Affiliate disclosure: The RX Index earns a commission when you sign up with some of the providers mentioned on this page. It does not affect what you pay, and it never determines our rankings or which providers we cover. Read the full disclosure.
Drug Pipeline · Head-to-Head Comparison ·
By The RX Index Research Team · · Sources: NEJM (REDEFINE 1), Novo Nordisk and Eli Lilly investor releases, Lilly Medical, FDA.gov · Affiliate disclosure · Editorial standards
CagriSema vs Retatrutide: Which Next-Gen GLP-1 Actually Matters Right Now?
Published:
Retatrutide currently has the bigger number — 28.7% average weight loss at 68 weeks in Eli Lilly’s Phase 3 TRIUMPH-4 readout. CagriSema is closer to your pharmacy shelf because Novo Nordisk filed its FDA application on , with a regulatory decision anticipated by late 2026. But here’s the part most CagriSema vs retatrutide comparison pages won’t lead with: neither drug is FDA-approved, and neither is legally available outside of clinical trials right now.
That’s not a reason to stop reading. It’s the reason this page exists. We built this comparison because most pages ranking for this query are either peptide vendors with a financial stake in selling grey-market retatrutide, or generic explainers that cherry-pick the biggest headline number without telling you what the trial actually measured. We don’t sell either drug. We pulled the data from published Phase 3 trials, FDA filing records, and the manufacturers’ own statements — then assembled the comparison nobody else has, including the safety signals that should change your thinking.
Need a path you can actually act on today? Take our free 60-second GLP-1 matching quiz — FDA-approved options available now.
Take Our Free 60-Second GLP-1 Matching Quiz →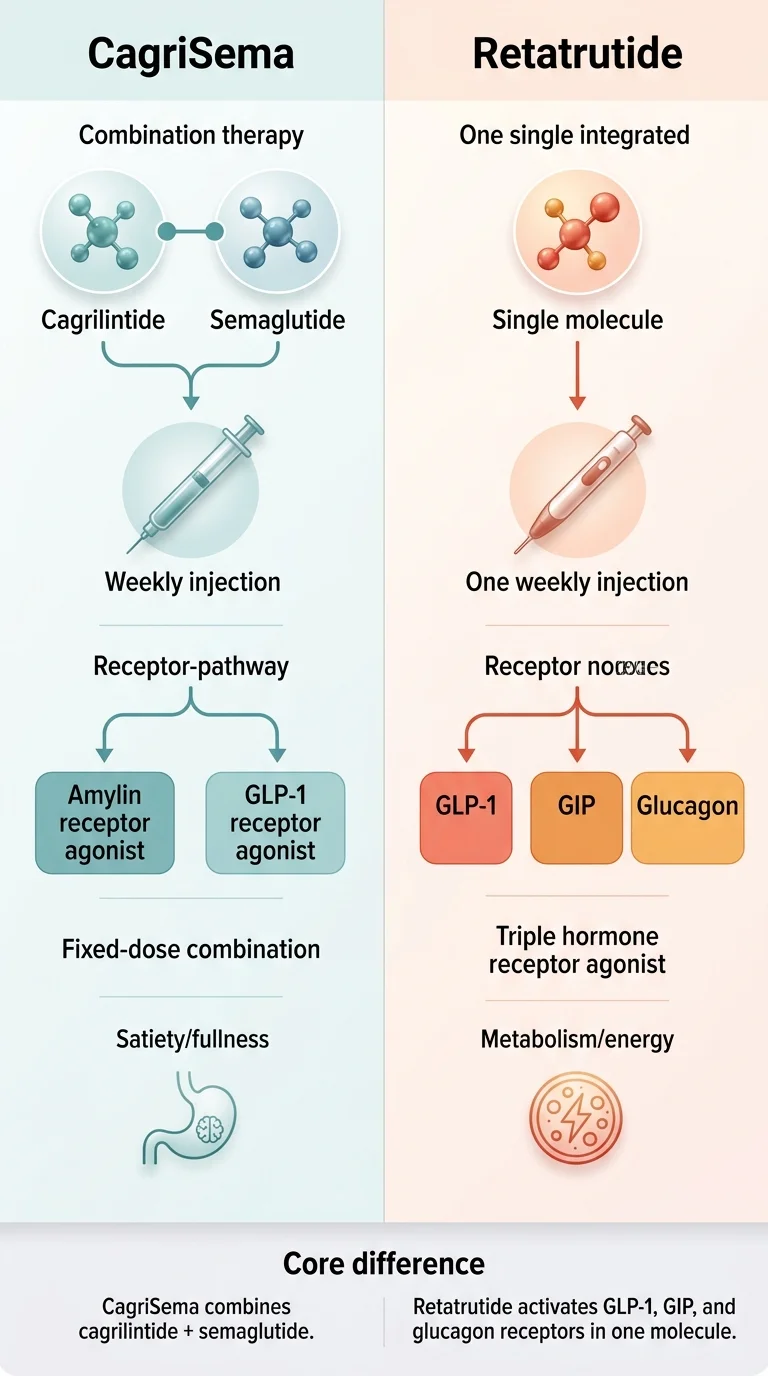
The RX Index 2026 Reality Check: CagriSema vs Retatrutide at a Glance
Pulled from primary sources. Every cell is sourced. Items we couldn’t verify are marked.
