Affiliate disclosure: The RX Index earns a commission when you sign up with some of the providers mentioned on this page. It does not affect what you pay, and it never determines our rankings or which providers we cover. Read the full disclosure.
Foundayo Side Effects: What to Expect, Real Rates by Dose, and When to Worry
Published:
By The RX Index Editorial Team · Last verified:
Disclosure: Some links on this page are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you.
Sources: FDA prescribing information (pi.lilly.com), Medication Guide, DailyMed, published Phase 3 trial data, Ro pricing pages (ro.co)
Foundayo side effects are mostly digestive — and the pill is not automatically easier on the stomach than injectable GLP-1 options. That's the single most important thing to know before you fill this prescription.
In FDA clinical trials, nausea hit 35% of patients at the highest approved dose (17.2 mg). Constipation reached 24%. Diarrhea, 25%. Vomiting, 24%. Overall, between 60% and 69% of patients experienced at least one GI side effect at maintenance doses, compared to 37% on placebo. Most were mild to moderate, and only 6–10% of people stopped treatment because of side effects. Source: Foundayo prescribing information, pooled ATTAIN-1 and ATTAIN-2 data
But there are things buried in the prescribing information that most pages don't surface — and they can change the decision entirely for certain people. This page covers the common and serious Foundayo side effects from the FDA label, gives you the numbers at each studied dose, tells you which symptoms are expected vs. which ones need action, and flags the hidden caution points that matter before you start.
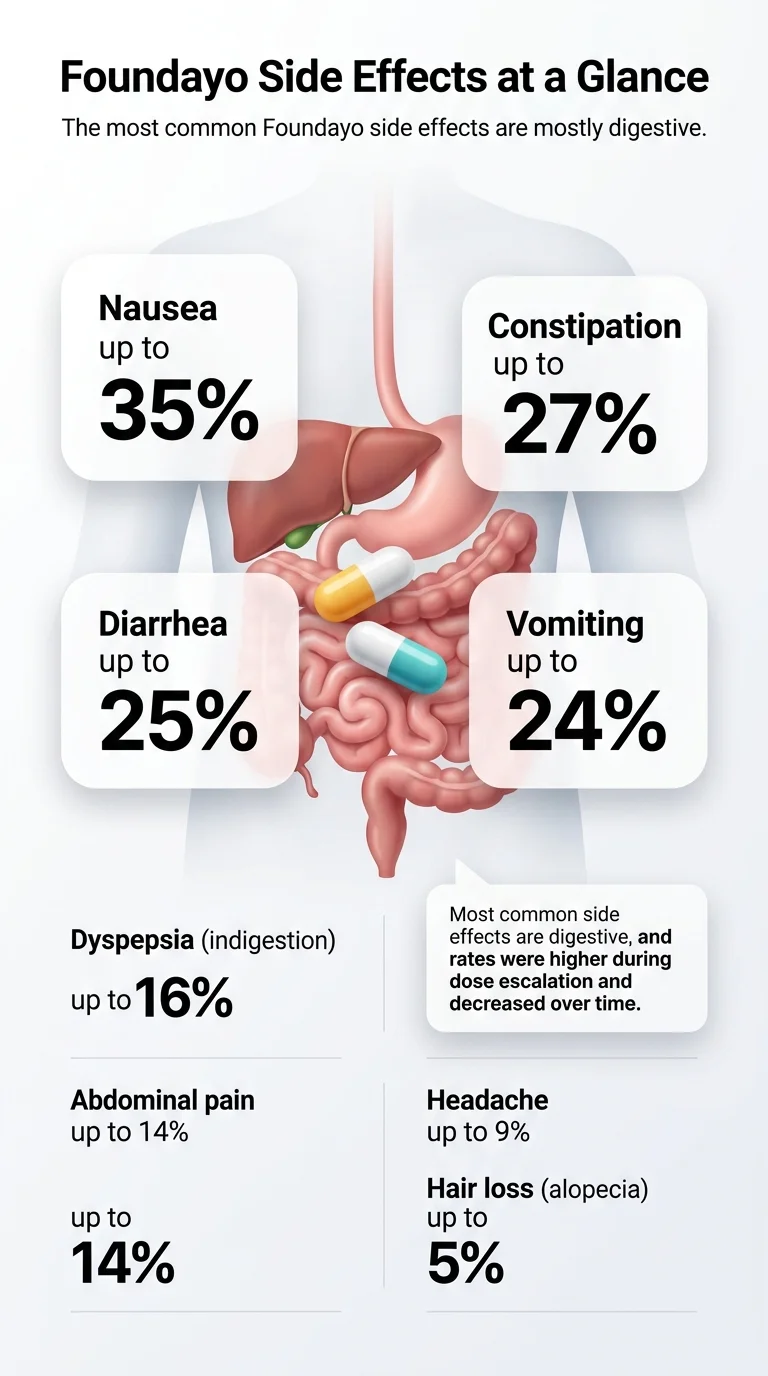
Foundayo at a glance
| Side effect | Rate at 17.2 mg | Worse during escalation? | Usually urgent? |
|---|---|---|---|
| Nausea | 35% | Yes | No |
| Constipation | 24% | Often | No |
| Diarrhea | 25% | Yes | Only if persistent / dehydrating |
| Vomiting | 24% | Yes | Can be — see triage below |
| Indigestion | 13% | Yes | No |
| Hair loss | 5% | Not the main early issue | No |
| Severe GI reactions | ~3% | Can occur at any dose | Yes |
| Discontinued due to all side effects | 10% | — | — |
Source: Foundayo prescribing information, pooled ATTAIN-1 and ATTAIN-2 trials. FDA-approved label, April 2026
