🚨 If you can't keep water down, have severe belly pain, are vomiting blood, or feel close to fainting —
Stop reading. Go to urgent care or the ER now. Bring the name, dose, and date of your last GLP-1.
Medical Information Guide — 2026 Verified
GLP-1 Gastroparesis: Symptoms, Red Flags, and What To Actually Do (2026)
Published:
Last verified: May 1, 2026 · Next scheduled re-verification: August 1, 2026
By The RX Index editorial team · Sourced from FDA / DailyMed prescribing information, peer-reviewed literature, and multi-society clinical guidelines · Editorial standards
GLP-1 gastroparesis is when a GLP-1 medication — like Ozempic, Wegovy, Rybelsus, Mounjaro, Zepbound, or Foundayo — slows your stomach down so much that food sits there for hours. These drugs are designed to slow your stomach a little. In a small share of users, that slowdown gets bad enough to cause real symptoms: throwing up undigested food, fullness that won't quit, severe bloating. Case reports and clinical commentary describe symptoms commonly improving within weeks to a few months after the medication is reduced or stopped — but severe symptoms need a clinician, not a wait-and-see approach.
What should you do right now?
| Your situation | What it likely means | What to do |
|---|---|---|
| Mild nausea, fullness, or burping after starting or upping your dose. You can still eat small meals and drink fluids. | Normal GLP-1 adjustment | Track it. Tell your prescriber at your next visit. |
| Repeated vomiting, food coming back up hours after you ate, severe bloating that won't pass, can't eat much for days, mild dehydration signs (dark urine, headache) | Possible delayed gastric emptying or gastroparesis | Call your prescriber today. Don't take your next dose without guidance. |
| Can't keep liquids down · severe belly pain · vomiting blood or coffee-ground material · very little or no urine output · confusion · no gas or stool with painful bloating · suspected aspiration during a procedure | Possible severe gastroparesis, ileus, obstruction, dehydration, or aspiration | Get urgent care or go to the ER now. Bring the name, dose, and date of your last GLP-1 shot or pill. |
This matrix is general guidance, not a diagnosis. When in doubt, default to the more cautious row.

Is GLP-1 gastroparesis the same thing as normal GLP-1 nausea?
Short answer: No. They live on the same line — but they're not the same point on it.
GLP-1 medications slow how fast food leaves your stomach. That's what they do. Feeling fuller faster, mild nausea, a few burps, smaller appetite — those are the medication doing its job. Gastroparesis is something else. It's a medical diagnosis. It means your stomach has slowed down so much that food sits there for hours. Severe bloating. Throwing up food you ate yesterday. Pain after meals that won't quit.
When symptoms are persistent, severe, or you're throwing up undigested food hours after a meal, that's no longer "the drug doing its job." That's a real symptom and a real signal to call your prescriber.
Normal GLP-1 effects vs. signs to take seriously
| What you're feeling | More likely normal | More likely a problem |
|---|---|---|
| Nausea | Mild, mostly around dose increases, getting better | Persistent, getting worse, blocking you from eating |
| Fullness | Smaller meals satisfy you faster | "Brick in my stomach" feeling that doesn't pass |
| Vomiting | None or rare | Repeated vomiting; vomiting up food eaten hours ago |
| Bloating | Comes and goes, mild | Severe, constant, painful |
| Constipation | Mild, manageable | No bowel movement or gas, getting worse |
| Hydration | Can keep fluids down | Dark urine, dizzy when standing, can't keep water down |
| Belly pain | Mild discomfort | Severe or sharp, especially with vomiting |
| Weight | Steady loss in line with prescriber's plan | Dropping faster than expected, can't eat enough to maintain |
Can GLP-1 medications actually cause gastroparesis?
Short answer: They can trigger gastroparesis-like symptoms and may worsen pre-existing delayed gastric emptying. Whether a specific GLP-1 caused chronic gastroparesis in a specific person is a clinical and legal question still being worked out. The honest middle ground: there's a real signal in the data that's bigger than zero — the American College of Gastroenterology calls the strongest study "hypothesis-generating" rather than proof of causation.
How GLP-1s slow your stomach
GLP-1 receptor agonists turn on receptors that slow the stomach's pumping action and tighten the pyloric muscle (the gate at the bottom of your stomach). Slower pump + tighter gate = food sits in the stomach longer. For most people, that means a longer feeling of fullness and steadier blood sugar. For a smaller group, it means food sits too long, and symptoms turn up.
What the research actually shows
A 2023 research letter in JAMA (Sodhi et al.) compared GLP-1 weight-loss users to people taking bupropion-naltrexone. The adjusted hazard ratio for gastroparesis among GLP-1 users was 3.67 vs. the comparator group. The American College of Gastroenterology summarized these findings as "hypothesis-generating."
