum weight loss is your priority.
Wegovy isn’t a weaker drug in every sense. It has an FDA cardiovascular outcomes indication Zepbound is still pursuing, a pill form Zepbound doesn’t offer, and for anyone with diagnosed heart disease, Wegovy is exactly right. The decision sits on what you need.
If you want Zepbound specifically: Ro carries tirzepatide through their GLP-1 program. Program pricing is $39 for the first month, then as low as $74/month with annual plan paid upfront, or $149/month ongoing without prepay. We have a dedicated guide with current Zepbound savings card and pricing details if you want the numbers before you click through.
Does this sound like you? A short eligibility screening on Ro tells you whether Zepbound is a fit for your body and your insurance. It’s not a commitment.
How does Cagrisema work compared to Wegovy?
Wegovy uses one hormone pathway. Cagrisema uses two.
Wegovy (semaglutide) mimics GLP-1 — a gut hormone released after meals. It slows stomach emptying, quiets hunger signaling in the hypothalamus, and stabilizes blood sugar. One mechanism, well-proven, FDA-approved since 2021 for chronic weight management.
Cagrisema adds amylin modulation on top of GLP-1. Cagrilintide is a long-acting synthetic version of amylin, a hormone produced in the same pancreatic beta cells that make insulin. Amylin works through the hindbrain (the area postrema) — a different brain region from GLP-1’s main targets. The idea: two appetite pathways, same injection.
In REDEFINE 1, that second pathway produced about 6 additional percentage points of weight loss. It also produced about 6 percentage points more total GI adverse events (79.6% vs 73.8% for semaglutide alone in the same trial). There’s no free lunch.
The REDEFINE trials used a dual-chamber pen that delivers both medications in a single injection. The commercial device format at launch — if and when the FDA approves Cagrisema — has not been publicly finalized.
For a deeper look at how Cagrisema works and what the amylin pathway means for long-term obesity treatment, see our What is Cagrisema? guide.
Side effects: Cagrisema vs Wegovy compared
Both drugs cause gastrointestinal side effects. Cagrisema causes meaningfully more total GI events than Wegovy in the same trial, consistent with two appetite pathways acting together on the gut during titration.
| Category | Cagrisema (REDEFINE 1) | Wegovy — semaglutide 2.4 mg (REDEFINE 1) | Wegovy — FDA label, adults 2.4 mg |
|---|---|---|---|
| Any GI adverse event | 79.6% | 73.8% | — |
| Nausea | Elevated vs semaglutide and placebo | Elevated vs placebo | 44% |
| Diarrhea | Elevated vs placebo | Elevated vs placebo | 30% |
| Vomiting | Elevated vs placebo | Elevated vs placebo | 24% |
| Constipation | Elevated vs placebo | Elevated vs placebo | 24% |
| Serious adverse events | Highest of the four trial arms | Lower | — |
| Discontinuation due to side effects | Rare but highest in Cagrisema arm | Rare | — |
Sources: Garvey et al., NEJM 2025 (REDEFINE 1); Wegovy prescribing information, FDA DailyMed.
Warnings and contraindications
Because Cagrisema contains semaglutide, semaglutide-related safety concerns and contraindications will be relevant to any future Cagrisema label. Cagrisema does not yet have approved prescribing information.
- Boxed warning — thyroid C-cell tumors: Wegovy’s approved label carries a boxed warning based on rodent studies showing thyroid C-cell tumors. Human risk is not established.
- Do not use if: You or a family member has a history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- Pancreatitis risk: Seek care for severe, persistent abdominal pain.
- Gallbladder disease: Rapid weight loss plus GLP-1 activity raises gallstone risk.
- Diabetic retinopathy: Rapid improvement in blood sugar can temporarily worsen existing retinopathy.
- Hypoglycemia: Particularly when combined with insulin or sulfonylureas.
Source: Wegovy prescribing information, FDA DailyMed, verified April 20, 2026. None of this is a reason to avoid GLP-1 therapy. It’s a reason to have a real conversation with a prescriber before starting.
What does Wegovy cost right now, and where can you get it?
Wegovy is sold today as a once-weekly injection pen, Wegovy HD, and a once-daily oral tablet. Each form has its own pricing structure.