| CagriSema | Retatrutide | |
|---|---|---|
| Manufacturer | Novo Nordisk | Eli Lilly |
| What it is | Fixed-dose combination: cagrilintide 2.4 mg (amylin analog) + semaglutide 2.4 mg (GLP-1 agonist) | Single-molecule triple agonist: GLP-1 + GIP + glucagon receptors |
| Best Phase 3 obesity result | 22.7% (full adherence) / 20.4% (all participants) at 68 weeks — REDEFINE 1 | 28.7% mean weight loss at 68 weeks — TRIUMPH-4 (population: obesity/overweight + knee osteoarthritis) |
| Head-to-head vs an approved drug | Lost to tirzepatide 15 mg in REDEFINE 4: 23.0% vs 25.5% at 84 weeks — did NOT meet non-inferiority | None published vs an approved drug |
| Best Phase 3 T2D result | 14.2% weight loss + 1.91-point A1C reduction at 68 weeks — REIMAGINE 2 | 16.8% weight loss + up to 2.0-point A1C reduction at 40 weeks (12 mg) — TRANSCEND-T2D-1 |
| Discontinuation due to adverse events | 5.9% — REDEFINE 1 | 12.2% at 9 mg / 18.2% at 12 mg — TRIUMPH-4 |
| Notable safety signal | Higher overall GI event rate (79.6%); no unusual non-GI signal | Dysesthesia (abnormal skin sensations) in 8.8% at 9 mg / 20.9% at 12 mg — not seen with CagriSema |
| Peer-reviewed Phase 3 obesity paper? | Yes — REDEFINE 1 published in NEJM | Not yet — Lilly says detailed results 'to be presented and published' |
| FDA application filed? | Yes — December 18, 2025 | No — Lilly has not yet submitted |
| Expected FDA decision | Anticipated by late 2026 (per Novo Nordisk) | Not yet projected by Lilly |
| Available today? | No — 'not approved in the US or EU' (Novo Nordisk) | No — 'currently available only in Lilly clinical trials' (Lilly) |
| Predicted commercial pricing | Not announced | Not announced |
Sources cited inline throughout the page below.
Retatrutide wins
Raw weight-loss upside
CagriSema wins
Market readiness & tolerability
Neither wins
What you can do today
What Are the Key Differences Between CagriSema and Retatrutide?
CagriSema is two established drugs combined in one weekly injection — semaglutide (the active ingredient in Wegovy) plus cagrilintide, a long-acting amylin analog that adds a second appetite pathway. Retatrutide is a single engineered molecule that activates three hormone receptors at once: GLP-1, GIP, and glucagon. The pharmacology strategies are fundamentally different, and that difference explains nearly everything about their efficacy, safety, and timeline gaps.
CagriSema: the “evolution of semaglutide” approach
Semaglutide powers Wegovy and Ozempic — it slows gastric emptying, reduces appetite, and improves insulin signaling. Millions already take it. Cagrilintide adds amylin receptor activation on top, increasing feelings of fullness through a complementary pathway (amylin is naturally co-released with insulin after meals to promote satiety).
Advantage: half the drug is already FDA-approved with years of real-world safety data. Trade-off: doesn’t engage the energy-expenditure pathway the way retatrutide does.
