GLP-1 FDA Indications Chart: Every FDA-Approved Use, by Drug
Last verified: May 12, 2026 — The Rx Index Editorial Team
Affiliate disclosure: The RX Index earns a commission when you sign up with some of the providers mentioned on this page. It does not affect what you pay, and it never determines our rankings or which providers we cover. Read the full disclosure.
You typed “GLP-1 FDA indications chart” because somewhere along the way, the brand names stopped making sense. Ozempic. Wegovy. Mounjaro. Zepbound. Foundayo. Same molecules, different brands, totally different approvals — and somebody (a doctor, a pharmacist, an insurance denial letter) just told you the difference matters.
Bottom line up front. Nine standalone GLP-1 or dual GIP/GLP-1 brand families are actively FDA-approved and on the U.S. market right now, plus two FDA-approved GLP-1/insulin combination products. Three older GLP-1s were approved but have been discontinued by their manufacturers. The full chart is right below — use the filter to narrow it by condition, age, and form preference.
Why does the same drug have two different approvals?
Because the FDA approves products, not molecules. Ozempic and Wegovy contain the same semaglutide. But Novo Nordisk filed them as separate products, with separate studies, for separate uses. So the FDA “indication” — the official approved use — is different. That's the entire knot this page untangles.
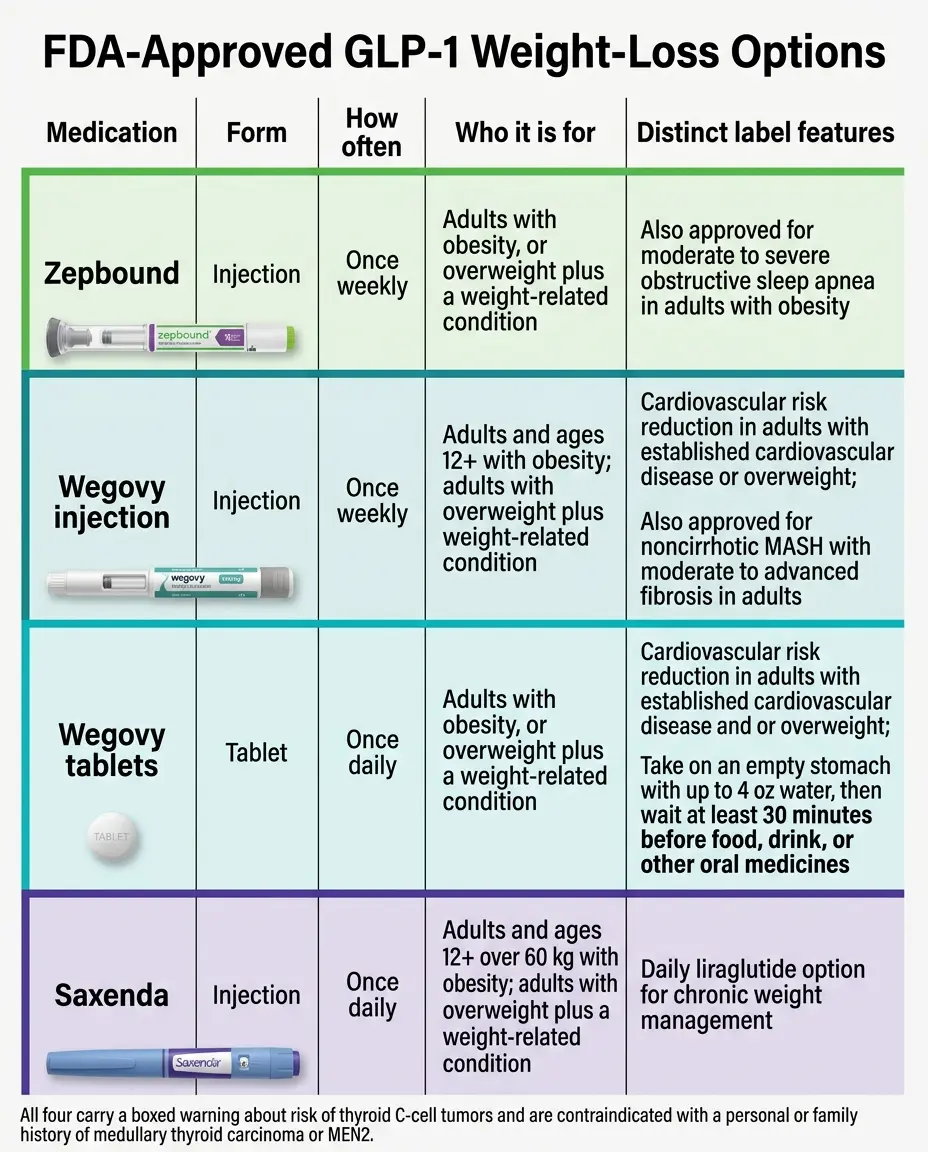
The master GLP-1 FDA indications chart
Below is every brand-name GLP-1 or GIP/GLP-1 product currently approved by the FDA. Every cell was checked against the drug's current prescribing information at DailyMed or the FDA's own press announcement establishing the indication. Use the filter below to narrow by condition, age range, and form preference.