If those numbers look manageable and you want to know whether Foundayo is a fit for your situation, our free 60-second quiz can help.
Get your personalized GLP-1 action plan →What are the most common Foundayo side effects?
The most common Foundayo (orforglipron) side effects are nausea, constipation, diarrhea, vomiting, indigestion (dyspepsia), abdominal pain, headache, abdominal bloating, fatigue, belching, heartburn (GERD), gas, and hair loss. Every one of these appeared in at least 5% of patients in the FDA's pooled clinical trial data, and all are more frequent at higher doses.
Here's what the numbers actually look like — pulled directly from the Foundayo prescribing information, Table 1, pooled ATTAIN-1 and ATTAIN-2 data. The same table your doctor sees.
Foundayo side-effect rates by studied dose group
| Side effect | Placebo | 5.5 mg | 9 mg | 17.2 mg |
|---|---|---|---|---|
| Any GI adverse reaction | 37% | 60% | 68% | 69% |
| Nausea | 10% | 26% | 34% | 35% |
| Constipation | 8% | 20% | 27% | 24% |
| Diarrhea | 11% | 21% | 23% | 25% |
| Vomiting | 3% | 13% | 21% | 24% |
| Indigestion (dyspepsia) | 4% | 12% | 16% | 13% |
| Abdominal pain | 7% | 13% | 14% | 14% |
| Headache | 7% | 7% | 9% | 9% |
| Abdominal bloating | 3% | 7% | 9% | 8% |
| Fatigue | 4% | 6% | 7% | 9% |
| Belching | 1% | 6% | 8% | 8% |
| Heartburn (GERD) | 2% | 6% | 6% | 7% |
| Gas (flatulence) | 2% | 5% | 6% | 6% |
| Hair loss (alopecia) | 2% | 4% | 4% | 5% |
| Discontinued due to any side effect | 3% | 6% | 9% | 10% |
| Discontinued due to GI side effects | 0.7% | 3% | 6% | 6% |
Source: Foundayo prescribing information, Table 1. Pooled ATTAIN-1 (N=3,127) and ATTAIN-2 (N=1,600+). The label reports rates for the 5.5 mg, 9 mg, and 17.2 mg dose groups — it does not separately tabulate rates for the 0.8 mg, 2.5 mg, or 14.5 mg titration doses. FDA-approved label
A few things jump out of this table that most pages don't mention:
The placebo rates give you context. 37% of people taking a placebo also reported GI symptoms. The incremental GI burden attributable specifically to Foundayo is the gap between the drug and placebo columns.
Lower doses have meaningfully lower rates. Nausea at 5.5 mg (26%) versus 17.2 mg (35%) is a 9-point gap. Not everyone needs the highest dose.