Retatrutide: the “triple threat” approach
Activates three receptors in a single molecule:
- • GLP-1 — appetite suppression and insulin benefits
- • GIP — improves insulin sensitivity and glucose metabolism (same as tirzepatide)
- • Glucagon — the differentiator. Associated with increased energy expenditure and hepatic fat clearance
That third receptor is why retatrutide produces the highest weight-loss numbers in the pipeline — and why it causes dysesthesia and higher dropout rates not seen with CagriSema.
Why this distinction matters for you
More receptors doesn’t automatically mean “better for everyone.” CagriSema builds on a drug millions of people already tolerate. Retatrutide is a new molecule with a mechanism we don’t yet have long-term data on. That’s not a reason to dismiss it — but it is a reason to read the safety section carefully.
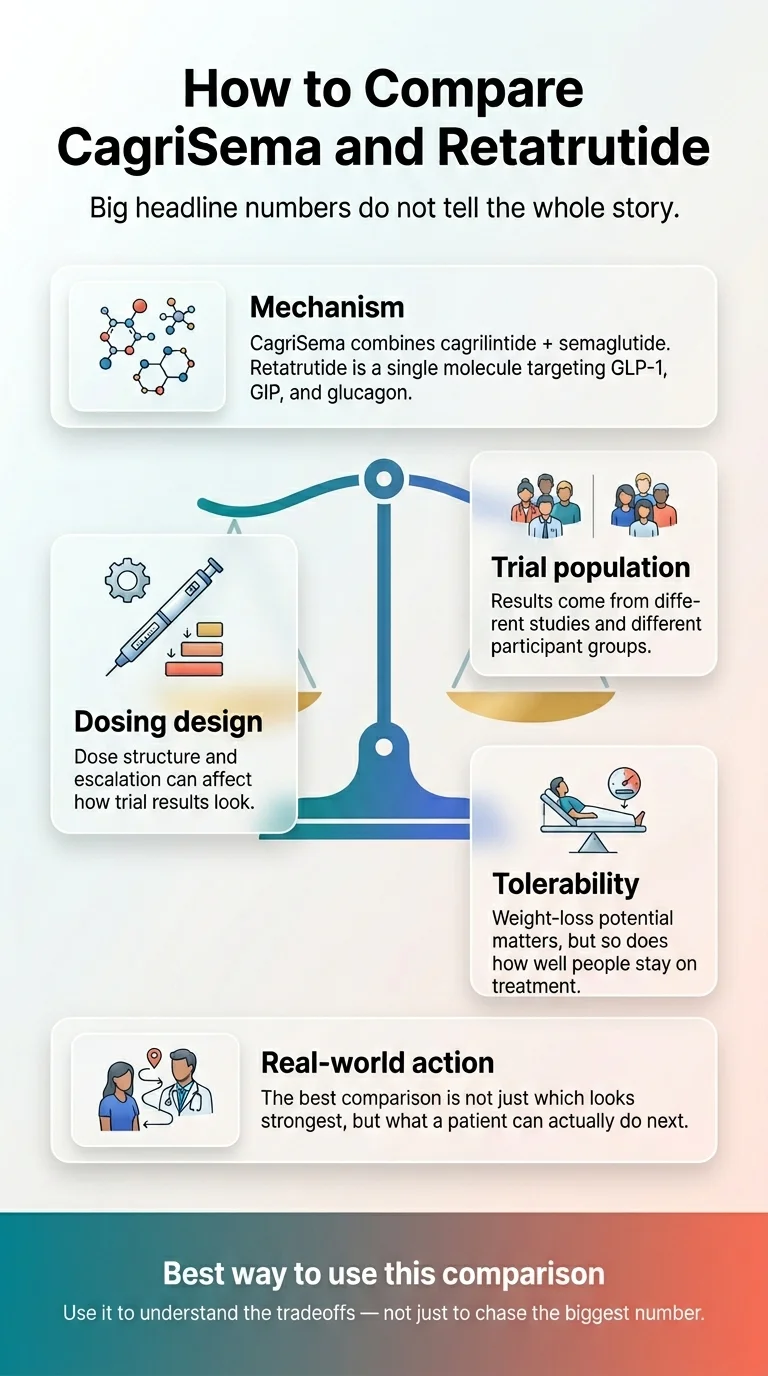
Why This Isn’t a Clean Apples-to-Apples Comparison
Most pages comparing CagriSema and retatrutide place the biggest numbers side by side and call it a day. That’s misleading. The headline weight-loss numbers come from different trials, in different patient populations, using different dosing designs, measured with different statistical approaches.
