GLP-1 Contraindication vs Warning vs Precaution: What the FDA Label Actually Means in 2026
Published: · Last reviewed:
By The RX Index Editorial Team · Updated quarterly and when the FDA issues a labeling change

What this page is. This page translates FDA label categories. It does not clear you to start, stop, pause, restart, or switch a GLP-1 medication. That conversation belongs with your prescriber.
If you just searched GLP-1 contraindication vs warning vs precaution because you read a Wegovy, Mounjaro, or Foundayo label and saw all three words in one place — they mean very different things. Mixing them up is the single biggest reason people either panic and stop a medicine they could safely take, or start one when they shouldn't.
Here is the short version. A GLP-1 contraindication is the FDA's “do not use” label entry. A warning or precaution is a real risk that needs monitoring, counseling, dose changes, or a stopping plan — but it does not automatically block treatment. A boxed warning is the FDA's loudest format; it sits at the top of the label in a bordered box and almost always points back to a Section 4 contraindication and a Section 5 warning. They are three different things even when they discuss the same underlying risk.
The 5 GLP-1 label categories at a glance
Answer capsule
A modern FDA prescribing information label has five safety-relevant categories: the Boxed Warning, Section 4 (Contraindications), Section 5 (Warnings and Precautions), Limitations of Use, and Section 8 (Use in Specific Populations). Section 4 is the true contraindications list. Everything else describes serious risk, limits of use, or population-specific issues that can still change a prescribing decision.
| Label category | What it legally means | What your prescriber does | A GLP-1 example |
|---|---|---|---|
| Boxed Warning | The FDA's most visible warning format — a bordered box at the top of the label for the most serious risks | Reads first; cross-references the related contraindication and warnings sections | Risk of thyroid C-cell tumors (every GLP-1 reviewed here except short-acting exenatide and lixisenatide) |
| Contraindication (Section 4) | “The drug should not be used because the risk clearly outweighs any possible benefit” | Does not prescribe | Personal or family history of medullary thyroid carcinoma (MTC) |
| Warning / Precaution (Section 5) | A serious risk with implications for prescribing, monitoring, dose changes, or stopping | Prescribes with monitoring, dose adjustment, or counseling | Acute pancreatitis — discontinue if suspected |
| Limitations of Use / “Not recommended” | Strong label advice short of a contraindication; drug was not studied here, or risk may be higher | Considers alternatives; may avoid the drug | Severe gastroparesis — “not recommended” for several GLP-1s |
| Use in Specific Populations (Section 8) | Pregnancy, lactation, kidney impairment, pediatrics, older adults | Reads product-by-product; some trigger a discontinuation discussion | Pregnancy is in Section 8 for every GLP-1 reviewed here |
What “contraindicated” actually means on a GLP-1 label
Answer capsule
A contraindication is the FDA's strongest “do not use” designation, defined under 21 CFR 201.57(c)(5). The FDA's October 2011 guidance states that “a drug should be contraindicated only in those clinical situations for which the risk from use clearly outweighs any possible therapeutic benefit” and that “only known hazards, and not theoretical possibilities, can be the basis for a contraindication.” For every currently marketed GLP-1 in the U.S., the Section 4 list is short: usually two or three items, never ten.
FDA verbatim, October 2011 Guidance for Industry
“A drug should be contraindicated only in those clinical situations for which the risk from use clearly outweighs any possible therapeutic benefit. Only known hazards, and not theoretical possibilities, can be the basis for a contraindication.”
— FDA Guidance for Industry, citing 21 CFR 201.57(c)(5)
Three things follow from that definition that most consumer-facing pages skip:
- 1The list has to be short. Section 4 must be a tight bulleted list of true do-not-use situations. If a drug has no known contraindications, the section must literally read "None." This is why every GLP-1 you look up has a Section 4 with only two or three lines, not ten.
- 2The bar is high. Theoretical risks cannot live here. Only known hazards make the cut. That is exactly why pregnancy lives in Section 8 (Use in Specific Populations) for every current GLP-1 -- the human data are limited, and the FDA chose not to put it in Section 4.
- 3The language is locked. The FDA's guidance is explicit that "do not use" and "[drug] is contraindicated in..." wording belongs in Section 4 and should not appear in Section 5. That is the cleanest legal-language test you can apply when reading any label.
Why “warning” and “precaution” are one section now
Answer capsule
“Warning” and “precaution” are not two separate FDA label categories anymore. They were merged into a single section titled WARNINGS AND PRECAUTIONS under the 2006 Physician Labeling Rule (PLR), governed by 21 CFR 201.57(c)(6). Every currently approved GLP-1 medication uses this modern format. The historical distinction — warning more serious than precaution — still lives in clinical conversation but is no longer how the regulatory label is structured.
Pre-2006 FDA labels had three separate sections: WARNINGS (more serious) and PRECAUTIONS (less serious) sat below CONTRAINDICATIONS. In 2006, the FDA rolled out the Physician Labeling Rule (PLR), which merged Warnings and Precautions into one Section 5 — with sub-headings (5.1, 5.2, 5.3…) for each individual risk. Every GLP-1 currently on the U.S. market uses this modern format. None of them have a separate “Precautions” section.
FDA verbatim — what belongs in Section 5
“The WARNINGS AND PRECAUTIONS section is intended to identify and describe a discrete set of adverse reactions and other potential safety hazards that are serious or are otherwise clinically significant because they have implications for prescribing decisions or for patient management. To include an adverse event in the section, there should be reasonable evidence of a causal association between the drug and the adverse event, but a causal relationship need not have been definitively established.”
— FDA Guidance for Industry, October 2011, citing 21 CFR 201.57(c)(6)
The practical reading rule: If you are looking at a current GLP-1 label and somebody calls something a “precaution,” they are using older clinical shorthand. In the actual FDA document, everything in Section 5 carries the same regulatory weight. A “precaution” is a “warning” is a Section 5 risk.
The boxed warning is a format, not a fourth category
Answer capsule
A boxed warning — sometimes called a “black box warning” — is the FDA's most visually prominent labeling format under 21 CFR 201.57(c)(1). It appears at the top of the prescribing information in a bordered box. It is not a separate risk category; it points to risks that already live in Section 4, Section 5, or both. For most GLP-1s, the same risk (thyroid C-cell tumors) appears in three places in the label — boxed warning, Section 4 contraindication, and Section 5.1 warning — because it is serious enough to warrant all three formats.
The FDA reserves the boxed warning for three situations:
- 1A risk so serious in proportion to the benefit that prescribers must consider it for every patient. The thyroid C-cell tumor finding in rodent studies is the GLP-1 example.
- 2A serious risk that can be prevented or reduced by specific prescriber action — patient selection, monitoring, dose changes, avoidance of certain other drugs.
- 3An FDA approval issued with use restrictions — the drug can only be safely used inside a managed-access program.
Key point: The Wegovy boxed warning cross-refers to two other label parts — the Section 4 contraindication for MTC/MEN 2, and the Section 5.1 discussion of thyroid C-cell tumors. You are not seeing three separate risks. You are seeing the same risk highlighted three times, in three label formats.
