Last verified: April 22, 2026 · REDEFINE 4 data confirmed · FDA status checked · Pricing verified
Comparison Guide · April 22, 2026
CagriSema vs Mounjaro (2026): Should You Wait or Start Tirzepatide Now?
Published:
By The RX Index Research Team · Last verified: April 22, 2026
Editorial disclosure: The RX Index earns a commission if you click through to Ro or Sesame Care. We do not have any financial relationship with Novo Nordisk or Eli Lilly. We publish what the data says regardless of who pays us.
The short answer
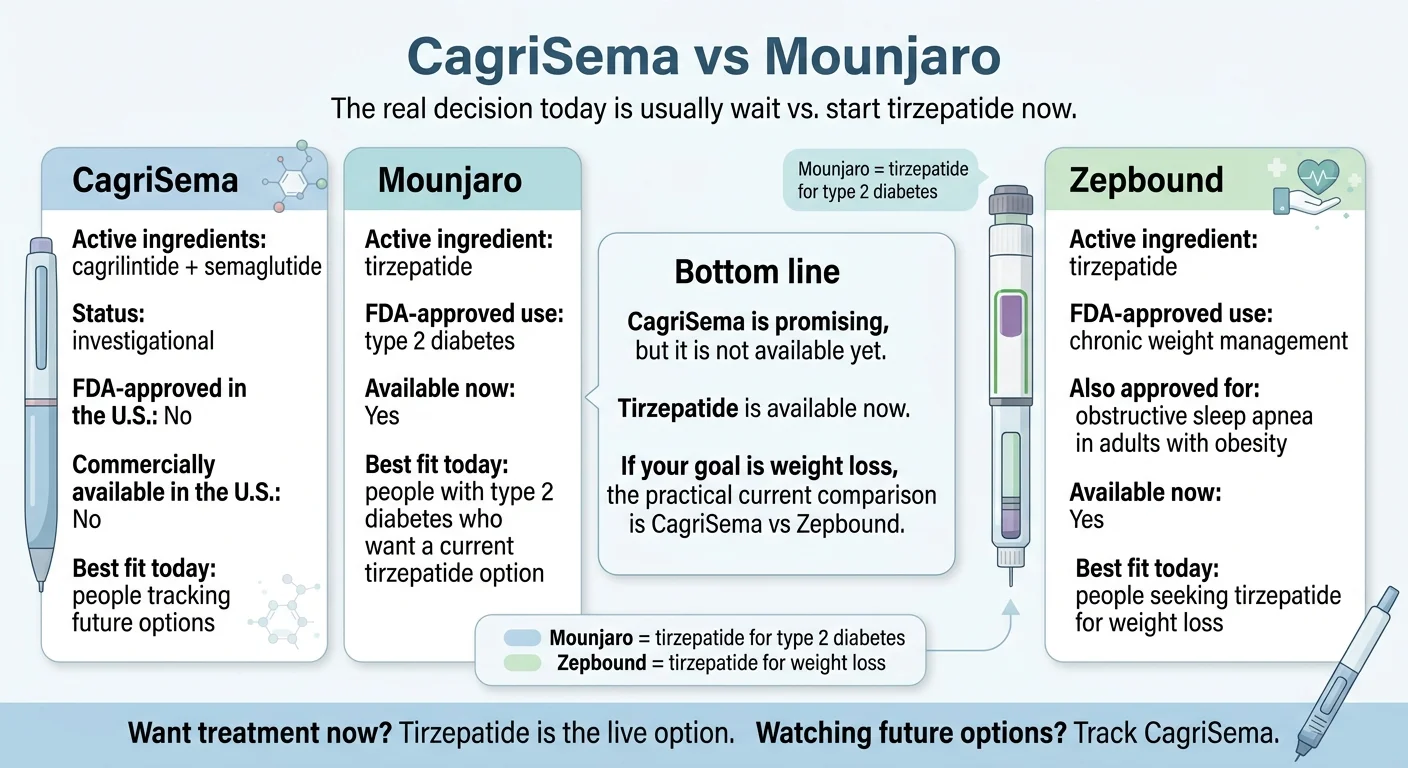
CagriSema is promising — but it is not FDA-approved and not commercially available in the United States as of April 2026. In the only direct head-to-head trial (REDEFINE 4, reported February 23, 2026), Mounjaro’s active ingredient tirzepatide beat CagriSema: 25.5% versus 23.0% mean weight loss at 84 weeks, and CagriSema failed to meet its non-inferiority endpoint.1 An FDA decision is expected in late 2026.2 For most people, the real decision is not “which drug” — it is wait vs. start.
 (sponsored affiliate link, opens in a new tab)
(sponsored affiliate link, opens in a new tab)The real decision today is usually wait vs. start tirzepatide now. CagriSema is not FDA-approved as of April 2026.
Considering tirzepatide for weight loss?
Ro carries Zepbound (same molecule as Mounjaro, approved for weight loss), handles prior authorization, and runs a free GLP-1 insurance coverage check that shows what your specific plan requires.
