Best Tirzepatide for Fatty Liver: 5 Paths [2026 FDA Verdict]
Last verified: May 12, 2026 — The RX Index Editorial Team
Sources: SYNERGY-NASH trial (NEJM 2024, updated September 2025), ESSENCE trial (NEJM 2025), AASLD November 2025 update, FDA prescribing labels for Zepbound, Mounjaro, Wegovy, and Rezdiffra, ClinicalTrials.gov NCT04166773 and NCT07189468, FDA enforcement announcements (March 2026, February 2026).
This page is educational and is not medical advice. Prescription eligibility, safety, and coverage depend on clinician review, diagnosis, medication history, and plan rules. We may earn commissions on some links; that never changes the ranking. Full editorial and affiliate policy linked in the footer.
Affiliate disclosure: The RX Index earns a commission when you sign up with some of the providers mentioned on this page. It does not affect what you pay, and it never determines our rankings or which providers we cover. Read the full disclosure.

Best Tirzepatide for Fatty Liver: The 60-Second Answer
The best tirzepatide for fatty liver is FDA-approved brand-name tirzepatide: Zepbound if your reason is weight management or sleep apnea, or Mounjaro if your reason is type 2 diabetes. Both contain the same molecule. The 15 mg dose hit 73.3% MASH resolution without worsening fibrosis in SYNERGY-NASH, vs 13.2% on placebo.
But here is what most pages bury and we won't: tirzepatide is not FDA-approved for fatty liver, MASLD, or MASH as of May 2026. The only GLP-1 with that approval is Wegovy injection (semaglutide), approved August 2025 for noncirrhotic MASH with F2–F3 fibrosis. There is also a non-GLP-1 option, Rezdiffra (resmetirom), approved March 2024 for the same liver indication.
Which path is right for you depends on three things: your fibrosis stage, whether you also have obesity or diabetes, and whether you have insurance. We walk you through every situation below, with the actual numbers, the real prices, and the honest tradeoffs.
If you already know you want to move and just need the eligibility check, Ro carries brand-name GLP-1 options including Zepbound and Wegovy injection through the Ro Body program, runs insurance verification in the intake, and supports prior authorization. Note: Ro does not currently offer Mounjaro through Ro Body, so fatty liver + type 2 diabetes readers who specifically want Mounjaro should use their PCP or endocrinologist.
The 60-Second Route Matrix: Find Your Situation
The right tirzepatide path depends on your fibrosis stage, whether you also have obesity, type 2 diabetes, or obstructive sleep apnea, and how you are paying. The matrix below maps seven common situations to the best first conversation, based on FDA approval status and real-world access paths as of May 12, 2026.
| If this is you | Best first conversation | Why |
|---|---|---|
| Fatty liver + obesity or overweight (BMI ≥27 with a related condition) — and you want tirzepatide | Zepbound through a brand-name GLP-1 provider | FDA-approved for weight management. SYNERGY-NASH liver data is a real bonus, but liver isn't on the label. |
| Fatty liver + type 2 diabetes | Mounjaro through your endocrinologist, PCP, or diabetes-care telehealth | Same molecule as Zepbound. FDA-approved for T2D in adults and pediatric patients 10+. Insurance more likely to cover. |
| Fatty liver + moderate-to-severe sleep apnea (and obesity) | Zepbound under the OSA indication | Zepbound got the OSA approval in December 2024. Often an easier insurance path than weight-loss-only. |
| Confirmed noncirrhotic MASH with F2–F3 fibrosis (FibroScan, MRE, or biopsy) | Wegovy injection or Rezdiffra | These are the only two FDA-approved MASH treatments. Wegovy injection is the GLP-1; Rezdiffra is not. |
| Cirrhosis (F4), jaundice, ascites, confusion, or worsening labs | Hepatologist or GI specialist — first, before any GLP-1 decision | This is not a telehealth decision. Don't let any page tell you otherwise. |
| Just heard of 'fatty liver' — no fibrosis testing yet | Get FIB-4 calculated from your existing labs, then a FibroScan if it's intermediate or high | You need a sense of your stage before picking a liver-specific path. We have the FIB-4 formula below. |
| Considering compounded tirzepatide because of cost | Read our compounded section first | Compounded GLP-1s are not FDA-approved. For a progressive liver condition, the FDA-approved path matters more than usual. |
If your row points to Zepbound or Wegovy injection, the cleanest next step is checking eligibility with a provider that carries both and runs insurance verification before the visit.
