Disclosure: Some links on this page are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you.
What Is CagriSema? FDA Status, Trial Results & What to Do Now
Published:

CagriSema in one glance
| Question | Answer |
|---|---|
| What it is | Cagrilintide 2.4 mg + semaglutide 2.4 mg fixed-dose combo |
| Made by | Novo Nordisk |
| How given | Once-weekly subcutaneous injection |
| FDA status | NDA filed Dec 18, 2025 — decision expected late 2026 |
| Best weight loss result | 22.7% over 68 weeks (REDEFINE 1, if-all-adhered) |
| Beat Wegovy (semaglutide alone)? | Yes — 22.7% vs 16.1% in the same trial |
| Beat Zepbound (tirzepatide 15 mg)? | No — 23.0% vs 25.5%, missed non-inferiority |
| Can you buy it now? | No. For most people, only via registered clinical trial. |
If you need weight-loss treatment now:
We’ll show you which FDA-approved option fits your budget, insurance, and goals. No email required to see your results.
Take our free 60-second GLP-1 matching quiz →What is CagriSema?
CagriSema is an investigational fixed-dose combination injection from Novo Nordisk that puts two weight-loss molecules — cagrilintide and semaglutide — into a single once-weekly shot. It’s being studied for chronic weight management in adults with obesity or overweight (the REDEFINE program) and for type 2 diabetes (the REIMAGINE program). It is not approved in the U.S. or the EU today.
The name is literally “Cagri” (cagrilintide) plus “Sema” (semaglutide). If it gets approved, it will be the first-ever injectable that pairs a GLP-1 receptor agonist with an amylin analogue in one fixed dose.
Every currently approved GLP-1 weight-loss drug hits one or two related appetite pathways. Wegovy and Ozempic hit GLP-1. Zepbound and Mounjaro hit GLP-1 and GIP. CagriSema is the first to pair GLP-1 (semaglutide) with amylin (cagrilintide) — a different hormone entirely, with a different signal to your brain about fullness. On paper, that’s why it should work better than semaglutide alone. And in the trials, it did. But “better than semaglutide alone” and “the best drug on the market” are not the same sentence.
Why are so many people searching for CagriSema right now?
Three dates reset the conversation: December 18, 2025 (Novo filed with the FDA), June 22, 2025 (REDEFINE 1 published in NEJM with a 22.7% headline), and February 23, 2026 (the head-to-head vs tirzepatide that missed its endpoint). That sequence created the exact mix of hype, confusion, and “wait, is this actually better?” that sent people to Google and here.
The New England Journal of Medicine paper from June 22, 2025 landed with coverage calling CagriSema the “next-generation” Novo Nordisk weight-loss drug. People plateaued on Wegovy started asking their doctors when they could switch. People brand-new to GLP-1s started asking if they should wait.
Then on February 23, 2026, the head-to-head data didn’t land the way Novo’s investors wanted. CagriSema delivered 23.0% weight loss vs 25.5% for tirzepatide and did not meet its pre-specified non-inferiority endpoint. Novo’s U.S. shares dropped sharply that morning. Patient forums lit up. And here you are.
What people in trial communities are actually saying
Three real comments from public trial-community threads. These are anecdotes from individuals — not typical results, not medical evidence:
- “ZERO hunger and food noise” — u/ClinTrial-Throwaway, r/Zepbound
- “the side effects are REAL for me” — u/RecordingTimely5707, r/Semaglutide
- “yeah so this sh*t works” — u/MoneyMedusa, r/Semaglutide
Genuine excitement at appetite suppression. Genuine frustration at GI side effects. Both are real. Neither is the whole story.
How does CagriSema work for weight loss?
CagriSema works by hitting two appetite hormones at the same time. Semaglutide activates GLP-1 receptors, which slow gastric emptying and suppress appetite. Cagrilintide mimics amylin — a hormone your pancreas releases alongside insulin after a meal — which increases the feeling of fullness. One drug tells your gut to slow down. The other tells your brain you’ve had enough. Together, they produce a stronger satiety signal than either one does alone.
