GLP-1 FDA Indication vs Off-Label Use: What's On-Label in 2026
Published: · Last reviewed:
By The RX Index Editorial Team · Verified monthly and on every FDA label change
This page classifies FDA-label status for GLP-1 medications. It is not medical advice and does not tell you whether a GLP-1 is right for you.
If you searched GLP-1 FDA indication vs off-label use because your prescription, denial letter, or telehealth provider used one of those words, here is the short answer first.
The 30-second answer
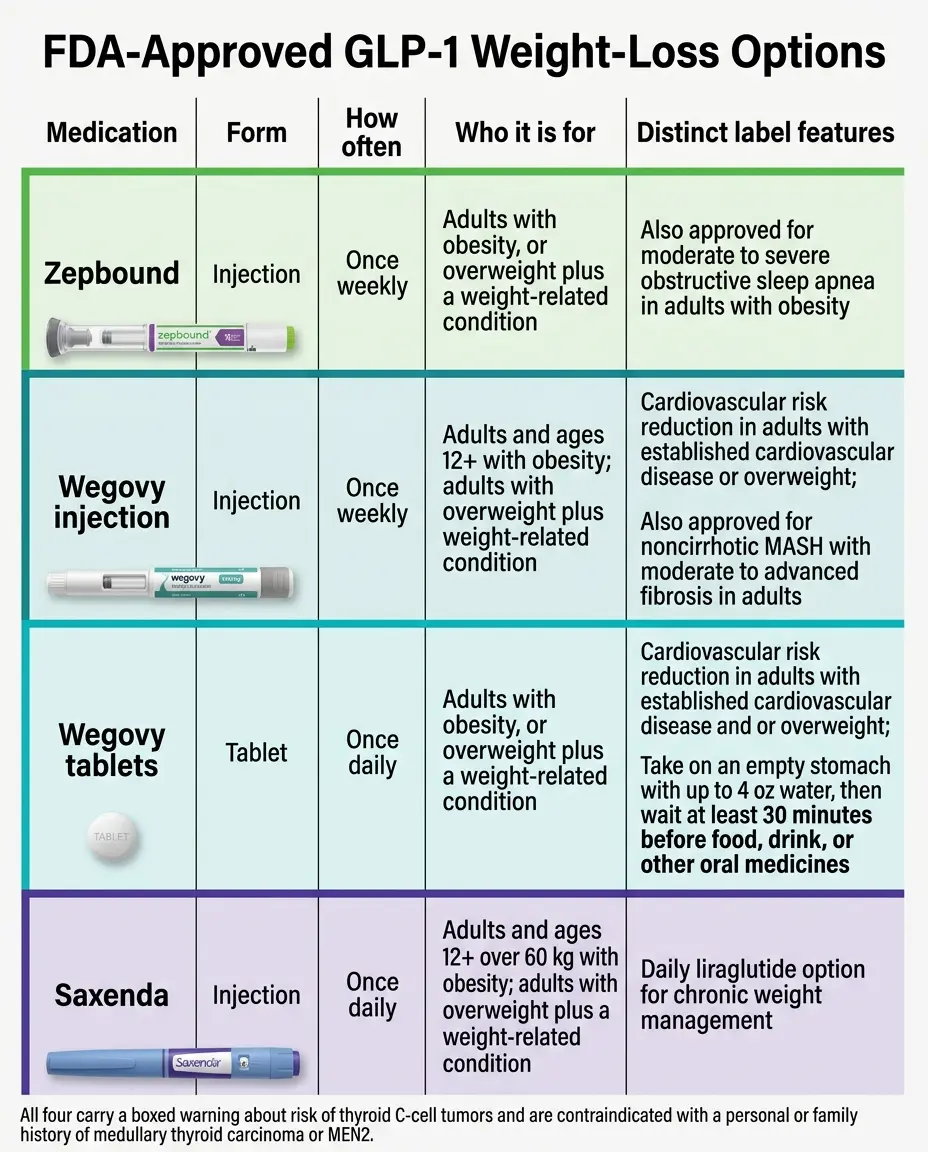
A GLP-1 use is FDA-approved only when the exact product, formulation, dose schedule, condition, and patient population all match the FDA-approved label for that specific product.
Using Ozempic for weight loss in someone without type 2 diabetes is generally off-label. Using Mounjaro for obesity alone is generally off-label. Using any GLP-1 specifically for PCOS, prediabetes, type 1 diabetes, binge-eating disorder, “food noise” at a healthy BMI, or alcohol cravings is off-label unless the same prescription separately matches an FDA-approved indication for that exact product.
Exact product, formulation, dose, diagnosis, and patient population all match the current FDA label.
FDA-approved drug prescribed for a condition, dose, or population not on its label. Legal but may not be covered.
Compounded, counterfeit, or research-grade GLP-1. Not an FDA-approved drug. Different category entirely.
| Your situation | Most likely status | What to do next |
|---|---|---|
| Exact drug, dose, and condition match a current FDA label | FDA-approved (on-label) | Verify the label, ask your clinician about fit and risks. |
| FDA-approved drug, but different condition, dose, or population | Off-label | Ask what evidence supports the use, and whether your insurance will cover it. |
| Compounded, counterfeit, “research peptide,” or no FDA-approved finished product | Not FDA-approved at all | Verify the source and avoid products not filled by a state-licensed pharmacy with a real prescription. |
What does “FDA-approved indication” mean for a GLP-1?
Quick answer
An FDA-approved indication is the specific condition, patient group, dose, and route the FDA approved a specific drug product to treat after reviewing its clinical-trial data. Every approved indication appears in Section 1 of that product's official prescribing information on DailyMed.
Think of an FDA approval as a very specific contract. The manufacturer ran a clinical trial, tested a certain dose in a certain group of people for a certain condition, and the FDA reviewed the data. Anything outside that contract — a different disease, a different age group, a different dose — wasn't tested in that trial. The FDA didn't say it doesn't work. The FDA said it didn't review it.
Why the same molecule can have very different FDA approvals
Two drugs can contain the exact same active ingredient and yet sit in completely different approval categories, because each product gets approved separately based on the trials its manufacturer submitted:
Semaglutide
Ozempic (T2D + CV + kidney indications), Wegovy (weight management, CV risk reduction, MASH), Rybelsus (T2D oral tablet)
Tirzepatide
Mounjaro (T2D glycemic control, adults and pediatric 10+), Zepbound (weight management and OSA in adults with obesity)
Liraglutide
Victoza (T2D + CV risk), Saxenda (weight management, adults and 12+); first FDA-approved generic liraglutide approved August 27, 2025
Same molecule. Different brand. Different label. Different FDA-label status and different coverage rules. This is the single most common source of confusion in GLP-1 medicine.
What does “off-label use” mean for a GLP-1?