Three things that make the cross-trial comparison unreliable
Different patient populations
TRIUMPH-4 — the trial behind retatrutide's 28.7% number — enrolled adults with obesity or overweight who also had knee osteoarthritis (per Lilly Medical's published TRIUMPH-4 summary). That's not the same as a general obesity population. REDEFINE 1 enrolled a broader obesity population without requiring a comorbidity like osteoarthritis (per the NEJM publication).
Different dosing designs
TRIUMPH-4 used a fixed dose escalation toward target dose, with dose reductions permitted for tolerability or BMI/weight-loss reasons. REDEFINE 1 also used flexible dosing — investigators could reduce the dose if side effects were intolerable, and the trial reports both an 'if all people adhered' estimate and a treatment-policy estimate capturing everyone randomized regardless of dose changes.
Different statistical estimands
REDEFINE 1 reports two numbers for a reason: 22.7% (trial-product estimand — effect if all people adhered at target dose) and 20.4% (treatment-policy estimand — effect across everyone randomized regardless of whether they stayed on treatment). REDEFINE 4 reports a treatment-regimen estimand (20.2% for CagriSema vs 23.6% for tirzepatide). We do not yet know which estimand Lilly will use as its primary for TRIUMPH-4 — the published topline was 28.7%.
What a fair comparison can and cannot say
- ✓ Can say: Retatrutide’s reported weight-loss numbers are higher than CagriSema’s across the comparable trial readouts so far. The difference is real and consistent across multiple trial designs.
- ✗ Cannot say: That retatrutide is “better” full stop. Efficacy is one variable. Tolerability, availability, cost, regulatory status, and timing are others — and on several of those, CagriSema has the edge.
- ✗ Cannot say: A precise “real-world adjusted” gap. Until there’s a head-to-head trial comparing CagriSema and retatrutide directly (none has been announced), any specific adjustment claim is speculation, not data.
Didn’t CagriSema Just Lose to Tirzepatide?
Yes — and this changes the entire conversation around CagriSema.
Most competing pages published before don’t include this data.
On , Novo Nordisk announced results from REDEFINE 4, an 84-week open-label head-to-head trial comparing CagriSema directly against tirzepatide 15 mg (the drug sold as Zepbound). The result, per Novo Nordisk’s own press release:
Tirzepatide (Zepbound)
25.5%
full adherence
23.6%
treatment-regimen estimand
CagriSema
23.0%
full adherence
20.2%
treatment-regimen estimand
CagriSema did not meet the primary endpoint of non-inferiority vs tirzepatide. In plain language: Novo was trying to prove CagriSema was “at least as good” as Zepbound, and the trial didn’t show that.
Why this matters practically
Tirzepatide is already FDA-approved and available right now as Zepbound. If CagriSema can’t match it — and CagriSema won’t launch until at earliest mid-to-late 2027 — the question becomes: why wait for a drug that performs comparably to (or slightly worse than) one you can get today?
Novo is pursuing a higher-dose CagriSema (cagrilintide 2.4 mg / semaglutide 7.2 mg) with a Phase 3 trial planned for . Those results won’t arrive for years.
Our editorial take
REDEFINE 4 weakens CagriSema’s case relative to retatrutide and relative to currently available options. Retatrutide’s 28.7% still stands clearly above both CagriSema and tirzepatide. If you’re excited about the next-gen pipeline specifically for the extra efficacy, retatrutide is the stronger candidate — it’s just further away from your pharmacy.
Tirzepatide (Zepbound) outperformed CagriSema in the head-to-head trial — and you can start it today.
Explore Zepbound and Wegovy Options Available Now on Ro → (sponsored affiliate link, opens in a new tab)Which Seems Easier to Tolerate?