Find My FDA-Approved GLP-1
| Brand | Active ingredient | Form | T2D adults | T2D 10+ | Weight adults | Weight 12+ | Heart risk (MACE) | Kidney (CKD) | Sleep apnea | MASH | Source · Grade |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Active — standalone GLP-1 and dual GIP/GLP-1 products | |||||||||||
| Ozempic injection | semaglutide | Weekly shot | ✅ | ✅ (T2D + known heart disease) | ✅ (T2D + CKD) | DailyMedA | |||||
| Ozempic tablets U.S. brand replacing Rybelsus; launched May 4, 2026 | semaglutide | Daily pill | ✅ | ✅ (T2D, high CV risk) | DailyMedA | ||||||
| Rybelsus Being transitioned to Ozempic tablets brand in U.S. | semaglutide | Daily pill | ✅ | ✅ (T2D, high CV risk) | DailyMedA | ||||||
| Wegovy injection | semaglutide | Weekly shot | ✅ | ✅ (age 12+, 95th percentile BMI) | ✅ (obesity/overweight + known heart disease) | ✅ (F2–F3 fibrosis, no cirrhosis) | DailyMedA | ||||
| Wegovy HD injection Approved March 19, 2026 under FDA National Priority Voucher program | semaglutide 7.2 mg | Weekly shot | ✅ (after tolerating 2.4 mg for ≥4 weeks) | DailyMed (Wegovy label)A | |||||||
| Wegovy tablets First oral GLP-1 for weight loss; approved December 22, 2025 | semaglutide 25 mg | Daily pill | ✅ | ✅ (obesity/overweight + known heart disease) | DailyMed (Wegovy label)A | ||||||
| Mounjaro Pediatric T2D added December 2025; MACE indication pending FDA decision | tirzepatide (GIP + GLP-1) | Weekly shot | ✅ | ✅ (age 10+, max 10 mg/week) | DailyMedA | ||||||
| Zepbound OSA indication added December 20, 2024 | tirzepatide (GIP + GLP-1) | Weekly shot | ✅ | ✅ (adults with obesity) | DailyMedA | ||||||
| Foundayo First non-peptide oral GLP-1; approved April 1, 2026. No empty-stomach rule. | orforglipron | Daily pill | ✅ | DailyMedA | |||||||
| Trulicity | dulaglutide | Weekly shot | ✅ | ✅ (age 10+) | ✅ (T2D + heart disease or multiple CV risk factors) | DailyMedA | |||||
| Victoza Generics available (Hikma, Teva) | liraglutide | Daily shot | ✅ | ✅ (age 10+) | ✅ (T2D + known heart disease) | DailyMedA | |||||
| Saxenda First generic (Teva) approved August 27, 2025 | liraglutide | Daily shot | ✅ | ✅ (age 12+, weight >60 kg) | DailyMedA | ||||||
| Active — GLP-1/insulin combination products | |||||||||||
| Soliqua 100/33 Fixed-ratio combo; not a standalone GLP-1. No thyroid boxed warning. | insulin glargine + lixisenatide | Daily shot | ✅ | DailyMedA | |||||||
| Xultophy 100/3.6 Fixed-ratio combo; not a standalone GLP-1 | insulin degludec + liraglutide | Daily shot | ✅ | novoMEDLINKB | |||||||
| Discontinued — formerly FDA-approved, no longer marketed | |||||||||||
| Adlyxin (lixisenatide) — discontinued Discontinued January 1, 2023 (Sanofi) | lixisenatide | Daily shot | (was T2D) | DailyMed PDFC | |||||||
| Byetta (exenatide) — discontinued Discontinued October 25, 2024 (AstraZeneca) | exenatide | Twice-daily shot | (was T2D) | DailyMedC | |||||||
| Bydureon BCise (exenatide ER) — discontinued Discontinued October 28, 2024 (AstraZeneca) | exenatide ER | Weekly shot | (was T2D adults + 10+) | (was 10+) | DailyMedC | ||||||
| Tanzeum (albiglutide) — discontinued Discontinued July 26, 2017 | albiglutide | Weekly shot | (was T2D) | MedlinePlusC | |||||||
✅ = FDA label explicitly lists this indication for this product. — = not listed. Grades: A = DailyMed confirmed; B = manufacturer PI confirmed, DailyMed pending; C = historical/discontinued reference only. Last verified: May 12, 2026.
What we actually verified for this page
- ✅ Each indication was confirmed against the current FDA prescribing information on DailyMed or the FDA press announcement that added it.
- ✅ Discontinuation status was checked against FDA notifications and manufacturer announcements.
- ✅ Approval dates were checked against the FDA approval letter or the manufacturer's confirming press release.
- ✅ Each row links to its primary source. DailyMed shows current labeling submitted to FDA by the manufacturer.
❌ We did not verify: insurance coverage, formulary status, pharmacy availability, current prices, prior-authorization rules, or whether any specific person should take a GLP-1. Those are between you, your pharmacist, your insurer, and your prescriber.
What GLP-1 medications are FDA-approved right now?
As of May 2026, eleven FDA-approved GLP-1 brand products are actively sold in the United States: nine standalone GLP-1 or dual GIP/GLP-1 brand families — Ozempic, Rybelsus/Ozempic tablets, Wegovy, Mounjaro, Zepbound, Foundayo, Trulicity, Victoza, and Saxenda — plus two GLP-1/insulin fixed-dose combinations (Soliqua and Xultophy). Three older GLP-1s (Adlyxin, Byetta, Bydureon BCise) and one even older drug (Tanzeum) were FDA-approved at one point but have been discontinued.