Quick answer
Off-label use is when a clinician prescribes an FDA-approved drug for a condition, dose, route, or patient group not listed in the drug's FDA label. The FDA says clinicians may generally do this when they judge it medically appropriate, but the FDA has not reviewed that specific use for safety and effectiveness. Off-label prescribing accounts for roughly 1 in 5 U.S. prescriptions.
The FDA regulates the drug itself and what the drug company is allowed to say about it — not the practice of medicine. That's why a licensed clinician has wide legal latitude to prescribe an approved drug for a use not on the label.
But there are three things off-label use is not:
Not an FDA endorsement. The FDA hasn't reviewed it.
Not automatically covered by insurance. Many plans deny coverage for off-label use unless the use is supported by recognized drug compendia such as DRUGDEX or AHFS DI.
Not the same as taking a compounded drug. These are entirely different categories.
What a manufacturer can and cannot do
A drug company is not allowed to promote a drug for an off-label use. That is why you will never see a Novo Nordisk commercial telling doctors to use Ozempic for weight loss, even though plenty of doctors do. The FDA sent warning letters in fall 2025 targeting misleading GLP-1 marketing — including claims that non-FDA-approved compounded products were generic versions of, the same as, or used the same active ingredient as FDA-approved drugs.
When is a GLP-1 product unapproved — not just off-label?
Quick answer
An unapproved GLP-1 product is one whose finished product has not gone through FDA approval. This includes counterfeit Ozempic, illegally imported drugs, “research peptides” sold online, and most compounded GLP-1 mixtures sold today. The FDA has not reviewed these for safety, effectiveness, or quality.
A real, FDA-approved finished drug, made in a regulated facility, prescribed by a licensed clinician for something other than its labeled indication. The product and its manufacturing are on the books; the specific use may not be.
A custom-mixed preparation made by a compounding pharmacy. The finished drug is not FDA-approved. The FDA has not reviewed it for safety, effectiveness, or quality. The pharmacy itself may be licensed, but the product is not an FDA-approved finished drug.
An unapproved drug sold illegally, often online, often labeled with disclaimers designed to dodge regulation. The FDA has warned consumers explicitly not to buy these.
A clinician prescribing Ozempic off-label is doing something legal and common. A website selling “research-grade” tirzepatide is doing something the FDA has flagged as a safety hazard. Calling both “off-label” smudges the line between routine medicine and a real risk.
How do I tell if my GLP-1 use is on-label, off-label, or unapproved?
Quick answer
Check five things against the current FDA label on DailyMed: the exact product, the formulation and route, the dose schedule, the diagnosis or labeled use, and the patient population. All five must match to be on-label.
The 5-part label match test
The decision tree (use this for any specific situation)
Step 1. Is the product an FDA-approved finished drug?
↳ No (compounded, counterfeit, “research,” online peptide)
→ Unapproved-product category. Talk to your clinician about an FDA-approved alternative.
↳ Yes → Step 2.
Step 2. Does the exact product match its current FDA label for your use?
↳ Yes → Step 3.
↳ No → Off-label. Legal, but coverage and safety questions are open.
Step 3. Does the patient population match the label? (Age, BMI, comorbidities.)
↳ Yes → Step 4.
↳ No → Off-label.
Step 4. Does the dose and route match the label?
↳ Yes → On-label.
↳ No → Off-label.
The GLP-1 FDA Indication vs Off-Label Use Matrix (May 2026)
Quick answer
The matrix covers all current FDA-approved GLP-1 products and formulations in the U.S. Each row was verified May 13, 2026 against FDA press releases and current DailyMed prescribing information.

Table 1: FDA-Approved GLP-1 Products and Their Approved Indications
| Brand (active ingredient) | Form | T2D glycemic control | T2D + CV risk reduction | T2D + CKD (kidney/CV death) | Chronic weight management | Weight + CV risk reduction | OSA in adults w/ obesity | MASH w/ moderate-advanced fibrosis |
|---|---|---|---|---|---|---|---|---|
| Ozempic (semaglutide) | Weekly injection | ✅ 2017 | ✅ 2020 (established CVD) | ✅ Jan 28, 2025 | ❌ off-label | ❌ off-label | ❌ off-label | ❌ off-label |
| Wegovy (semaglutide) | Weekly injection | ❌ off-label | — | — | ✅ 2021 (adults), 2022 (12+) | ✅ Mar 2024 | ❌ off-label | ✅ Aug 15, 2025 |
| Wegovy HD (semaglutide 7.2 mg) | Weekly injection | ❌ off-label | — | — | ✅ Mar 19, 2026 | ❌ off-label at this dose | ❌ off-label | ❌ off-label |
| Wegovy pill (oral semaglutide 25 mg) | Daily tablet | ❌ off-label | — | — | ✅ Dec 2025 | ✅ Dec 2025 | ❌ off-label | ❌ off-label |
| Rybelsus (oral semaglutide) | Daily tablet | ✅ 2019 | ✅ 2025 (T2D + high CV risk) | ❌ off-label | ❌ off-label | ❌ off-label | ❌ off-label | ❌ off-label |
| Mounjaro (tirzepatide) | Weekly injection | ✅ 2022 (adults and pediatric 10+) | ❌ off-label | ❌ off-label | ❌ off-label | ❌ off-label | ❌ off-label | ❌ off-label |
| Zepbound (tirzepatide) | Weekly injection | ❌ off-label | — | — | ✅ Nov 2023 | ❌ off-label | ✅ Dec 20, 2024 | ❌ off-label |
| Foundayo (orforglipron) | Daily tablet | ❌ not approved for T2D | — | — | ✅ Apr 1, 2026 | ❌ off-label | ❌ off-label | ❌ off-label |
| Trulicity (dulaglutide) | Weekly injection | ✅ 2014 (adults and pediatric 10+) | ✅ 2020 (established CVD or multiple CV risk factors) | ❌ off-label | ❌ off-label | — | ❌ | ❌ |
| Victoza (liraglutide) | Daily injection | ✅ 2010 (adults and pediatric 10+) | ✅ 2017 (established CVD) | ❌ off-label | ❌ off-label | — | ❌ | ❌ |
| Saxenda (liraglutide; generic approved Aug 27, 2025) | Daily injection | ❌ off-label | — | — | ✅ 2014 (adults), 2020 (12+) | — | ❌ | ❌ |
| Byetta / Bydureon BCise (exenatide) | Twice-daily / weekly injection | ✅ 2005 / 2017 | ❌ off-label | ❌ | ❌ off-label | — | ❌ | ❌ |
| Adlyxin (lixisenatide) | Daily injection | ✅ 2016 | ❌ off-label | ❌ | ❌ off-label | — | ❌ | ❌ |
| Soliqua 100/33 (insulin glargine + lixisenatide) | Daily injection | ✅ T2D combination | ❌ off-label | ❌ | ❌ off-label | — | ❌ | ❌ |
| Xultophy 100/3.6 (insulin degludec + liraglutide) | Daily injection | ✅ T2D combination | ❌ off-label | ❌ | ❌ off-label | — | ❌ | ❌ |
Legend: ✅ = FDA-approved for this use, with year of first approval shown. ❌ off-label = the drug is FDA-approved (for at least one indication) but this use is off-label. — = not applicable to this product category. Verified May 13, 2026.