Run Ro’s free GLP-1 insurance coverage check → (sponsored affiliate link, opens in a new tab)What we actually verified for this page
- ✅ REDEFINE 4 head-to-head results — Novo Nordisk press release, February 23, 20261
- ✅ CagriSema FDA filing and status — Novo Nordisk press release, December 18, 20252
- ✅ Mounjaro FDA status and current pricing — Eli Lilly Mounjaro FAQ and pricing info3
- ✅ Zepbound FDA status and LillyDirect self-pay tiers — Lilly Zepbound savings page4
- ✅ Ro pricing and what Ro actually carries — Ro public pricing page5
- ✅ FDA stance on compounding cagrilintide — FDA concerns page on unapproved GLP-1 drugs6
CagriSema vs Mounjaro at a glance
The table most pages haven’t updated yet — verified April 22, 2026.
| CagriSema | Mounjaro | Zepbound | |
|---|---|---|---|
| Active ingredient(s) | Cagrilintide 2.4 mg + semaglutide 2.4 mg | Tirzepatide | Tirzepatide (same molecule as Mounjaro) |
| Mechanism | GLP-1 + amylin | Dual GIP + GLP-1 | Dual GIP + GLP-1 |
| Manufacturer | Novo Nordisk | Eli Lilly | Eli Lilly |
| FDA status (April 2026) | Investigational. NDA filed Dec 18, 2025. Decision expected late 20262 | Approved for type 2 diabetes (May 2022) | Approved for chronic weight management (Nov 2023) and obstructive sleep apnea with obesity (Dec 2024) |
| Available to fill today? | No | Yes (diabetes) | Yes (weight loss / OSA) |
| Best weight-loss trial number | 22.7% at 68 weeks (REDEFINE 1, trial product estimand)8 | n/a (weight-loss data reported under Zepbound) | 20.9% at 72 weeks (SURMOUNT-1, 15 mg); 22.9% at 176 weeks (three-year SURMOUNT-1)9 |
| Head-to-head vs tirzepatide (REDEFINE 4, 84 weeks) | 23.0% (trial product) / 20.2% (treatment regimen) | — | 25.5% (trial product) / 23.6% (treatment regimen) — tirzepatide won1 |
| List price (28-day supply) | Not priced (not on market) | ~$1,112.16 per 28-day supply3 | ~$1,086.37 per 28-day supply (LillyDirect self-pay paths are lower)4 |
| Self-pay path | None today | Limited cash-pay options | LillyDirect vials: $299 (2.5 mg) / $399 (5 mg) / $449 (7.5–15 mg) with 45-day refill4 |
| Who it’s best for today | Pipeline watchers willing to wait | Adults with T2D who want a live FDA-approved GIP/GLP-1 option | Adults with obesity (or overweight + a weight-related condition) who want tirzepatide for weight loss now |
Considering tirzepatide for weight loss?
The single highest-leverage move is running your real coverage before you decide anything else. Ro’s free tool checks your plan’s coverage and prior-authorization requirements so you know what you’re actually working with.
Run Ro’s free GLP-1 insurance coverage check → (sponsored affiliate link, opens in a new tab)Ro is a legitimate FDA-approved-brand telehealth provider that offers Zepbound and Foundayo with pricing that matches LillyDirect. The RX Index earns a commission if you use this link — that does not change the trial data above.
Do you actually mean Mounjaro — or Zepbound?
Answer capsule: Mounjaro and Zepbound contain the same active ingredient, tirzepatide, at the same doses. Mounjaro is FDA-approved for type 2 diabetes. Zepbound is FDA-approved for chronic weight management in adults with obesity or overweight with a weight-related condition, and separately for moderate-to-severe obstructive sleep apnea in adults with obesity. If you’re comparing “CagriSema vs Mounjaro” because you want weight loss and don’t have diabetes, your practical comparison is CagriSema vs Zepbound — and tirzepatide already won that one in REDEFINE 4.
This is the single most important thing to get out of the way, because half the internet’s comparison pages quietly conflate these — and it changes everything about your cost, your coverage, and your access path.