Both drugs produce the familiar GLP-1 gastrointestinal side effects — nausea, diarrhea, vomiting, constipation — mostly during dose escalation, mostly mild-to-moderate, mostly fading with time. That part isn’t dramatically different from what current Wegovy or Zepbound users experience. The differences that matter are in the details.
Side-by-Side Safety Signals (Phase 3 Trial Data)
| Side effect | CagriSema (2.4/2.4 mg) | Retatrutide 12 mg | Retatrutide 9 mg | Semaglutide 2.4 mg (reference) |
|---|---|---|---|---|
| Any GI event | 79.6% | Approximately 60–65% | Lower than 12 mg | ~44% (STEP 1) |
| Discontinuation due to adverse events | 5.9% | 18.2% | 12.2% | ~7% (STEP 1) |
| Dysesthesia | Not reported | 20.9% | 8.8% | Not reported |
| Injection site reactions | 10–15% | 8–12% | 8–12% | ~5% |
| Heart rate | Similar to semaglutide | Modest increase | Modest increase | Modest increase |
Sources: REDEFINE 1 (NEJM, Garvey et al. 2025), TRIUMPH-4 (Lilly Medical published summary; Lilly investor release December 2025), STEP 1 (NEJM 2021).
The number that jumps off the page
Retatrutide’s discontinuation rate at 12 mg — 18.2% — is roughly three times CagriSema’s 5.9%. Nearly 1 in 5 people on retatrutide 12 mg stopped treatment because of side effects. At the 9 mg dose, discontinuation drops to 12.2% — still higher than CagriSema, but more manageable.
Dysesthesia — unique to retatrutide
Dysesthesia means abnormal touch sensations: tingling, burning, sensitivity to pressure or temperature. In TRIUMPH-4, it affected 20.9% at 12 mg and 8.8% at 9 mg, per Lilly Medical. This signal does not appear with CagriSema, semaglutide, or tirzepatide. Events were generally mild and rarely led to discontinuation — but we don’t yet have long-term data.
Our honest assessment
If you’re sensitive to side effects or the idea of tingling/burning skin sensations concerns you, CagriSema has the cleaner profile. If you prioritize maximum weight loss and can tolerate initial side effects that typically improve with time, retatrutide’s profile is manageable for most people — but the 12 mg dose is rough for nearly 1 in 5 patients.
When Will CagriSema and Retatrutide Actually Be Available?
Neither drug is something you can walk into a pharmacy and fill today. Here’s what’s actually confirmed by primary sources, separated cleanly from what’s still projection.
CagriSema Timeline (Confirmed)
Retatrutide Timeline (Confirmed)
What this means practically
CagriSema has a clear head start. It has filed; retatrutide has not. CagriSema has a regulatory decision projected for this calendar year; retatrutide does not yet. But even an FDA decision in late 2026 means commercial launch in 2027 at the earliest after manufacturing scale-up, and broad insurance coverage takes longer still. Submission isn’t approval. Approval isn’t launch. Launch isn’t insurance coverage.
Can You Get CagriSema or Retatrutide Right Now?
No — and we need to be blunt about this.
CagriSema is “not approved in the US or EU,” per Novo Nordisk’s filing announcement. You cannot legally obtain CagriSema through any pharmacy, telehealth provider, or compounding pharmacy.
Retatrutide is not FDA-approved and Eli Lilly’s own published statement says it is “currently available only in Lilly clinical trials.”
FDA consumer alert on retatrutide
The FDA has issued a published consumer alert about illegally sold unapproved retatrutide products — including products labeled “for research purposes” sold online. Per FDA, these products have not been evaluated for safety, effectiveness, or quality. The TRIUMPH-4 weight-loss numbers were achieved under controlled medical supervision with verified pharmaceutical product — there is no reason to assume the same results or safety from an unregulated vial purchased from a peptide website.
If you genuinely want access to these drugs before FDA approval, the only responsible path is clinical trial enrollment. Search ClinicalTrials.gov for active REDEFINE (CagriSema) or TRIUMPH (retatrutide) trials. Trial participation provides free medication and medical monitoring; the trade-offs are strict eligibility criteria and the possibility of receiving placebo or comparator.
Three real options. Pick the one that matches where you are right now:
Need treatment this month?
See our guide to FDA-approved GLP-1 telehealth providers →Want to try CagriSema or retatrutide before approval?