Exact label criteria (key thresholds and population requirements)
| Product & indication | Key criteria from the label |
|---|---|
| Wegovy injection (adult weight management) | BMI ≥30, or BMI ≥27 with at least one weight-related comorbidity (high blood pressure, T2D, or high cholesterol) |
| Wegovy injection (pediatric weight management) | Patients 12 years and older with a BMI at or above the 95th percentile for age and sex |
| Wegovy injection (CV risk reduction) | Adults with established cardiovascular disease and either obesity or overweight |
| Wegovy injection (MASH) | Adults with noncirrhotic MASH and moderate-to-advanced fibrosis (stages F2 or F3) |
| Wegovy HD 7.2 mg (weight management) | Adults who tolerated the 2.4 mg maintenance dose for at least four weeks and need additional weight reduction |
| Zepbound (adult weight management) | BMI ≥30, or BMI ≥27 with at least one weight-related comorbidity |
| Zepbound (OSA) | Adults with moderate-to-severe obstructive sleep apnea and obesity |
| Foundayo / orforglipron (weight management) | Adults with obesity (BMI ≥30) or overweight (BMI ≥27) with at least one weight-related comorbidity |
| Saxenda / generic liraglutide (adult) | BMI ≥30, or BMI ≥27 with at least one weight-related comorbidity |
| Saxenda (pediatric) | Patients 12 years and older with body weight above 60 kg and a BMI corresponding to ≥30 kg/m² for adults |
| Ozempic (CKD) | Adults with type 2 diabetes and chronic kidney disease |
Table 2: Use-Based Classification — What's Approved, Off-Label, or Unapproved
Most readers have a use in mind, not a brand. This table maps the other direction.
| What you're trying to do | FDA-approved products (when criteria align) | Likely off-label if you use… | Unapproved (different category) |
|---|---|---|---|
| Type 2 diabetes glycemic control | Ozempic, Rybelsus, Mounjaro, Trulicity, Victoza, Byetta, Bydureon BCise, Adlyxin, Soliqua, Xultophy | Wegovy, Wegovy HD, Wegovy pill, Zepbound, Foundayo, Saxenda | Compounded, counterfeit, or research GLP-1 |
| Chronic weight management (adults) | Wegovy, Wegovy HD, Wegovy pill, Zepbound, Foundayo, Saxenda | Ozempic, Mounjaro, Rybelsus, Trulicity, Victoza, Byetta/Bydureon, Adlyxin | Compounded, counterfeit, or research GLP-1 |
| Adolescent weight management (12+) | Wegovy injection (12+), Saxenda (12+ with body weight >60 kg and BMI corresponding to adult obesity) | Any adult-only or diabetes-only GLP-1 | Compounded products marketed to minors |
| CV risk reduction in adults with T2D + established CVD or high CV risk | Ozempic, Trulicity, Victoza, Rybelsus (criteria differ by product label) | Any GLP-1 without that specific labeled indication | — |
| CV risk reduction in adults with obesity/overweight + established CVD | Wegovy injection, Wegovy pill | Ozempic, Mounjaro, Zepbound for this indication | — |
| Kidney/CV death risk in T2D + CKD | Ozempic injection only | Any other GLP-1 | — |
| Moderate-to-severe OSA in adults with obesity | Zepbound only | Any other GLP-1, including Mounjaro | — |
| Noncirrhotic MASH with moderate-to-advanced fibrosis | Wegovy injection only | Any other GLP-1 | — |
| PCOS | None — no GLP-1 is FDA-approved for PCOS | Any GLP-1, when prescribed specifically for PCOS | — |
| Prediabetes alone | None — no GLP-1 is FDA-approved for prediabetes alone | Any GLP-1, when the only diagnosis is prediabetes | — |
| Type 1 diabetes adjunct | None — no GLP-1 is FDA-approved for T1D | Any GLP-1, when used alongside insulin in T1D | — |
| Binge-eating disorder | None — no GLP-1 is FDA-approved for BED | Any GLP-1, when prescribed specifically for BED | — |
| Alcohol use disorder / alcohol cravings | None — no GLP-1 is FDA-approved for AUD | Any GLP-1, when prescribed specifically for AUD | Research peptides sold online |
| Normal-BMI weight loss / food noise | None match weight-management labels at normal BMI | Any GLP-1, when used outside obesity/overweight criteria | Compounded or research products marketed for this |
Is Ozempic FDA-approved for weight loss?
Quick answer
No. Ozempic is FDA-approved for type 2 diabetes glycemic control (2017), MACE reduction in adults with T2D and established CVD (2020), and kidney-risk and cardiovascular-death reduction in adults with T2D and chronic kidney disease (January 28, 2025). Using Ozempic for weight loss in someone without type 2 diabetes is off-label. Wegovy is the semaglutide product FDA-approved for weight management.
Ozempic and Wegovy both contain semaglutide. They are made by the same company. They cause similar weight loss. But they are two different drugs in the eyes of the FDA, because they were studied in two different trials for two different purposes.
When a clinician prescribes Ozempic to someone without type 2 diabetes for weight loss: (1) it is legal — off-label prescribing is part of U.S. medicine; (2) your insurance probably won't cover it — most plans gate Ozempic to a T2D diagnosis. If you don't have one documented, prior authorization will likely fail and your prescriber will typically move you to Wegovy or another weight-management-labeled product.
Is Mounjaro FDA-approved for weight loss?
Quick answer
No. Mounjaro is FDA-approved to improve glycemic control in adults and pediatric patients 10 years and older with type 2 diabetes (2022). Zepbound is the tirzepatide product FDA-approved for chronic weight management (November 2023) and for moderate-to-severe OSA in adults with obesity (December 20, 2024).
| Question | Answer |
|---|---|
| Same active ingredient? | Yes, tirzepatide. |
| Same FDA label? | No. |
| Same coverage rules? | Usually no. Mounjaro gates to T2D. Zepbound gates to obesity/overweight criteria, OSA + obesity, or both. |
| Can a clinician prescribe Mounjaro off-label for weight loss? | Yes, legally. The FDA didn't approve that use. |
| Will insurance cover Mounjaro for weight loss? | Usually not without a T2D diagnosis, because many plans gate Mounjaro to its type 2 diabetes indication. |
The most common off-label GLP-1 uses (and what the evidence really says)
For each scenario: the regulatory status, the evidence signal, and what to ask. This is not a recommendation. It is a classification so you can have the right conversation with your clinician.