A few brand families include more than one product:
- Wegovy includes the original injection (up to 2.4 mg), Wegovy HD (7.2 mg injection), and Wegovy tablets (25 mg oral).
- Ozempic includes the original injection (0.5/1/2 mg) and Ozempic tablets (1.5/4/9 mg oral) — the latter introduced as part of Novo Nordisk's transition from the Rybelsus brand in the United States.
The class includes seven distinct molecules:
| Molecule | Brand names (U.S.) | Notes |
|---|---|---|
| Semaglutide | Ozempic, Ozempic tablets, Rybelsus, Wegovy, Wegovy HD, Wegovy tablets | All made by Novo Nordisk |
| Tirzepatide | Mounjaro, Zepbound | Eli Lilly. Technically a dual GIP/GLP-1, not a pure GLP-1, but grouped with the class for comparison. |
| Liraglutide | Victoza, Saxenda | Novo Nordisk for branded; generics from Hikma and Teva |
| Dulaglutide | Trulicity | Eli Lilly |
| Orforglipron | Foundayo | Eli Lilly. The newest — the only non-peptide GLP-1 on the market. |
| Exenatide | Byetta, Bydureon BCise | AstraZeneca. Both discontinued in 2024. |
| Lixisenatide | Adlyxin (standalone discontinued); Soliqua (still active as combo) | Sanofi. Standalone discontinued in 2023. |
The first FDA-approved GLP-1 was Byetta (exenatide), approved April 28, 2005. The newest is Foundayo (orforglipron), approved April 1, 2026. See our GLP-1 FDA approvals timeline for the full chronology.
Which GLP-1s are pills, and which are injections?
The FDA-approved oral GLP-1 products are Ozempic tablets, Rybelsus, Wegovy tablets, and Foundayo. Every other active product is an injection. For most of GLP-1 history, the only options were injections. That changed fast between September 2019 (Rybelsus, the first oral GLP-1) and April 2026 (Foundayo, the first non-peptide oral GLP-1).
The oral options
| Product | Use | Administration | Dose-timing rules |
|---|---|---|---|
| Ozempic tablets / Rybelsus | Type 2 diabetes | Daily pill | Take in the morning on an empty stomach with up to 4 oz of plain water, then wait at least 30 minutes before food, drink, or other oral medications. |
| Wegovy tablets (25 mg) | Chronic weight management | Daily pill | Same empty-stomach rule as Ozempic tablets/Rybelsus. |
| Foundayo | Chronic weight management | Daily pill | DailyMed says take once daily, with or without food. No 30-minute waiting rule in the label. |
The practical difference: Foundayo has no empty-stomach or 30-minute waiting rule. Wegovy tablets and Ozempic tablets/Rybelsus do. If you live with someone who eats breakfast right when they wake up, or if you take other morning medications, that label difference can change how realistic each option is for your routine.
The injection options range from twice daily (Byetta, now discontinued) to daily (Victoza, Saxenda, Soliqua, Xultophy) to once weekly (Ozempic injection, Wegovy injection, Wegovy HD, Mounjaro, Zepbound, Trulicity, plus the discontinued Bydureon BCise).
Which GLP-1s are FDA-approved for type 2 diabetes?
Five standalone GLP-1 or dual GIP/GLP-1 brand families are currently FDA-approved to treat type 2 diabetes in adults: Ozempic injection, Ozempic tablets/Rybelsus, Mounjaro, Trulicity, and Victoza. Three of those — Mounjaro, Trulicity, and Victoza — are also FDA-approved for kids ages 10 and older. Two combination products (Soliqua and Xultophy) are FDA-approved for adults with type 2 diabetes. Wegovy, Zepbound, Saxenda, and Foundayo are not approved for type 2 diabetes, even though some share active ingredients with diabetes-approved drugs.
Adult type 2 diabetes
If your doctor is choosing a GLP-1 for adult type 2 diabetes, the FDA-approved standalone options are:
- Ozempic injection (semaglutide, 0.5/1/2 mg weekly)
- Ozempic tablets (semaglutide, 1.5/4/9 mg daily; the 1.5 mg dose is the recommended starting dose for the first 30 days and is not effective for glycemic control on its own)
- Rybelsus (semaglutide, 3/7/14 mg daily) — same molecule as Ozempic tablets, in a different dose strength set; brand being transitioned to Ozempic tablets in the U.S.
- Mounjaro (tirzepatide, 2.5–15 mg weekly)
- Trulicity (dulaglutide, 0.75–4.5 mg weekly)
- Victoza (liraglutide, up to 1.8 mg daily)
Each is approved as an “adjunct to diet and exercise” — meaning the FDA expects it to be used alongside lifestyle changes, not instead of them.
Pediatric type 2 diabetes (age 10 and up)
This area changed in December 2025. Three GLP-1s are now FDA-approved for kids 10 and older with type 2 diabetes:
- Mounjaro — approved December 2025, based on the SURPASS-PEDS trial. Kids on Mounjaro lowered their A1C by an average of 2.2% over 30 weeks. One detail most charts miss: the pediatric maximum dose is 10 mg weekly, not the 15 mg adult maximum.
- Trulicity — approved for pediatric use in 2022, the first weekly GLP-1 cleared for this age group.