Sponsored affiliate link. Ro is a telehealth provider that carries Zepbound and Wegovy injection through Ro Body, plus insurance verification and prior-authorization support. Ro does not currently offer Mounjaro through Ro Body.
The Honest Truth: Tirzepatide Is Not FDA-Approved for Fatty Liver
As of May 2026, tirzepatide is FDA-approved for type 2 diabetes (Mounjaro), chronic weight management (Zepbound), and moderate-to-severe obstructive sleep apnea with obesity (Zepbound). It is not FDA-approved for fatty liver disease, MASLD, MASH, or NASH.
We are putting this above any pitch because pages that hide this fact are everywhere, and a few are flat wrong. We have seen large health sites claim tirzepatide is already FDA-approved for MASH. As of the date on this page, that is not true.
What tirzepatide is approved for today
- Mounjaro: type 2 diabetes in adults and pediatric patients 10 years and older (approved May 13, 2022; pediatric indication added later)
- Zepbound: chronic weight management in adults with obesity (BMI ≥30) or overweight (BMI ≥27) plus a weight-related condition (approved November 8, 2023)
- Zepbound: moderate-to-severe obstructive sleep apnea in adults with obesity (approved December 2024)
What is actually FDA-approved for fatty liver disease (MASH)
- Rezdiffra (resmetirom) — first MASH drug ever approved, March 14, 2024. Not a GLP-1. Works through the thyroid hormone receptor beta.
- Wegovy injection (semaglutide 2.4 mg) — second MASH drug approved, August 2025. The only GLP-1 with this indication. Note: this is the injection; Wegovy tablets have separate obesity/cardiovascular labeling.
What is coming
Eli Lilly's Phase 3 SYNERGY-OUTCOMES trial for tirzepatide in MASH started enrolling in October 2025 (ClinicalTrials.gov NCT07189468). Results are expected around 2027. If the Phase 3 confirms the Phase 2 numbers, tirzepatide will likely get a MASH approval after that. But “likely to be approved” and “is approved” are different things — and they lead to very different insurance conversations.
Why this fact actually matters to you
Three real reasons:
- Insurance. Your insurer reviews the diagnosis code on your prescription. “Fatty liver” or “MASH” as the indication for tirzepatide gets denied because it is off-label. The same medication prescribed for obesity (Zepbound) or T2D (Mounjaro) — diagnoses you also have — gets covered far more often.
- Your doctor's comfort level. Some clinicians won't prescribe off-label for liver disease. They will route you to Wegovy injection or Rezdiffra because those have the indication.
- Compounded products. When someone tells you compounded tirzepatide is “clinically proven for fatty liver,” they are combining two things that don't go together. The SYNERGY-NASH trial used Eli Lilly's brand tirzepatide. Compounded products were not what was studied.
What SYNERGY-NASH Actually Showed (Dose by Dose)
The full SYNERGY-NASH results table
| Endpoint at 52 weeks | Placebo | Tirzepatide 5 mg | Tirzepatide 10 mg | Tirzepatide 15 mg |
|---|---|---|---|---|
| MASH resolution without worsening fibrosis | 13.2% | 51.8% | 62.8% | 73.3% |
| ≥1-stage fibrosis improvement without MASH worsening | 32.8% | 59.1% | 53.3% | 54.2% |
| Both endpoints achieved | ~10% | 32% | 33% | 39% |
| Mean weight loss | −0.1% | −10.7% | −13.0% | −15.6% |
Table uses the efficacy-estimand percentages reported by Eli Lilly for SYNERGY-NASH. Source: Loomba R, Hartman ML, et al. Tirzepatide for Metabolic Dysfunction-Associated Steatohepatitis with Liver Fibrosis. N Engl J Med 2024;391(4):299-310. Published June 8, 2024; updated September 4, 2025. Funded by Eli Lilly. NCT04166773. These are not head-to-head results against ESSENCE or MAESTRO-NASH.