The discontinuation rate is the honesty check. Only 6–10% of people stopped treatment due to side effects across the 72-week trials. If 90% of patients at the highest dose stayed on, the side effects were tolerable for the large majority.
Additional side effects below the 5% threshold: tachycardia in 3% vs. 0.9% placebo (mean +4–5 bpm); hypotension in 2% vs. 0.5%; dizziness in 4% vs. 3%; dysgeusia (altered taste) in 0.9% vs. 0.3%.
Why a daily pill still causes stomach issues
This surprises people. A pill feels like it should be gentler than a weekly injection. But Foundayo works because it activates GLP-1 receptors — the same receptors that slow your stomach emptying, reduce appetite, and change how your gut processes food. The mechanism that produces weight loss is the same mechanism that produces nausea and digestive changes.
Of the patients who experienced GI side effects, the FDA label reports that 60% were mild, 36% were moderate, and 4% were severe. The vast majority of people are dealing with discomfort, not danger.

Which Foundayo side effects are normal, and which ones mean call a doctor?
Most people searching “Foundayo side effects” aren't looking for a list — they're looking for a decision framework. Is what I'm feeling okay, or should I be worried?
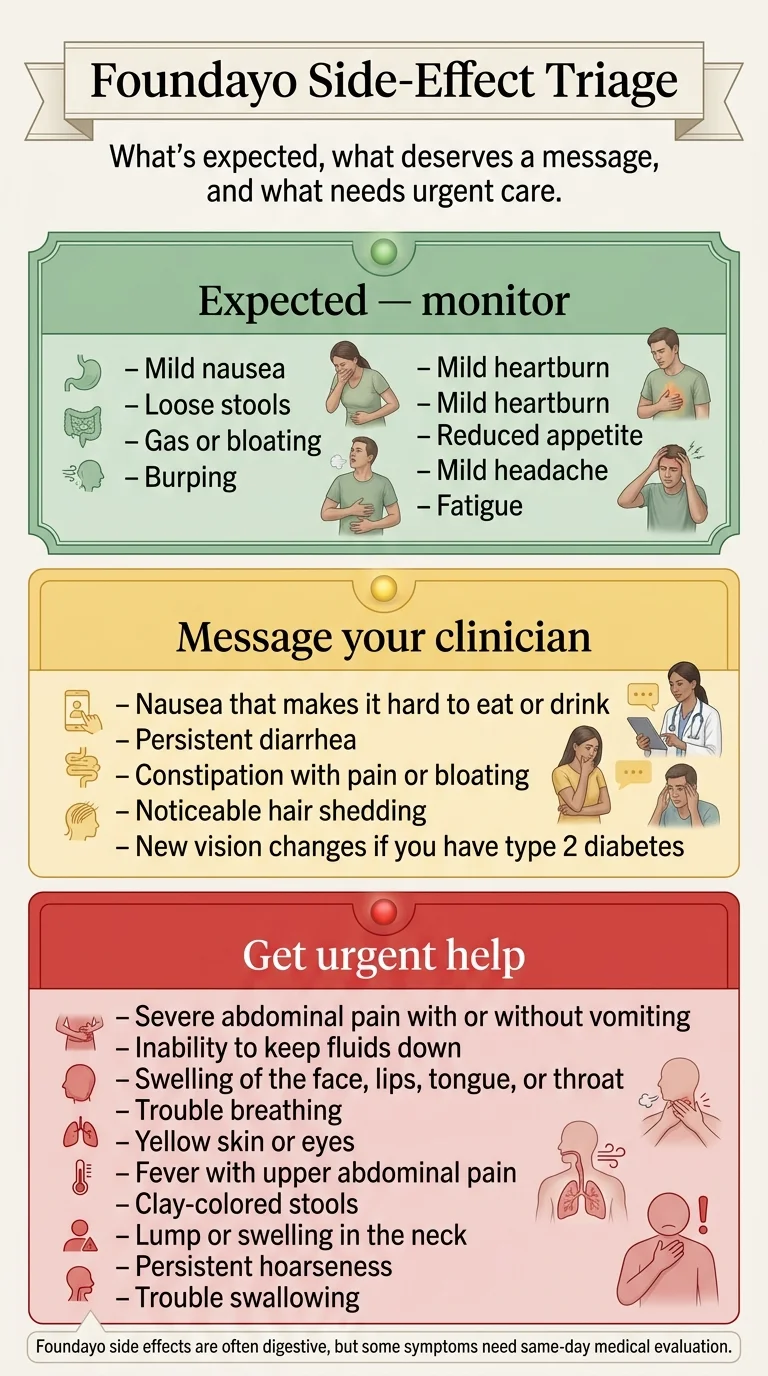
Foundayo Side-Effect Triage
- Mild nausea
- Loose stools
- Gas, bloating, burping
- Mild heartburn
- Reduced appetite
- Mild headache
- Fatigue
These are the most common side effects during dose escalation. Eat smaller meals, stay hydrated, and let your body adjust. The prescribing information confirms these symptoms decrease over time.