Which GLP-1s carry the thyroid C-cell boxed warning?
| Product (current U.S. label) | Boxed warning for thyroid C-cell tumors? | Section 4 MTC/MEN 2 contraindication? | Source |
|---|---|---|---|
| Wegovy injection / Wegovy HD / Wegovy tablets (semaglutide) | Yes | Yes | DailyMed; accessdata.fda.gov |
| Ozempic injection (semaglutide) | Yes | Yes | DailyMed |
| Rybelsus / Ozempic tablets (oral semaglutide) | Yes | Yes | DailyMed; FDA approval letter 2026 |
| Mounjaro (tirzepatide) | Yes | Yes | DailyMed |
| Zepbound (tirzepatide) | Yes | Yes | DailyMed |
| Saxenda (liraglutide) | Yes | Yes | DailyMed; label revised 05/2025 and 02/2026 |
| Victoza (liraglutide) | Yes | Yes | DailyMed |
| Trulicity (dulaglutide) | Yes | Yes | DailyMed |
| Bydureon BCise (exenatide ER) | Yes | Yes | DailyMed |
| Byetta (exenatide IR) | No | No (hypersensitivity contraindication only) | DailyMed |
| Adlyxin (lixisenatide) | No | No (hypersensitivity contraindication only) | DailyMed; accessdata.fda.gov 2026 PDF |
| Soliqua (insulin glargine + lixisenatide) | No | No MTC/MEN 2 entry; hypoglycemia + hypersensitivity contraindications | DailyMed |
| Xultophy (insulin degludec + liraglutide) | Yes (from liraglutide) | Yes | DailyMed |
| Foundayo (orforglipron) | Yes (despite no rodent C-cell tumors with orforglipron specifically) | Yes | DailyMed; FDA approval letter 04/2026 |
The Foundayo entry is the most interesting current example. Orforglipron is not pharmacologically active in rats or mice and did not produce thyroid C-cell tumors in rodent studies — but it is pharmacologically active at the human GLP-1 receptor, and the label carries the MTC/MEN 2 contraindication and boxed warning due to class-level uncertainty. Byetta and Adlyxin show the other side: the thyroid C-cell boxed warning is not class-mandatory.
Can I take a GLP-1 with a family history of thyroid cancer?
Answer capsule
It depends on the type. GLP-1 labels specifically name medullary thyroid carcinoma (MTC) and MEN 2 as the Section 4 contraindication. Other thyroid cancer types — including the most common ones — are not the named contraindication. The American Thyroid Association lists papillary thyroid cancer at roughly 70–80% of all thyroid cancers, follicular at 10–15%, medullary at about 2%, and anaplastic at less than 2%. If your family history is unclear, confirm the pathology before treating GLP-1 eligibility as a yes/no answer.
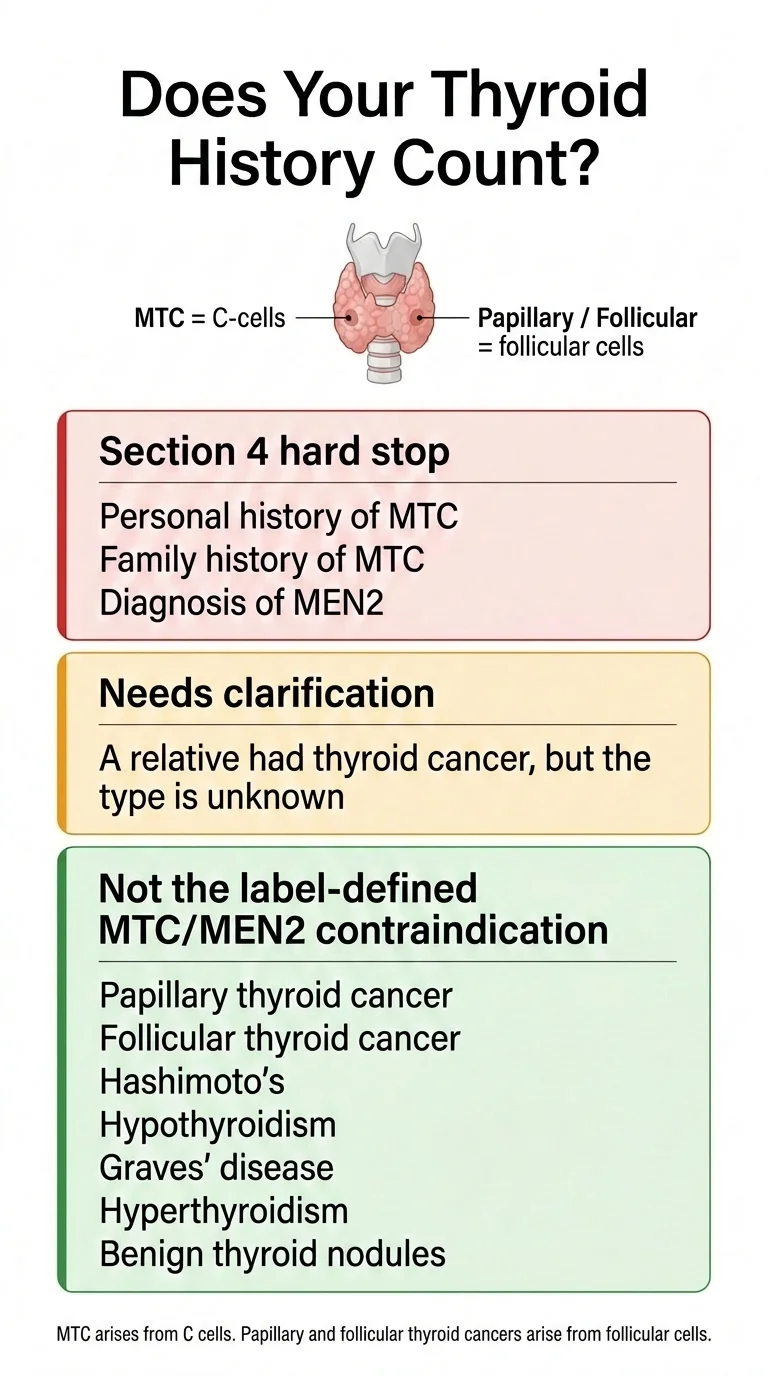
The Section 4 contraindication on every GLP-1 label that carries it names two specific things: personal or family history of medullary thyroid carcinoma (MTC), and Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). MTC is biologically distinct from the more common thyroid cancers (papillary, follicular, oncocytic/Hürthle cell), which start in follicular cells — not C-cells.
- •Confirm the type before assuming. If the affected family member is alive, ask about the pathology. If they aren't, medical records or oncology summaries may identify the subtype.
- •MTC family history is Section 4 territory. If the family history is MTC or includes MEN 2, every GLP-1 with the MTC/MEN 2 contraindication is off the table per label.