How CagriSema’s dual-pathway approach compares to approved GLP-1 drugs
| Drug | Molecule(s) | Hormone pathways targeted |
|---|---|---|
| Wegovy / Ozempic | Semaglutide | GLP-1 |
| Zepbound / Mounjaro | Tirzepatide | GLP-1 + GIP |
| CagriSema (investigational) | Cagrilintide + semaglutide | GLP-1 + amylin |
| Saxenda | Liraglutide | GLP-1 (short-acting) |
What do the CagriSema trials actually show?
CagriSema produced strong weight loss across multiple Phase 3 trials — but the numbers are different for obesity without diabetes, obesity with diabetes, and head-to-head against tirzepatide. In REDEFINE 1 (obesity, no diabetes): 22.7%. In REDEFINE 2 (obesity + T2D): 15.7%. In REDEFINE 4 (vs tirzepatide 15 mg): 23.0% vs 25.5% for tirzepatide — missed non-inferiority.
The RX Index REDEFINE Clinical Trial Dashboard
| Trial | Who | Duration | N | Weight loss (if-all-adhered) | Weight loss (real-world) | Endpoint |
|---|---|---|---|---|---|---|
| REDEFINE 1 | Obesity/overweight, no T2D, ≥1 comorbidity | 68 wks | 3,417 | 22.7% vs 2.3% placebo | 20.4% vs 3.0% placebo | ✓ Met |
| REDEFINE 2 | Obesity/overweight + T2D | 68 wks | 1,206 | 15.7% vs 3.1% placebo | 13.7% vs 3.4% placebo | ✓ Met |
| REDEFINE 4 | Obesity, BMI ≥30 | 84 wks | 809 | 23.0% vs 25.5% tirz | 20.2% vs 23.6% tirz | ✗ Missed non-inferiority |
| REDEFINE 3 | Established CVD ±T2D | Event-driven | ~7,000 | In progress | In progress | Pending |
| REDEFINE 11 | Obesity | 80 wks + 80-wk extension | 600 | Expected H1 2027 | Expected H1 2027 | Pending |
| REDEFINE 9 | Obesity/overweight | 68 wks | 300 | Ongoing (lower doses) | Ongoing | Pending |
| High-dose (2.4/7.2 mg) | Obesity | TBD | TBD | Trial starts H2 2026 | — | Pending |
REDEFINE 1 (obesity, no type 2 diabetes)
This was the big one. 3,417 adults, 68 weeks, four arms: CagriSema, semaglutide alone, cagrilintide alone, and placebo. Among people who stayed on treatment (trial product estimand), CagriSema produced 22.7% body weight loss vs 16.1% for semaglutide alone and 11.8% for cagrilintide alone. About 40.4% of CagriSema patients on that same estimand lost at least 25% of their body weight. Published in NEJM, June 2025.
REDEFINE 2 (obesity + type 2 diabetes)
1,206 adults with type 2 diabetes and BMI ≥27. Weight loss was lower than in REDEFINE 1 — as it typically is for any GLP-1 drug in a diabetic population — at 15.7% (if-all-adhered) or 13.7% (treatment policy). Glycemic control was strong: 73.5% of CagriSema patients hit an HbA1c of 6.5% or lower, vs 15.9% on placebo. Published in NEJM, 2025.
REDEFINE 4 (head-to-head vs tirzepatide) — the honest tradeoff
This is the trial that reset expectations. 809 adults with obesity, 84 weeks, open-label, CagriSema vs tirzepatide 15 mg. CagriSema delivered 23.0% weight loss among adherent patients vs 25.5% for tirzepatide. Under real-world adherence: 20.2% vs 23.6%. The trial did not achieve its pre-specified primary endpoint of demonstrating non-inferiority.
Is CagriSema better than Wegovy?