What stands out from this data
The 15 mg dose produced the highest MASH resolution rate among the major GLP-1 and GIP/GLP-1 MASH results compared in this guide. 73.3% beats Wegovy injection's Phase 3 ESSENCE number (62.9%). It also beats Rezdiffra's MAESTRO-NASH Phase 3 numbers (25.9% at 80 mg, 29.9% at 100 mg).
But — and this is the part the headlines skip — these are cross-trial comparisons in different populations with different study designs. They are not the same as head-to-head data. There has not been a randomized comparison of tirzepatide vs Wegovy injection vs Rezdiffra for fatty liver.
The fibrosis improvement signal is real but more modest. All three doses produced about 53–59% fibrosis improvement, vs 32.8% on placebo. That is clinically meaningful — fibrosis is what predicts liver-related death and transplant — but the study was not powered to detect this endpoint.
A post-hoc subgroup analysis published in JHEP Reports (June 2025) found that patients with more advanced fibrosis or higher MASH activity responded better at 10 mg and 15 mg than at 5 mg. If your liver is in worse shape, titrating up may matter more.
Honest limitations of SYNERGY-NASH
- It was a Phase 2 trial with 190 patients. ESSENCE (the Wegovy injection MASH trial) had 800 participants in its interim analysis.
- 52 weeks is short. Long enough to measure histologic change. Too short to measure cirrhosis progression, liver transplant, or death.
- The trial used Eli Lilly's branded tirzepatide. These results do not apply to compounded versions.
- Most patients in the trial were on the maximum tolerated dose by week 24. Real-world dose escalation usually goes slower because of GI side effects.
Which Tirzepatide Brand and Dose Is Best for Your Fatty Liver?
Mounjaro vs Zepbound: same molecule, different routes
| Factor | Mounjaro | Zepbound |
|---|---|---|
| Molecule | Tirzepatide | Tirzepatide (same) |
| Manufacturer | Eli Lilly | Eli Lilly |
| FDA indication | Type 2 diabetes (adults, pediatric 10+) | Chronic weight management; moderate-to-severe OSA with obesity |
| Adult dose strengths | 2.5, 5, 7.5, 10, 12.5, 15 mg | 2.5, 5, 7.5, 10, 12.5, 15 mg |
| Insurance path | T2D diagnosis (HbA1c ≥6.5% or fasting glucose ≥126 on two occasions) | Obesity (BMI ≥30) or overweight + comorbidity; or OSA with AHI ≥15 |
| Self-pay option | No direct LillyDirect cash-pay equivalent | LillyDirect Zepbound Self Pay Journey Program ($299–$449/mo) |
What dose works best?
The SYNERGY-NASH data shows a clear dose-response. Higher doses produced higher MASH resolution rates: 51.8% at 5 mg, 62.8% at 10 mg, and 73.3% at 15 mg. But you don't start at 15 mg. The FDA-approved titration goes like this:
| Timeline | Dose | Note |
|---|---|---|
| Weeks 1–4 | 2.5 mg weekly | Starting dose only, not maintenance |
| Weeks 5–8 | 5 mg weekly | |
| Weeks 9–12 | 7.5 mg weekly | |
| Weeks 13–16 | 10 mg weekly | Most patients tolerate up to here |
| Weeks 17–20 | 12.5 mg weekly | |
| Week 21+ | 15 mg weekly | Maximum dose; highest MASH resolution (73.3%) |
Most patients tolerate up to 10 mg. The 15 mg dose has more nausea, diarrhea, and vomiting. Your doctor will hold you at whatever dose you tolerate.
Practical translation for fatty liver: If your fibrosis is more advanced (F3) or your MASH is more active, and you can get to the higher doses, the trial data says you will likely get more benefit. This is a clinician decision, not a self-titration decision. Bring the SYNERGY-NASH dose data to your appointment as informed-patient context, not a target you set yourself.
The FIB-4 Quick Check: Do You Actually Have Significant Liver Scarring?