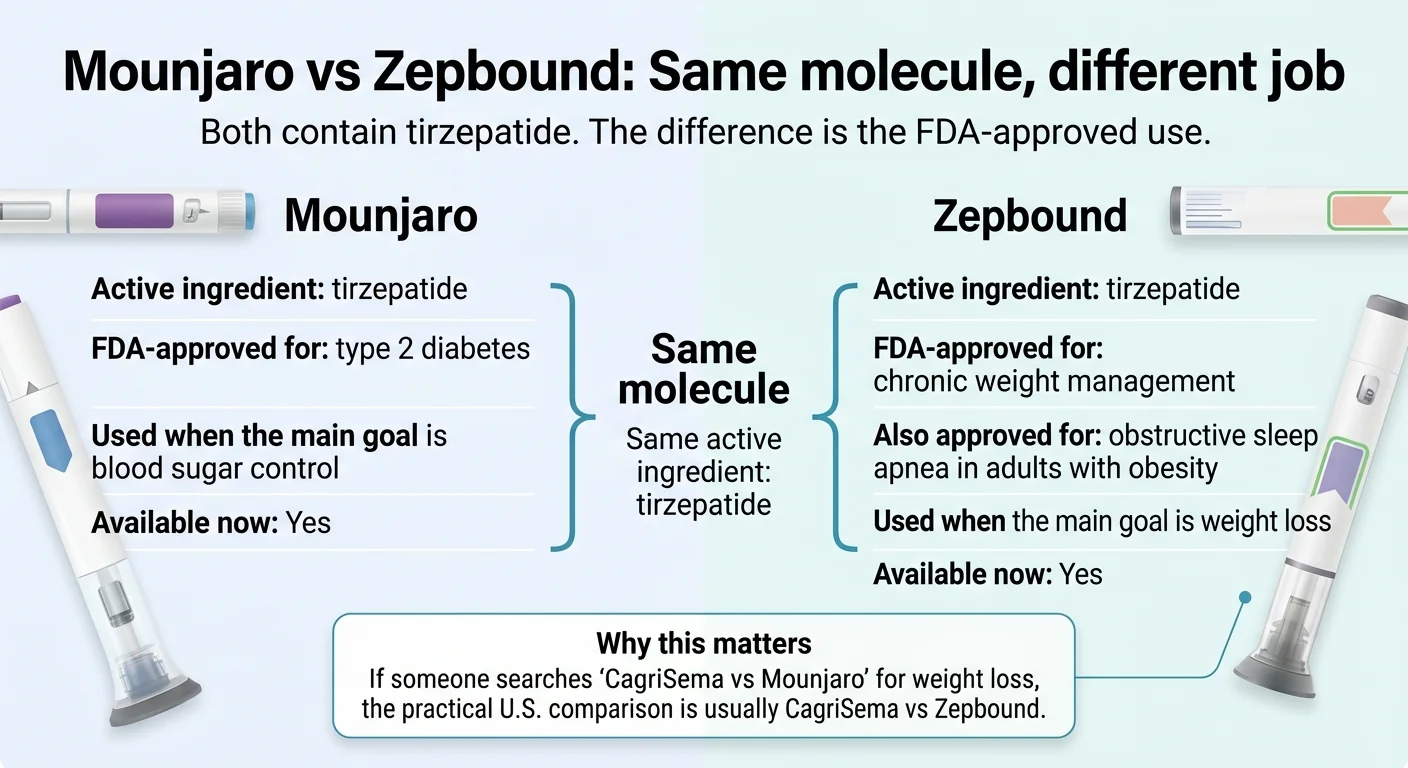
Mounjaro and Zepbound contain the same active ingredient. The difference is what the FDA approved each brand to treat.
Why the confusion exists
Mounjaro came first, in May 2022, as a diabetes drug. It blew up online — Reddit and TikTok were talking about the weight-loss side effect before Eli Lilly had an approved weight-loss label. By the time Zepbound launched in November 2023 with the weight-management indication, the public conversation had already locked in on the name “Mounjaro.” So when someone types “cagrisema vs mounjaro” today, they often mean “cagrisema vs tirzepatide-for-weight-loss” — which is Zepbound’s territory.
Why this changes your answer
- If your goal is diabetes control: Mounjaro is the relevant brand. Insurance coverage is more common. You’re not in a hurry on CagriSema because CagriSema isn’t approved for diabetes yet either.
- If your goal is weight loss: Zepbound is the relevant brand. LillyDirect self-pay vials start at $299/month for the 2.5 mg starter — far more transparent cash pricing than Mounjaro offers.
- If you’re still debating: the practical “best current head-to-head winner” is tirzepatide regardless of which brand your prescription is written under.
For the deeper wait-vs-Zepbound specifics, skip to the wait-or-start framework below. If your real question is about long-term type 2 diabetes management, our best GLP-1 for diabetes guide is the right next page.
Which one is better right now? The REDEFINE 4 result, explained
Answer capsule: In REDEFINE 4 — an 84-week open-label phase 3 trial of 809 adults with obesity reported February 23, 2026 — tirzepatide 15 mg produced a mean 25.5% weight loss compared with CagriSema’s 23.0% (trial product estimand). Under the treatment-regimen estimand, tirzepatide led 23.6% to 20.2%. CagriSema did not meet its primary endpoint of demonstrating non-inferiority to tirzepatide.1
What REDEFINE 4 actually tested
- Design: Open-label, randomized, phase 3, 84 weeks
- Participants: 809 adults with obesity (BMI ≥30) and at least one weight-related comorbidity; mean baseline body weight 114.2 kg
- Arms: CagriSema (cagrilintide 2.4 mg + semaglutide 2.4 mg, once weekly) vs. tirzepatide 15 mg, once weekly
- Primary endpoint: Non-inferiority of CagriSema vs. tirzepatide on weight change at 84 weeks — not met
- Reported: Novo Nordisk press release, February 23, 20261
The two numbers that matter
Trial product estimand (efficacy if everyone stays on treatment):
- CagriSema: 23.0%
- Tirzepatide: 25.5%
Treatment-regimen estimand (real-world, regardless of discontinuation):
- CagriSema: 20.2%
- Tirzepatide: 23.6%
Under both estimands, tirzepatide wins by roughly 2.5 to 3.4 percentage points. That is not a rounding error.