- Victoza — first approved for kids in June 2019. Generic versions from Hikma and Teva are also approved for pediatric T2D.
No GLP-1 is FDA-approved for type 2 diabetes in children younger than 10.
Heart attack and stroke risk reduction (MACE)
Some GLP-1s carry an extra FDA-approved use: reducing the risk of major adverse cardiovascular events — what doctors call MACE (cardiovascular death, non-fatal heart attack, or non-fatal stroke). The populations differ by product:
- Ozempic injection — adults with type 2 diabetes and known heart disease, based on the SUSTAIN-6 trial (26% MACE reduction vs. placebo).
- Ozempic tablets / Rybelsus — adults with type 2 diabetes who are at high risk for these events (a broader label population than the injection's known-heart-disease language).
- Trulicity — adults with type 2 diabetes plus either established cardiovascular disease or multiple cardiovascular risk factors. This is the broadest label of the three — the REWIND trial enrolled people without prior heart disease.
- Victoza — adults with type 2 diabetes and established cardiovascular disease, based on the LEADER trial.
The newer drugs — Mounjaro and Foundayo — do not yet have a MACE indication. Eli Lilly has submitted Mounjaro's SURPASS-CVOT trial data to the FDA. A decision is pending.
Chronic kidney disease in type 2 diabetes
Only one GLP-1 is FDA-approved to slow chronic kidney disease (CKD). Ozempic injection — added January 28, 2025 based on the FLOW trial. The exact label wording: it reduces the risk of “kidney disease worsening, kidney failure, and death due to cardiovascular disease” in adults with type 2 diabetes and CKD. The FLOW trial reported a 24% reduction in kidney-related events. This is one of the most overlooked label details — if you have T2D plus CKD, this is the only GLP-1 with a kidney-specific indication.
GLP-1 plus insulin combination products
- Soliqua 100/33 — insulin glargine plus lixisenatide. Adults with type 2 diabetes.
- Xultophy 100/3.6 — insulin degludec plus liraglutide. Adults with type 2 diabetes.
These are not the same as taking a standalone GLP-1. They're fixed-dose combos, dosed in units rather than milligrams. Pharmacy benefit forms sometimes lump them with the GLP-1 class, but they're a separate category clinically.
Approved for weight management GLP-1s? Find a provider.
If the chart shows you qualify for Wegovy, Zepbound, or Foundayo, Ro's insurance concierge can check your specific coverage before you pay anything.
Which GLP-1s are FDA-approved for chronic weight management?
Six FDA-approved GLP-1 products are indicated for chronic weight management in 2026: Wegovy injection, Wegovy HD injection, Wegovy tablets, Zepbound, Saxenda, and Foundayo. Wegovy injection and Saxenda are also approved for adolescents ages 12 and older. Ozempic, Mounjaro, Trulicity, and Victoza are not FDA-approved for weight loss — even though weight loss commonly happens when people take them for diabetes.
Why the distinction matters for insurance: Many plans cover weight-management drugs only when they have the actual FDA chronic weight management indication. Prescribing Ozempic for weight loss is legal, but it's off-label — and that affects what plans will pay for.
Adult chronic weight management — BMI thresholds
To match the FDA-approved population for any weight-management GLP-1, you generally need:
- A BMI of 30 or higher (the FDA's threshold for obesity), or
- A BMI of 27 or higher (called overweight) plus at least one weight-related condition like high blood pressure, high cholesterol, type 2 diabetes, or obstructive sleep apnea.
The six adult weight-management options
Wegovy injection
(semaglutide, up to 2.4 mg weekly)Approved June 2021STEP 1 trial: ~15% body weight loss at 68 weeks.
Wegovy HD injection
(semaglutide 7.2 mg weekly)Approved March 19, 2026STEP UP: 20.7% weight loss at 72 weeks. Only for adults who have already tolerated 2.4 mg for at least 4 weeks.
Wegovy tablets
(semaglutide 25 mg daily)Approved December 22, 2025OASIS 4 trial: 16.6% weight loss at 64 weeks with full adherence. First oral GLP-1 for weight loss. Empty-stomach rule applies.
Zepbound
(tirzepatide, 2.5–15 mg weekly)Approved November 8, 2023~21% weight loss at the 15 mg dose at 72 weeks. Also FDA-approved for obstructive sleep apnea.
Saxenda
(liraglutide, up to 3.0 mg daily)Approved December 23, 2014~8% weight loss at 56 weeks. A daily injection rather than weekly. First generic (Teva) approved August 27, 2025.
Foundayo
(orforglipron, daily pill)Approved April 1, 2026ATTAIN-1 at highest dose: 12.4% weight loss at 72 weeks. No empty-stomach rule — take with or without food.
Adolescent chronic weight management (age 12 and up)
- Wegovy injection — for kids 12+ with BMI at or above the 95th percentile for their age and sex.
- Saxenda — for kids 12+ with body weight greater than 60 kg (about 132 lb) and obesity by international cut-offs.
No GLP-1 is FDA-approved for weight management in children under 12.
Cardiovascular risk reduction in obesity and overweight
In March 2024, Wegovy became the first weight-loss medication to receive an FDA indication for reducing major cardiovascular events in adults with established cardiovascular disease who also have obesity or overweight — even without diabetes. Wegovy tablets carry the same indication. Wegovy HD does not yet — its approval is for weight loss alone. The approval was based on the SELECT trial, which showed a 20% reduction in MACE versus placebo over a median 33 months. This means Wegovy is currently the only GLP-1 with an FDA-approved cardiovascular benefit for non-diabetic people.