Search ClinicalTrials.gov for “REDEFINE” or “retatrutide TRIUMPH” — the only legitimate pathNot sure which option fits?
Take our 60-second GLP-1 matching quiz →Should You Wait for One of These — or Start Treatment Now?
This is the question that actually matters, and the one most competing pages dodge. They compare the drugs, declare a winner, and leave you with no actionable next step. Here’s our framework based on where you are right now.
If you’re not on any GLP-1 yet
Start now. Don’t wait.
Waiting 12–24 months for an investigational drug isn’t a weight-loss strategy. Semaglutide (Wegovy) produces ~15% weight loss. Tirzepatide (Zepbound) produces ~20–22%. Every month spent waiting for a marginally stronger drug is a month carrying excess weight with all its associated health consequences. You can always switch when next-gen options launch. Starting now doesn’t lock you in — it gives you a head start.
Take our 60-second GLP-1 matching quiz →If you’re on semaglutide and you’ve plateaued
Consider switching to tirzepatide (Zepbound) before waiting for CagriSema. CagriSema just failed to beat tirzepatide head-to-head in REDEFINE 4. The drug you can get right now performed at least as well as the drug you’d be waiting 12+ months for. If you have insurance, a provider with prior-authorization support can often unlock Zepbound coverage at a copay much lower than cash price.
Ro carries Zepbound and includes an insurance concierge that submits prior-authorization paperwork for you. Get started for $39, then as low as $74/month with annual plan paid upfront. Medication costs depend on treatment and insurance.
See current pricing through Ro → (sponsored affiliate link, opens in a new tab)If you’re on tirzepatide and you’ve plateaued
This is the one scenario where tracking the pipeline genuinely makes sense. You’re already on the strongest FDA-approved option. CagriSema uses a different mechanism (amylin) that might work for people who’ve maximized GIP/GLP-1 benefit. Retatrutide adds glucagon, which could push results further. Your best move: explore clinical trial enrollment for either REDEFINE (CagriSema) or TRIUMPH (retatrutide) studies — the only legitimate way to try these drugs before approval, and it’s free.
If you have type 2 diabetes and A1C is the priority
Both next-gen drugs show strong diabetes data. CagriSema’s REIMAGINE 2 trial showed a 1.91-point A1C reduction at 68 weeks. Retatrutide’s TRANSCEND-T2D-1 showed up to 2.0-point A1C reductions at 40 weeks. Both compelling — but neither is accessible today. Currently available GLP-1 medications are already highly effective for glycemic control. If A1C management is urgent, starting treatment now is medically more sound than waiting.
See our guide: Best GLP-1 Telehealth Providers →If you’re tempted by “research peptide” retatrutide offers online
Don’t. The FDA has issued a specific consumer alert about illegally sold unapproved retatrutide products. These are unregulated chemicals with no verified purity, potency, or sterility. The TRIUMPH-4 numbers were achieved under controlled medical supervision with verified pharmaceutical product — there’s no reason to assume the same results or safety from an unregulated vial purchased online. If you want strong results from something you can actually start now, an FDA-approved GLP-1 through a licensed telehealth provider is a dramatically safer path.
What You Can Actually Do Right Now
You can’t get CagriSema or retatrutide today. But you can start treatment with currently available, FDA-approved GLP-1 medications that produce real, significant weight loss. The choice comes down to one question: do you have insurance that might cover a GLP-1, or are you paying entirely cash?
Path 1: FDA-Approved Medication With Insurance Support — Ro
For most people considering next-gen drugs, the smart move is to start now with an FDA-approved option that can deliver real results while these pipeline drugs work through approval. Ro is the strongest pick.
| Ro | |
|---|---|
| Medication type | FDA-approved (Wegovy, Zepbound, Foundayo, Ozempic) |
| Compounded GLP-1 offered? | No |
| Membership intro | $39 first month |
| Membership ongoing | As low as $74/month (annual plan paid upfront) or $149/month |
| Insurance support | Yes — dedicated insurance concierge handles prior authorization |
| Free coverage check | Yes — no membership required |
| What's included | Licensed clinician, dose titration, side-effect support, unlimited messaging, lab testing, coaching |
Pricing verified from ro.co/weight-loss/pricing, .