- •The degree of relation isn't spelled out in labels. Labels say "personal or family history of MTC." Whether a grandparent, aunt, or uncle's MTC counts the same as a parent or sibling's is a clinical interpretation. Bring the family history to the prescriber.
- •Hashimoto's, hypothyroidism, and benign thyroid nodules are not MTC. They are different conditions and are not the labeled contraindication.
GLP-1 contraindication vs warning vs precaution — every label, side-by-side
Answer capsule
Across the U.S. FDA labels reviewed for this guide, the most common Section 4 contraindications are personal or family history of MTC or MEN 2, and serious hypersensitivity to the drug or its excipients. Product-specific exceptions matter: Soliqua and Xultophy add hypoglycemia episodes; exenatide products add prior exenatide-induced immune-mediated thrombocytopenia; Byetta, Adlyxin, and Soliqua do not carry the thyroid C-cell boxed warning.
Every entry below was verified against the current U.S. FDA label. The “label revised” date is the date shown on the label source; verified May 13, 2026.
Section 4 contraindications by product (verified May 13, 2026)
| Product (active ingredient) | Class | Boxed warning | Section 4 Contraindications | Source / label revised |
|---|---|---|---|---|
| Ozempic injection (semaglutide) | GLP-1 RA | Yes | MTC personal/family history; MEN 2; known serious hypersensitivity to semaglutide or excipients | DailyMed — current label |
| Wegovy injection / Wegovy HD / Wegovy tablets (semaglutide) | GLP-1 RA | Yes | MTC personal/family history; MEN 2; known serious hypersensitivity to semaglutide or any excipient in the formulation taken | DailyMed; accessdata.fda.gov 2026 PDF |
| Rybelsus / Ozempic tablets (oral semaglutide) | GLP-1 RA | Yes | MTC personal/family history; MEN 2; prior serious hypersensitivity to semaglutide or excipients | DailyMed combined oral semaglutide label record; FDA approval letter 2026 |
| Mounjaro (tirzepatide) | Dual GIP/GLP-1 RA | Yes | MTC personal/family history; MEN 2; known serious hypersensitivity to tirzepatide or excipients | DailyMed — current label |
| Zepbound (tirzepatide) | Dual GIP/GLP-1 RA | Yes | MTC personal/family history; MEN 2; known serious hypersensitivity to tirzepatide or excipients | DailyMed — current label |
| Saxenda (liraglutide) | GLP-1 RA | Yes | MTC personal/family history; MEN 2; hypersensitivity to liraglutide or product components. (Pregnancy removed from Section 4 in May 2025.) | DailyMed — label revised 05/2025; SI/B removed 02/2026 |
| Victoza (liraglutide) | GLP-1 RA | Yes | MTC personal/family history; MEN 2; known serious hypersensitivity to liraglutide or excipients | DailyMed — current label |
| Trulicity (dulaglutide) | GLP-1 RA | Yes | MTC personal/family history; MEN 2; known serious hypersensitivity to dulaglutide or excipients | DailyMed — current label |
| Bydureon BCise (exenatide ER) | GLP-1 RA | Yes | MTC personal/family history; MEN 2; known serious hypersensitivity to exenatide; history of drug-induced immune-mediated thrombocytopenia from exenatide | DailyMed — current label |
| Byetta (exenatide IR) | GLP-1 RA | No | Known serious hypersensitivity to exenatide or excipients; history of drug-induced immune-mediated thrombocytopenia from exenatide | DailyMed — current label |
| Adlyxin (lixisenatide) | GLP-1 RA | No | Known serious hypersensitivity to lixisenatide or excipients | DailyMed; accessdata.fda.gov 2026 PDF |
| Soliqua (insulin glargine + lixisenatide) | Combo | No | Episodes of hypoglycemia; known serious hypersensitivity to insulin glargine, lixisenatide, or excipients | DailyMed — current label |
| Xultophy (insulin degludec + liraglutide) | Combo | Yes (liraglutide component) | MTC personal/family history; MEN 2; episodes of hypoglycemia; known serious hypersensitivity to insulin degludec, liraglutide, or excipients | DailyMed — current label |
| Foundayo (orforglipron, oral) | GLP-1 RA | Yes (despite no rodent C-cell tumors with orforglipron specifically) | MTC personal/family history; MEN 2; known serious hypersensitivity to orforglipron or excipients | DailyMed; FDA approval letter 04/2026 |
Section 5 (Warnings and Precautions) — the shared backbone with product variation

Section 5 has a recurring backbone, but it is not identical product to product. The risks below appear in different combinations depending on the product:
| Section 5 Risk | Products where it appears | Key label instruction |
|---|---|---|
| Thyroid C-cell tumors | Every product that carries the boxed warning | Cross-references Section 4 and the boxed warning |
| Acute pancreatitis | Every GLP-1 reviewed here | Discontinue if suspected; do not restart if confirmed |
| Acute gallbladder disease (cholelithiasis, cholecystitis) | Most semaglutide, tirzepatide, liraglutide, dulaglutide, lixisenatide labels | Evaluate if symptoms develop |
| Hypoglycemia | Every GLP-1 reviewed (concomitant insulin or sulfonylureas) | Dose adjustment may be needed for insulin/secretagogue |
| Acute kidney injury | Every GLP-1 reviewed | Monitor renal function in patients with renal impairment reporting severe GI reactions |
| Hypersensitivity reactions (anaphylaxis, angioedema) | Every GLP-1 reviewed | Separate from Section 4 contraindication; ongoing monitoring required |
| Severe gastrointestinal reactions | Several labels; Saxenda Section 5.7 added 10/2025 | Product-specific monitoring and discontinuation language |
| Diabetic retinopathy complications | Primarily T2DM-labeled products | Monitor patients with retinopathy in T2DM with rapid glycemic improvement |
| Pulmonary aspiration during anesthesia or deep sedation | Multiple labels updated 2024–2026 | Counsel patients; inform procedure team in advance |
| Heart rate increase | Some labels | Follow product-specific monitoring language |
| Suicidal behavior and ideation | Previously Saxenda, Wegovy, Zepbound | Removed from all three in early 2026 following FDA Drug Safety Communication (Jan 13, 2026) |
The 5-second legal-language test
Answer capsule
The fastest way to tell a contraindication apart from a warning or precaution is to read the verb. The FDA's 2011 guidance reserves “do not use” and “[drug] is contraindicated in…” language for Section 4 and says that terminology should not appear in Section 5. Section 5 uses verbs like “use with caution,” “monitor for,” “consider reducing the dose,” and “discontinue if [condition] develops.”