A 2024 Cleveland Clinic analysis using the TriNetX database (~88 million patient records) found a statistically significant increased risk of new gastroparesis diagnoses among Type 2 diabetes patients on GLP-1 receptor agonists at 6, 9, 12, 18, and 24 months.
A separate review in the Journal of Clinical Endocrinology & Metabolism (December 2024) noted that some smaller studies haven't found a clear link, and that the body of evidence is still evolving.
How common is GLP-1 gastroparesis, really?
Short answer: Uncommon, but real. Published estimates vary because studies use different populations, different definitions, and different denominators — which is why you'll see one source say "very rare" and another say "5%." They're often measuring different things.
| Source | Population | What was measured | Number |
|---|---|---|---|
| Sodhi et al., JAMA 2023 | GLP-1 weight-loss users vs. bupropion-naltrexone users (claims database) | Adjusted hazard ratio for new gastroparesis diagnosis | aHR 3.67 (95% CI 1.15–11.90) |
| All of Us cohort analysis | 10,328 adults with diabetes or obesity who were new GLP-1 RA users | Reported gastroparesis among GLP-1 RA cohort | 5.1% |
| All of Us cohort, by drug | Same cohort, by specific GLP-1 | Highest gastroparesis prevalence (exenatide) | 6.9% (vs. lower for semaglutide) |
| National Jewish Health (Dr. Pranav Periyalwar) | Clinical experience | Gastroparesis side effects on GLP-1s | “Less than 2%” |
| Wegovy injection label | Phase 3 weight-loss trial | Severe GI adverse reactions | 4.1% vs. 0.9% placebo |
| Zepbound label | Phase 3 weight-loss trial | Severe GI adverse reactions, by dose | 1.7% (5 mg), 2.5% (10 mg), 3.1% (15 mg) vs. 1% placebo |
Gastroparesis on a GLP-1 is uncommon. It is more likely than on no drug at all. It is less common than the lawsuit ads suggest. If you're experiencing real symptoms, the population statistics don't matter for your specific case — what matters is what you do next.
What does each FDA label say about gastroparesis?
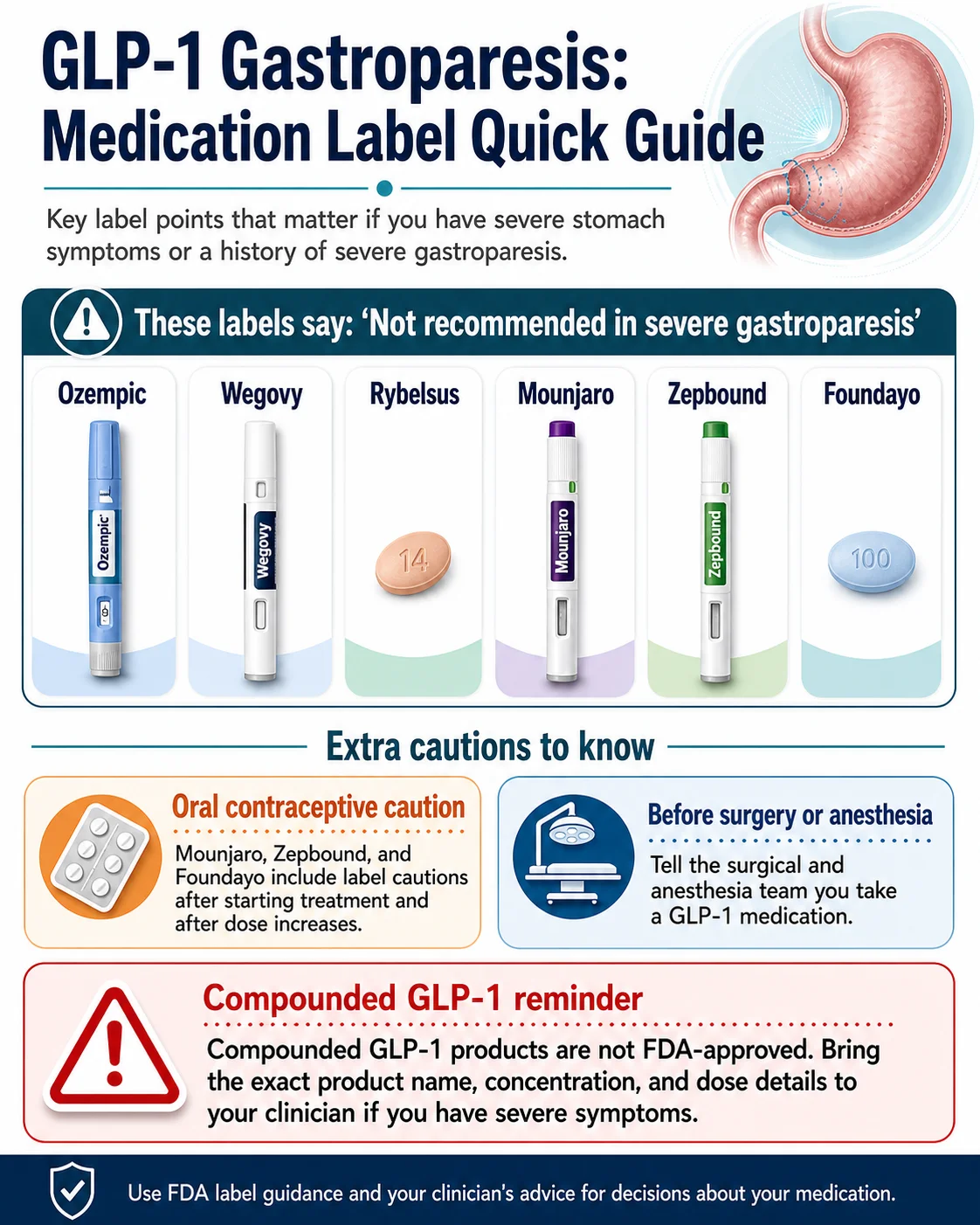
Short answer: Every major GLP-1 medication has FDA-approved label language about delayed gastric emptying, severe gastrointestinal adverse reactions, or both. The current Ozempic, Wegovy, Rybelsus, Mounjaro, Zepbound, and Foundayo labels all explicitly say the medication is “not recommended in patients with severe gastroparesis.”
| Medication | What the FDA label says | Severe GI rate (per label) | What this means for you |
|---|---|---|---|