The FIB-4 Formula
FIB-4 = (Age × AST) ÷ (Platelets × √ALT)
Pull out your most recent labs. You will need your age, AST (aspartate aminotransferase), ALT (alanine aminotransferase), and platelet count (from your CBC). If you don't have these yet, ask your doctor for a liver function panel and a CBC. Both are routine.
| FIB-4 score | What it typically means | Suggested next step |
|---|---|---|
| Under 1.3 | Low risk of significant fibrosis | Routine monitoring. Weight management. Lifestyle changes. Talk to your doctor about whether GLP-1 therapy fits your overall picture. |
| 1.3 to 2.67 | Indeterminate risk | Get a FibroScan (VCTE), MRE, or ELF blood test. Don't treat tirzepatide as a liver-protection shortcut until you know your stage. |
| Over 2.67 | High risk of advanced fibrosis | Request hepatology referral. Discuss FDA-approved MASH treatment options (Wegovy injection or Rezdiffra) along with GLP-1 weight management. |
FIB-4 thresholds and interpretation per AASLD Practice Guidance. This is a screening tool, not a diagnosis. A FIB-4 score does not replace a FibroScan, MRE, ELF blood test, or biopsy.
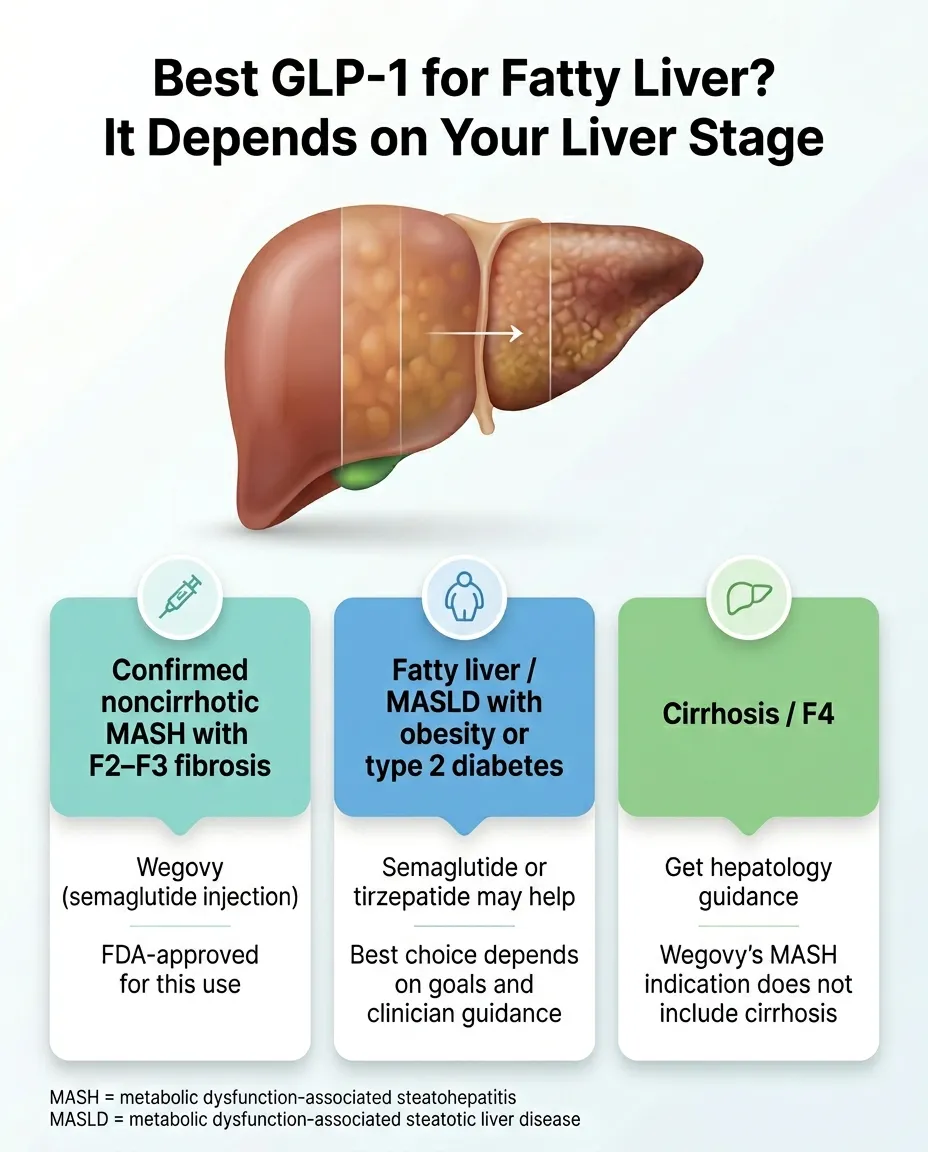
What this means for your tirzepatide decision: If your FIB-4 is low and you have obesity or overweight, you are in the “GLP-1 for weight management with fatty liver as a bonus” group. Zepbound makes a lot of sense.