On trial data, yes — CagriSema beat semaglutide alone in REDEFINE 1 by about 6.6 percentage points (22.7% vs 16.1%, trial product estimand). But “better in a trial” and “the better decision for you right now” are two different sentences. Wegovy exists and you can get it. CagriSema does not, and you cannot.
If you’re already on Wegovy and losing weight well, the right move is almost always to stay on it. CagriSema’s trial edge does not beat your actual, working medicine.
Is CagriSema better than Zepbound or Mounjaro?
On current head-to-head evidence: no. In REDEFINE 4, CagriSema did not meet its pre-specified non-inferiority endpoint vs tirzepatide 15 mg. CagriSema produced 23.0% weight loss vs 25.5% for tirzepatide (if-all-adhered), or 20.2% vs 23.6% under real-world adherence.
Zepbound (tirzepatide, FDA-approved for chronic weight management) and Mounjaro (tirzepatide, FDA-approved for type 2 diabetes) are both on U.S. pharmacy shelves. On self-pay via LillyDirect, Zepbound single-dose vials are priced at $299/month for 2.5 mg, $399/month for 5 mg, and $449/month for 7.5 mg through 15 mg (with a 45-day refill requirement to maintain the $449 price on higher doses). No waiting, no FDA timeline to track.
Is CagriSema FDA approved?
No. CagriSema is not approved in the United States or the European Union as of April 2026. Novo Nordisk submitted the New Drug Application to the FDA on December 18, 2025, based on REDEFINE 1 and REDEFINE 2. The company has publicly stated that an FDA decision is anticipated by late 2026.
CagriSema FDA timeline
| Date | Event |
|---|---|
| 2022 | Phase 2 data: 15.6% weight loss at 32 weeks |
| December 2024 | REDEFINE 1 topline announced (22.7%) |
| March 2025 | REDEFINE 2 topline announced (15.7% in T2D) |
| June 22, 2025 | REDEFINE 1 and REDEFINE 2 published in New England Journal of Medicine |
| December 18, 2025 | Novo Nordisk files NDA with the FDA |
| February 23, 2026 | REDEFINE 4 topline: 23.0% vs tirzepatide’s 25.5%, missed non-inferiority |
| Late 2026 (expected) | FDA decision anticipated |
| H2 2026 (planned) | High-dose CagriSema 2.4/7.2 mg trial begins |
| H1 2027 (expected) | REDEFINE 11 longer-duration data |
| 2027 (expected if approved) | Commercial launch |
When will CagriSema be available?
There is no exact launch date. Novo filed in December 2025, a decision is anticipated by late 2026, and commercial launch — if approved — would most likely begin in 2027. Exact timing depends on FDA approval date, final label negotiations, and manufacturing scale-up.
Can you buy CagriSema now? And what about compounding?
No. There is no lawful routine retail path to obtain CagriSema in the U.S. in 2026. Here is every claimed path and what it actually is:
| Source claiming to offer it | What they say | Reality | Risk level |
|---|---|---|---|
| Telehealth clinic | “Cagrilintide-based weight-loss protocol” | No lawful routine prescribing path for an investigational drug outside a registered clinical trial | Illegal for standard prescription workflow |
| Peptide “research” supplier | “Cagrilintide, research use only” | “Research use only” disclaimers do not create a lawful consumer pathway. Selling investigational compounds for human consumption violates federal law. | Very high (purity/contamination) |
| International online pharmacy | “Order from overseas, no prescription needed” | Personal importation of unapproved drugs is generally prohibited under the FD&C Act. | Very high (counterfeit, customs, sterility) |
| “Custom” injectable on social media | DM-based sales, influencer referrals | No lawful pathway exists. | Very high (zero regulatory oversight) |
| Registered REDEFINE clinical trial | Participation in active Novo Nordisk Phase 3 study | The primary lawful U.S. pathway for most patients before FDA approval | Monitored clinical environment |
| FDA Expanded Access / Right to Try | Physician-led request outside a trial | Possible in narrow circumstances — not a routine consumer path. Case-by-case, physician-initiated, not guaranteed. | Physician-supervised |
⚠ Scam warning
If someone is offering you CagriSema, cagrilintide, or a “cagrilintide + semaglutide custom combo” today — it is not a legitimate retail product. The pricing doesn’t matter. The marketing doesn’t matter. There is no lawful routine way to obtain this drug in the U.S. in 2026 outside a clinical trial or a narrow physician-led expanded-access request.