The most common off-label use, period. A clinician prescribes Ozempic, Mounjaro, Rybelsus, Trulicity, or another diabetes-indicated GLP-1 to a patient who doesn't have type 2 diabetes, with weight loss as the goal. Legal. Also usually denied by insurance unless the prescriber switches to the FDA-approved weight-management product and the patient meets the label criteria.
No GLP-1 is currently FDA-approved for PCOS. Clinicians do prescribe GLP-1s off-label for women with PCOS, especially those with obesity or insulin resistance. Truveta Research reported that among 445,258 patients with PCOS, semaglutide or tirzepatide prescribing rose from 2.4% in 2021 to 17.6% in 2025. Nearly all also had obesity or type 2 diabetes -- which matters, because those are approved metabolic indications. The prescription may be on-label for the metabolic indication even when the conversation is about PCOS.
No GLP-1 is FDA-approved for type 1 diabetes. Trials have studied GLP-1 receptor agonists as adjuncts to insulin in T1D, but use for T1D remains off-label. The risk profile includes a real concern about hypoglycemia when GLP-1s are combined with insulin without careful dose adjustment. Ask specifically about hypoglycemia monitoring, insulin dose adjustment, and whether a CGM is part of the plan.
No GLP-1 is FDA-approved for prediabetes by itself. Prescribing a GLP-1 to someone with prediabetes -- but without a qualifying T2D diagnosis or weight-management criteria -- is off-label. Coverage is plan-specific, but a prediabetes-only GLP-1 prescription usually faces the same denial problem as other off-label uses.
No GLP-1 is FDA-approved for binge-eating disorder. No GLP-1 is FDA-approved for healthy-BMI patients who describe persistent food noise. Lisdexamfetamine (Vyvanse) is FDA-approved for moderate-to-severe binge-eating disorder in adults. GLP-1s are not. This is one of the higher-risk off-label scenarios -- the safety data in this population is limited because clinical trials excluded it.
No GLP-1 is FDA-approved for alcohol use disorder or substance use disorder. A 2026 BMJ cohort study of U.S. veterans with T2D found GLP-1 use was associated with a 14% lower composite risk of substance use disorders. That's an observational signal, not an FDA indication. If a provider is recommending a GLP-1 specifically for cravings or alcohol use, that is explicitly off-label and explicitly investigational.
Heart failure with preserved ejection fraction (HFpEF), peripheral artery disease (PAD), Parkinson's disease, Alzheimer's disease, and osteoarthritis-related knee pain are all active research topics, but none is an FDA-approved GLP-1 indication today.
Compounded GLP-1s are not the same as off-label use
Quick answer
Compounded GLP-1s are mixtures prepared by compounding pharmacies under federal sections 503A or 503B. They are not FDA-approved drugs. As of April 30, 2026, the FDA proposed to permanently exclude semaglutide, tirzepatide, and liraglutide from the 503B Bulks List. The public comment period closes June 29, 2026.
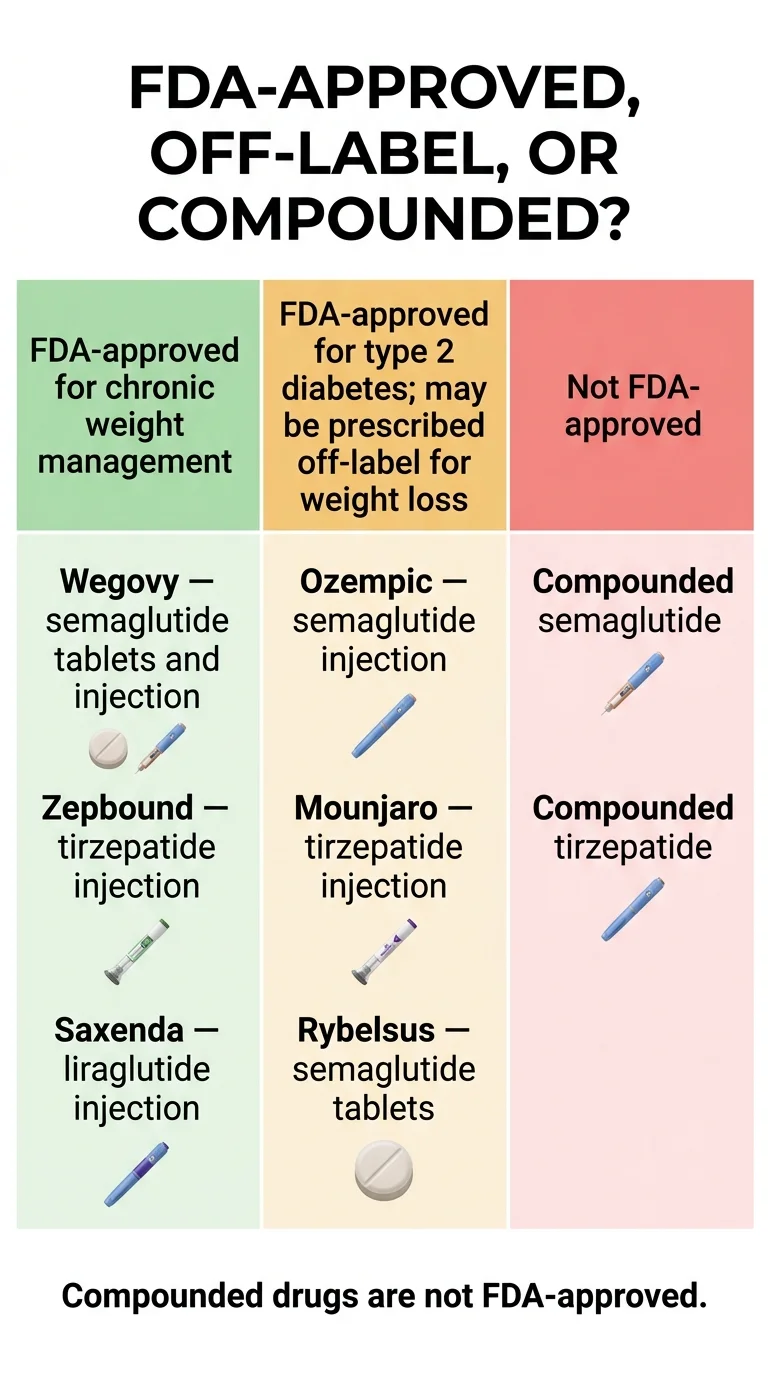
The 503A vs 503B distinction
| Pathway | Who it covers | FDA-approved? |
|---|---|---|
| 503A | Traditional pharmacy compounding for a specific patient with a specific prescription | No |
| 503B (outsourcing facility) | Larger facilities that compound in bulk for hospitals and clinics | No |
| FDA-approved generic | Has gone through FDA's ANDA review process (example: generic liraglutide, approved August 27, 2025) | Yes |