| Ozempic (semaglutide injection) | Warns about delayed gastric emptying. Not recommended in patients with severe gastroparesis. Label revised 10/2025; Major Changes include severe GI adverse reactions and pulmonary aspiration during anesthesia. | 0.4% on 0.5 mg, 0.8% on 1 mg, 0% placebo | If you already have severe gastroparesis, the label says Ozempic is not recommended. If you develop severe symptoms, call your prescriber. |
| Wegovy (semaglutide for weight management) | Same active ingredient as Ozempic. Not recommended in patients with severe gastroparesis. Same class GI warnings. | 4.1% on injection vs. 0.9% placebo · 2% on tablet vs. 0% placebo | The weight-loss formulation has higher reported severe-GI rates than the diabetes dose. Take the label seriously, especially when titrating up. |
| Rybelsus (oral semaglutide tablet) | Not recommended in patients with severe gastroparesis. Same class GI warnings. | 0.6% on 7 mg, 2% on 14 mg, 0.3% placebo | “It's just a pill” doesn't mean the stomach effect goes away. The mechanism — slowing emptying — is the same. |
| Mounjaro (tirzepatide injection) | Not recommended in patients with severe gastroparesis. Label notes delayed gastric emptying can affect oral medications, including oral hormonal contraceptives after starting and after dose increases. | 1.3% on 5 mg, 0.4% on 10 mg, 1.2% on 15 mg, 0.9% placebo | If you're on the pill, this label has specific guidance. See the birth control section below. |
| Zepbound (tirzepatide for weight management) | Not recommended in patients with severe gastroparesis. | 1.7% on 5 mg · 2.5% on 10 mg · 3.1% on 15 mg · 1% placebo | Severe GI rate goes up with dose. Most bad weeks happen right after a titration step. |
| Foundayo (orforglipron tablet — FDA-approved 2026) | Not recommended in patients with severe gastroparesis. Addresses delayed gastric emptying, oral medication absorption, and pulmonary aspiration warnings. Advises non-oral or barrier contraception for 30 days after starting and 30 days after each dose escalation. | ~3% vs. 1% placebo | The newest oral GLP-1. Note the 30-day contraceptive window is longer than the 4-week window on Mounjaro and Zepbound. |
| Compounded semaglutide / tirzepatide | Per the FDA, compounded drugs are not FDA-approved and do not undergo FDA review for safety, effectiveness, or quality. The FDA has reported dosing errors and adverse events. On April 30, 2026, the FDA proposed excluding semaglutide, tirzepatide, and liraglutide from the 503B bulks list. | No standardized rate — products vary by compounding pharmacy | If you're using one and have severe symptoms, tell your prescriber the exact product, concentration, syringe units, and pharmacy. |
Sources: DailyMed prescribing information for each medication, verified May 1, 2026. FDA postmarket safety communication on unapproved GLP-1 drugs; FDA April 30, 2026 press announcement.