What a 2.5-point gap actually looks like at your weight
Pure math using the 23.0% and 25.5% trial-product estimands:
| Starting weight | CagriSema (23.0%) | Tirzepatide (25.5%) | Gap, pounds |
|---|---|---|---|
| 200 lb | ~46 lb lost | ~51 lb lost | ~5 lb |
| 250 lb | ~57.5 lb lost | ~63.75 lb lost | ~6.25 lb |
| 300 lb | ~69 lb lost | ~76.5 lb lost | ~7.5 lb |
| 350 lb | ~80.5 lb lost | ~89.25 lb lost | ~8.75 lb |
What Novo Nordisk said, and what it actually means
Novo’s Chief Scientific Officer pointed out that more patients reached the full 15 mg tirzepatide dose than reached the full CagriSema dose, and that the open-label design — meaning everyone knew which drug they were taking — may have influenced adherence toward the more famous drug.1 Novo is starting a higher-dose CagriSema trial (2.4 mg cagrilintide / 7.2 mg semaglutide) in the second half of 2026 to test whether the ceiling is higher than what REDEFINE 4 showed.7
The honest read: CagriSema may do better at higher doses we haven’t tested yet. But “may do better, eventually, at a dose that doesn’t exist in trials yet” is not the same as “beats the drug on the market right now.” Today’s best available head-to-head evidence is REDEFINE 4, and tirzepatide won.
Before we go further — the damaging admission
Ro does not currently offer Mounjaro. If your priority is specifically the Mounjaro brand — usually because you have type 2 diabetes and your doctor already wrote that prescription — Ro is not your fit, and you should look at Sesame Care or stick with your current pharmacy. We’re telling you that plainly because we’d rather lose this click than get it dishonestly.
But here’s why Ro still makes sense for most people reading this page: Ro carries Zepbound (same tirzepatide molecule, approved for weight loss) at LillyDirect-matched pricing, plus an insurance concierge that handles prior-authorization paperwork and a free coverage checker.
If you already know you want tirzepatide for weight loss
Check what your specific plan actually covers. Ro’s free tool returns your coverage status and any prior-authorization requirements — no sales call, no commitment.
See if your plan covers Zepbound (free coverage check) → (sponsored affiliate link, opens in a new tab)Can you actually get CagriSema in 2026?
Answer capsule: No. As of April 2026, CagriSema is not FDA-approved and is not commercially available in the United States. Novo Nordisk filed a New Drug Application on December 18, 2025, and an FDA decision is expected in late 2026.2 The only legitimate way to access CagriSema today is through participation in an authorized clinical trial.
The CagriSema timeline at a glance
- June 22, 2025: REDEFINE 1 and REDEFINE 2 results published in The New England Journal of Medicine
- December 18, 2025: Novo Nordisk submits NDA to FDA based on REDEFINE 1 and REDEFINE 22
- February 23, 2026: REDEFINE 4 head-to-head vs. tirzepatide reports — primary endpoint not met1
- Late 2026: Expected FDA decision2
- H2 2026: Higher-dose CagriSema (2.4 mg / 7.2 mg) phase 3 trial planned to start7
- H1 2027: REDEFINE 11 longer-duration data expected7
- 2027: REDEFINE 8 — includes an 80-week extension and a dose-tapering algorithm for weight-loss maintenance7
Three things to watch out for right now
- 1. Do not buy “CagriSema” from unregulated online sellers. Any site claiming to sell CagriSema today is operating outside FDA oversight. The FDA has stated directly that cagrilintide is among the ingredients that cannot be used in compounded drugs under federal law.6
- 2. “Compounded CagriSema” is not a real legal product. If you see a telehealth provider or research-peptide site marketing “compounded CagriSema,” “CagriSema peptide blend,” or similar, treat it as a red flag. This is not the same legal category as the compounded semaglutide or tirzepatide people were getting during the FDA shortage windows.6
- 3. Waiting is not free. If you’re overweight or have a weight-related condition and you put off treatment for 6 to 12 months while you wait for CagriSema, you are giving up 6 to 12 months of potential weight loss on a drug that’s already approved, already available, and that won the only head-to-head trial. That’s a real cost, even if it doesn’t show up on a receipt.
If this section eliminated waiting for CagriSema as a realistic option for you
The logical next step is figuring out whether tirzepatide is the right move — specifically for your goal, insurance, and timeline. Our 60-second quiz hands you a personalized plan based on your exact situation.
Get your free 60-second GLP-1 action plan →How CagriSema and Mounjaro actually work (plain English)
Answer capsule: CagriSema combines semaglutide (a GLP-1 receptor agonist — same molecule as Wegovy and Ozempic) with cagrilintide (an amylin analog) in one weekly injection. Mounjaro’s active ingredient, tirzepatide, hits two different receptors — GIP and GLP-1 — simultaneously. CagriSema is GLP-1 + amylin. Tirzepatide is GLP-1 + GIP. Different partner mechanisms, similar goal.