Wegovy injection pen (self-pay through NovoCare)
| Pathway | 0.25 mg / 0.5 mg | 1 mg / 1.7 mg / 2.4 mg | Wegovy HD 7.2 mg |
|---|---|---|---|
| List price | $1,349.02/mo | $1,349.02/mo | $1,349.02/mo |
| NovoCare intro offer (new patients, 2 fills through June 30, 2026) | $199/mo | — | — |
| NovoCare standard cash-pay | $349/mo | $349/mo | $399/mo |
| Commercial insurance + Wegovy Savings Offer | As low as $25/mo | As low as $25/mo | As low as $25/mo |
Wegovy tablet (self-pay through NovoCare)
| Pathway | 1.5 mg | 4 mg | 9 mg | 25 mg |
|---|---|---|---|---|
| List price | $1,349.02/mo | $1,349.02/mo | $1,349.02/mo | $1,349.02/mo |
| NovoCare cash-pay | $149/mo | $149/mo through Aug 31, 2026, then $199/mo | $299/mo | $299/mo |
| Commercial insurance + Wegovy Savings Offer | As low as $25/mo | As low as $25/mo | As low as $25/mo | As low as $25/mo |
Source: novocare.com and wegovy.com, verified April 20, 2026. Intro and savings offers apply to new patients per program terms. Government-insurance beneficiaries are excluded from the $25/month commercial savings offer. Full terms at WegovyTerms.com.
Need the pill-specific details? See our dedicated Wegovy pill guide covering dose escalation, pharmacy options, and how the pill compares to the pen for needle-averse readers.
If you’re on government insurance
Medicare, Medicaid, VA, and TRICARE readers have separate rules. The Wegovy Savings Offer excludes government beneficiaries. See our Wegovy Medicare coverage guide. If you have an HSA or FSA, our HSA/FSA for GLP-1 guide walks through what qualifies.
Should you wait for Cagrisema or start Wegovy now?
For most people eligible for weight-loss medication, don’t wait.
Starting a proven FDA-approved option today produces 12–18 months of real progress before Cagrisema even launches. And because Zepbound already outperformed Cagrisema in head-to-head testing, “wait for Cagrisema” isn’t a clean upgrade case — it’s a choice to defer treatment for a drug that may not even be the strongest option when it arrives.
| Your situation | Our recommendation | Why |
|---|---|---|
| Currently on Wegovy and losing weight steadily | Don’t switch. Stay on Wegovy. | Switching a working medication for a theoretical one you can’t access is the highest-regret move in treatment decisions. |
| Never started and eligible for Wegovy | Start now. Wegovy or Zepbound. | Eighteen months of actual weight loss beats 18 months of standing still. |
| Plateaued on Wegovy after 12+ months | Talk to your prescriber about Zepbound — not Cagrisema later. | REDEFINE 4 showed Cagrisema 23.0% vs tirzepatide 25.5%. Waiting for Cagrisema to break a plateau means choosing the slightly weaker drug 12+ months later. |
| T2D and obesity together | Start Wegovy or Ozempic now; put Cagrisema on your watchlist. | This is Cagrisema’s strongest data population per REIMAGINE 2. But “later” isn’t a treatment plan. |
| Uninsured or insurance doesn’t cover weight-loss drugs | Start Wegovy via NovoCare or Success by Sesame. | Wegovy has a full cash-pay infrastructure at $149–$399/month. Cagrisema will launch at or above list price with no equivalent program for its first 12+ months. |
| Treatment-naive, wanting strongest weight loss available | Talk to your prescriber about Zepbound (tirzepatide). | Zepbound delivered 25.5% weight loss vs Cagrisema’s 23.0% in REDEFINE 4. It’s FDA-approved and available now via Ro. |
| Genuinely willing to wait with no progress | Put Cagrisema on your watchlist and check back here quarterly. | If Cagrisema is approved, you’ll be able to act informed instead of guessing. Waiting is a real option — just make it on purpose. |
The “start now and switch later” case
The most common question we get is some version of: “Can I start Wegovy now and switch to Cagrisema later if it’s better for me?”