What the label language actually means: A label saying "not recommended in patients with severe gastroparesis" is not the FDA saying the drug causes gastroparesis. It's a precaution: don't start this drug if you already have a stomach that doesn't empty properly. A label adding "delayed gastric emptying" or pulmonary aspiration language is the FDA acknowledging that postmarket reports have raised the issue. A label change isn't a verdict on causation. It's a careful update to the warnings.
Symptom severity self-check
Short answer: Use this matrix to sort your symptoms into one of four buckets. Each bucket has a different next step. When in doubt, go to the more cautious bucket. This is general guidance based on FDA label warnings, NIDDK warning signs, and standard dehydration / obstruction red flags — it does not replace a clinician's judgment.
The four-tier triage
| Tier | What you're feeling | What it likely is | What to do |
|---|---|---|---|
| Expected GLP-1 effect | Mild nausea in your first weeks or after a dose bump · feeling full faster · mild bloating that fades between meals · slightly less appetite | Your medication is working as designed | Eat smaller, lower-fat meals · drink fluids · note it for your next prescriber visit · no need to call today |
| Worth a message | Persistent nausea past 2 weeks · vomiting more than rarely · bloating that doesn't fade · weight dropping faster than plan · sulfur burps that won't go away | Possible delayed gastric emptying beyond the intended therapeutic effect | Send a portal message or call within a few days · keep a symptom log · ask whether to lower or pause the dose |
| Call prescriber today | Vomiting daily · throwing up undigested food from hours earlier · can't tolerate solid food · belly pain after meals · early dehydration signs (dark urine, dizzy, headache) | Possible gastroparesis or significant delayed gastric emptying | Call today. If you can't reach them, go to urgent care. Do not take your next scheduled dose without guidance. |
| ER now | Can't keep water down · severe or sharp belly pain · vomiting blood or coffee-ground material · very little or no urine output · confusion · suspected bowel obstruction · choking on stomach contents during a procedure | Possible severe gastroparesis, ileus, bowel obstruction, severe dehydration, or aspiration | Go now. Bring the name, dose, and date of your last GLP-1 shot or pill. Tell every clinician you take a GLP-1. |
Only a clinician can diagnose gastroparesis (typically with a gastric emptying test). This matrix is to help you decide what to do today — not to replace a real evaluation.
Is GLP-1 gastroparesis reversible?
Short answer: In most reported cases, yes — case reports and clinical commentary describe symptoms commonly improving within weeks to a few months after the medication is reduced or stopped. This is different from gastroparesis caused by long-term diabetic nerve damage, which is often permanent.
Recovery patterns from the case-report literature
These are patterns described in published case reports — not promises. Your timeline depends on the drug, dose, how long you've been on it, your underlying health, and whether other things are also slowing your stomach.
- ◆Days to ~2 weeks in milder cases, especially with prompt dose reduction or pause, on shorter-acting GLP-1s.
- ◆2–8 weeks for moderate cases on weekly-dosed GLP-1s (semaglutide, tirzepatide). These drugs have long half-lives, so the medication keeps working in your body for a while after your last dose.
- ◆2–6 months for some severe cases, or for people whose underlying diabetic gastroparesis was unmasked rather than caused by the drug.
- ◆Longer or persistent in a smaller subset, especially those with pre-existing diabetes or other risk factors.
The literature does not support the lawsuit-ad framing that GLP-1 gastroparesis is universally permanent. It also doesn't support the "don't worry about it, it always reverses" framing. The real picture: usually reversible, sometimes slow, occasionally lingering, and worth taking seriously.
Should you stop your GLP-1?
Short answer: This is a decision that belongs with your prescriber, not the internet. Severe or red-tier symptoms = don't take another dose until you get medical guidance. Stopping abruptly without a plan can cause weight regain and blood sugar swings, especially if you're using the drug for Type 2 diabetes.
Path 1 — Reduce the dose
Often the first move when symptoms started right after a titration step. Many people tolerate the lower dose just fine and stay on the drug long-term.
Path 2 — Pause for a defined period
Sometimes the right move is a 2–4 week break, see how you feel, then re-evaluate. Your appetite will return, and so will some weight if you've been losing — that's part of the calculation.
Path 3 — Stop and switch (or not)
For severe symptoms, pre-existing severe gastroparesis, or when other tries haven't worked, stopping the drug is the answer. From there, your clinician decides whether a different approach makes sense.