CagriSema, plain English
- Semaglutide mimics GLP-1, a gut hormone that slows stomach emptying, blunts hunger, and prompts insulin release
- Cagrilintide mimics amylin, a pancreatic hormone that drives fullness after eating through a different brain pathway than GLP-1
- Running both pathways at once is why CagriSema outperformed semaglutide alone in REDEFINE 1 (20.4% vs. 14.9% weight loss at 68 weeks, treatment-regimen estimand)8
Mounjaro, plain English
- Tirzepatide is an engineered peptide that binds two incretin receptors at the same time: GIP and GLP-1
- GIP activation appears to amplify GLP-1’s effects on appetite and glucose control
- This is the mechanism behind tirzepatide’s SURMOUNT-1 results: 20.9% mean weight loss at 72 weeks on the 15 mg dose, and 22.9% at 176 weeks in SURMOUNT-1’s extended follow-up9
So what’s the practical difference for you?
Both are “GLP-1 plus something else” — that’s the whole category. CagriSema’s something else is amylin biology; tirzepatide’s is GIP biology. On the current evidence, tirzepatide’s GIP partner appears slightly stronger. Whether cagrilintide’s amylin partner catches up at higher CagriSema doses is the open scientific question Novo is chasing in the H2 2026 high-dose trial.
Side effects and safety — two different kinds of evidence
Important distinction: Mounjaro and Zepbound have FDA-approved prescribing information, including a boxed warning. CagriSema does not. CagriSema is investigational, so what we have is clinical trial safety data — not an FDA-approved label. We keep these separate because they’re different legal and scientific categories.
CagriSema — trial safety signals (REDEFINE 1 and REDEFINE 2)
Safety data reported by Novo Nordisk from phase 3 trials:8
- GI adverse events (REDEFINE 1): 79.6% of CagriSema participants vs. 39.9% on placebo
- Nausea (REDEFINE 1): 55% vs. 12.6% placebo
- Constipation (REDEFINE 1): 30.7% vs. 11.6% placebo
- Vomiting (REDEFINE 1): 26.1% vs. 4.1% placebo
- Discontinuation due to adverse events (REDEFINE 1): 5.9% vs. 3.5% placebo
- Discontinuation due to adverse events (REDEFINE 2, with T2D): 8.4% vs. 3% placebo
Novo reports most GI events were mild to moderate and diminished over time, consistent with the broader GLP-1 class. CagriSema does not yet have an FDA-approved label, so there are no labeled contraindications or boxed warnings — only what the investigational protocol and clinical trial reports describe.
Mounjaro / Zepbound — FDA-labeled safety information
Per the FDA prescribing information for tirzepatide:10
Boxed warning: Thyroid C-cell tumors
Tirzepatide causes dose- and treatment-duration-dependent thyroid C-cell tumors in rats. Human relevance is unknown. Tirzepatide is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Common side effects:
Nausea, diarrhea, decreased appetite, vomiting, constipation, indigestion (dyspepsia), abdominal pain, and injection-site reactions.
Other labeled warnings:
Pancreatitis; acute kidney injury (usually from dehydration); gallbladder disease; hypersensitivity reactions including anaphylaxis and angioedema; hypoglycemia when combined with insulin or sulfonylureas; potential reduced effectiveness of oral hormonal contraceptives — the label recommends a backup contraceptive method for 4 weeks after starting and for 4 weeks after each dose escalation.
What this means practically
On published trial data, CagriSema’s GI tolerability profile looks broadly similar to tirzepatide’s. But tirzepatide has years of post-market surveillance and an FDA-approved label; CagriSema’s long-term and real-world safety will be known only after approval and wider use. Individual response varies a lot, and this is a prescriber conversation — not a decision to make from any comparison article, ours included.
What CagriSema and Mounjaro actually cost in 2026
Answer capsule: CagriSema has no commercial pricing because it is not sold. Mounjaro’s list price is approximately $1,112.16 per 28-day supply; with commercial insurance, eligible patients can pay as little as $25 for up to a 3-month prescription via the Mounjaro Savings Card.3 Zepbound — same molecule, weight-loss label — offers transparent self-pay pricing through LillyDirect: $299/month for the 2.5 mg starter, $399/month for 5 mg, and $449/month for 7.5 mg–15 mg (with a 45-day refill requirement).4
Mounjaro pricing — verified April 2026
| Scenario | Typical monthly cost |
|---|---|
| List price (WAC), 28-day supply | ~$1,112.163 |
| Commercial insurance covers Mounjaro + Savings Card | As little as $25 for up to a 3-month prescription3 |
| Medicare Part D (per Lilly’s FAQ) | About 8 out of 10 Medicare Part D patients pay $0–$50 for a 28-day supply3 |
| Medicaid (per Lilly’s FAQ) | Average co-pay of about $5–$11 per fill3 |
Medicare, Medicaid, and other government-program enrollees cannot use the Mounjaro Savings Card under federal law. If you have no prescription coverage at all, expect to pay close to the retail list price.3
Zepbound pricing — verified April 2026
| Scenario | Typical monthly cost |
|---|---|
| LillyDirect self-pay, 2.5 mg (starter) | $299/month4 |
| LillyDirect self-pay, 5 mg | $399/month4 |
| LillyDirect self-pay, 7.5 mg / 10 mg / 12.5 mg / 15 mg | $449/month4 (with 45-day refill requirement) |
| If you miss the 45-day refill window | Regular cash prices apply — $499 for 7.5 mg; $699 for 10 mg, 12.5 mg, or 15 mg4 |
| Commercial insurance covers Zepbound + Savings Card | As little as $25 per fill4 |
CagriSema pricing
Novo Nordisk has not announced CagriSema pricing. There is no reliable price to report. We will update this page the day a launch price is published.