FDA verbatim, October 2011
“Terminology that generally infers a contraindication (e.g., ‘Do not use’ or ‘Drug X should not be used’) should not appear in the WARNINGS AND PRECAUTIONS section.”
| Verb pattern | Section it lives in | A GLP-1 example |
|---|---|---|
| “is contraindicated in…” / “do not use in…” / “must not be administered to…” | Section 4 (Contraindication) | “WEGOVY is contraindicated in patients with a personal or family history of MTC…” |
| “use with caution in…” / “consider…” / “monitor for…” / “discontinue if…” / “instruct patients to…” | Section 5 (Warning / Precaution) | “Discontinue promptly if pancreatitis is suspected.” |
| “has not been studied in…” / “is not recommended in…” | Limitations of Use or Section 5 callout | “ADLYXIN is not recommended in patients with end-stage renal disease.” |
| Bold, bordered, top of label | Boxed Warning format — read the cross-reference | The thyroid C-cell tumor box on Wegovy points to Section 4 and Section 5.1 |
Where the test fails: Telehealth intake forms and third-party summary articles often simplify “discontinue if pancreatitis is suspected” into “history of pancreatitis is a contraindication.” That is not what the label says. If you have been told a Section 5 risk is a hard stop, you have grounds to ask which section of the actual prescribing information that claim comes from.
Is pancreatitis a contraindication for GLP-1 medications?
Answer capsule
No. Acute pancreatitis is a Section 5 warning on every currently marketed GLP-1 receptor agonist, not a Section 4 contraindication. Labels instruct prescribers to discontinue the drug if pancreatitis is suspected and not to restart it if pancreatitis is confirmed. The label is silent on whether a history of pancreatitis prohibits new initiation — which is why this is treated as a discussion item, not an automatic disqualifier.

This is the most common single misclassification — including on intake forms from large telehealth providers. What the labels actually say (wording is nearly identical across semaglutide, tirzepatide, liraglutide, dulaglutide, exenatide, and orforglipron products):
- →Acute pancreatitis has been observed in clinical trials. Some events have been severe.
- →Inform patients of the symptoms -- severe, persistent abdominal pain that may radiate to the back, often with nausea or vomiting.
- →Discontinue the drug promptly if pancreatitis is suspected.
- →Do not restart if pancreatitis is confirmed.
That is a Section 5 warning. It tells the prescriber what to do during therapy, not whether to start. There is no FDA-label rule that a prior episode of pancreatitis is a contraindication.
Clinically, many prescribers do treat a history of pancreatitis as a relative contraindication — that is a judgment call, not a label requirement. If you have been declined for GLP-1 therapy because of a prior pancreatitis episode, you can ask: which section of the prescribing information does this decision come from? Was the prior episode severe, mild, or unknown cause? Is an alternative a better fit, or am I being declined out of caution?
Is gastroparesis a contraindication, warning, or “not recommended”?
Answer capsule
Severe gastroparesis is not a formal Section 4 contraindication for any current GLP-1 medication, but several product labels state the drug is “not recommended” in patients with severe gastroparesis or was not studied in this population. That language is stronger than a simple precaution — it is a clear signal to choose a different therapy or to involve a gastroenterologist before starting.
GLP-1 receptor agonists slow stomach emptying by design — that is part of the mechanism that creates fullness. For someone whose stomach already empties too slowly, adding a drug that slows it further can mean nausea, vomiting, food intolerance, and in severe cases hospitalization.
A short checklist if you have any GI history:
- •Have you been formally diagnosed with gastroparesis?
- •Have you been hospitalized for vomiting or dehydration in the past two years?
- •Do you have severe reflux, frequent food retention, or inability to keep meals down?
- •Do you take other medications affected by stomach emptying -- oral contraceptives, anti-seizure medicines, or certain antibiotics?
- •Do you have an upcoming endoscopy, colonoscopy, or anesthesia procedure?
If any of those apply, the GLP-1 conversation is more nuanced than a yes/no eligibility form can capture. The label has told the prescriber to think twice. Ask why this product, why this dose, and what the monitoring plan looks like.
Is pregnancy a GLP-1 contraindication?
Answer capsule
No. Pregnancy is not a Section 4 contraindication on any of the current U.S. labels reviewed here. Pregnancy is addressed in Section 8 (Use in Specific Populations), but the action language differs by product and indication. The Saxenda Section 4 pregnancy contraindication — which existed from 2014 through May 2025 — was removed in a May 2025 label update.
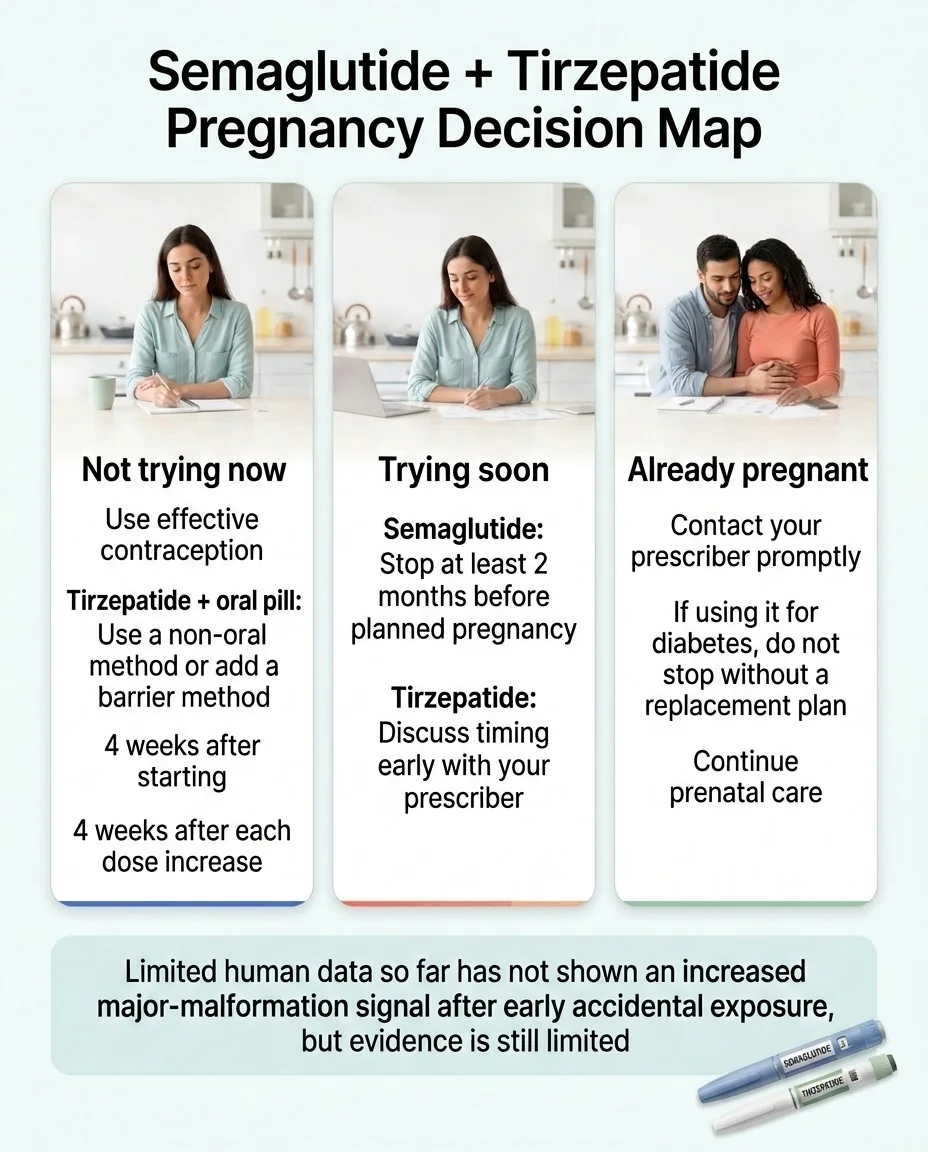
How pregnancy is handled, product by product
| Product | Section 4 lists pregnancy? | Section 8 action language | Planned-pregnancy timing |
|---|---|---|---|
| Saxenda (liraglutide, weight loss) | No (removed 05/2025) | Weight loss offers no benefit to a pregnant patient; may cause fetal harm; discontinue when pregnancy is recognized | Discuss with prescriber |
| Wegovy (semaglutide, weight loss/CV) | No | Discontinue when pregnancy is recognized; advise of fetal risk | Stop at least 2 months before planned pregnancy |
| Ozempic / Rybelsus (semaglutide, diabetes) | No | Limited human data; advise of fetal risk | Stop at least 2 months before planned pregnancy |
| Zepbound (tirzepatide, weight loss) | No | Weight loss offers no benefit to a pregnant patient; may cause fetal harm; discontinue when pregnancy is recognized | Discuss with prescriber |