Which GLP-1 is FDA-approved for sleep apnea?
Zepbound (tirzepatide) is the only FDA-approved GLP-1 — and the only FDA-approved prescription medication of any kind — for obstructive sleep apnea (OSA). The indication, added December 20, 2024, covers moderate-to-severe OSA in adults with obesity, used along with a reduced-calorie diet and increased physical activity. Wegovy, Ozempic, Mounjaro, Trulicity, Victoza, Saxenda, and Foundayo are not FDA-approved for sleep apnea.
What this approval changes in practice is the prior-authorization question. Zepbound for OSA is an FDA-labeled OSA-in-adults-with-obesity indication, not just a weight-management claim. The relevant ICD-10 code is G47.33. Whether your specific plan covers Zepbound for OSA still depends on your plan's criteria — coverage rules vary by plan and change over time.
The approval was based on the SURMOUNT-OSA trial (469 adults with moderate-to-severe OSA and obesity). After 52 weeks, participants on tirzepatide had a statistically significant reduction in apnea-hypopnea index (AHI) versus placebo. Some achieved remission or mild OSA with resolution of symptoms.
Which GLP-1 is FDA-approved for fatty liver disease (MASH)?
Wegovy injection 2.4 mg (semaglutide) is the only FDA-approved GLP-1 for MASH — short for metabolic dysfunction-associated steatohepatitis, a severe form of fatty liver disease that used to be called NASH. The indication, granted August 15, 2025 under accelerated approval, covers adults with non-cirrhotic MASH and moderate-to-advanced liver fibrosis (scarring). Patients with cirrhosis are explicitly excluded. Wegovy tablets, Wegovy HD, and all other GLP-1s do not currently have a MASH indication.
A few important details that most pages miss:
- The approval specifies F2 or F3 fibrosis — the second and third stages on the standard 0-to-4 liver scarring scale. Stage F4 is cirrhosis, which is excluded.
- This is an accelerated approval, meaning the FDA cleared it based on liver-biopsy improvements before the long-term outcomes data is in. Part 2 of the ESSENCE trial is expected to read out around 2029 and will determine whether the approval converts to standard approval.
- The November 2025 update to AASLD practice guidance now recommends semaglutide for MASH patients with F2-F3 fibrosis, identified using non-invasive tests like FibroScan, MR elastography, or the ELF blood test.
In the ESSENCE trial's Part 1, 63% of patients on Wegovy achieved resolution of MASH without worsening of fibrosis, compared to 34% on placebo, at 72 weeks. Fibrosis improvement without worsening of MASH occurred in 37% versus 22%. Average weight loss in the semaglutide arm was 10.5%.
Same active ingredient, different FDA indication — the most common confusion
Same active ingredient ≠ same FDA-approved indication.
Three of the major GLP-1 molecules — semaglutide, tirzepatide, and liraglutide — are each sold under two or more brand names with different FDA indications. The molecule does the same thing biologically, but the FDA approves products, not molecules.
| Molecule | Diabetes brand(s) | Weight management brand(s) | Other label uses |
|---|---|---|---|
| Semaglutide | Ozempic injection, Ozempic tablets, Rybelsus | Wegovy injection, Wegovy HD, Wegovy tablets | Ozempic injection: CKD in T2D · Wegovy injection: MASH · Wegovy injection/tablets: MACE in obesity/overweight |
| Tirzepatide | Mounjaro | Zepbound | Zepbound: OSA in adults with obesity |
| Liraglutide | Victoza, Xultophy (combo) | Saxenda | Victoza: MACE in T2D |
- Ozempic is not FDA-approved for weight loss. Many people take it for that anyway, off-label, and lose weight. But the prescription is off-label, and insurance for weight loss reflects that.
- Mounjaro is not FDA-approved for weight loss either. Zepbound is the tirzepatide product for that.
- Wegovy is not FDA-approved for diabetes. Ozempic is the semaglutide product for that.
- The doses are not interchangeable. Wegovy maxes out at 2.4 mg (or 7.2 mg for Wegovy HD). Ozempic maxes out at 2 mg. Rybelsus tops out at 14 mg; Ozempic tablets top out at 9 mg. The labels say Rybelsus and Ozempic tablets are not substitutable on a mg-to-mg basis.
What's new in GLP-1 FDA approvals since 2025?
Between January 2025 and May 2026, the FDA approved or expanded eight major GLP-1 actions. In rough chronological order:
Based on the FLOW trial, Ozempic injection became the only GLP-1 approved to reduce kidney disease progression in adults with T2D and CKD.
Accelerated approval based on Part 1 of the ESSENCE trial. The first GLP-1 cleared for a liver disease.
Via FDA labeling revision, Mounjaro became approved for kids 10+ with type 2 diabetes, based on SURPASS-PEDS. Pediatric maximum dose: 10 mg weekly.
First oral GLP-1 for chronic weight management. Launched in early January 2026.
Same molecule as Rybelsus, in reformulated doses (1.5, 4, 9 mg). Launched May 4, 2026. Rybelsus brand continues outside the U.S.