Provider-stated vs. verified pricing — quarterly audit
Current as of April 22, 2026
| Path | Provider-stated price | What we verified | Insurance tool? |
|---|---|---|---|
| Lilly — Mounjaro direct | ~$1,112.16/month list | Verified against Lilly’s official Mounjaro pricing / FAQ page3 | Savings Card portal |
| Lilly — Zepbound LillyDirect | $299 / $399 / $449 by dose (45-day refill terms) | Verified against zepbound.lilly.com/savings4 | Savings Card portal |
| Ro — Zepbound & Foundayo | $39 first month, then $149/month ongoing, or as low as $74/month with annual plan | Verified against Ro’s public pricing5 | Yes — free GLP-1 Insurance Coverage Checker5 |
| Sesame Care | Varies by appointment + medication | Verified against Sesame Care’s public listings | Marketplace-style listings |
| Compounded “CagriSema” | Not legally available | FDA states cagrilintide cannot be used in compounding6 | — |
| Unauthorized online “CagriSema” sellers | Not legally available | FDA actively warns against unauthorized GLP-1 products6 | — |
If cost is the real blocker
The cheapest legitimate path for most people is either insurance + Savings Card ($25) or Zepbound LillyDirect self-pay ($299–$449). Ro’s free coverage check tells you in a few minutes which path your plan supports.
Check Zepbound coverage and prior-auth requirements on Ro (free) → (sponsored affiliate link, opens in a new tab)Disclosure: The RX Index earns a commission if you use Ro or Sesame Care links. That compensation does not change the prices above.
Should you wait for CagriSema or start tirzepatide now?
Answer capsule: For most people, the answer is start tirzepatide now. CagriSema lost the head-to-head comparison at the doses actually tested, it is not available, an FDA decision is 6+ months away, launch is later, and launch pricing is unknown. Tirzepatide is approved, available, and produced 20.9% mean weight loss at 72 weeks in SURMOUNT-1 and 22.9% at 176 weeks in the three-year follow-up.9
The four situations we see most in our traffic
| If you are… | What the current evidence supports | Watch-out |
|---|---|---|
| Already on tirzepatide and it’s working | Stay the course. REDEFINE 4 gives you no clinical reason to switch. | Don’t change a medication that’s working based on marketing for an unapproved drug. |
| Considering starting tirzepatide now | Start. The head-to-head winner is already on the market. Waiting 6–12 months for CagriSema means giving up 6–12 months of results for a drug that currently trails tirzepatide. | If cost is the blocker, Zepbound LillyDirect self-pay ($299–$449/month) is the cheapest legitimate FDA-approved path. |
| On semaglutide (Ozempic, Wegovy) and plateaued | Talk to your prescriber about switching to tirzepatide. SURMOUNT-5 showed tirzepatide produced significantly greater weight loss than injectable semaglutide (20.2% vs. 13.7% at 72 weeks).11 | Do not stop semaglutide and start a new medication without clinician input. |
| Specifically want CagriSema for pipeline reasons | Watch and wait. Decision expected late 2026. Enroll in a REDEFINE or REIMAGINE trial if you qualify. | Do not buy “CagriSema” from unregulated sellers. It is not legally available.6 |
The one-line summary
A future drug can’t help you today. If CagriSema launches, performs well at higher doses, and later evidence shows it’s the better option for your situation, you can switch then. Starting tirzepatide today gets you months of results that CagriSema simply cannot deliver, because it doesn’t exist commercially yet.
You are not missing out by starting now. You are getting a head start on the drug that just won the head-to-head.
Still weighing wait vs. start?
Our 60-second matching quiz walks through your goal, insurance, timeline, and medical-history flags, then hands you a specific next step — not another article.
Take the free 60-second GLP-1 matching quiz →What real people are actually worried about
We read hundreds of Reddit, GLP-1 forum, and community threads while building this page. The questions people actually ask are narrower than the headlines — and they deserve a direct answer.
“The difference is only about 3kg. Is that really enough to care?” (r/MounjaroAus, February 2026)
The head-to-head gap is 2.5 percentage points on the trial-product estimand (25.5% vs 23.0%) and 3.4 points on the treatment-regimen estimand (23.6% vs 20.2%). At 250 lb starting weight, that’s roughly 6 lbs over 84 weeks. Whether that matters to you is personal. What’s not personal is that tirzepatide won the only controlled comparison — and it’s available right now.