| Mounjaro (tirzepatide, diabetes) | No | Use only if the potential benefit justifies the potential risk to the fetus (benefit-risk language) | Discuss with prescriber |
| Trulicity (dulaglutide, diabetes) | No | Use only if the potential benefit justifies the potential risk (benefit-risk language) | Discuss with prescriber |
| Byetta / Bydureon BCise (exenatide, diabetes) | No | Product-specific Section 8 wording | Discuss with prescriber |
| Adlyxin (lixisenatide, diabetes) | No | Use only if the potential benefit justifies the potential risk | Discuss with prescriber |
| Foundayo (orforglipron, weight loss) | No | Product-specific Section 8 wording | Discuss with prescriber |
Do GLP-1s affect oral contraceptives?
Some GLP-1 labels include specific instructions. Zepbound tells females using oral contraceptives to switch to a non-oral contraceptive method or add a barrier method for 4 weeks after initiation and for 4 weeks after each dose escalation. Adlyxin tells patients to take oral contraceptives at least 1 hour before the Adlyxin dose or 11 hours after it. If you depend on an oral contraceptive for pregnancy prevention, a backup barrier method during initiation and dose escalation is a reasonable default until you and your prescriber have confirmed the specific timing rules for your product.
Is kidney disease a GLP-1 contraindication?
Answer capsule
Kidney disease is generally not a Section 4 contraindication on current GLP-1 labels, but label language is product-specific. Most GLP-1 labels carry a Section 5 warning about acute kidney injury tied to dehydration from vomiting or diarrhea. Adlyxin (lixisenatide) is “not recommended” in end-stage renal disease, and exenatide products carry stronger renal-use limitations than semaglutide or tirzepatide products.
- Semaglutide products (Ozempic, Wegovy, Rybelsus)No renal-impairment contraindication. Section 5 warns about acute kidney injury, primarily from dehydration tied to GI side effects. Monitor renal function in patients with renal impairment reporting severe GI reactions.
- Tirzepatide products (Mounjaro, Zepbound)Same pattern as semaglutide -- no Section 4 renal contraindication; Section 5 acute kidney injury warning tied to volume depletion.
- Liraglutide products (Saxenda, Victoza)Section 5 acute kidney injury warning; no Section 4 renal contraindication.
- Dulaglutide (Trulicity)Section 5 acute kidney injury warning; no Section 4 renal contraindication.
- Exenatide products (Byetta, Bydureon BCise)Stronger renal-use limitations than other GLP-1s -- Byetta is not recommended in severe renal impairment, and creatinine clearance thresholds appear in the labels.
- Lixisenatide (Adlyxin)Section 5 acute kidney injury warning, and the label states Adlyxin is not recommended in end-stage renal disease (ESRD) due to limited data.
The practical rule: if you have any meaningful kidney impairment, the answer is product-specific. A patient with stage 3 chronic kidney disease may be appropriate for a semaglutide or tirzepatide product but a poor candidate for Byetta. The label of the specific drug — not a generic GLP-1 summary — is the source.
Is type 1 diabetes or DKA a GLP-1 contraindication?
Answer capsule
For most diabetes-labeled GLP-1 products, type 1 diabetes and diabetic ketoacidosis (DKA) are handled as Limitations of Use or “has not been studied” language, not as Section 4 contraindications. The practical point is still serious: GLP-1s are not insulin and should not be treated as DKA therapy.
- •A person with type 1 diabetes is not “contraindicated” per Section 4 of most labels — they are outside the studied indication. Some endocrinologists prescribe a GLP-1 off-label for type 1 patients with obesity, with careful insulin management. That is a clinical decision, not a label authorization.
- •DKA is a medical emergency. GLP-1s are not therapy for it. If you have a history of DKA, the relevant question for GLP-1 initiation is glycemic stability and insulin management — not whether the GLP-1 is “contraindicated.”
- •For weight-loss-indicated GLP-1s (Wegovy, Zepbound, Saxenda, Foundayo), type 1 diabetes wasn't the studied population. Off-label use in type 1 diabetes is a separate conversation with an endocrinologist who manages insulin.
What should you do about a GLP-1 before surgery, anesthesia, or deep sedation?
Answer capsule
Most current GLP-1 labels include a Section 5 warning about pulmonary aspiration during anesthesia or deep sedation, because delayed gastric emptying can leave food or fluid in the stomach even after the standard fasting period. The labels do not set a universal “stop the drug X days before surgery” rule — they instruct patients to inform the anesthesia and procedure team about which GLP-1 they take, when the last dose was, and any GI symptoms.
The Zepbound label explicitly states that available data are insufficient to determine whether modifying fasting or temporarily discontinuing the drug reduces aspiration risk — the FDA saying: the risk is real but the right peri-operative protocol is not yet settled.
If you have a scheduled surgery, endoscopy, colonoscopy, or any procedure involving anesthesia:
- 1Tell the procedure or anesthesia team before the day of surgery, ideally at the pre-op visit.
- 2Have ready: which GLP-1 you take (brand and active ingredient), the dose, whether it is weekly or daily, and the date of your most recent dose.
- 3Report any current GI symptoms -- nausea, vomiting, fullness, reflux -- even if you think they are minor.
- 4Ask whether they want you to change your fasting plan, modify medication timing, or adjust anything else.
- 5Do not stop the drug on your own without that conversation. Stopping abruptly may have other consequences (rebound hunger, blood sugar swings) that need to be planned for.