If your FIB-4 is intermediate or high, you may still qualify for a GLP-1 through obesity, OSA, or diabetes — but you might also be a better fit for FDA-approved Wegovy injection or Rezdiffra. Get staged first.
If your FIB-4 is high enough to suggest cirrhosis, this is not a telehealth question. See a hepatologist.
Mounjaro vs Zepbound vs Compounded vs Wegovy Injection vs Rezdiffra: The Full Comparison
For fatty liver, your real options come down to four drugs across five access paths. Wegovy injection and Rezdiffra are FDA-approved specifically for noncirrhotic MASH with F2–F3 fibrosis. Mounjaro and Zepbound (both tirzepatide) are not approved for fatty liver but have strong Phase 2 evidence. Compounded tirzepatide has no FDA approval and was not what SYNERGY-NASH studied.
| Drug | Brand | FDA-approved for MASH? | Strongest fatty-liver evidence | Avg weight loss | Cash-pay price (US) | Access path |
|---|---|---|---|---|---|---|
| Tirzepatide | Zepbound | × No | SYNERGY-NASH Phase 2: 73.3% MASH resolution at 15 mg | ~15.6% at 15 mg | LillyDirect Self Pay: $299/mo (2.5 mg), $399 (5 mg), $449 (7.5–15 mg with offer terms) | Insurance PA (obesity or OSA); LillyDirect self-pay; brand-name telehealth |
| Tirzepatide | Mounjaro | × No | Same molecule as Zepbound; SYNERGY-NASH evidence applies | Comparable | List similar to Zepbound; diabetes-route insurance typically broader | Endocrinologist, PCP, or diabetes-care telehealth with insurance PA citing T2D |
| Semaglutide | Wegovy injection | \u2713 Yes (Aug 2025) | ESSENCE Phase 3 (n=800 interim): 62.9% MASH resolution at 72 weeks | ~10.5% | ~$1,349/mo list; NovoCare savings programs reduce | Strongest insurance path for MASH; PA citing MASH (K75.81) |
| Resmetirom | Rezdiffra | \u2713 Yes (Mar 2024) | MAESTRO-NASH Phase 3: 25.9–29.9% MASH resolution at 80/100 mg | None (not a weight-loss drug) | ~$47,400/yr list (~$3,950/mo) | Specialty pharmacy; hepatology-led prescription |
| Compounded tirzepatide | varies | × No | Not studied for MASH. SYNERGY-NASH used brand Lilly tirzepatide. | Variable; not FDA-evaluated | ~$179–$299/mo typical cash | Compounding-focused telehealth providers |
All pricing reflects publicly listed US prices verified directly on manufacturer and provider websites on May 12, 2026. Out-of-pocket cost varies based on insurance, savings programs, and pharmacy. We re-verify quarterly and on major regulatory or pricing changes.
What this table tells you
- If your liver is your primary reason and you have insurance: Wegovy injection has the FDA approval for noncirrhotic MASH with F2–F3 fibrosis. That is the path with the easiest prior authorization.
- If you also want maximum weight loss and have obesity or OSA: Zepbound's SYNERGY-NASH numbers are higher than Wegovy injection's ESSENCE numbers at the highest dose, and Zepbound produces more total weight loss. The catch: liver is not on Zepbound's label.
- If you have type 2 diabetes: Mounjaro is the same molecule as Zepbound with the T2D indication. Insurance coverage for T2D is typically broader and easier than for obesity.