7.2 mg weekly injection for adults who have tolerated 2.4 mg for at least 4 weeks. Fourth approval under FDA's National Priority Voucher pilot program.
First non-peptide oral GLP-1. First new molecular entity approved under the National Priority Voucher program — 50 days after filing, the fastest new-drug approval since 2002.
Pending decisions (as of May 12, 2026)
- Mounjaro MACE indication — Eli Lilly submitted based on SURPASS-CVOT, which reported an 8% lower rate of MACE-3 events versus Trulicity. FDA decision pending.
- Foundayo for type 2 diabetes — Eli Lilly has said it plans to submit to the FDA for T2D by the end of Q2 2026.
- Ozempic tablets 25 mg for T2D — Novo Nordisk filed a supplemental application; FDA decision expected by end of 2026.
We'll update this chart the day each one lands.
Which GLP-1s have a generic available?
Two GLP-1 molecules have FDA-approved generics as of May 2026: exenatide and liraglutide. Generic exenatide exists, but its branded reference products (Byetta and Bydureon BCise) have been discontinued. Generic liraglutide is the active generic story:
| Molecule | Brand referenced | Generic applicant | FDA approval date | What it's labeled for | Branded still active? |
|---|---|---|---|---|---|
| Liraglutide | Victoza | Hikma | December 23, 2024 | T2D in adults and kids 10+ | Yes |
| Liraglutide | Victoza | Teva | 2024 (earlier in year) | T2D in adults and kids 10+ | Yes |
| Liraglutide | Saxenda | Teva | August 27, 2025 | Adult and adolescent chronic weight management | Yes |
| Exenatide | Byetta | Various | 2024 | T2D in adults | No — Byetta discontinued |
The bigger picture: Ozempic, Wegovy, Mounjaro, Zepbound, Trulicity, Rybelsus/Ozempic tablets, and Foundayo have no FDA-approved generics. Any product sold online as a “generic” version of these is either compounded (a separate regulatory category) or counterfeit.
Which GLP-1s have been discontinued?
Three FDA-approved GLP-1s have been voluntarily discontinued by their makers: Adlyxin (lixisenatide, discontinued January 1, 2023 by Sanofi), Byetta (exenatide, discontinued October 25, 2024 by AstraZeneca), and Bydureon BCise (exenatide extended-release, discontinued October 28, 2024 by AstraZeneca). Tanzeum (albiglutide) was discontinued in 2017. The active ingredient in Adlyxin is still available combined with insulin in Soliqua.
| Brand | Molecule | Originally approved | Discontinued | Source |
|---|---|---|---|---|
| Tanzeum | albiglutide | April 15, 2014 | July 26, 2017 | Manufacturer market withdrawal |
| Adlyxin | lixisenatide | July 27, 2016 | January 1, 2023 | Sanofi business announcement, October 2022 |
| Byetta | exenatide | April 28, 2005 | October 25, 2024 | AstraZeneca manufacturer notification |
| Bydureon BCise | exenatide ER | January 27, 2012 | October 28, 2024 | AstraZeneca manufacturer notification |
FDA-approved vs. off-label vs. compounded — the categories matter
A drug is FDA-approved when its specific manufacturer has shown the FDA that the product is safe and effective for a specific use in a specific population. An off-label prescription is a legal prescription of an FDA-approved drug for a use the FDA hasn't approved. A compounded drug is one a pharmacy makes individually — these are not FDA-approved at all.
FDA-approved
What this entire chart is about. The drug, the indication, the dose, the form, the population — every piece was reviewed and cleared by the FDA. The label tells doctors and pharmacists exactly what's been studied and approved.
Off-label
Doctors are allowed to prescribe an FDA-approved drug for uses outside its FDA-approved indication. This is legal and common in medicine. Example: Mounjaro for weight loss in someone without diabetes. The drug is FDA-approved (for diabetes), but using it for weight loss is off-label. Insurance often doesn't cover off-label use.
Compounded
A licensed pharmacy mixes the ingredients individually. Compounded GLP-1s are not FDA-approved. The FDA has clarified that compounders generally cannot make products that are essentially copies of commercially available FDA-approved drugs. The branded GLP-1 shortages that allowed broader compounding have ended (tirzepatide injection in December 2024, semaglutide injection in February 2025).
Common compounded GLP-1 claims vs. what the FDA actually says
| Claim you might see online | What the FDA actually says |
|---|---|
| "Same as Ozempic / Wegovy / Mounjaro" | Compounded drugs are not FDA-approved and cannot be marketed as generic versions or the same as FDA-approved drugs. |
| "FDA-approved semaglutide" (when sold by a compounder) | The molecule semaglutide is FDA-approved as Novo Nordisk's branded products. A compounded preparation containing semaglutide is not an FDA-approved drug. |
| "Research-use only" or "not for human consumption" semaglutide/tirzepatide vials | The FDA has warned about these — they are unapproved drug products, often sold illegally to consumers despite the 'research-use' label. |
| "Available without a prescription" GLP-1 | All FDA-approved GLP-1s in the U.S. are prescription-only. |
How to use this chart with your doctor or insurer
This chart is built to answer one question: “What is this product FDA-labeled to do, for which population?” It's not built to tell you which drug is best for you, whether your insurance will cover it, whether your pharmacy stocks it, or whether the dose is right for your situation.