“Debating whether to try CagriSema instead since some have said it has better appetite suppression.” (GLP-1 Forum discussion thread)
Anecdotal reports of how a drug “feels” don’t tell us it’s better — they tell us how it felt for one person. REDEFINE 4 is the controlled comparison, and tirzepatide won. If you specifically value appetite suppression, there is no current controlled evidence that CagriSema does it better than tirzepatide. That may change with dose-optimization trials. It hasn’t yet.
These quotes reflect individual opinions, not typical outcomes, and are not medical evidence. We included them because if you’re comparing CagriSema to Mounjaro in 2026, these are the exact worries running through most people’s heads.
What could change this verdict later?
Answer capsule: The current verdict is not the forever verdict. Novo Nordisk has two ongoing programs that could move the answer: a higher-dose CagriSema phase 3 trial (cagrilintide 2.4 mg / semaglutide 7.2 mg) expected to start in H2 2026, and REDEFINE 11 — with data expected in H1 2027. Launch pricing and insurance coverage will also be deciding factors.7
Three specific things that could flip the answer
- 1. Higher-dose CagriSema works dramatically better. If the 2.4/7.2 formulation pushes CagriSema’s mean weight loss meaningfully above tirzepatide at equal adherence, that changes the head-to-head math. Watch for data readouts in 2027 and beyond.
- 2. Long-duration or maintenance data shows a different profile. REDEFINE 4 ran 84 weeks. REDEFINE 8 and REDEFINE 11 run longer and include maintenance assessments. If CagriSema maintains weight loss better than tirzepatide over multiple years, the calculus could shift for long-term users.
- 3. Launch pricing and coverage favor CagriSema. If CagriSema launches with a lower list price or a more aggressive direct-to-patient self-pay program than LillyDirect’s Zepbound vials, the cost-per-percent-lost math could flip for cash-pay patients — even at slightly lower efficacy.
What we’ll update here when it changes
- The FDA decision (expected late 2026)
- Launch price and self-pay availability
- Any new head-to-head data
- Any label changes to Mounjaro, Zepbound, or Wegovy
Last verified: April 22, 2026. Next scheduled refresh: whichever comes first — 30 days or any material CagriSema regulatory news.
Type 2 diabetes readers — is this comparison different for you?
Answer capsule: Yes. If your primary goal is blood sugar control, Mounjaro is the current FDA-approved tirzepatide brand for type 2 diabetes, and CagriSema is not yet approved for diabetes in the U.S. either. Novo’s REIMAGINE program is pursuing a diabetes indication, but the obesity indication is the lead FDA filing.8 For most T2D patients, Mounjaro is the right current choice and CagriSema’s timeline should not delay that.
What the Mounjaro label says
Mounjaro is FDA-approved as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes. It is not approved for weight loss, even though weight loss is a common effect. Dosing starts at 2.5 mg once weekly and titrates every 4 weeks in 2.5 mg increments as tolerated, to a maximum of 15 mg weekly.10
Who should not use Mounjaro
- Personal or family history of medullary thyroid carcinoma (MTC)
- Multiple endocrine neoplasia syndrome type 2 (MEN 2)
- Known serious hypersensitivity to tirzepatide or any excipient
- Consult your prescriber if you have a history of pancreatitis, severe gastrointestinal disease, diabetic retinopathy, or are pregnant or planning pregnancy
For T2D readers, the key question is insurance coverage and whether your prescriber is already familiar with Mounjaro’s diabetes protocol. If you need a direct comparison of GLP-1 options for blood sugar control, our best GLP-1 for diabetes guide covers that in detail.
Need Mounjaro specifically (type 2 diabetes)?
As noted above, Ro does not carry Mounjaro. Sesame Care offers access to multiple GLP-1 providers and publishes transparent marketplace pricing — a good option if you need Mounjaro for diabetes management.
See Mounjaro options on Sesame Care → (sponsored affiliate link, opens in a new tab)Frequently asked questions
Is CagriSema better than Mounjaro?
No — not by the current evidence. In the only head-to-head trial (REDEFINE 4, reported February 23, 2026), Mounjaro’s active ingredient tirzepatide outperformed CagriSema at the doses tested: 25.5% vs. 23.0% mean weight loss at 84 weeks under the trial-product estimand. CagriSema did not meet its primary endpoint of non-inferiority. Tirzepatide is the current head-to-head winner.
When will CagriSema be FDA approved?
Novo Nordisk filed the NDA on December 18, 2025. An FDA decision is expected in late 2026. No PDUFA date has been publicly confirmed.
Can I buy CagriSema right now?
No. CagriSema is not FDA-approved and is not commercially available in the United States as of April 2026. The only legitimate access is through authorized clinical trials. The FDA has stated that cagrilintide cannot be used in compounded drugs under federal law.
Is Mounjaro the same as Zepbound?