What happened to the GLP-1 suicidal ideation warning in early 2026
Answer capsule
On January 13, 2026, the FDA issued a Drug Safety Communication requesting removal of suicidal behavior and ideation (SI/B) warning language from the Section 5 labels of Saxenda, Wegovy, and Zepbound. The FDA's comprehensive review found no increased risk of SI/B associated with GLP-1 use. Current U.S. labels for all three products now show the warning as removed (Wegovy 01/2026; Saxenda and Zepbound 02/2026). No Section 4 contraindication changed.
| Product | SI/B warning previously in label? | Current status | Section 4 changed? |
|---|---|---|---|
| Wegovy (semaglutide) | Yes | Removed 01/2026 | No |
| Saxenda (liraglutide) | Yes | Removed 02/2026 | No |
| Zepbound (tirzepatide) | Yes | Removed 02/2026 | No |
| Diabetes-indicated GLP-1s (Ozempic, Mounjaro, Trulicity, etc.) | No | N/A — never had SI/B warning | No |
Why this matters for understanding the label categories:
- Section 5 is dynamic. Warnings can be added when evidence appears and removed when evidence does not bear them out. The standard is “reasonable evidence of causal association,” and that standard can shift with better data.
- Section 4 is harder. Contraindications change much more rarely. The threshold is higher and the data must show a known hazard.
- A removed warning is not a removed problem. People with active mental health conditions still benefit from screening, monitoring, and clinical care during GLP-1 treatment. The FDA's action was about labeling accuracy, not about whether psychiatric history matters in a prescribing conversation.
Things people commonly miscategorize
Answer capsule
The most common GLP-1 label misreadings run in two directions: things people call contraindications that are actually Section 5 warnings (pancreatitis, kidney disease, gallbladder disease, surgery, diabetic retinopathy, pregnancy), and things people underestimate as “just warnings” that are actually Section 4 contraindications (documented serious hypersensitivity to the specific drug, and for combination products, episodes of hypoglycemia).
Pattern one: calling Section 5 risks contraindications
| Common claim | What the FDA label actually says | Section |
|---|---|---|
| "Pancreatitis is a contraindication" | Discontinue if pancreatitis suspected; do not restart if confirmed | 5 |
| "Kidney disease is a contraindication" | Monitor renal function; counsel about dehydration (Adlyxin is "not recommended" in ESRD) | 5 |
| "Gallbladder disease is a contraindication" | Evaluate if symptoms develop | 5 |
| "Diabetic retinopathy is a contraindication" | Monitor patients with retinopathy in T2DM | 5 |
| "Upcoming surgery is a contraindication" | Pulmonary aspiration warning; counsel before procedures | 5 |
| "Pregnancy is a contraindication" | Use in Specific Populations (Section 8); Saxenda's Section 4 pregnancy entry was removed in May 2025 | 8 |
| "Depression / anxiety is a contraindication" | Was a Section 5 warning for Saxenda/Wegovy/Zepbound; removed in early 2026 following FDA Drug Safety Communication | Removed |
Pattern two: missing Section 4 entries that actually apply
- •A documented anaphylaxis or angioedema reaction to a specific GLP-1 is a Section 4 contraindication for that drug. Mild rash or injection site reaction is not. Patients sometimes report a "reaction" without knowing which threshold it crossed.
- •For Soliqua and Xultophy, the insulin component creates a Section 4 contraindication during episodes of hypoglycemia. This is product-specific to the combination products.
- •For exenatide products (Byetta and Bydureon BCise), a history of exenatide-induced immune-mediated thrombocytopenia is a Section 4 contraindication that no other GLP-1 has.
- •Family history of MTC is Section 4 language for nearly every GLP-1 label. If the affected relative is not a parent, sibling, or child (a grandparent, aunt, or uncle), don't guess the degree rule from an intake form -- bring the family history to the prescriber.
Compounded GLP-1s are a different conversation
Answer capsule
Compounded versions of semaglutide, tirzepatide, and related GLP-1 receptor agonists are not FDA-approved products and do not carry FDA-reviewed labels. The Section 4 contraindications, Section 5 warnings, and boxed warnings described on this page apply to FDA-approved branded products only. The underlying biological risks are still relevant for compounded molecules of the same active ingredient — but the verification chain is different.
The FDA has publicly identified specific safety concerns with unapproved compounded GLP-1 drugs:
- •Compounded versions are not FDA-reviewed for safety, effectiveness, or quality before they are marketed.
- •The FDA has received adverse event reports specifically involving compounded products.
- •Some compounded products use salt forms of semaglutide (such as semaglutide sodium and semaglutide acetate) that are not the same active ingredient as the approved drug and have not been evaluated for safety.
- •Dosing errors are more common when compounded products use different concentrations or measurement formats than approved branded pens.
- •Storage and stability have been concerns for some compounded preparations.
The Section 4 risks identified on branded labels (MTC, MEN 2, hypersensitivity) are still real risks for compounded molecules of the same active ingredient. Your prescriber should still screen for them. But with a compounded product, you are relying on the prescribing provider and the compounding pharmacy — there is no FDA-reviewed label of record to consult.
How to verify any GLP-1 label claim yourself in 30 seconds
Answer capsule
Two primary sources are sufficient: DailyMed (dailymed.nlm.nih.gov) and accessdata.fda.gov (Drugs@FDA). For historical changes, use the FDA Drug Safety-Related Labeling Changes (SrLC) Database. Any consumer summary that conflicts with the current label on those sources should be questioned.
- 1Go to dailymed.nlm.nih.gov. Type the brand name in the search box (Wegovy, Mounjaro, Foundayo, etc.).
- 2Open the current label. The first result is the active version. Look at the Effective Date near the top -- it tells you how current the label is.
- 3Click "Contraindications" in the section navigation. Or scroll to Section 4. Read the bullet list. That is what the FDA currently considers the do-not-use list for that drug.
- 4Click "Warnings and Precautions" or scroll to Section 5. Read the sub-section titles (5.1, 5.2, 5.3...). That is the current warning catalog.
- 5For historical changes, look at the Recent Major Changes section at the top. It lists every Section 4, Section 5, or boxed-warning revision with a date, or use the FDA's Drug Safety-Related Labeling Changes (SrLC) Database.
Why third-party summaries lag the actual label:
- • Labels update through a formal supplemental new drug application (sNDA) or FDA-initiated change. They are legal documents.
- • Marketing pages and consumer summaries simplify, sometimes lag by weeks or months, and occasionally miscategorize.
- • Telehealth intake forms further simplify. They are designed for triage at scale, not for label-level precision.
Bring this to your prescriber — the conversation checklist
This checklist mirrors the same five categories used in the label itself. Print it and fill it in before your visit.
- Which GLP-1 has been suggested or prescribed? (Brand and active ingredient)
- Is the product FDA-approved or compounded?