Honest answer: nobody can promise that path today because Cagrisema doesn’t have an approved label, switching protocols, or insurance rules yet. But here’s the shape. GLP-1 medications in the same class can generally be titrated between under a prescriber’s guidance. Starting Wegovy now builds tolerance to GI side effects, establishes your GLP-1 response, and gets the weight coming off. If and when Cagrisema is approved, you’ll have a year-plus of real data about how your body responds to semaglutide — which makes any “is this worth switching” conversation with your prescriber real instead of theoretical.
Still not sure which path fits you? Take our free 60-second GLP-1 matcher. We’ll ask about your goals, your insurance, and your timing, and route you to the right FDA-approved option for your situation. No guessing.
Where can you actually get Wegovy or Zepbound right now?
Three legitimate routes. We’ll match each to the reader it fits best.
Ro — best fit when you want structure, insurance help, and access to the strongest current data
Ro operates a consumer GLP-1 weight-loss program with FDA-approved medications, including Zepbound (tirzepatide) and Foundayo (orforglipron). Program pricing is $39 first month, then $149/month ongoing, or as low as $74/month with annual plan paid upfront. Medication cost is separate. What makes Ro the right fit for most readers searching this page: real insurance infrastructure. Ro’s free GLP-1 Insurance Coverage Checker and insurance concierge handle prior-authorization paperwork — uncommon in telehealth and meaningful if you’ve ever tried to do a PA yourself. (Note: Ro currently does not coordinate GLP-1 coverage for government insurance plans — Medicare, Medicaid, VA, TRICARE.)
Best fit: you want the strongest data-supported current GLP-1 (Zepbound), you want structured monthly support, and you want someone working your insurance benefits alongside you.
Success by Sesame — best fit for the price-conscious self-pay shopper
Sesame runs a GLP-1 subscription called Success by Sesame. Pricing: $59/month with annual plan, or $99/month month-to-month. Medication cost is separate. Formulary includes Wegovy, Zepbound, Ozempic, and Mounjaro.
Best fit: you want transparent subscription pricing, you don’t need a high-touch monthly program, and you want medication choice that spans multiple FDA-approved GLP-1s.
NovoCare Pharmacy — best fit when you’ve decided on Wegovy specifically
If you’ve decided on Wegovy and you have a prescription (or your current doctor will write one), NovoCare is the manufacturer-direct path. Pricing is what we showed in the cost matrix above. Home delivery included.
Best fit: you already have a prescribing relationship and want the shortest path from “I have a prescription” to “I have my medication.”
Why you can’t legally buy Cagrisema today (and what to avoid)
Cagrisema is investigational. Cagrilintide — the other half of Cagrisema — cannot legally be used in compounding.
FDA statement (verbatim)
“Retatrutide and cagrilintide cannot be used in compounding under federal law. Additionally, these are not components of FDA-approved drugs and have not been found safe and effective for any condition.”
— U.S. Food and Drug Administration, “FDA’s Concerns with Unapproved GLP-1 Drugs Used for Weight Loss”
Any telehealth company, compounding pharmacy, peptide site, or online marketplace selling compounded cagrisema, compounded cagrilintide, or a cagrisema-style compounded combination is operating outside federal law.
Red-flag signals
- “Compounded cagrisema” or any compounded cagrilintide product on a consumer telehealth site.
- “Cagrilintide for research use only” sold alongside weight-loss consultations — the disclaimer does not make it legal to use in humans.
- Prices that are dramatically low for an investigational drug.
- Any phrase like “same active ingredients as Cagrisema” applied to a compounded product.
- A pharmacy offering to ship cagrilintide to consumers — investigational drugs can’t be shipped to consumers outside a trial.
The only legitimate way to access Cagrisema today
Clinical trial enrollment. The REDEFINE program has multiple active trials and the REIMAGINE program continues in type 2 diabetes. Search “Cagrisema” or “cagrilintide semaglutide” on ClinicalTrials.gov and talk to your prescriber about whether a site near you is enrolling.
Cagrisema vs Zepbound, Ozempic, and Mounjaro — the adjacent comparisons
Cagrisema vs Zepbound (tirzepatide)
Zepbound is the currently-approved drug Cagrisema was aiming to beat. In REDEFINE 4, it didn’t. Cagrisema: 23.0% weight loss at 84 weeks. Zepbound: 25.5%. Non-inferiority not met. Zepbound is approved for chronic weight management and available now. See our Cagrisema vs Zepbound guide for the full head-to-head and our Zepbound savings card guide for current pricing.