See which currently FDA-approved option fits your situation →What are the side effects and safety tradeoffs?
The main tradeoff is gastrointestinal tolerability, and it’s real. In REDEFINE 1, 79.6% of CagriSema patients had a GI adverse event, compared to 39.9% on placebo. Nausea hit 55%, constipation 30.7%, and vomiting 26.1%. Most events were mild to moderate and diminished over time. Treatment discontinuation from adverse events was 6% on CagriSema vs 3.7% on placebo — manageable, but higher than placebo.
| Metric | REDEFINE 1 (CagriSema vs placebo) | REDEFINE 2 (CagriSema vs placebo) |
|---|---|---|
| Any GI adverse event | 79.6% vs 39.9% | 72.5% vs 34.4% |
| Nausea | 55.0% vs 12.6% | Similar class-consistent range |
| Constipation | 30.7% vs 11.6% | Similar class-consistent range |
| Vomiting | 26.1% vs 4.1% | Similar class-consistent range |
| Discontinuation due to AEs | 6% vs 3.7% | 8.4% vs 3% |
Can you get CagriSema now through a clinical trial?
Possibly — if you meet eligibility criteria for an active REDEFINE protocol in your area. Multiple CagriSema trials are listed on ClinicalTrials.gov, including REDEFINE 3 (cardiovascular outcomes) and REDEFINE 11 (longer-duration), plus the planned high-dose trial starting H2 2026. Recruiting status varies by site and protocol. See every REDEFINE NCT identifier and current status in our GLP-1 clinical trials tracker.
Before enrolling, ask:
- Is this protocol currently recruiting at a site near me?
- What are the inclusion/exclusion criteria? (Most exclude recent certain cancers, pancreatitis, or severe GI conditions.)
- How long is the treatment period? How often do I have to come in?
- What happens at end of trial — continued access or treatment stops?
- Is there compensation for time and travel?
Enrollment is a commitment. You’ll have scheduled clinic visits, blood draws, study surveys, and specific protocols. You might get the placebo, not the active drug. If you’re trying to actually lose weight on a predictable timeline, trial enrollment is usually not the right path.