- Dose, frequency (daily/weekly), planned start date
- Personal history of medullary thyroid carcinoma (MTC)? Yes / No / Unsure
- Family history of MTC -- parent, sibling, child, grandparent, aunt, or uncle? Yes / No / Unsure
- Diagnosis of MEN 2 (Multiple Endocrine Neoplasia syndrome type 2)? Yes / No / Unsure
- Any prior serious allergic reaction to a GLP-1 medication (anaphylaxis, angioedema)? Yes / No
- For exenatide products only: history of exenatide-induced immune-mediated thrombocytopenia? Yes / No / N/A
- For combination products (Soliqua, Xultophy): currently experiencing low blood sugar episodes? Yes / No / N/A
- History of acute pancreatitis? Yes / No / Unsure -- date and cause if known
- Currently on insulin or sulfonylurea? Yes / No
- Kidney disease (chronic kidney disease, history of acute kidney injury, dialysis)? Yes / No
- Gallbladder disease or gallstones? Yes / No
- Diabetic retinopathy (any stage)? Yes / No / N/A
- Type 2 diabetes? Yes / No
- History of severe nausea/vomiting requiring hospitalization? Yes / No
- Pregnant or planning pregnancy within 6 months? Yes / No
- Breastfeeding? Yes / No
- Currently using an oral contraceptive (relevant for tirzepatide and lixisenatide products)? Yes / No
- Age (younger than 18 or older than 75 may trigger product-specific considerations)
- Severe gastroparesis or chronic gastric motility disorder? Yes / No
- Type 1 diabetes or history of diabetic ketoacidosis (DKA)? Yes / No
- End-stage renal disease (relevant for lixisenatide and exenatide products)? Yes / No
- Currently using any other GLP-1 product (this rules out combining)? Yes / No
- Any scheduled surgery, endoscopy, colonoscopy, or other procedure with anesthesia or deep sedation? Yes / No -- date if known
- Last GLP-1 dose date and time (if continuing therapy)
- Any persistent or severe abdominal pain?
- Any new severe nausea, vomiting, or inability to keep food down?
- Any new vision changes (if diabetic)?
- Any unusual swelling, breathing problems, or rash since last dose?
When to call your clinician urgently
Answer capsule
Specific symptoms during GLP-1 therapy warrant prompt medical attention: severe persistent abdominal pain (possible pancreatitis or gallbladder); serious allergic reaction symptoms; severe dehydration from ongoing vomiting or diarrhea; new vision changes in diabetic patients; and right-upper-quadrant pain with fever or jaundice (possible gallbladder).
Possible acute pancreatitis
Same-daySevere, persistent abdominal pain, often radiating to the back, sometimes with nausea or vomiting. Stop the GLP-1 and seek same-day medical care.
Serious allergic reaction
Call 911Swelling of face, lips, tongue, or throat. Difficulty breathing or swallowing. Severe rash or itching. Fainting or rapid heartbeat.
Severe dehydration
Same-dayRepeated vomiting or diarrhea preventing fluid intake. Dizziness on standing. Reduced urination. Dark urine.
New diabetic retinopathy symptoms
Same-day ophthalmologySudden vision changes, blurred vision, dark spots, or vision loss -- particularly in patients with type 2 diabetes whose blood sugar has recently improved rapidly.
Gallbladder symptoms
Same-dayRight-upper-abdomen pain, especially after meals, with fever, yellowing of the skin or eyes, or clay-colored stools.
Anesthesia / surgery within 48 hours
Call nowIf you are within 48 hours of a planned procedure with anesthesia or deep sedation and have not yet told the anesthesia team about your GLP-1, call now.
This is a list to help you decide when to contact a clinician, not a list to self-diagnose from. If you are unsure, calling is the right call.
Ready to find your best GLP-1 path?
Now that you understand the label categories, our 60-second quiz matches you with the right provider for your coverage, eligibility, and situation.
→ Find My GLP-1 PathWhat we actually verified
This page is built on three classes of primary source: the FDA's October 2011 guidance for industry on Warnings and Precautions, Contraindications, and Boxed Warning Sections (the active FDA guidance as of May 13, 2026); the current FDA-approved prescribing information for each named GLP-1 medication via DailyMed and accessdata.fda.gov; and the FDA's January 13, 2026 Drug Safety Communication requesting removal of the suicidal ideation warning.
Verified against primary sources (May 13, 2026)
- Verbatim regulatory definitions for Contraindications, Warnings and Precautions, and Boxed Warning
- Section 4 and Section 5 entries for every GLP-1 in the matrix against current DailyMed labels
- May 2025 Saxenda label update removing pregnancy from Section 4
- February 2026 removal of the SI/B warning from Saxenda's Section 5.9
- Foundayo orforglipron approval and label content via April 2026 FDA approval letter
- January 13, 2026 FDA Drug Safety Communication on SI/B removal
- Current Wegovy (01/2026), Zepbound (02/2026), Saxenda (02/2026) removal dates
- Pulmonary aspiration label language for Zepbound, including FDA wording about insufficient data
- Thyroid cancer subtype percentages from the American Thyroid Association
- Zepbound and Adlyxin label instructions regarding oral contraceptive timing
What we did not verify:
- • Whether any individual reader is eligible for GLP-1 therapy
- • International (non-U.S.) prescribing information
- • Compounded products against FDA-approved labels (they have no FDA-reviewed labels)
- • Current market availability of every product (Byetta and Bydureon BCise applicants notified the FDA of marketing-discontinuation plans in 2024)
Sources
All retrieved May 13, 2026. Primary sources are verified via FDA.gov and DailyMed.
- FDA. Warnings and Precautions, Contraindications, and Boxed Warning Sections of Labeling for Human Prescription Drug and Biological Products. Guidance for Industry, October 2011.
- 21 CFR 201.56 and 201.57 -- the federal regulation governing prescription drug labeling under the Physician Labeling Rule.
- FDA Drug Safety Communication, January 13, 2026: FDA Requests Removal of Suicidal Behavior and Ideation Warning from GLP-1 RA Medications.
- FDA. FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss.
- American Thyroid Association. Thyroid Cancer Subtypes.
- Wegovy (semaglutide injection / Wegovy HD / Wegovy tablets) full prescribing information, accessdata.fda.gov and DailyMed, current label.
- Ozempic (semaglutide injection) full prescribing information, DailyMed, current label.
- Rybelsus / Ozempic tablets (oral semaglutide) full prescribing information, DailyMed; FDA approval letter 2026.
- Mounjaro (tirzepatide) full prescribing information, DailyMed, current label.
- Zepbound (tirzepatide) full prescribing information, DailyMed, current label.
- Saxenda (liraglutide) full prescribing information, DailyMed, current label (revised 05/2025; SI/B removed 02/2026).
- Victoza (liraglutide) full prescribing information, DailyMed, current label.
- Trulicity (dulaglutide) full prescribing information, DailyMed, current label.
- Bydureon BCise (exenatide extended-release) full prescribing information, DailyMed, current label.
- Byetta (exenatide immediate-release) full prescribing information, DailyMed, current label.
- Adlyxin (lixisenatide) full prescribing information, DailyMed and accessdata.fda.gov 2026 PDF, current label.
- Soliqua (insulin glargine + lixisenatide) full prescribing information, DailyMed, current label.