- If your fibrosis is more advanced and weight loss is not your goal: Rezdiffra is the non-GLP-1, liver-specific FDA-approved option. At ~$47,400/yr list, it requires hepatology involvement, but for someone whose main problem is liver scarring without metabolic disease, it has a place in the conversation.
Sponsored affiliate links. Ro evaluates you for Zepbound or Wegovy injection; Sesame lists self-pay GLP-1 prices.
The One Honest Tradeoff With Tirzepatide for Fatty Liver
Tirzepatide is one of the most promising medications being studied for MASH — and it is not the FDA-approved fatty-liver answer today. That gap matters most for people with already-diagnosed F2–F3 MASH who have commercial insurance, because for them, Wegovy injection or Rezdiffra has an easier prescription and coverage path.
If you have F2–F3 MASH plus insurance, Wegovy injection is the cleaner liver-specific path, full stop. If that is your priority, ask about Wegovy injection first. But if you also have significant obesity, sleep apnea, or type 2 diabetes — and most people with MASH do — tirzepatide produces more weight loss than semaglutide in head-to-head comparison data, and that weight loss is what is driving most of the liver improvement either way.
Both paths are legitimate. Your fibrosis stage, your other conditions, and your insurance decide which one fits.
If you are more in the “I have F2–F3 MASH and want the FDA-approved liver path” group, read our companion guide: Best GLP-1 for Fatty Liver in 2026 → It leads with Wegovy injection and goes deeper on the semaglutide side of the decision.
Sponsored affiliate link. Ro evaluates you for both FDA-approved options and routes you to the one that fits your stage and insurance.
How to Actually Get FDA-Approved Tirzepatide for Fatty Liver (Your 3 Paths)
Ro (our primary recommendation for fatty-liver readers)
Ro carries both Zepbound and Wegovy injection through Ro Body, runs insurance verification built into intake, and supports prior authorization paperwork. It also offers a free GLP-1 Insurance Coverage Checker without starting an account.
- Membership pricing: $39 first month, then as low as $74/month (annual) or $149/month (monthly). Medication cost is separate.
- Best for: confirmed F2–F3 MASH readers wanting Wegovy injection PA support, obesity + fatty liver readers wanting Zepbound with insurance help, readers comparing both options.
- One real-world note: Ro does not currently offer Mounjaro through Ro Body.
Sponsored affiliate link. Opens in a new tab.
Sesame Care (if you want provider choice or transparent self-pay pricing)
Sesame lists Success by Sesame at $59/month with annual subscription, medication separate, and publishes current cash-pay prices for Wegovy injection, Zepbound KwikPen, and other GLP-1 options.
Sponsored affiliate link.
LillyDirect self-pay for Zepbound
If you don't have insurance, or your plan excludes weight-loss medication entirely, LillyDirect is the FDA-approved cash-pay option directly from Eli Lilly.
| Dose | Self Pay Journey Program price | Regular cash price |
|---|---|---|
| 2.5 mg | $299/month | (starting dose, not maintenance) |
| 5 mg | $399/month | |
| 7.5 mg | $449/month (terms met) | $499/month |
| 10 mg | $449/month (terms met) | $699/month |
| 12.5 mg | $449/month (terms met) | $699/month |
| 15 mg | $449/month (terms met) | $699/month |
LillyDirect requires a valid prescription from a licensed clinician and ships directly to your door. Verify current pricing at LillyDirect →
Your existing doctor (PCP, endocrinologist, or hepatologist)
If you already have a clinician managing your fatty liver, or if you have type 2 diabetes and need Mounjaro specifically, bring everything in this guide to your next appointment. Print or screenshot this list:
- What is my current fibrosis stage? Do I have a FibroScan, FIB-4, ELF, or biopsy result on file?
- If I am in F2–F3, are we considering Wegovy injection or Rezdiffra (the FDA-approved options) before discussing tirzepatide?
- If I have obesity, would Zepbound under the obesity indication be appropriate given my SYNERGY-NASH-relevant fibrosis level?
- If I have T2D, would Mounjaro be the right brand to start with?
- What dose are we targeting and over what timeline?