Cagrisema vs Ozempic
Ozempic is semaglutide for type 2 diabetes — the same active ingredient as Wegovy at different dose tiers. It’s FDA-approved for T2D, not weight loss, though prescribers sometimes use it off-label. If your goal is T2D control with weight loss as a secondary benefit, you’re in the Ozempic conversation, not the Cagrisema conversation, until Cagrisema has a T2D indication.
Cagrisema vs Mounjaro
Mounjaro is Zepbound’s T2D sibling — same molecule, tirzepatide, different FDA-approved indication. It’s approved for type 2 diabetes, sometimes prescribed off-label for weight loss. In the indirect comparison, tirzepatide-based drugs have produced more weight loss in head-to-head testing than Cagrisema.
Cagrisema vs Saxenda
Saxenda is liraglutide — an older, daily-injection GLP-1 — with roughly 7% average weight loss. Cagrisema produced roughly three times that in trials. Saxenda’s niche today is mostly pediatric use and patients who want daily dose flexibility.
What other GLP-1 drugs are coming in 2026 and 2027?
Short pipeline context to put Cagrisema in perspective.
- Foundayo (orforglipron): FDA-approved in 2026. The first oral small-molecule GLP-1. Available through Ro. Real option for readers who want to avoid injections.
- Retatrutide: Eli Lilly’s investigational triple-receptor agonist (GLP-1 + GIP + glucagon). Phase 3 TRIUMPH-4 produced 28.7% weight loss at 68 weeks — the highest number reported so far in this class. Lilly has not publicly filed an NDA as of April 2026. See our Cagrisema vs Retatrutide guide.
- Cagrisema: What you’re reading about. NDA decision expected in 2026.
The headline: obesity pharmacotherapy is moving fast. Waiting for one specific drug is a bet against the rest of the pipeline.
Frequently asked questions
Is Cagrisema better than Wegovy?
On trial weight-loss data: yes. Cagrisema produced 22.7% mean weight loss vs 16.1% for semaglutide alone in REDEFINE 1 and beat Wegovy 14.2% vs 10.2% head-to-head in REIMAGINE 2 (type 2 diabetes). Cagrisema also has higher total gastrointestinal adverse events, isn’t FDA-approved, and missed non-inferiority against Zepbound. “Better” depends on availability, side-effect tolerance, cost, and what you’re comparing against.
Is Cagrisema FDA-approved yet?
No. Novo Nordisk filed the New Drug Application on December 18, 2025. FDA review is expected in 2026 per company guidance. No PDUFA date has been publicly confirmed.
When will Cagrisema be available?
If approval lands in 2026, realistic US patient availability is late 2026 at the earliest, with broader access likely 2027 depending on launch logistics and insurance coverage timing.
How much will Cagrisema cost?
Novo Nordisk has not disclosed pricing. No public launch price or savings program exists yet.
Can I switch from Wegovy to Cagrisema?
Not today — Cagrisema isn’t available. If and when it’s approved, switching will be a conversation with your prescriber based on how you’re doing on Wegovy, your side-effect tolerance, and whether Cagrisema offers meaningful additional benefit for your situation.
Does Cagrisema contain the same active ingredient as Wegovy?
No. Cagrisema contains two active ingredients: cagrilintide (an amylin analogue) plus semaglutide (the active ingredient in Wegovy). The added cagrilintide is what makes it a different drug.
Can I buy compounded Cagrisema now?
No. The FDA has explicitly stated that cagrilintide cannot be used in compounding under federal law. Any site selling “compounded cagrisema” or compounded cagrilintide is operating outside federal law.
Is Cagrisema better than Zepbound?
No. In REDEFINE 4, the only Phase 3 trial directly comparing them, Zepbound produced 25.5% weight loss vs Cagrisema’s 23.0% at 84 weeks. Cagrisema missed its non-inferiority goal. If your priority is maximum weight loss, Zepbound (available now) is the stronger data-driven choice.
Does Cagrisema reduce cardiovascular risk like Wegovy?