The honest move when symptoms cross the "call your prescriber today" line: don't push through, don't stop alone in a panic, call your prescriber. That's the right path almost every time.
Surgery, anesthesia, birth control, and other oral medications
Short answer: GLP-1s slow your stomach. That means anything you swallow gets absorbed differently, and food can sit in your stomach during procedures. Most articles skip this section. We won't.
Surgery and anesthesia (this changed recently)
What changed in October 2024
In October 2024, a multi-society guidance from the ASA, AGA, ASMBS, ISPCOP, and SAGES shifted the position: most patients can continue taking their GLP-1 medication before elective surgery, with individualized precautions for higher-risk situations. This was a meaningful reversal from the 2023 ASA guidance that had recommended holding the medication.
Your action items haven't changed:
- → Tell your surgical team. Every member. Day of, the morning of, before they put you under.
- → Bring the brand, dose, and date of your last dose. Don't say "I take one of those weight loss shots." Say "I take Wegovy 2.4 mg, last dose was Tuesday."
- → Don't change your dosing on your own. If your prescriber and surgical team agree to hold a dose, hold it.
- → If you feel full at the start of the procedure, say something. Even after fasting. That's the warning sign.
The Ozempic and Foundayo labels both include explicit pulmonary aspiration warnings during procedures requiring anesthesia or deep sedation. That hasn't changed. The conversation about when to hold the dose changed.
Birth control pills
The Mounjaro, Zepbound, and Foundayo labels include specific guidance about oral hormonal contraceptives — slowed gastric emptying changes how the pill is absorbed:
Mounjaro & Zepbound (tirzepatide)
Consider a non-oral or barrier method for 4 weeks after starting and 4 weeks after each dose escalation.
Foundayo (orforglipron)
Use a non-oral or barrier method for 30 days after starting and 30 days after each dose escalation.
This guidance does not appear in the same form on semaglutide labels, but the general principle — delayed gastric emptying can change how some oral medications are absorbed — still applies. Ask your prescriber.
Other oral medications
Narrow-therapeutic-index drugs — warfarin, levothyroxine for thyroid, anticonvulsants — where the dose-to-effect window is small can be sensitive to absorption changes. If you take any of these and start a GLP-1, your prescriber may want to monitor levels more closely during your titration period.
How to talk to your prescriber (the script)
Short answer: A 10-minute appointment is not enough time to figure this out from scratch. Walk in with a written log, the right questions in the right order, and a clear ask.
Bring with you
- Symptom log. Dates, times, what you ate, when you vomited, what came up. If undigested food shows up — what it was and how long after the meal.
- Your medication info. Brand name, dose, weeks at this dose, total weeks on the drug, date of your last dose.
- Other medications and supplements — including birth control, blood thinners, thyroid medication, seizure medication.
- Weight and weight change over the past month.
- Whether you can keep liquids down. Yes / no / sometimes.
The copy-paste prescriber message
Questions to ask, in order
- Based on my symptom pattern, do you think this is expected GLP-1 effects, delayed gastric emptying, or possible gastroparesis?
- Should I get a gastric emptying study? (This is the main objective test.)
- What are my options — lower the dose, pause, switch, stop?
- If I pause or stop, what should I expect for my weight and blood sugar in the next 4–8 weeks?
- What symptoms would mean I need to go to the ER before our next appointment?
- Are there other medications you can prescribe to help with the symptoms — anti-nausea (like ondansetron), prokinetics?
- Do my other oral medications need monitoring or backup — especially birth control if I'm on tirzepatide or Foundayo?
- If symptoms keep going after stopping, what's next — gastroenterology referral?
Decisions to come prepared to make
- Are you willing to lower or pause the dose?
- Do you want testing for peace of mind even if symptoms ease?
- Do you want a gastroenterology referral if your prescriber recommends one?
- What's your plan for managing weight or blood sugar during a pause?
How is gastroparesis diagnosed and treated?
Short answer: Diagnosis combines symptoms with an objective test that measures how fast your stomach empties — most commonly a 4-hour gastric emptying scintigraphy, a wireless motility capsule, or a breath test. Treatment depends on cause and severity.
Diagnosis
Per Mayo Clinic and NIDDK, the main diagnostic tools are:
- Gastric emptying scintigraphy (GES). The standard test. You eat a small meal with a tiny amount of radioactive material. A camera tracks how the meal moves out of your stomach over 4 hours. More than 10% of the meal still in your stomach at 4 hours is generally considered abnormal.
- Wireless motility capsule (WMC). A swallowable capsule that records pH and movement as it travels through your digestive tract.
- Breath test. Measures emptying based on substances detected in your breath after a labeled meal.