- What liver labs do you want before I start, and how often will we recheck?
- Do I have any contraindications — MTC family history, MEN2, history of pancreatitis, pregnancy plans?
- If we go with off-label tirzepatide for the liver benefit, will you support a prior authorization with a letter of medical necessity citing SYNERGY-NASH?
Should you use compounded tirzepatide for fatty liver?
Direct answer: not as your first choice for fatty liver.
Compounded tirzepatide is not FDA-approved, has not been evaluated by the FDA for safety or efficacy, and the SYNERGY-NASH trial used Eli Lilly's branded tirzepatide — those results do not apply to compounded products.
- In March 2026, the FDA warned 30 telehealth companies about illegal marketing of compounded GLP-1s, including claims implying sameness with FDA-approved products.
- The FDA proposed excluding semaglutide, tirzepatide, and liraglutide from the 503B bulks list. If finalized, that would restrict outsourcing facilities from compounding these drugs from bulk substances.
- The FDA issued a February 2026 warning letter to MEDVi concerning false or misleading claims about compounded semaglutide and tirzepatide products.
For a progressive liver condition, the FDA-approved path is the one we would guide a family member toward first, every time. If you want to compare compounded options honestly, see the broader compounded GLP-1 provider comparison →
Will Insurance Cover Tirzepatide for Fatty Liver?
The brand-name + indication strategy
| Drug | Diagnosis needed | Documentation | Coverage notes |
|---|---|---|---|
| Mounjaro | Type 2 diabetes (ICD-10 E11.x) | HbA1c ≥6.5%, or fasting glucose ≥126 mg/dL on two separate occasions | Generally accessible on commercial plans; covered on most Medicare Part D formularies |
| Zepbound (obesity) | Obesity (E66.x) or overweight with comorbidity | BMI ≥30, or BMI ≥27 + weight-related condition (hypertension, dyslipidemia, T2D) | Variable; many require step-therapy; many plans exclude weight-loss meds entirely |
| Zepbound (OSA) | Moderate-to-severe OSA (G47.33) + obesity | Sleep study with AHI ≥15 | Often easier than obesity indication. If you have OSA and obesity, this may be the smarter PA path. |
| Wegovy injection | Noncirrhotic MASH with F2–F3 fibrosis (K75.81) or obesity or CVD | FibroScan, MRE, ELF, or biopsy showing F2–F3; AASLD Nov 2025 guidance | MASH indication is new (Aug 2025); PA reviewers are still learning it. Cite AASLD guidance in letter of medical necessity. |
The step-by-step PA strategy for fatty-liver readers
- Get your fibrosis stage confirmed. Request a FibroScan, MRE, ELF blood test, or biopsy if you haven't had one. For PA purposes, results within the last year are usually accepted.
- Identify which on-label condition you also have. You probably have at least one of: T2D, obesity, overweight + comorbidity, or moderate-to-severe OSA.
- Pick the brand and indication that fits. Mounjaro for T2D, Zepbound for obesity or OSA, Wegovy injection for MASH (if you're F2–F3).
- Make sure your medical record codes match. 'Fatty liver' or 'elevated enzymes' is too vague. 'MASH' with the K75.81 code, 'obesity' with the right BMI code, or 'T2D' with HbA1c numbers attached.
- Submit the PA. Your doctor or telehealth provider handles this. If they ask you to follow up with your insurer, do it.
- If denied, appeal. Appeals can overturn GLP-1 denials when the diagnosis, documentation, and formulary criteria line up. Include the letter of medical necessity and the relevant trial data.
The 2026 insurance landscape
- CVS Caremark removed Zepbound from its Standard Control, Advanced Control, and Value formularies effective July 1, 2025.
- Health New England dropped GLP-1 medication coverage for weight loss for Individual and Small Group members effective January 1, 2026.
- The Medicare GLP-1 Bridge is scheduled to run from July 1, 2026 through December 31, 2027 with a $50 copay, covering Foundayo, Wegovy injection/tablets, and Zepbound KwikPen. This is not a blanket MASH coverage pathway.
- TrumpRx is live as of May 2026 listing Zepbound at $299/month.