Yes, chemically. Both contain the same active ingredient — tirzepatide — at the same doses. The difference is the FDA-approved indication: Mounjaro for type 2 diabetes, Zepbound for chronic weight management and obstructive sleep apnea with obesity. They work identically in the body.
Is Mounjaro approved for weight loss?
No. Mounjaro is FDA-approved for type 2 diabetes. The weight-loss version of the same drug is Zepbound, approved for chronic weight management in adults with obesity or overweight with at least one weight-related condition.
How much weight do people lose on CagriSema vs Mounjaro?
In REDEFINE 4 (84 weeks), CagriSema produced a mean 23.0% weight loss and tirzepatide produced 25.5% under the trial-product estimand. In SURMOUNT-1’s three-year extension, tirzepatide 15 mg maintained an average 22.9% weight loss at 176 weeks. REDEFINE 1 (not head-to-head) showed CagriSema at 22.7% at 68 weeks. Individual results vary substantially.
Does insurance cover Mounjaro or Zepbound?
Commercial insurance commonly covers Mounjaro for type 2 diabetes with prior authorization. Zepbound coverage for weight loss is less consistent and varies widely by plan. Ro’s free GLP-1 Insurance Coverage Checker reports your plan’s coverage status and any prior-authorization requirements.
Should I wait for CagriSema?
Usually no. CagriSema is 6+ months from an FDA decision in the best case, launch is later, launch pricing is unknown, and it trailed tirzepatide in the only head-to-head trial at tested doses. For most people ready to start GLP-1 therapy, starting tirzepatide now is the evidence-based choice. If you’re specifically pipeline-watching or already stable on another GLP-1, waiting and monitoring makes sense.
Can you take CagriSema and Mounjaro together?
No. Tirzepatide’s FDA-approved prescribing information states it should not be coadministered with other tirzepatide-containing products or any GLP-1 receptor agonist. CagriSema contains semaglutide (a GLP-1 receptor agonist), so combining the two is outside the label and outside any studied, approved use.
Related guides
Still not sure which path is right for you?
You read a long comparison. You have the head-to-head data. You know CagriSema isn’t on the market. You understand the Mounjaro-vs-Zepbound confusion. If you’re still unsure which move makes sense for your specific situation — your insurance, your goal, your timeline, your medical history — take the quiz. It’s 60 seconds, it’s free, and it hands you a specific personalized next step instead of another article.
Take the free 60-second GLP-1 matching quiz →Ready to check your Zepbound coverage?
Ro’s free GLP-1 Insurance Coverage Checker is the fastest way to find out what your plan actually covers and what prior authorization requires — before you commit to anything.
Check Zepbound coverage on Ro (free) → (sponsored affiliate link, opens in a new tab)Sources
- 1 Novo Nordisk A/S. “CagriSema demonstrated 23% weight loss in an open-label head-to-head REDEFINE 4 trial in people with obesity, the primary endpoint was not achieved.” Press release. February 23, 2026. globenewswire.com
- 2 Novo Nordisk. “Novo Nordisk files for FDA approval of CagriSema.” PR Newswire, December 18, 2025.
- 3 Eli Lilly. Mounjaro FAQ and official pricing. mounjaro.lilly.com/faq; pricinginfo.lilly.com/mounjaro
- 4 Eli Lilly. Zepbound Savings and LillyDirect pricing. zepbound.lilly.com/savings; pricinginfo.lilly.com/zepbound
- 5 Ro. Public pricing and GLP-1 Insurance Coverage Checker. ro.co/weight-loss/glp1-insurance-checker
- 6 U.S. Food and Drug Administration. “FDA’s Concerns with Unapproved GLP-1 Drugs Used for Weight Loss.” fda.gov
- 7 Novo Nordisk REDEFINE program details — REDEFINE 8, REDEFINE 9, REDEFINE 11, and planned high-dose (2.4 mg / 7.2 mg) phase 3 trial. Summarized in Novo Nordisk 2026 press materials.
- 8 Garvey WT, et al. Cagrilintide-Semaglutide in Adults with Overweight or Obesity (REDEFINE 1). N Engl J Med 2025; 393:635–647. And Davies MJ, et al. Cagrilintide-Semaglutide in Adults with Overweight or Obesity and Type 2 Diabetes (REDEFINE 2). N Engl J Med 2025; 393:648–659.
- 9 Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1). N Engl J Med 2022; 387:205–216. Three-year follow-up announced by Eli Lilly November 2024; published in NEJM 2025.
- 10 Mounjaro (tirzepatide) U.S. Prescribing Information. Eli Lilly. pi.lilly.com/us/mounjaro-uspi.pdf; pi.lilly.com/us/zepbound-uspi.pdf
- 11 Aronne LJ, et al. SURMOUNT-5: tirzepatide vs. injectable semaglutide for obesity. Announced by Eli Lilly December 2024.
Written by The RX Index Research Team. The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. This article is informational and does not constitute medical advice. Speak with a licensed healthcare provider before starting, changing, or stopping any prescription medication. Last verified: April 22, 2026.