If you're walking into a doctor's appointment with this page open, here's the short checklist:
- What am I being treated for? Diabetes, weight, heart, kidney, sleep apnea, MASH, or some combination?
- Which product on this chart is FDA-labeled for that? Cross-check the row.
- Does my age and population match? Adult, adolescent 12+, child 10+.
- Does my insurance care about FDA indication or just drug class? Most prior-authorization forms ask for both diagnosis code and drug name. A mismatch is a common cause of denial.
- Is there a brand of the same molecule that fits my situation better? Someone with both T2D and obesity, for example, may want to discuss Ozempic vs. Wegovy with their doctor.
What this chart can't tell you:
- Whether the drug is right for you medically.
- Whether your specific insurance plan covers it.
- Whether your pharmacy has it in stock right now (shortages and rolling launches affect this).
- Whether an off-label prescription would be reasonable for your situation.
- Whether a compounded version is “the same” — it isn't, regulatorily, even if it contains the same molecule.
How we verified every cell in this chart
Every indication on this chart was checked against the current FDA-approved prescribing information at DailyMed or the FDA press announcement that added the indication, and cross-checked against the manufacturer's confirming press release. We did not interpolate from competitor pages or summary articles. Each cell is graded A, B, or C based on the strength of the source.
Our source hierarchy, in order:
- DailyMed prescribing information (dailymed.nlm.nih.gov). DailyMed contains the labeling submitted to FDA by manufacturers, currently in use. The “Indications and Usage” section is where the FDA-approved indication is officially defined.
- FDA approval letters and press announcements (fda.gov). Used to confirm dates and the specific indication wording for newly-added uses.
- Manufacturer prescribing information PDFs (e.g., pi.lilly.com, novo-pi.com). Used when DailyMed has not yet updated for the most recent label change.
- Manufacturer press releases (PRNewswire archives, investor.lilly.com, novonordisk.com). Used only as supporting evidence and to verify dates.
Verification grades
- ACurrent FDA / DailyMed label text confirmed. The strongest grade. This is most rows.
- BManufacturer prescribing information confirmed, DailyMed cross-check pending. Used briefly when a label change is recent and DailyMed hasn't synced yet.
- CHistorical or discontinued reference only. The drug had FDA approval at some point but is not currently marketed.
We refresh the chart at least monthly and immediately after any FDA action that changes a label. The “Last verified” date at the top of this page is updated every time we re-confirm — not just the first time we published.
Frequently asked questions
Is Ozempic FDA-approved for weight loss?
No. Ozempic injection and Ozempic tablets are FDA-approved for type 2 diabetes. Ozempic injection is also approved for reducing major cardiovascular events in adults with T2D and known heart disease, and for reducing kidney disease worsening and cardiovascular death in adults with T2D and CKD. Ozempic is not FDA-approved for weight loss. Wegovy contains the same molecule (semaglutide) and is the FDA-approved product for chronic weight management.
Is Mounjaro FDA-approved for weight loss?
No. Mounjaro is FDA-approved for type 2 diabetes in adults and, since December 2025, in kids ages 10 and older. Zepbound contains the same molecule (tirzepatide) and is the FDA-approved product for chronic weight management. Zepbound is also FDA-approved for moderate-to-severe obstructive sleep apnea in adults with obesity — Mounjaro is not.
Is Rybelsus being discontinued?
In the United States, Rybelsus is being transitioned to the new Ozempic tablets brand (the same molecule, semaglutide, in slightly reformulated 1.5/4/9 mg doses). Novo Nordisk says patients currently on Rybelsus should continue as prescribed and discuss transitioning with their healthcare provider. Rybelsus and Ozempic tablets are not interchangeable on a milligram-for-milligram basis. Outside the U.S., Rybelsus remains the brand name.
What is Foundayo?
Foundayo is the brand name for orforglipron, the first non-peptide oral GLP-1 receptor agonist. The FDA approved it on April 1, 2026 for chronic weight management in adults with obesity, or adults with overweight plus a weight-related medical problem. DailyMed says Foundayo can be taken once daily with or without food — there is no empty-stomach or 30-minute waiting rule in the label. It is made by Eli Lilly. It is not currently FDA-approved for type 2 diabetes, although Lilly has said it plans to submit a T2D filing by the end of Q2 2026.
Is there an FDA-approved GLP-1 pill?
Yes. Four oral GLP-1 products are FDA-approved in the United States: Ozempic tablets and Rybelsus (semaglutide, for type 2 diabetes), Wegovy tablets (semaglutide 25 mg, for chronic weight management), and Foundayo (orforglipron, for chronic weight management). All other FDA-approved GLP-1s are injections.
Which GLP-1 is FDA-approved for sleep apnea?
Only Zepbound (tirzepatide). The indication, added December 20, 2024, covers moderate-to-severe obstructive sleep apnea in adults with obesity. No other GLP-1 — including Mounjaro, which contains the same molecule — has an FDA-approved sleep apnea indication.
Which GLP-1 is FDA-approved for fatty liver / MASH?
Only Wegovy injection (semaglutide 2.4 mg). The accelerated approval, granted August 15, 2025, covers adults with non-cirrhotic MASH and moderate-to-advanced fibrosis (stages F2-F3). Wegovy tablets, Wegovy HD, and all other GLP-1s do not currently have a MASH indication.
Which GLP-1 is approved for kidney disease?