ClinicalTrials.gov is the official registry. Search “CagriSema” or protocol IDs like “NCT06131437” (REDEFINE 4) for the current list.
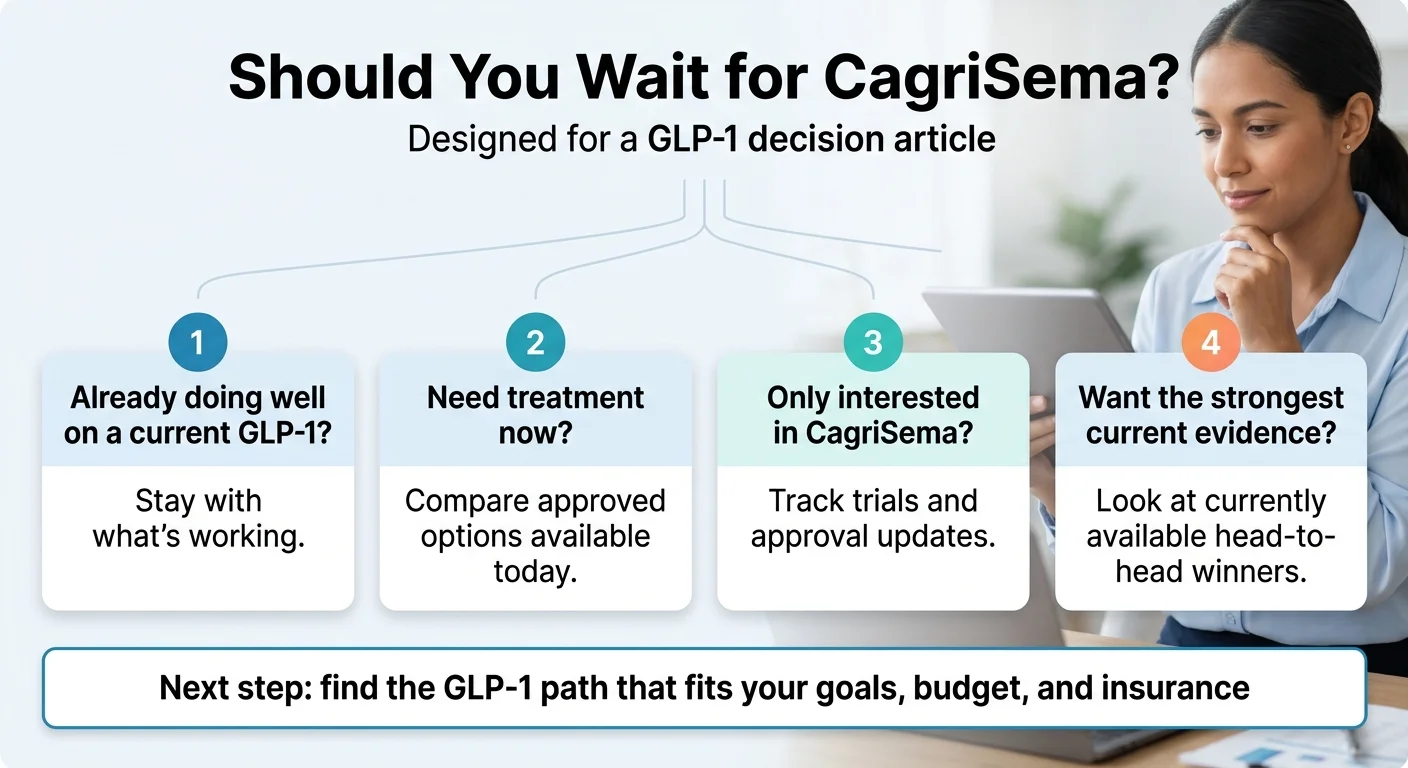
What’s still unknown about CagriSema?
Honest uncertainty is part of why the rest of this page is trustworthy. Here’s what we don’t know yet:
- Exact FDA PDUFA date. Novo has said “late 2026” but hasn’t published a specific date. We’ll update the moment one is announced.
- Commercial launch date. Tied to approval date + manufacturing readiness.
- Launch price. No Novo Nordisk disclosure yet.
- Launch distribution channels. NovoCare Pharmacy and telehealth partners are likely given Novo’s current strategy, but specific channel plans haven’t been announced.
- Insurance coverage. Coverage typically builds over 12–24 months for new obesity drugs; CagriSema specifics are unknown.
- Final FDA label. Age range, BMI thresholds, contraindications, and boxed warnings are all answered only at approval.
- Real-world long-term safety. Trial data covers up to 84 weeks. Post-market surveillance will shape the full picture.
- REDEFINE 11 data — expected H1 2027.
- High-dose (2.4/7.2 mg) trial results — trial starts H2 2026; data likely not until 2028.
Frequently asked questions about CagriSema
Is CagriSema FDA approved?
No. As of April 2026, CagriSema is not approved in the U.S. or EU. Novo Nordisk filed the NDA with the FDA on December 18, 2025, and has said a decision is anticipated by late 2026.
When will CagriSema be available?
There is no exact launch date. Novo filed in December 2025 and has said an FDA decision is anticipated by late 2026; commercial launch, if approved, would most likely begin in 2027.
What is CagriSema made of?
It’s a fixed-dose combination of cagrilintide 2.4 mg (a long-acting amylin analogue) and semaglutide 2.4 mg (the GLP-1 receptor agonist in Wegovy and Ozempic), in a single once-weekly subcutaneous injection.