Why we lead with Ro for this page specifically: if you’ve been researching pipeline drugs, you’re someone who takes this decision seriously. Ro takes it seriously back. Their insurance concierge is the difference between paying $1,000+/month at retail and paying a $25–$75 copay if your plan covers Zepbound or Wegovy.
Check If Your Insurance Covers a GLP-1 Through Ro → (sponsored affiliate link, opens in a new tab)Free coverage check, no membership required. Get started for $39, then as low as $74/month with annual plan paid upfront. Medication costs depend on treatment and insurance.
Path 2: Cash-Pay Without Insurance
If you’re paying entirely cash and insurance isn’t part of your equation, the GLP-1 telehealth landscape has shifted significantly in 2026 with FDA’s restrictions on mass-marketed compounded GLP-1 products. We cover the current cash-pay landscape — including which providers still offer compounded GLP-1 under what conditions — on our dedicated guide.
Best GLP-1 telehealth providers →Path 3: Not Sure Which Direction Is Right
That’s exactly what our quiz is built for. Tell us your insurance status, budget, medication preference, and goals. We’ll match you with the path — and the provider — that fits your actual situation. Not a sales pitch. A decision shortcut.
Take Our Free 60-Second GLP-1 Matching Quiz →“I’m excited about retatrutide, an injectable medication that not only targets GLP-1 but also glucose-dependent insulinotropic polypeptide (GIP). Clinical trials show it can significantly reduce body weight. Another combination of two drugs, semaglutide and cagrilintide (known as CagriSema) also has had very good results.”
The pipeline is genuinely exciting. It’s also still the pipeline. The right move for most people is to start with what works today and re-evaluate when these drugs launch.
How We Built This Comparison — And What We Actually Verified
What we verified from primary sources
- •CagriSema obesity results — REDEFINE 1, published in NEJM (Garvey WT et al., 2025)
- •CagriSema vs tirzepatide — REDEFINE 4, Novo Nordisk press release Feb 23, 2026
- •CagriSema diabetes results — REIMAGINE 2, Novo Nordisk press release Feb 2, 2026
- •Retatrutide obesity results — TRIUMPH-4 topline, Eli Lilly investor release Dec 2025; AE rates per Lilly Medical's published TRIUMPH-4 summary
- •Retatrutide diabetes results — TRANSCEND-T2D-1, Eli Lilly investor release Mar 19, 2026
- •CagriSema NDA filing — Novo Nordisk press release Dec 18, 2025
- •Retatrutide availability statement — lilly.com, 'What to know about retatrutide'
- •FDA consumer alert on unapproved GLP-1 products — fda.gov
- •Ro pricing — ro.co/weight-loss/pricing, verified April 2026
What we could NOT verify or chose not to speculate on
- •Official commercial pricing for either drug — no manufacturer has announced retail pricing
- •Insurance formulary decisions — no insurer has announced coverage plans
- •Full TRIUMPH-4 detailed results — Lilly has released topline numbers only as of April 2026
- •A precise 'real-world adjusted' cross-trial gap — requires a direct head-to-head trial, which has not been announced
- •Long-term safety data — both drugs studied only in trials up to 84 weeks
Why we avoided speculative pricing:
Several competing pages publish predicted monthly costs ($1,200–$1,600/month) as though they’re facts. They’re educated guesses. We refuse to build any reader recommendation around pricing that doesn’t yet exist.
Who we are: The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. Our revenue comes from affiliate partnerships with currently available GLP-1 providers. We have no financial relationship with Novo Nordisk, Eli Lilly, or any entity selling CagriSema or retatrutide.
Frequently Asked Questions
Is retatrutide better than CagriSema?
Retatrutide produces more weight loss in clinical trials — 28.7% at 68 weeks in TRIUMPH-4 vs CagriSema's 22.7% at 68 weeks in REDEFINE 1. But CagriSema is closer to FDA approval, has a much lower discontinuation rate (5.9% vs 18.2% at 12 mg), and doesn't cause dysesthesia. Neither is available for routine public use today.
Is CagriSema FDA-approved yet?
No. As of April 15, 2026, CagriSema is not approved in the US or EU. Novo Nordisk filed its NDA with the FDA on December 18, 2025, with a regulatory decision anticipated by late 2026 per Novo's own filing announcement.
Is retatrutide FDA-approved yet?