The compounding timeline that changed everything (2024–2026)
| Date | What happened |
|---|---|
| Dec 2024 | FDA declared the tirzepatide shortage resolved. |
| Feb 2025 | FDA declared the semaglutide shortage resolved. |
| Mar 19 / May 22, 2025 | FDA's enforcement-discretion grace periods for 503B compounding of tirzepatide and semaglutide ended. |
| Apr 24, 2025 | Federal court denied the Outsourcing Facilities Association's preliminary injunction motion, letting FDA's enforcement deadlines stand. |
| Aug 27, 2025 | FDA approved the first generic version of liraglutide (Saxenda equivalent). Teva launched it August 28, 2025. |
| Sep 2025 | FDA issued warning letters targeting misleading GLP-1 marketing -- including "generic," "same as," and "same active ingredient" claims by compounders. |
| Dec 2025 | FDA approved Wegovy oral tablet 25 mg for chronic weight management and CV risk reduction. |
| Mar 19, 2026 | FDA approved Wegovy HD (semaglutide 7.2 mg) for adults who tolerated the 2.4 mg dose and need more weight loss. |
| Apr 1, 2026 | FDA approved Foundayo (orforglipron) -- first non-peptide oral GLP-1, first approval under the National Priority Voucher Program. |
| Apr 30, 2026 | FDA proposed to permanently exclude semaglutide, tirzepatide, and liraglutide from the 503B Bulks List. Comment period closes June 29, 2026. |
Table 3: The 503A/503B Compounded GLP-1 Picture (as of May 13, 2026)
| Active ingredient | Drug shortage status | 503B bulk compounding | 503A patient-specific compounding |
|---|---|---|---|
| Semaglutide | Resolved February 2025 | Wind-down ended May 22, 2025; FDA's April 30, 2026 proposal would permanently exclude semaglutide from the 503B Bulks List | Permitted only for a documented patient-specific clinical need (e.g., allergy to an inactive ingredient). Not for cost or convenience. |
| Tirzepatide | Resolved December 2024 | Wind-down ended March 19, 2025; same April 30, 2026 proposal applies | Same narrow 503A pathway as semaglutide |
| Liraglutide injection | Verify against the FDA Drug Shortages database for current status | Compoundable by 503B facilities while a shortage is in effect; April 30, 2026 proposal would exclude from bulks list once shortage resolves | Permitted under standard 503A rules |
Language the FDA specifically called out in 2025 warning letters:
- “Compounded semaglutide contains the same active ingredient as Ozempic and Wegovy.”
- “Compounded tirzepatide is a generic version of Mounjaro/Zepbound.”
- “Our compounded GLP-1 is an FDA-equivalent alternative to FDA-approved options.”
The FDA flagged these as false or misleading. A compounded product and an FDA-approved drug are different things even when the molecule looks similar on paper.
How does FDA indication status affect insurance and prior authorization?
Quick answer
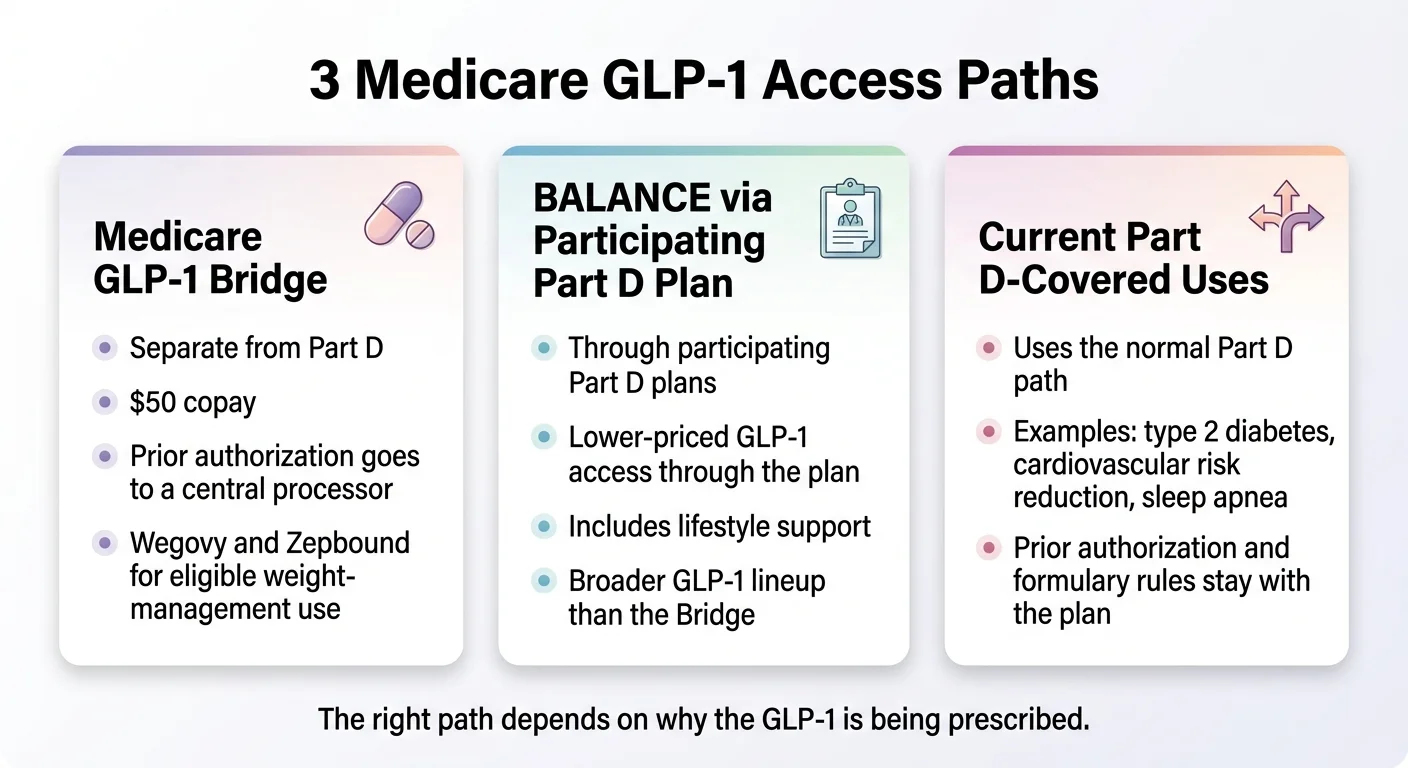
Insurance plans often use the FDA-approved indication as a major coverage gate, but coverage also depends on the plan's formulary, exclusions, prior authorization rules, diagnosis code, step therapy, and quantity limits. Medicare Part D has historically excluded weight-loss drugs, but the FDA's MACE-reduction (March 2024) and MASH (August 2025) approvals for Wegovy created Medicare-coverable indications. The CMS Medicare GLP-1 Bridge program is scheduled to launch July 1, 2026 through December 31, 2027, providing eligible Part D beneficiaries access to certain weight-loss GLP-1s at approximately $50 per month.