- Nausea that prevents eating or drinking for an extended period
- Persistent diarrhea or constipation that isn't resolving
- Noticeable hair shedding
- Heart racing or pounding (tachycardia reported in 3% of patients)
- Any new vision changes (if you have type 2 diabetes)
These warrant clinical evaluation. Your prescriber may adjust your dose, pause escalation, or add supportive treatment. Do not stop Foundayo on your own without talking to them first.
- Severe abdominal pain radiating to your back, with or without vomiting (possible pancreatitis)
- Inability to keep any fluids down (dehydration → kidney risk)
- Signs of allergic reaction: swelling of face, lips, tongue, or throat; difficulty breathing
- Yellowing of skin or eyes, fever, clay-colored stools (possible gallbladder disease)
- Lump or swelling in the neck, persistent hoarseness, trouble swallowing (thyroid concern)
These require same-day emergency evaluation. Do not wait.
The RX Index editorial triage framework, built from the Foundayo prescribing information and Medication Guide. Action thresholds are editorial guidance — not FDA-specified cutoffs. This is not a substitute for clinical judgment. Prescribing information · Medication Guide
Not sure whether Foundayo is still the right fit? Our 60-second quiz matches you to the GLP-1 approach that fits your tolerance, insurance, and preferences — whether that's Foundayo, an injectable, or something else entirely.
Get your personalized GLP-1 action plan →Are Foundayo side effects worse when you first start?
Yes — and during every dose increase. The FDA label states directly that nausea, vomiting, and diarrhea “were higher during the orforglipron dosage escalation period and decreased over time.” Source: prescribing information, Section 6.1
Foundayo's dosing schedule is designed around this reality. You start at 0.8 mg and increase no sooner than every 30 days:
Foundayo dose escalation schedule
What this means practically
Each dose increase can bring a brief return or intensification of GI symptoms, which then tend to ease as your body adapts to the new level of GLP-1 receptor activation. The prescribing information supports the pattern — symptoms track with dose changes and decrease over time — but does not specify an exact number of days or weeks for resolution. Individual timelines vary.
The missed-dose trap most people don't know about
If you miss Foundayo for 7 or more consecutive days, the Medication Guide instructs you to contact your prescriber before restarting. You may need to reinitiate at a lower dose to avoid a spike in GI side effects — essentially restarting part of the escalation. This is a practical reason to stay consistent, even on days when you don't feel like taking it. Source: Medication Guide
How long do Foundayo side effects last?
There is no single answer, and we're not going to manufacture one. The FDA label says GI side effects were worse during dose escalation and decreased over time — that's the supported claim. The clinical trials ran for 72 weeks, and by the end, discontinuation rates due to GI issues had leveled off.
| What the label says | What it does NOT say |
|---|---|
| GI symptoms were higher during dose escalation | A specific number of weeks for resolution |
| GI symptoms decreased over time | That every patient's symptoms resolve on the same timeline |
| 60% mild, 36% moderate, 4% severe | That severe symptoms always resolve without intervention |
We include this transparency box because many pages give clean “4–8 week” timelines without sourcing them. We'd rather give you the real picture.
If your side effects are not improving at a stable dose — meaning you're not in the middle of a dose increase and symptoms are still significant — that's a conversation with your prescriber. They may recommend a dose hold, a slower escalation, or a different approach. Don't quit on your own.