No. Retatrutide is investigational and currently available only through Eli Lilly clinical trials, per Lilly's own published statement. Lilly has not yet submitted an NDA. Timing of any future FDA submission and approval depends on completion of the remaining TRIUMPH Phase 3 trials.
Can you buy retatrutide online?
Not legally — and the FDA has issued a consumer alert about illegally sold unapproved retatrutide products. Per FDA, products labeled 'for research purposes' sold online have not been evaluated for safety, effectiveness, or quality, and consumers are urged not to use them.
Did CagriSema beat tirzepatide (Zepbound)?
No. In REDEFINE 4 (results announced February 23, 2026), CagriSema achieved 23.0% weight loss at 84 weeks vs 25.5% for tirzepatide 15 mg — and did not meet the primary endpoint of non-inferiority. Tirzepatide (Zepbound) is already FDA-approved and available.
Which one has worse side effects?
It depends on the type. CagriSema has a higher overall GI event rate (79.6% in REDEFINE 1) but a lower discontinuation rate (5.9%). Retatrutide has a higher discontinuation rate (12.2% at 9 mg / 18.2% at 12 mg) and a unique side effect — dysesthesia (abnormal skin sensations) — affecting 8.8% at 9 mg and 20.9% at 12 mg. CagriSema does not cause dysesthesia.
How much will CagriSema or retatrutide cost?
There is no official commercial retail price for either drug yet. Manufacturer pricing has not been announced. Any specific monthly figures published elsewhere are analyst projections, not facts. We'll update this page when official pricing is released.
Should I wait for CagriSema or retatrutide instead of starting Wegovy or Zepbound?
For most people, no. Currently available GLP-1s produce 15–22% weight loss. Waiting 12–24 months means 12–24 months of not being on effective treatment. You can switch when next-gen options launch. The exception: if you've already plateaued on the strongest approved options and want to explore clinical trial enrollment.
What's the safest next step if I'm interested in these drugs?
Three options: (1) Start treatment now with an FDA-approved GLP-1 through a legitimate provider; you can switch later. (2) Search ClinicalTrials.gov for active REDEFINE (CagriSema) or TRIUMPH (retatrutide) trials — the only legitimate way to access these drugs before approval. (3) Bookmark this page; we update it with every major regulatory or trial milestone.
Still not sure which GLP-1 program is right for you?
We’ll match you with the right path — whether that’s FDA-approved with insurance support, or watching the pipeline until something new launches. No commitment. Just clarity.
Take Our Free 60-Second Matching Quiz →Browse Available GLP-1 Programs on Ro → (sponsored affiliate link, opens in a new tab)Sources
- Garvey WT, et al. Coadministered Cagrilintide and Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine (REDEFINE 1)
- Eli Lilly investor release: “Lilly’s triple agonist retatrutide delivered weight loss average of 28.7% in TRIUMPH-4” ()
- Lilly Medical TRIUMPH-4 summary — published clinical design and adverse event rates
- Novo Nordisk press release: NDA filing for CagriSema (); REDEFINE 4 results (); REIMAGINE 2 results ()
- Eli Lilly investor release: TRANSCEND-T2D-1 results ()
- lilly.com: “What to know about retatrutide” — Lilly availability statement
- FDA consumer guidance on unapproved GLP-1 and retatrutide products — fda.gov
- STEP 1 (NEJM, ) for semaglutide 2.4 mg reference rates
Related guides
- What Is CagriSema? — full 2026 FDA status, REDEFINE trial tracker, and mechanism explained
- Best GLP-1 Telehealth Providers Compared for 2026
- Wegovy Providers That Take HSA or FSA — verified payment paths
- Oral GLP-1 Providers That Take HSA or FSA (2026)
- GLP-1 Copay Assistance Programs 2026 — savings cards, PAPs, Medicare Bridge
- Does Medicare Cover Wegovy? — 2026 rules and the July GLP-1 Bridge
By The RX Index Research Team — a pricing intelligence and comparison resource for GLP-1 telehealth providers. Next scheduled review: This page was produced by reviewing primary source manufacturer press releases, NEJM publications, Lilly Medical published summaries, FDA guidance documents, and Ro’s pricing page. This page is for informational purposes only and does not constitute medical advice.