A prior-authorization checklist that actually works
| Ask this | Why it matters |
|---|---|
| Is the denial because of my diagnosis code? | Diagnosis-code mismatches are a common denial reason and one of the first things to check. |
| Is this product excluded for weight loss in my plan? | Some plans carve out obesity drugs entirely. An appeal usually cannot override a plan-wide exclusion unless the plan has an exception path. |
| Does the plan require BMI or comorbidity documentation? | Weight-management labels and plan rules often require it in writing. |
| Is there a preferred GLP-1 on my formulary? | The plan may cover Zepbound but not Wegovy, or vice versa. |
| Is this denial because the use is off-label? | If yes, the appeal needs evidence -- typically compendia citations. |
| Is there a step-therapy requirement? | You may need to try a less expensive option first. |
| What's the formal appeal process and timeline? | Denial isn't always the end of the road. |
Scripts for calling your insurance company and pharmacy
“I'm calling about a denial for [drug name] prescribed for [condition]. Can you tell me the specific denial reason, the covered diagnosis criteria for this drug under my plan, whether the denial was due to off-label use, whether a different GLP-1 is preferred on my formulary for my diagnosis, and what the appeal process is — including the deadline?”
“Can you confirm the NDC and product submitted on my claim, whether the claim rejected for prior authorization, a plan exclusion, a quantity limit, or a non-covered diagnosis, and whether the insurer listed a preferred alternative in the rejection message?”
What to ask your clinician before any off-label GLP-1
The FDA itself suggests that patients asked to take a drug off-label should ask several specific questions. Here they are, tightened for a real appointment.
Is the drug you're prescribing FDA-approved for my specific condition and dose?
If it's off-label, what evidence supports this use, and what are the FDA-approved alternatives?
Will my insurance likely cover this prescription, and if not, what's the cash price?
Is this an FDA-approved finished drug or a compounded product? If compounded, what medical need cannot be met by an FDA-approved drug, which state-licensed pharmacy will fill it, and what quality safeguards apply?
How will you monitor for side effects, especially for off-label use?
What do we do if I can't tolerate the dose or it stops working?
Are there clinical trials I might qualify for instead?
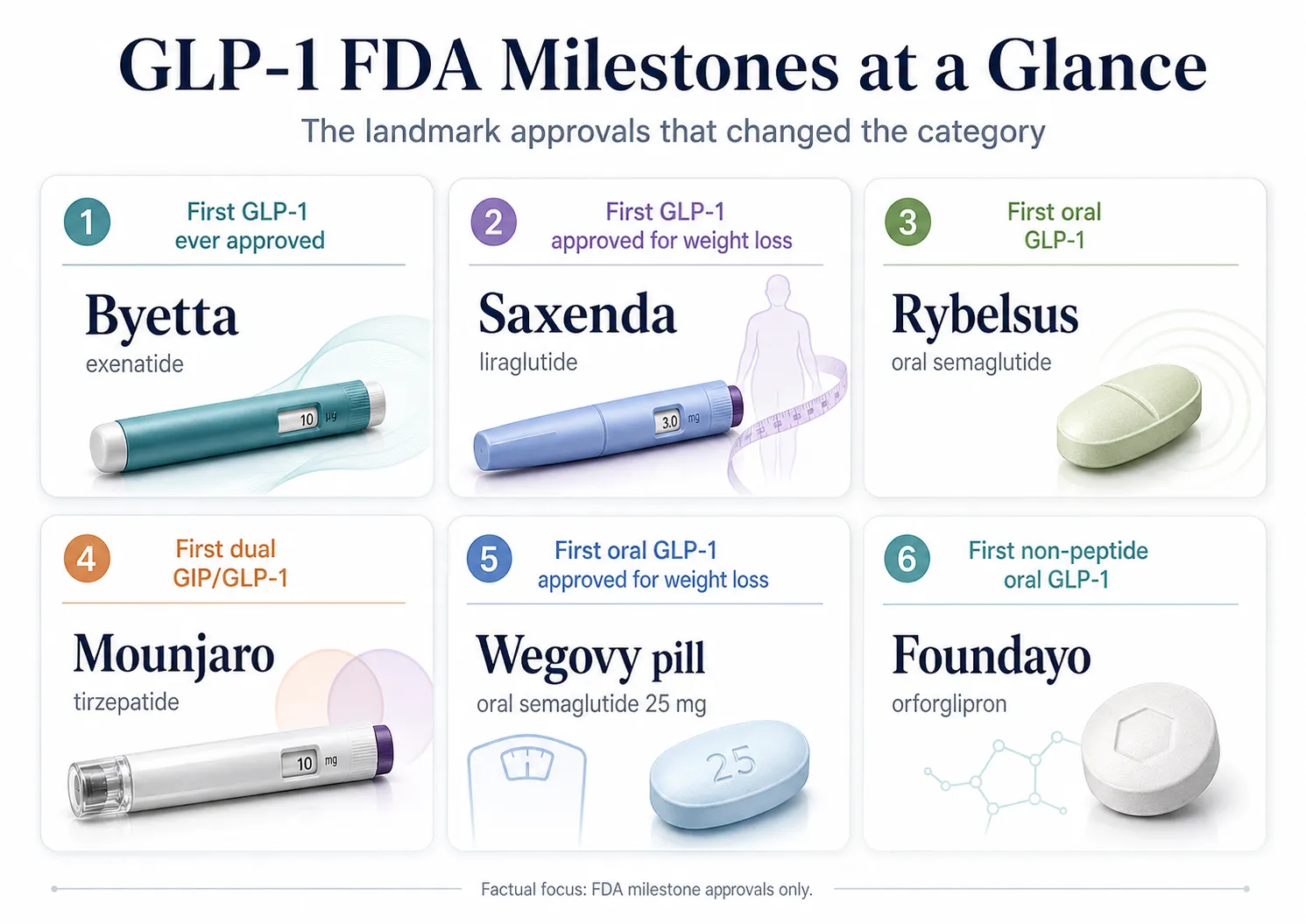
What changed in GLP-1 land in 2024–2026 (the short version)
If you read a guide on this topic from before late 2024, it's probably wrong in at least two places.