Does Foundayo cause hair loss?
Yes, hair loss is on the FDA label. In the pooled clinical trials, alopecia was reported in 4–5% of Foundayo patients versus 2% on placebo. The label adds an important detail: hair loss was reported more frequently in women (7%) than men (0.9%) on Foundayo, compared to 3% of women and 0.7% of men on placebo. The label also states that hair loss adverse reactions “were associated with weight reduction.” Source: prescribing information, Section 6.1
That weight-reduction association is consistent with what's seen across other weight-loss interventions — GLP-1 medications, bariatric surgery, and aggressive calorie restriction can all be accompanied by hair shedding. Dermatology literature describes this pattern as telogen effluvium, a temporary form of diffuse shedding triggered by physiological stress.
It's not full hair loss. The pattern is diffuse thinning — more hair in your brush or shower drain, not bald patches.
It's associated with weight loss broadly. The label ties it to weight reduction, not to orforglipron specifically attacking hair follicles.
If you notice significant shedding, tell your prescriber. They can evaluate whether it's weight-loss-associated hair changes or whether other causes (thyroid function, nutrient deficiencies) need investigation.
The 5% rate on the label is real, but 95% of patients at the highest dose did not report hair loss.
Is Foundayo easier on the stomach because it's a pill?
No — and this is the biggest misconception we see. The assumption that “daily pill = smoother ride than weekly injection” is intuitive but not supported by the data.
Foundayo does NOT look gentler on the stomach just because it's a pill. In clinical trials, GI adverse reactions occurred in 60–69% of Foundayo patients at maintenance doses versus 37% on placebo. If lowest GI friction is your top priority, this page alone won't answer that question — you need a head-to-head comparison with your other options.
There is now a head-to-head Phase 3 trial (ACHIEVE-3) comparing orforglipron to oral semaglutide in adults with type 2 diabetes. Lilly reported that orforglipron delivered superior blood sugar control and weight loss compared to oral semaglutide. Source: Eli Lilly press release, published in The Lancet However, no head-to-head obesity trial between Foundayo and the Wegovy pill has been published for direct tolerability comparison.
What Foundayo does offer over the Wegovy pill: no fasting requirement, no empty-stomach timing, take it any time of day with or without food. That's a real practical advantage for adherence — even if it doesn't translate to fewer stomach issues.
The hidden Foundayo cautions most side-effects pages bury
This section exists because the prescribing information contains several high-impact warnings that most pages either skip or bury at the bottom. These aren't rare edge cases — they affect common patient populations.