Not established. Wegovy has a specific FDA indication for reducing major cardiovascular events in adults with heart disease. Cagrisema’s cardiovascular outcomes trial (REDEFINE 3) is ongoing.
Can I get Cagrisema through a clinical trial?
Yes — trial enrollment is the only legal US access path today. Search ClinicalTrials.gov for “Cagrisema” or “cagrilintide semaglutide.” Check individual study pages for enrollment criteria and terms.
What’s the difference between Wegovy pill and Wegovy pen?
Both are semaglutide. The pen is a once-weekly subcutaneous injection (doses 0.25–2.4 mg plus HD 7.2 mg). The tablet is a once-daily oral form (1.5, 4, 9, 25 mg). Self-pay pricing differs: the tablet starts at $149/month, the standard pen is $349/month through NovoCare.
Should I wait for Cagrisema if I’m already on Wegovy and it’s working?
No. Don’t switch a working treatment for a drug you can’t access. If you plateau or Cagrisema is approved and your prescriber agrees it’s a meaningful upgrade, you can evaluate then. Real progress beats theoretical progress.
Is waiting for Cagrisema ever the right call?
Yes — in narrow cases. If you have no urgency, strong financial resources to wait, and a clear preference for Novo Nordisk’s dual-hormone approach, waiting is a defensible choice. Just know it’s a 12+ month commitment of no progress, and Zepbound is already producing more weight loss than Cagrisema did in head-to-head testing.
Still not sure which GLP-1 program is right for you?
We get it — “cagrisema vs wegovy” wasn’t really your question. The real question was: what should I actually do? Take our free 60-second matching quiz. We’ll ask about your goals, your insurance situation, your budget, your timeline, and your medical history, and route you to the right FDA-approved GLP-1 option for your specific situation.
Sources and further reading
Primary sources for this page, all verified April 20, 2026:
- U.S. Food and Drug Administration — “FDA’s Concerns with Unapproved GLP-1 Drugs Used for Weight Loss” (fda.gov)
- NovoCare — Wegovy pricing and availability (novocare.com/patient/medicines/wegovy.html)
- Wegovy.com — official pricing page (wegovy.com/obesity/what-to-pay-for-wegovy.html)
- Novo Nordisk — CagriSema NDA submission press release, December 18, 2025
- Garvey WT, et al. — “Coadministered Cagrilintide and Semaglutide in Adults with Overweight or Obesity” — New England Journal of Medicine, June 2025 (NEJMoa2502081)
- Davies MJ, et al. — “Cagrilintide–Semaglutide in Adults with Overweight or Obesity and Type 2 Diabetes” — New England Journal of Medicine, June 2025 (NEJMoa2502082)
- Novo Nordisk — REIMAGINE 2 topline results press release, February 2, 2026
- Novo Nordisk — REDEFINE 4 topline results press release, February 23, 2026
- Wegovy prescribing information — FDA DailyMed
- ClinicalTrials.gov — NCT05567796 (REDEFINE 1), NCT05394519 (REDEFINE 2), REIMAGINE and REDEFINE series
- Ro — Weight-loss program pricing (ro.co/weight-loss)
- Sesame Care — Success by Sesame subscription details (sesamecare.com)
How we built this page
The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We don’t accept payment for placement in our recommendations. Our affiliate relationships are disclosed — Ro and Success by Sesame are partners whose offers we recommend when the evidence supports the fit.
For this page, we reviewed Novo Nordisk’s complete regulatory and trial record through April 2026, the FDA’s published guidance on unapproved GLP-1s, current Wegovy prescribing information from FDA DailyMed, live pricing pages at NovoCare, Ro, and Success by Sesame, and peer-reviewed Phase 3 data in the New England Journal of Medicine for REDEFINE 1 and 2.
When something couldn’t be verified against a primary source, we labeled it as an estimate or omitted it. When the easy conclusion would have been “Wegovy is better because we can link to Wegovy,” we told you Zepbound has better head-to-head data instead.
Spot an outdated price, a changed FDA status, or a mistake? Email us. We update this page monthly — and weekly during the Cagrisema review window.
This article is informational and does not replace medical advice. Talk to a licensed clinician before starting or changing any GLP-1 medication.
Last verified: April 20, 2026.