| Date | What changed |
|---|---|
| Dec 20, 2024 | FDA approved Zepbound for moderate-to-severe obstructive sleep apnea in adults with obesity. First prescription drug for OSA. |
| Jan 28, 2025 | FDA approved Ozempic to reduce risk of sustained eGFR decline, end-stage kidney disease, and cardiovascular death in adults with T2D and CKD. Based on the FLOW trial. |
| Aug 15, 2025 | FDA granted accelerated approval to Wegovy injection 2.4 mg for noncirrhotic MASH with moderate-to-advanced fibrosis. Based on Part 1 of the ESSENCE trial. |
| Aug 27, 2025 | FDA approved the first generic liraglutide (Saxenda equivalent). Teva launched it August 28, 2025. |
| Sep 2025 | FDA issued warning letters targeting misleading GLP-1 marketing -- including 'generic,' 'same as,' and 'same active ingredient' claims. |
| Dec 2025 | FDA approved Wegovy oral tablet 25 mg for chronic weight management and CV risk reduction in adults with obesity/overweight plus established CVD. Based on the OASIS 4 trial. |
| Mar 19, 2026 | FDA approved Wegovy HD (semaglutide 7.2 mg injection) for chronic weight management in adults who tolerated the 2.4 mg maintenance dose. STEP UP trial showed 20.7% mean weight loss at 72 weeks. |
| Apr 1, 2026 | FDA approved Foundayo (orforglipron) for chronic weight management in adults. First non-peptide oral GLP-1. First new molecular entity under the National Priority Voucher Program. |
| Apr 30, 2026 | FDA proposed to permanently exclude semaglutide, tirzepatide, and liraglutide from the 503B Bulks List. Comment period closes June 29, 2026. |
Last verified: May 13, 2026. Our “Last verified” date for this page is May 13, 2026. If you are reading this and the date is more than a few months ago, check the current FDA labels before relying on it.
Find your on-label GLP-1 path
Our 60-second quiz matches you with FDA-approved GLP-1 options that fit your diagnosis, coverage, and situation — before you spend time on a prescription that will get denied.
→ Find My GLP-1 PathHow to verify a GLP-1's FDA approval yourself
This page should make you self-sufficient. Here is the five-step process used to update the matrix.
What we verified
Checked against primary sources on May 13, 2026:
- FDA's official definition of off-label use and “FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss” page
- FDA's compounding policy clarifications, resolved shortage announcements, and April 30, 2026 proposal to exclude semaglutide, tirzepatide, and liraglutide from the 503B Bulks List
- DailyMed prescribing information (Section 1, Indications and Usage) for all 16 GLP-1 products in the matrix
- CMS Medicare GLP-1 Bridge program page, including eligible products, $50 copay, and exclusion rules
- FDA press announcements for each approval: Zepbound OSA, Ozempic CKD, Wegovy MASH, generic liraglutide, Wegovy oral 25 mg, Wegovy HD, Foundayo
- 2026 BMJ cohort study of GLP-1 use and substance use disorder outcomes in U.S. veterans with type 2 diabetes
- Truveta Research's 2025 analysis of GLP-1 prescribing trends in patients with PCOS
Source hierarchy:
Regulatory facts come from FDA, DailyMed, and CMS first. Manufacturer press releases were used only as secondary confirmation of approval dates. Reddit, telehealth provider marketing pages, and compounded-product vendor websites were not used as evidence.
Sources
FDA — definitions and warnings
- • FDA, “Understanding Unapproved Use of Approved Drugs ‘Off-Label’” — fda.gov
- • FDA, “FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss” — fda.gov
- • FDA, “FDA Clarifies Policies for Compounders as National GLP-1 Supply Begins to Stabilize” — fda.gov
- • FDA, “FDA Intends to Take Action Against Non-FDA-Approved GLP-1 Drugs” — fda.gov
- • FDA, “FDA Proposes to Exclude Semaglutide, Tirzepatide, and Liraglutide on 503B Bulks List” (April 30, 2026) — fda.gov
FDA — recent approvals
- • Zepbound for OSA (December 20, 2024)
- • Ozempic for CKD/CV death in T2D + CKD (January 28, 2025)
- • Wegovy for noncirrhotic MASH with moderate-to-advanced fibrosis (August 15, 2025)
- • First generic liraglutide (August 27, 2025)
- • Wegovy oral 25 mg tablet (December 2025)
- • Wegovy HD 7.2 mg (March 19, 2026)
- • Foundayo (orforglipron) (April 1, 2026)
DailyMed prescribing information (Section 1)
- • Ozempic, Rybelsus, Wegovy, Mounjaro, Zepbound
- • Trulicity, Victoza, Saxenda, Byetta, Bydureon BCise
- • Adlyxin, Soliqua 100/33, Xultophy 100/3.6
- • Foundayo (orforglipron)
Other authoritative sources
- • CMS, “Medicare GLP-1 Bridge” program — cms.gov
- • AHRQ, “Off-Label Drugs: What You Need to Know” — ahrq.gov
- • BMJ, GLP-1 receptor agonists and risk of substance use disorders in U.S. veterans with T2D (2026 cohort study)
- • Truveta Research, “Rising use of GLP-1 medications among women with PCOS”
- • American Diabetes Association, Standards of Medical Care in Diabetes
Frequently asked questions
Is it legal for a doctor to prescribe Ozempic for weight loss?
Yes. Off-label prescribing of FDA-approved drugs is legal in the United States. Ozempic is FDA-approved for type 2 diabetes, MACE reduction in T2D plus established CVD, and kidney-risk reduction in T2D plus CKD. Using it for weight loss without a T2D diagnosis is off-label but not illegal.
Is Mounjaro FDA-approved for weight loss?
No. Mounjaro is FDA-approved to improve glycemic control in adults and pediatric patients 10 years of age and older with type 2 diabetes. Zepbound is the tirzepatide product FDA-approved for chronic weight management (November 2023) and for moderate-to-severe OSA in adults with obesity (December 20, 2024).
Are Wegovy and Ozempic the same drug?
They contain the same active ingredient (semaglutide), but they are not the same drug in the eyes of the FDA. Wegovy is FDA-approved for weight management at higher doses, plus CV risk reduction in certain adults and noncirrhotic MASH with moderate-to-advanced fibrosis. Ozempic is FDA-approved for type 2 diabetes at lower doses, plus cardiovascular and kidney indications in that population. The labels, dose ranges, and insurance coverage rules differ.
Is Zepbound the same as Mounjaro?
Both contain tirzepatide, but they are not interchangeable from a regulatory perspective. Mounjaro is FDA-approved for type 2 diabetes in adults and pediatric patients 10 years and older. Zepbound is FDA-approved for chronic weight management and for OSA in adults with obesity. Insurance plans typically treat them as different drugs.