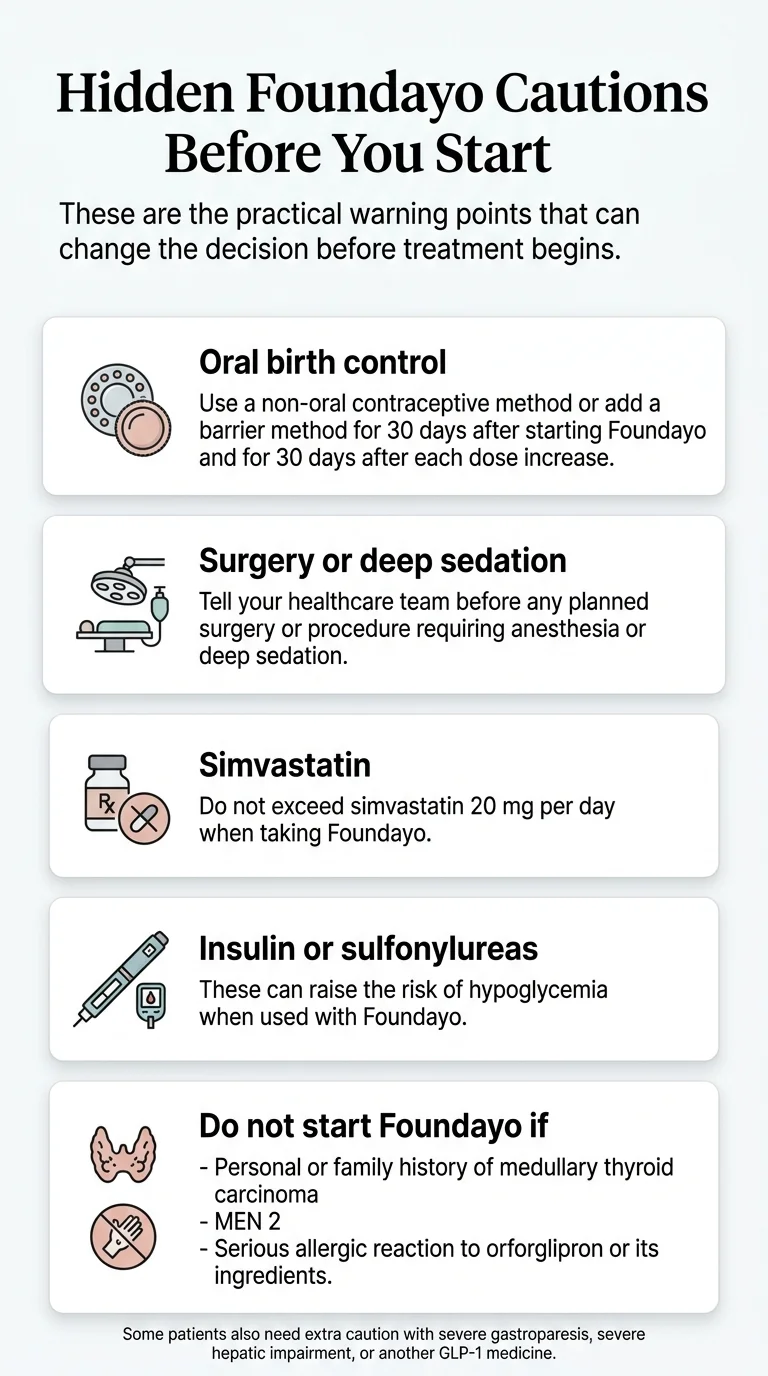
Oral birth control: the label instruction you need to know
The Foundayo prescribing information states that Foundayo delays gastric emptying and may affect the rate of absorption of other orally administered drugs. The specific clinical instruction: women using oral contraceptives should switch to a non-oral contraceptive method or add a barrier method for 30 days after starting Foundayo and for 30 days after each dose increase. Source: prescribing information, Sections 7.3 and 8.3
Foundayo's dose escalation spans 5+ months. During that time, you'd need backup contraception for at least 30 days at each of the six dose levels.
Surgery and deep sedation: tell your healthcare providers
Foundayo slows how quickly your stomach empties. Even after standard pre-procedure fasting, food or liquid may still be in your stomach, increasing the risk of pulmonary aspiration (food entering the lungs) during anesthesia. The prescribing information instructs patients to tell healthcare providers about planned surgeries or procedures. Source: prescribing information, Section 5.9
Simvastatin: dose cap required
Foundayo increases exposure to simvastatin acid approximately 2-fold to 2.5-fold. The prescribing information says: do not exceed simvastatin 20 mg/day when taken with Foundayo. If you're on a higher simvastatin dose, your prescriber may need to switch you to a different statin. Source: prescribing information, Section 7.2
Insulin and sulfonylureas: hypoglycemia risk increases
Foundayo alone has low hypoglycemia risk in patients without diabetes. But if you take insulin or a sulfonylurea, the combination increases your risk of low blood sugar — including severe hypoglycemia. Your prescriber may need to lower the dose of your diabetes medication when starting Foundayo. Know the signs: shakiness, sweating, rapid heartbeat, confusion, weakness.
CYP3A4 drug interactions
Foundayo is metabolized by the CYP3A4 enzyme. Strong CYP3A4 inhibitors (clarithromycin, ketoconazole, certain HIV medications) cap your maximum Foundayo dose at 9 mg. Strong CYP3A4 inducers such as carbamazepine should be avoided entirely. Moderate CYP3A4 inducers (such as efavirenz) require monitoring and possible dose escalation. Bring your complete medication and supplement list to your prescriber. Source: prescribing information, Sections 2.2 and 7.1
Severe hepatic impairment
Foundayo is not recommended for patients with severe hepatic (liver) impairment. If you have significant liver disease, this needs to be part of the conversation with your prescriber before starting. Source: prescribing information, Section 8.6
Reading this section and realizing one of these applies to you? That doesn't mean GLP-1 therapy is off the table. It means Foundayo specifically needs a closer look — and the right provider can help you figure out which option actually fits.