Only Ozempic injection (semaglutide). The indication, added January 28, 2025, covers adults with type 2 diabetes and chronic kidney disease, to reduce the risk of kidney disease worsening, kidney failure, and death due to cardiovascular disease. No other GLP-1 currently has a CKD-specific indication.
Which GLP-1s are FDA-approved for kids?
For type 2 diabetes (ages 10 and older): Mounjaro, Trulicity, Victoza. For chronic weight management (ages 12 and older): Wegovy injection and Saxenda. No GLP-1 is FDA-approved for children under 10.
Are compounded GLP-1s FDA-approved?
No. Compounded GLP-1s are not FDA-approved drugs. The FDA regulates them under a separate framework (sections 503A and 503B). The branded GLP-1 shortages that previously allowed broader compounding have ended (tirzepatide injection in December 2024, semaglutide injection in February 2025), and the FDA has clarified that compounders generally cannot produce products that are essentially copies of commercially available FDA-approved drugs.
What was the first FDA-approved GLP-1?
Byetta (exenatide), approved by the FDA on April 28, 2005, for type 2 diabetes. AstraZeneca voluntarily discontinued Byetta in October 2024.
What is the newest FDA-approved GLP-1?
Foundayo (orforglipron), approved April 1, 2026 for chronic weight management. It is the first non-peptide oral GLP-1 and the first new molecular entity approved under the FDA's National Priority Voucher pilot program.
Update log
This log shows every change we've made to the chart since the current version was published.
| Date verified | Product | What changed | Source |
|---|---|---|---|
| May 12, 2026 | Full chart | Initial publication of the May 2026 verified chart, incorporating all approvals through Foundayo (April 1, 2026) and the Ozempic tablets launch (May 4, 2026). | FDA, DailyMed, manufacturer press releases |
| May 4, 2026 | Ozempic tablets | Launched in U.S. retail pharmacies. | Novo Nordisk press release, May 1, 2026 |
| April 1, 2026 | Foundayo | First non-peptide oral GLP-1 approved. | FDA press announcement |
| March 19, 2026 | Wegovy HD | Higher-dose Wegovy 7.2 mg approved. | FDA press announcement |
| February 4, 2026 | Ozempic tablets | Approved as U.S. brand replacement for Rybelsus. | Novo Nordisk press release |
| December 22, 2025 | Wegovy tablets | First oral GLP-1 for chronic weight management approved. | Novo Nordisk press release |
| December 2025 | Mounjaro | Pediatric T2D indication added (ages 10+). | FDA prescribing information revision |
| August 15, 2025 | Wegovy injection | MASH (F2-F3 fibrosis) indication added under accelerated approval. | FDA press announcement |
| January 28, 2025 | Ozempic injection | CKD-progression indication added. | Novo Nordisk press release |
Next refresh target: Mounjaro MACE indication (pending FDA decision), Foundayo type 2 diabetes filing (expected end of Q2 2026), Ozempic tablets 25 mg for T2D (expected end of 2026).
Who created this page
This page was written and verified by the Rx Index editorial team. We're a U.S.-based research team that maintains independent reference pages on prescription medications. We do not prescribe, dispense, or sell any of the drugs listed on this chart. We are not a healthcare provider, and nothing on this page is medical advice.
Disclosure: The Rx Index participates in affiliate programs with telehealth providers. Affiliate relationships do not influence which drugs appear on this chart or what indications are listed. The chart is sourced to the FDA and cannot be edited for a commercial partner.
Have a correction? If you spot something on this chart that doesn't match a current FDA label, please write to corrections at therxindex.com with the discrepancy and your source. We'll review and re-verify within 7 days.
Related guides on The Rx Index
- GLP-1 FDA Approvals Timeline (2005–2026) — full chronological history
- Best GLP-1 for Diabetes — decision guide for type 2 diabetes
- Best GLP-1 for PCOS — off-label conversation for women with PCOS
- Is GLP-1 Safe? The 2026 Safety Guide
- GLP-1 Cost Without Insurance
- Best Brand-Name GLP-1 Providers
- Does FEHB Cover Zepbound? 2026 Plan-by-Plan Guide
Sources cited on this page
DailyMed (NLM): Current prescribing information for Ozempic injection, Ozempic tablets/Rybelsus, Wegovy, Mounjaro, Zepbound, Foundayo, Trulicity, Victoza, Saxenda, Soliqua.
FDA press announcements: Foundayo (April 1, 2026), Wegovy HD (March 19, 2026), Wegovy MASH (August 15, 2025), Zepbound OSA (December 20, 2024), First generic liraglutide (December 23, 2024).
FDA compounding and safety: FDA's concerns about unapproved GLP-1 drugs; FDA clarification of compounder policies; FDA shortage resolution document; FDA exenatide pediatric postmarketing review.
Major peer-reviewed trials: SUSTAIN-6, REWIND, LEADER, STEP 1, STEP UP, OASIS 4, ESSENCE, SURMOUNT-OSA, SURPASS-PEDS, ATTAIN-1, FLOW, SELECT, SURPASS-CVOT.
This page is a regulatory reference, not medical advice. FDA approvals change. Last verified May 12, 2026. If you're reading this more than 90 days after that date, check the update log above or contact your pharmacist or prescriber for current label information.
Published: · Last reviewed:
Last verified: May 12, 2026.