Will insurance cover off-label GLP-1 use?
Sometimes, but not by default. Most plans deny off-label coverage unless the use is supported by recognized drug compendia (specialty references like DRUGDEX, AHFS DI, or NCCN) or unless an exception is granted on appeal. The most common path is to switch to the FDA-approved product for your specific use.
Are compounded GLP-1 medications FDA-approved?
No. Compounded drugs are not FDA-approved. The FDA does not review them for safety, effectiveness, or quality before they are marketed. The FDA says compounded drugs should only be used when an FDA-approved drug cannot meet the patient's medical need. As of mid-2026, federal enforcement has narrowed the legal pathways for large-scale compounding of semaglutide, tirzepatide, and liraglutide.
Are compounded GLP-1s FDA-approved generics?
No. A generic drug is an FDA-approved finished product that has gone through the FDA's review process. A compounded drug is a custom mixture prepared by a compounding pharmacy that has not. The FDA explicitly says companies cannot lawfully claim non-FDA-approved compounded GLP-1 products are generic versions of, the same as, or use the same active ingredient as FDA-approved drugs. The first FDA-approved generic liraglutide (Saxenda equivalent) was approved on August 27, 2025 -- that is a generic. Compounded semaglutide and tirzepatide are not.
Is compounded semaglutide the same as Ozempic or Wegovy?
No. Compounded semaglutide products are not FDA-approved drugs and are not equivalent to Ozempic or Wegovy. The FDA called out claims of equivalence in its 2025 warning letters as false or misleading.
Is retatrutide FDA-approved?
No. Retatrutide is an investigational drug in clinical trials. It is not FDA-approved and cannot legally be used in compounding under federal law. Any product sold as 'compounded retatrutide' is not a legitimate compounded medication.
Are GLP-1s FDA-approved for PCOS?
No. No GLP-1 is currently FDA-approved for polycystic ovary syndrome. Clinicians sometimes prescribe GLP-1s off-label for PCOS patients with obesity or insulin resistance based on growing evidence. In many cases, the prescription is on-label for a metabolic indication the patient also has -- such as type 2 diabetes or chronic weight management -- even when the goal is to address PCOS symptoms.
Are GLP-1s FDA-approved for prediabetes?
No. No GLP-1 is FDA-approved for prediabetes alone. Prescribing a GLP-1 for prediabetes without a qualifying T2D or weight-management indication is off-label.
Are GLP-1s FDA-approved for type 1 diabetes?
No. No GLP-1 is FDA-approved for type 1 diabetes. Use in T1D is off-label and typically managed by an endocrinologist with attention to hypoglycemia risk and insulin dose adjustment.
Are GLP-1s FDA-approved for binge-eating disorder or food noise?
No. No GLP-1 is FDA-approved for binge-eating disorder or for healthy-BMI patients with food noise. Lisdexamfetamine -- sold as Vyvanse and FDA-approved generics -- is FDA-approved for moderate-to-severe binge-eating disorder in adults. GLP-1s are not.
Are GLP-1s FDA-approved for alcohol use disorder?
No. No GLP-1 is FDA-approved for alcohol use disorder or substance use disorder. A 2026 BMJ cohort study of U.S. veterans with type 2 diabetes reported a 14% lower composite risk of substance use disorders among GLP-1 users compared with non-users -- a signal in observational data, not an FDA indication.
What GLP-1 is FDA-approved for sleep apnea?
Zepbound (tirzepatide) is the only GLP-1 currently FDA-approved for moderate-to-severe obstructive sleep apnea in adults with obesity. The approval came on December 20, 2024.
What GLP-1 is FDA-approved for liver disease?
Wegovy (semaglutide 2.4 mg injection) received accelerated FDA approval on August 15, 2025 for adults with noncirrhotic metabolic dysfunction-associated steatohepatitis (MASH) with moderate-to-advanced liver fibrosis. It is not approved for earlier-stage or unstaged fatty liver disease.
How do I verify a GLP-1's FDA-approved indication myself?
Open DailyMed (dailymed.nlm.nih.gov), search the brand name, open the current label, and read Section 1 (Indications and Usage). Cross-check recent approvals on Drugs@FDA or FDA press announcements.
What's the difference between off-label and compounded?
Off-label use is when an FDA-approved drug is prescribed for a condition or population not on its label. The drug itself is still FDA-approved and regulated. Compounded means a pharmacy prepared a custom mixture that is not an FDA-approved finished drug and has not been reviewed by the FDA for safety, effectiveness, or quality.
Does FDA approval mean the drug is right for me?
No. FDA approval means the drug was reviewed for safety and effectiveness in the approved population, dose, and condition. Whether it's the right choice for your specific situation depends on your medical history, your goals, your other medications, and the conversation with your clinician.
Does the Medicare GLP-1 Bridge cover weight-loss GLP-1s?
Starting July 1, 2026 through December 31, 2027, the CMS Medicare GLP-1 Bridge program will provide eligible Medicare Part D beneficiaries access to certain weight-loss GLP-1 medications -- including all Foundayo formulations, all Wegovy formulations, and the Zepbound KwikPen formulation only -- at approximately $50 per month. The Bridge operates outside the Part D benefit's normal coverage and payment flow; the $50 copay does not count toward Part D true out-of-pocket costs, and beneficiaries already covered for the same drug under basic Part D do not qualify for Bridge coverage for that drug.
What should I do if my GLP-1 prescription is off-label and my insurance denied it?
Call your insurance company and ask for the exact denial reason, the covered diagnosis criteria, and the appeal process. Call your pharmacy and confirm the product submitted and the rejection code. Talk to your prescriber about whether an FDA-approved alternative for your specific use exists and whether your situation supports a formal appeal.
Related guides on The RX Index
- Ozempic vs Wegovy: Full Side-by-Side Comparison
- How to Switch from Mounjaro to Zepbound (2026 Guide)
- Compounded GLP-1 Alternatives 2026: Verified Prices and Paths
- Best GLP-1 Providers That Accept Insurance (2026)
- Best GLP-1 Providers That Help With Prior Authorization
- GLP-1 Contraindication vs Warning vs Precaution: FDA Label Explained
- Find My GLP-1 Path — 60-Second Matching Quiz
Know your label. Get the right prescription.
Our quiz takes 60 seconds and matches you to the FDA-approved GLP-1 path that fits your diagnosis, coverage, and budget — without the off-label denial risk.
→ Find My GLP-1 PathThis page is published by The RX Index editorial team. It is for informational and regulatory-classification purposes only. It is not medical advice. Talk to a licensed clinician before starting, stopping, or changing any medication. Methodology · Editorial Standards
Last verified: May 13, 2026. This page is reviewed at least monthly and immediately whenever the FDA, CMS, or DailyMed updates a GLP-1 label or coverage rule.