Take the 60-second quiz to find your best GLP-1 path →Who should NOT start Foundayo?
Foundayo is contraindicated — meaning it should not be used at all — in three specific groups. These are hard stops from the FDA label, not “talk to your doctor” suggestions.
Do not take Foundayo if you:
- Have a personal or family history of medullary thyroid carcinoma (MTC)
- Have Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Have had a serious allergic reaction to orforglipron or any ingredient in Foundayo
Concomitant use with another GLP-1 receptor agonist is not recommended. If you're already on Ozempic, Wegovy, Zepbound, Mounjaro, or any other GLP-1, do not add Foundayo on top.
Foundayo requires extra clinical caution for:
| Situation | Label guidance |
|---|---|
| Pregnancy | Foundayo may cause fetal harm. Stop if pregnancy recognized. Use effective contraception. Call 1-800-LillyRx for pregnancy registry. |
| Breastfeeding | Not recommended — unknown if orforglipron passes into breast milk. |
| Severe gastroparesis | Not recommended — Foundayo further slows gastric emptying. |
| Severe hepatic impairment | Not recommended per the label. |
| History of pancreatitis | Risk-benefit calculation requires careful clinical judgment. |
| Diabetic retinopathy | Monitor for vision changes during treatment. |
What serious risks are on the Foundayo label?
Every medication has a serious-risk section. We're including this because the prescribing information requires it and because informed patients make better decisions — not to scare you away from a treatment that helps the large majority of people who take it.
The thyroid boxed warning
Foundayo carries an FDA boxed warning — the most serious type of drug safety label — about thyroid C-cell tumors. This same boxed warning appears on Wegovy and Zepbound.
An important technical distinction: unlike peptide-based GLP-1 drugs (semaglutide, tirzepatide), orforglipron is not pharmacologically active at rodent GLP-1 receptors and did not produce thyroid tumors in rodent studies. The boxed warning is retained as a precaution. If you notice a lump in your neck, persistent hoarseness, or trouble swallowing — contact your prescriber immediately. Source: prescribing information, boxed warning and Section 5.1
Pancreatitis
Acute pancreatitis has been reported with Foundayo. In the pooled label data, 6 adjudication-confirmed acute pancreatitis events occurred in 6 Foundayo-treated patients versus 2 events in 1 placebo-treated patient. The warning sign is severe abdominal pain that radiates to your back, with or without vomiting. If this happens: stop Foundayo and get emergency care. Source: prescribing information, Section 5.2
Dehydration and kidney injury
Persistent vomiting or diarrhea can cause dehydration, which can lead to acute kidney injury — in some cases requiring hemodialysis. The label instructs prescribers to monitor renal function in patients reporting adverse reactions that could lead to volume depletion, especially during dose initiation and escalation. Hydration isn't just a comfort tip.
Gallbladder disease
Treatment with GLP-1 receptor agonists is associated with increased occurrence of acute gallbladder disease. In pooled trials, cholelithiasis (gallstones) was reported in 1% of Foundayo patients vs. 0.7% placebo. Watch for upper abdominal pain, nausea with fever, or yellowing of skin/eyes.
Severe allergic reactions
Rare, but serious. Anaphylaxis and angioedema have been reported with GLP-1 receptor agonists. Stop Foundayo immediately if you develop facial or throat swelling or difficulty breathing.
What should you eat to reduce Foundayo side effects?
The prescribing information doesn't include a specific diet plan, but the GI management principle is straightforward: work with the medication's mechanism, not against it. Foundayo slows gastric emptying — a large, heavy meal in a slowed-down stomach is the recipe for nausea and bloating.
Eat smaller, more frequent meals
Five or six smaller portions throughout the day instead of three large meals. This is the single most effective change.
Favor low-fat, easy-to-digest foods
Oatmeal, rice, bananas, lean proteins (chicken, fish, eggs), cooked vegetables, and crackers tend to be well-tolerated — especially early on. High-fat foods independently slow gastric emptying, compounding the effect.