- Endoscopy and imaging. Used to rule out blockages, ulcers, gallstones, or other causes that look like gastroparesis on the surface but aren't.
A legal note: in MDL 3094, Judge Karen Marston ruled (August 15, 2025) that plaintiffs claiming GLP-1-induced gastroparesis must have a properly performed gastric emptying study at the time of diagnosis to support their claim. That's a legal requirement. For your medical purposes, your clinician decides what testing makes sense.
Treatment
For drug-induced gastroparesis specifically, the standard first move is to discontinue or reduce the suspected medication. Most cases improve with that change.
For symptom management while recovering:
- Diet. Smaller, more frequent meals. Lower fat. Lower fiber. Soft, well-cooked foods. More liquid calories.
- Hydration. Sip fluids throughout the day rather than chugging at meals. Add electrolytes if you've been vomiting.
- Medications. Metoclopramide is the only FDA-approved prokinetic, but it has a black-box warning and is generally limited to ~12 weeks. Off-label options include erythromycin. Anti-nausea medications like ondansetron are commonly used.
- For severe, refractory cases. Per AGA's 2025 clinical guidance, gastric electrical stimulation, G-POEM, and botulinum toxin injections are not recommended for routine use — reserved for selected refractory cases under specialist care.
Most GLP-1-related cases never need any of the severe-end interventions. They get better with dose reduction or stopping the drug.
Who is at higher risk?
Short answer: People with long-standing diabetes, pre-existing gastroparesis or significant GI disease, prior gastric or vagus nerve surgery, older age, female sex, and people who escalated their GLP-1 dose rapidly or skipped the recommended titration steps.
- ◆Long-standing Type 2 diabetes: especially over 10 years. Diabetes can damage the vagus nerve over time, which causes gastroparesis on its own. A GLP-1 added to that picture amplifies the slowdown.
- ◆Pre-existing gastroparesis or significant GI motility disorders: All major GLP-1 labels now say not recommended in severe gastroparesis. This is why.
- ◆Prior gastric or vagus nerve surgery: bariatric surgery, fundoplication, certain cancer surgeries.
- ◆Older adults: Slower baseline motility, more medications interacting, higher risk of dehydration complications.
- ◆Women: Gastroparesis is roughly four times more common in women than men in the general population.
- ◆Rapid dose escalation: This is the one risk factor you can actually do something about. The titration schedule exists for a reason. Don't skip steps.
- ◆Compounded GLP-1 products with dosing errors: The FDA has documented adverse-event reports involving wrong syringe sizes and miscalculated doses with compounded products.
Compounded GLP-1s and gastroparesis risk
Short answer: Compounded GLP-1 medications are not FDA-approved drugs. They don't go through the FDA's review for safety, effectiveness, and quality. The FDA has reported dosing errors and adverse events with compounded products. As of April 30, 2026, the FDA has proposed excluding semaglutide, tirzepatide, and liraglutide from the 503B bulks list.
What's accurate:
- Compounded drugs are not FDA-approved and do not undergo FDA review for safety, effectiveness, or quality.
- The FDA has reported dosing errors and adverse events in compounded semaglutide and tirzepatide products, including products containing salt forms (semaglutide sodium, semaglutide acetate) that the FDA notes are different active ingredients than those in approved drugs.
- On April 30, 2026, the FDA proposed excluding semaglutide, tirzepatide, and liraglutide from the 503B bulks list.
- If you're using a compounded GLP-1 and you develop severe symptoms, your clinician will want to know exactly what product, concentration, syringe units, and pharmacy you used.
Compounded products may be legitimate options for some users in some situations — but on a gastroparesis page, the additional variables don't fit the moment. If you specifically want a compounded option after talking to your prescriber, we have separate guides for that.
Already past the symptoms — finding the right program
If your symptoms are settled and you're now evaluating which GLP-1 program fits someone with a careful stomach history, two programs stand out for different reasons. We do not feature compounded providers on this page — see the compounded section above for why.
For FDA-approved medication + insurance support
Ro
Ro publicly offers FDA-approved GLP-1 options including Wegovy pill, Wegovy pen, Zepbound KwikPen, and Foundayo. Their program includes an insurance concierge that handles prior-authorization paperwork, plus a free GLP-1 Insurance Coverage Checker tool. Ro Body membership: $39 first month, then $149/month, or as low as $74/month with annual plan. Medication cost is charged separately.
The honest case: FDA-approved medication backed by the labels we used to build the matrix above, an insurance concierge that can help you use your coverage. The honest tradeoff: not the cheapest cash-pay option.