- Xultophy (insulin degludec + liraglutide) full prescribing information, DailyMed, current label.
- Foundayo (orforglipron) full prescribing information, DailyMed; FDA approval letter dated April 2026.
- FDA. Drug Safety-Related Labeling Changes (SrLC) Database.
Frequently asked questions
What is the difference between a GLP-1 contraindication and a warning?
A GLP-1 contraindication is a Section 4 entry on the FDA prescribing information meaning the drug should not be used because the risk clearly outweighs any benefit. A warning or precaution is a Section 5 entry -- a serious risk that requires monitoring, counseling, or stopping rules during therapy, but does not automatically block prescribing. The two categories use different verbs: Section 4 uses 'is contraindicated'; Section 5 uses 'use with caution' or 'discontinue if.' That verb is the fastest way to tell them apart.
Is a precaution the same as a warning?
In modern FDA labels (Physician Labeling Rule format, 2006 and after), yes. Warnings and Precautions are merged into a single Section 5 of the prescribing information. The older clinical distinction -- warning more serious than precaution -- is still used in conversation but is no longer reflected in the regulatory document.
What is the GLP-1 boxed warning?
For most GLP-1 receptor agonists, the boxed warning describes a risk of thyroid C-cell tumors based on rodent studies. The labels state that human relevance has not been determined and contraindicate use in patients with personal or family history of medullary thyroid carcinoma (MTC) or MEN 2. Byetta, Adlyxin, and Soliqua do not carry this boxed warning. The boxed warning is a formatting designation; it cross-references the Section 4 contraindication and the Section 5.1 thyroid C-cell tumor discussion.
Is pancreatitis a contraindication for GLP-1 medications?
No. Pancreatitis is a Section 5 warning on every current GLP-1 label. The labels instruct prescribers to discontinue the drug if pancreatitis is suspected and not to restart it if pancreatitis is confirmed. A history of pancreatitis is not listed as a Section 4 contraindication on any current GLP-1 label, though many clinicians treat it as a relative contraindication based on clinical judgment.
Can I take a GLP-1 with a family history of thyroid cancer?
It depends on the type. The Section 4 contraindication specifically names medullary thyroid carcinoma (MTC) and MEN 2. The American Thyroid Association lists papillary thyroid cancer at about 70 to 80 percent of thyroid cancers, follicular at 10 to 15 percent, medullary at about 2 percent, and anaplastic at less than 2 percent. If the family cancer type is unclear, confirm the pathology before treating it like a yes/no GLP-1 answer.
Should I stop my GLP-1 before surgery?
Tell your anesthesia or procedure team in advance -- at the pre-op visit, not the morning of surgery -- exactly which GLP-1 you take, the dose, the schedule, and when you last took it. Most current GLP-1 labels include a Section 5 warning about pulmonary aspiration during anesthesia or deep sedation. The labels do not standardize a universal 'stop X days before' rule, and the Zepbound label explicitly notes that available data are insufficient to determine whether fasting modifications or temporary discontinuation reduce aspiration risk. Your anesthesia and surgery team determines the protocol for your procedure.
Is pregnancy a contraindication for GLP-1 medications?
No. Pregnancy is not listed in Section 4 on the current U.S. labels reviewed here, but Section 8 wording differs by product and indication. Weight-loss labels such as Saxenda and Zepbound tell prescribers to discontinue the drug when pregnancy is recognized; semaglutide labels recommend stopping at least 2 months before a planned pregnancy due to the drug's long washout period; diabetes labels such as Mounjaro and Trulicity use benefit-risk language rather than universal discontinue-on-recognition wording. Some older summaries still describe Saxenda as the exception with pregnancy in Section 4; that contraindication was removed in May 2025.
Is kidney disease a contraindication for GLP-1 medications?
Generally no. Kidney disease is not a Section 4 contraindication on most current GLP-1 labels, but the language is product-specific. Most labels carry a Section 5 warning about acute kidney injury tied to dehydration from vomiting or diarrhea. Adlyxin is not recommended in end-stage renal disease, and exenatide products (Byetta, Bydureon BCise) carry stronger renal-use limitations than semaglutide or tirzepatide products.
Do GLP-1s affect oral contraceptives?
Some GLP-1 labels include specific instructions. Zepbound tells females using oral contraceptives to switch to a non-oral method or add a barrier method for 4 weeks after initiation and 4 weeks after each dose escalation. Adlyxin tells patients to take oral contraceptives at least 1 hour before the Adlyxin dose or 11 hours after it. Other GLP-1s use more general language about delayed gastric emptying potentially affecting absorption of concomitant oral medications.
Why was the suicidal ideation warning removed from Wegovy and Zepbound in 2026?
On January 13, 2026, the FDA issued a Drug Safety Communication requesting removal of suicidal behavior and ideation warning language from Saxenda, Wegovy, and Zepbound. A comprehensive FDA review found no increased risk of suicidal ideation or behavior associated with GLP-1 use. The Section 4 contraindications for those drugs did not change; only the Section 5 warning was affected. Current U.S. labels for all three products now reflect the removal.
Do compounded GLP-1 medications have the same contraindications?
The contraindications described here apply to FDA-approved branded products. Compounded GLP-1 products are not FDA-reviewed and do not carry FDA-approved labels. The underlying biological risks (thyroid C-cell tumor signal, hypersensitivity, pancreatitis) are still relevant for the same active ingredients, and your prescriber should still screen against them. But there is no FDA-reviewed label of record to consult for compounded products. The FDA has issued specific concerns about unapproved GLP-1 products, including issues with salt forms, dosing errors, and storage.
If a telehealth intake form rejected me for a condition, was that a label-based denial?
Not necessarily. Telehealth intake forms typically simplify Section 4 (true contraindications) and Section 5 (warnings) into a single yes/no eligibility list. Many providers set tighter clinical criteria than the FDA label requires, often because remote prescribing limits the kinds of monitoring they can do. If you were declined for a Section 5 condition (pancreatitis history, kidney function concerns, etc.), the denial reflected clinical judgment rather than a Section 4 label rule. You can request reconsideration with documentation, or seek a different provider.
Related guides on The RX Index
- GLP-1 Providers That Minimize Side Effects: Dosing, Support, Monitoring
- Best FDA-Approved Brand-Name GLP-1 Telehealth Providers 2026
- GLP-1 Providers That Accept TRICARE: What Actually Works in 2026
- GLP-1 Cost Guide — Verified Pricing Across Every Major Provider
- Find My GLP-1 Path — 60-Second Personalized Matching Quiz
Know your label. Find the right path.
Our 60-second quiz matches you to the right GLP-1 provider for your coverage, health history, and goals — with no affiliate recommendation until we understand your situation.
→ Find My GLP-1 PathThis page is published by The RX Index editorial team. It is not medical advice and is not a substitute for an evaluation by a qualified prescribing clinician familiar with your full medical history. This page is a label-interpretation guide, not a medical recommendation. Methodology · Editorial Standards
Last verified: May 13, 2026. Next scheduled review: August 2026.