How does CagriSema work?
Semaglutide activates GLP-1 receptors to slow digestion and reduce appetite. Cagrilintide mimics amylin to increase feelings of fullness. Together, they produce stronger satiety than semaglutide alone.
Is CagriSema better than Wegovy?
On REDEFINE 1 trial data, yes — CagriSema produced 22.7% average weight loss vs 16.1% for semaglutide alone in the same trial (trial product estimand). But Wegovy is available now; CagriSema is not.
Is CagriSema better than Zepbound?
Not on current head-to-head evidence. In REDEFINE 4, CagriSema produced 23.0% weight loss vs 25.5% for tirzepatide 15 mg and missed its non-inferiority endpoint.
Can you buy CagriSema now?
No. CagriSema is not approved, and FDA states cagrilintide cannot be used in compounding under federal law. For most people, the only realistic pre-approval path is enrollment in a registered clinical trial.
Can CagriSema be compounded?
No. FDA has publicly stated that cagrilintide cannot be used in compounding under federal law. Any pharmacy claiming to compound CagriSema or cagrilintide is operating outside the law.
Are there any ways to get CagriSema outside a clinical trial?
In narrow circumstances, FDA Expanded Access or state Right-to-Try programs may allow physician-led requests for pre-approval access. These are case-by-case, physician-initiated, not guaranteed, and not a routine consumer purchase path.
What are the side effects of CagriSema?
Mostly gastrointestinal: in REDEFINE 1, nausea occurred in 55% of CagriSema patients, constipation in 30.7%, and vomiting in 26.1%. Most were mild to moderate and diminished over time. Treatment discontinuation from adverse events was 6% on CagriSema vs 3.7% on placebo.
Will CagriSema be covered by insurance?
Unknown — it’s not approved yet. Commercial coverage for new obesity drugs typically takes 12–24 months to build at launch. Medicare does not generally cover weight-loss GLP-1s.
Should I wait for CagriSema or start a GLP-1 now?
For most people, starting now beats waiting. The currently FDA-approved weight-management options (Wegovy and Zepbound) work today, the head-to-head evidence doesn’t show CagriSema clearly winning, and CagriSema’s launch is at least 12–18 months away.
Still not sure which GLP-1 program is right for you?
You came here to find out what CagriSema is, whether you can get it, and whether to wait. You now know: it’s real, you can’t get it through normal channels, and waiting usually isn’t the answer. The next step is figuring out which currently-available path fits your situation — injection or pill, insurance or self-pay, maximum weight loss or gentle onboarding.
Take the free 60-second GLP-1 matching quiz →One minute. No email required to see results.
Related guides
- Next-Generation GLP-1 Drugs Coming Soon (2026) — the full pipeline including retatrutide, cagrisema, maritide, and more
- Foundayo Cost Without Insurance: Full Pricing Guide — the first new-molecular-entity oral GLP-1 pill, already approved April 2026
- Get Foundayo Covered by Insurance in 2026 — every coverage lane for the only oral GLP-1 approved in 2026
- How to Bypass Step Therapy for GLP-1 Medications — for when your plan forces you onto a different drug first
Sources
- Novo Nordisk. “Novo Nordisk files for FDA approval of CagriSema…” PR Newswire, December 18, 2025.
- Novo Nordisk. “CagriSema demonstrated 23% weight loss in an open-label head-to-head REDEFINE 4 trial…” February 23, 2026.
- REDEFINE 1 and REDEFINE 2 results. New England Journal of Medicine, June 2025.
- FDA guidance on unapproved GLP-1 compounding (cagrilintide).
- ClinicalTrials.gov — REDEFINE protocols, including NCT06131437.
- NovoCare Pharmacy — current Wegovy self-pay pricing.
- LillyDirect — current Zepbound Self Pay Journey Program pricing.
About this page. The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We don’t sell medications. We compare what’s real, verify it, and publish it. Last verified: April 20, 2026 · Next scheduled review: May 1, 2026. Corrections: corrections@therxindex.com.