Check Ro program details and eligibility (opens in new tab)For self-pay with HSA/FSA flexibility
Eden
Eden offers FDA-approved branded medications (including Ozempic, Wegovy, and Zepbound), no membership fees, no insurance required, HSA/FSA eligibility, and free shipping. Verified May 1, 2026 on tryeden.com.
The honest case: a self-pay program with FDA-approved branded medication, no membership fee, and HSA/FSA flexibility. The honest tradeoff: insurance support is less hands-on than Ro's. If you have insurance you want to use first, start with Ro.
Check Eden program details and eligibility (opens in new tab)Affiliate disclosure: The RX Index may earn a commission if you start a program through these links. That commission does not change the price you pay. Commercial relationships are disclosed and do not override medical-safety criteria. See our editorial standards.
Frequently Asked Questions
Can Ozempic cause gastroparesis?
Population studies have found a meaningfully increased risk of gastroparesis among Ozempic and other GLP-1 users compared to non-users; the JAMA 2023 research letter (Sodhi et al.) reported an adjusted hazard ratio of 3.67 vs. bupropion-naltrexone. The current Ozempic label says the medication is not recommended in patients with severe gastroparesis. Whether Ozempic causes gastroparesis in the strict pharmacological sense is being actively litigated in MDL 3094.
How do I know if I have gastroparesis or just normal Ozempic side effects?
The clearest distinguishing features are persistent or repeated vomiting (especially of undigested food eaten hours earlier), inability to keep food or fluids down, severe bloating that doesn't pass between meals, and significant unintentional weight loss faster than your prescriber expected. Use the symptom severity matrix on this page. Call your prescriber if you fall into the "call today" or "urgent care" tiers.
Is gastroparesis from Ozempic permanent?
In most reported cases, gastroparesis associated with Ozempic and other GLP-1s improves within weeks to several months after the medication is reduced or stopped. This is different from gastroparesis caused by long-standing diabetic nerve damage, which is often permanent. A minority of people report symptoms that linger; talk to your prescriber if symptoms persist beyond a few months.
How long does GLP-1 gastroparesis last after stopping?
Case reports describe a range from days to several months, with most cases improving within weeks. Weekly-dosed GLP-1s like semaglutide and tirzepatide have long half-lives, so symptom resolution may take longer than with shorter-acting drugs. People with pre-existing diabetes or other risk factors may have longer recovery.
Can Mounjaro or Zepbound cause gastroparesis at the same rate as Ozempic?
Some pharmacovigilance analyses have found tirzepatide showed a smaller signal for gastroparesis than semaglutide or liraglutide, though absolute rates are still being studied. Mounjaro and Zepbound users have reported gastroparesis cases, and both labels include the standard "not recommended in severe gastroparesis" language. The Zepbound label reports severe GI adverse reactions of 1.7%, 2.5%, and 3.1% across the 5 mg, 10 mg, and 15 mg doses, vs. 1% on placebo.
Does Foundayo (orforglipron) cause gastroparesis?
Foundayo is the newest oral GLP-1 receptor agonist tablet (FDA-approved in 2026). The label addresses delayed gastric emptying, oral medication absorption, and pulmonary aspiration warnings during anesthesia. The label says Foundayo is not recommended in patients with severe gastroparesis. Severe GI adverse reactions on Foundayo were reported at about 3% versus 1% placebo.
Can I take a GLP-1 if I already have gastroparesis?
If you have severe gastroparesis, the labels for Ozempic, Wegovy, Rybelsus, Mounjaro, Zepbound, and Foundayo all advise against use. For mild or controlled gastroparesis, the decision belongs to a clinician — typically a gastroenterologist working with your prescriber.
Should I stop my GLP-1 before surgery?
Tell your surgical team — including your anesthesiologist — that you take a GLP-1, the brand, the dose, and your last dose date. Per the October 2024 multi-society guidance from the ASA, AGA, ASMBS, ISPCOP, and SAGES, most patients can continue their GLP-1 before elective surgery, with risk-stratified precautions for higher-risk situations. The plan should come from your surgical team, not the internet.
Do GLP-1s affect birth control pills?
The Mounjaro and Zepbound labels recommend a non-oral contraceptive method or a backup barrier method for 4 weeks after starting and 4 weeks after each dose escalation. The Foundayo label uses 30 days for both windows. The semaglutide labels (Ozempic, Wegovy, Rybelsus) don't have the same specific guidance, but the general principle — slowed oral absorption — still applies. Ask your prescriber.
Are compounded GLP-1s riskier for gastroparesis?