Stay hydrated consistently — not in big gulps
Sip water throughout the day. The Medication Guide specifically warns that nausea, vomiting, and diarrhea can lead to dehydration and kidney problems. If you can't keep fluids down for an extended period, contact your prescriber.
What to avoid
Large fatty meals, greasy or fried foods, carbonated beverages (worsen bloating), and alcohol — especially during the early weeks and after each dose increase.
Managing side effects while on Foundayo?
Ro Body members get clinical support from prescribers who specialize in GLP-1 medication management — including dose adjustments and side-effect guidance. Foundayo available starting at $149/month.
See if Foundayo is available through Ro → (sponsored affiliate link, opens in a new tab)How we verified this page
We built this page because the current top results for “Foundayo side effects” either repeat the label without interpreting it, give neat timelines they can't source, or bury the practical caution points that change real decisions.
What we actually checked:
What this page does not claim
This page does not provide medical advice. We do not claim to replace the prescribing information or your prescriber's judgment. Side-effect rates come from clinical trials with controlled populations — your personal experience may differ. Triage thresholds are editorial guidance, not FDA-specified instructions.
How often we recheck: Pricing monthly. Label data quarterly — or immediately if the FDA issues a safety communication. The “Last verified” date at the top reflects our most recent check.
Frequently asked questions about Foundayo side effects
What are the most common Foundayo side effects?
The most common Foundayo side effects are nausea (up to 35%), constipation (up to 24%), diarrhea (up to 25%), vomiting (up to 24%), indigestion, abdominal pain, headache, bloating, fatigue, belching, heartburn, gas, and hair loss. All rates are from the FDA prescribing information at the 17.2 mg dose group.
Are Foundayo side effects worse when you first start treatment?
Yes. The FDA label confirms that nausea, vomiting, and diarrhea were more common during dose escalation and decreased over time. Each dose increase (every 30+ days) can bring a return of symptoms.
How long do Foundayo side effects last?
The prescribing information says GI symptoms are worse during dose escalation and decrease over time, but does not specify a fixed timeline. Individual experience varies based on dose, tolerability, and other factors.
Does Foundayo cause hair loss?
Hair loss was reported in 4–5% of patients (7% of women specifically). The Foundayo label states that hair loss adverse reactions were associated with weight reduction.
Can Foundayo make birth control pills less effective?
The prescribing information advises switching to a non-oral contraceptive method or adding a barrier method for 30 days after starting Foundayo and for 30 days after each dose increase, because Foundayo delays gastric emptying and may affect absorption of oral medications.
What if I miss Foundayo for 7 or more days?
Contact your healthcare provider about how to restart treatment. You may need to reinitiate at a lower dose to reduce the risk of GI side effects.
Is Foundayo easier to tolerate than the Wegovy pill?
Not clearly. Foundayo's GI adverse reaction rates (60–69%) are in the same range as other oral GLP-1 options. There is a head-to-head trial (ACHIEVE-3) in type 2 diabetes but no head-to-head obesity trial for direct tolerability comparison. The practical advantage is convenience — no fasting requirement, take any time of day.
Should I stop Foundayo before surgery?
Tell all your healthcare providers that you take Foundayo before any planned surgery or procedure requiring anesthesia or deep sedation. Foundayo slows stomach emptying, which increases aspiration risk.
What if I take insulin or a sulfonylurea with Foundayo?
Your prescriber may need to lower the dose of your diabetes medication when you start Foundayo, because the combination increases hypoglycemia risk. Do not adjust diabetes medications on your own.
Can you drink alcohol on Foundayo?
No specific interaction is listed in the prescribing information. However, alcohol can irritate the stomach, worsen nausea, and its effects may feel stronger due to slowed gastric emptying. Use caution, especially during the early weeks.
Still not sure which GLP-1 program is right for you?
Take our free 60-second matching quiz. You'll get a personalized action plan based on your side-effect concerns, insurance status, medication preferences, and whether Foundayo, an injectable, or a different option is actually the best fit for where you are right now.
Take the quiz — get your personalized GLP-1 action plan →