Compounded products are not FDA-approved and do not undergo FDA review for safety, effectiveness, or quality. The FDA has reported dosing errors and adverse events with compounded GLP-1s. On April 30, 2026, the FDA proposed excluding semaglutide, tirzepatide, and liraglutide from the 503B bulks list. If you're using a compounded product and have severe symptoms, tell your prescriber the exact product, concentration, syringe units, and pharmacy.
What test confirms gastroparesis?
A 4-hour gastric emptying scintigraphy is the standard. You eat a small meal containing a tiny amount of radioactive material, and a camera tracks how the meal empties from your stomach over 4 hours. Other options include a wireless motility capsule and a breath test. Per Mayo Clinic and NIDDK, more than 10% of the meal still in your stomach at 4 hours is generally considered abnormal.
Is "stomach paralysis" just a lawsuit term?
"Stomach paralysis" is a plain-language term for gastroparesis. It's also been heavily used in lawsuit ads. The medical reality: in most GLP-1 cases, the stomach isn't fully paralyzed; it's slowed. The useful question isn't whether the lawsuit ad is right; it's whether your symptoms are mild, persistent, severe, or urgent. Use the symptom severity matrix on this page.
Can vomiting undigested food from earlier meals confirm gastroparesis?
It's a strong symptom signal but not a diagnosis on its own. Normal digestion breaks down food within 2–4 hours; seeing whole food pieces in vomit hours later is consistent with delayed gastric emptying. It needs to be combined with other symptoms and clinical evaluation. Tell your prescriber if this is happening.
How do I report a GLP-1 side effect to the FDA?
You can file a MedWatch report at the FDA's online voluntary reporting page or by calling 1-800-FDA-1088. The FDA explicitly encourages patients and clinicians to report adverse events, including with compounded GLP-1 products. Filing a MedWatch report is separate from any legal action — you can do one, both, or neither.
Still not sure? Here's a shorter path.
Most people who land on this page need one of three things.
- If you're having severe symptoms right now — vomiting blood, can't keep water down, severe belly pain, suspected obstruction — stop reading and get to urgent care or the ER. Bring the name, dose, and date of your last GLP-1.
- If your symptoms aren't urgent but you need to talk to your prescriber — use the prescriber script above. That's the script so a 10-minute appointment doesn't waste your symptoms.
- If symptoms are settled and you're figuring out which GLP-1 program fits — take our free matching quiz.
If you're currently having severe vomiting, dehydration, severe abdominal pain, or can't keep fluids down — don't take a quiz. Get medical care first.
Sources and verification
This page was built from primary sources. Key claims are attributed to their source inline. Pricing and FDA label statements were verified May 1, 2026.
FDA prescribing information (DailyMed) — verified May 1, 2026
- Ozempic (semaglutide injection) prescribing information
- Wegovy (semaglutide) prescribing information
- Rybelsus (oral semaglutide) prescribing information
- Mounjaro (tirzepatide) prescribing information
- Zepbound (tirzepatide) prescribing information
- Foundayo (orforglipron) prescribing information
FDA postmarket safety communications
- FDA — Concerns with Unapproved GLP-1 Drugs Used for Weight Loss
- FDA MedWatch Voluntary Reporting Program (1-800-FDA-1088)
Peer-reviewed literature
- Sodhi et al., JAMA (2023) — Risk of Gastrointestinal Adverse Events Associated With GLP-1 Receptor Agonists for Weight Loss
- Journal of Clinical Endocrinology & Metabolism (December 2024) — Clinical Consequences of Delayed Gastric Emptying With GLP-1 RAs and Tirzepatide
- Pharmaceuticals (PMC, 2024) — GLP-1 RAs Associated GI Adverse Events: NIH "All of Us" Cohort
- Cureus (2018) — Liraglutide-Induced Acute Gastroparesis
- Cureus (2025) — Unmasking Semaglutide-Induced Gastroparesis
Clinical guidelines
- ASA / AGA / ASMBS / ISPCOP / SAGES — October 2024 Multi-Society Perioperative GLP-1 Guidance
- American Gastroenterological Association — 2025 Clinical Guidance on the Management of Gastroparesis
- NIDDK — Gastroparesis: Definition, Symptoms, Diagnosis, and Treatment
- Mayo Clinic — Gastroparesis: Diagnosis and Treatment
Published:
Last verified: May 1, 2026 · Next scheduled re-verification: August 1, 2026
Provider pricing verified May 1, 2026 on ro.co/weight-loss/pricing and tryeden.com/weight-loss.
Related guides
About The RX Index. The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We are not a medical practice. The content on this site is educational and is not medical advice. Always talk to a licensed clinician about your specific situation. We may earn a commission when readers start a program through links on our site; commercial relationships are disclosed and do not override medical-safety criteria. See our editorial standards.