For Medicare details, see our guide: Medicare GLP-1 Bridge Program: Eligibility, Cost, and Coverage
When tirzepatide PA fails and Wegovy injection is the right pivot
If your Zepbound PA gets denied and you have confirmed F2–F3 MASH, ask your provider to switch to Wegovy injection with the MASH indication. The PA path is materially easier — the indication is literally on the label.
Sponsored affiliate link. Ro's clinical team supports eligibility verification and prior-authorization paperwork as part of the membership.
Tirzepatide vs Wegovy Injection for Fatty Liver: When Each One Wins
The head-to-head comparison (with the cross-trial caveat)
| Dimension | Wegovy injection (semaglutide 2.4 mg) | Tirzepatide (Zepbound / Mounjaro) |
|---|---|---|
| FDA-approved for noncirrhotic MASH F2–F3? | ✓ Yes (Aug 2025) | ✗ No (Phase 3 SYNERGY-OUTCOMES underway; results ~2027) |
| Strongest trial | ESSENCE Phase 3, n=800 interim | SYNERGY-NASH Phase 2, n=190 |
| MASH resolution rate | 62.9% vs 34.3% placebo | 51.8% / 62.8% / 73.3% by dose vs 13.2% placebo |
| Fibrosis improvement rate | 36.8% vs 22.4% placebo | 53–59% vs 32.8% placebo |
| Average weight loss | ~10.5% | ~10.7% to 15.6% by dose |
| Mechanism | GLP-1 receptor agonist (single) | Dual GIP + GLP-1 receptor agonist |
| Injection frequency | Once weekly | Once weekly |
| Liver-indication insurance path | Strongest (FDA-approved for MASH) | Off-label for liver; on-label for T2D / obesity / OSA |
Which one wins for which person
| If you are… | Best path |
|---|---|
| Confirmed noncirrhotic F2–F3 MASH + commercial insurance | Wegovy injection — FDA-approved liver indication, easier PA |
| Obesity + fatty liver, weight loss is the priority | Zepbound — SYNERGY-NASH liver benefit + bigger weight loss |
| Type 2 diabetes + fatty liver | Mounjaro — T2D coverage is easiest; same molecule as Zepbound |
| Moderate-to-severe sleep apnea + obesity + fatty liver | Zepbound under the OSA indication |
| Simple MASLD with F0–F1 fibrosis (no significant scarring) | Either works. Pick what your insurance covers. Weight loss is what matters. |
| Cirrhosis (F4) | Hepatologist first. Not a telehealth decision. |
| Unsure of your stage | Get FIB-4 + FibroScan first. Then decide. |
Sponsored affiliate link. Ro carries Zepbound and Wegovy injection through Ro Body and supports prior authorization for either.
Is Tirzepatide Safe for Your Liver?
What typically happens to your liver on tirzepatide
In SYNERGY-NASH, all three tirzepatide doses significantly reduced ALT and AST compared to placebo. Imaging measures of liver fat (MRI-PDFF) showed substantial reductions. The mechanism: weight loss reduces visceral and hepatic fat, insulin sensitivity improves, systemic inflammation drops. The dual GIP/GLP-1 action also has direct effects on adipose tissue that may benefit the liver beyond weight loss alone.
The rare exception
Drug-induced liver injury with tirzepatide is rare but has been reported. Michigan Medicine published a case report of tirzepatide-associated hepatitis with jaundice — the first known case. The NIH's LiverTox database notes isolated reports for both semaglutide and tirzepatide. The Zepbound and Mounjaro labels do not require routine liver-enzyme monitoring for every patient; for fatty-liver readers, baseline and follow-up labs make sense.
The gallstone risk during rapid weight loss
Rapid weight loss — from any cause including GLP-1 medications, bariatric surgery, or aggressive dieting — can trigger gallstone formation. Gallstones can occasionally cause cholestasis, which affects liver enzymes. This is a monitoring issue during the first 6–12 months. If you experience right-upper-quadrant abdominal pain, nausea after fatty meals, or jaundice, talk to your doctor.
Absolute contraindications — do not start tirzepatide if you have:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2 (MEN2)
- Known serious hypersensitivity to tirzepatide or any product component
Have a separate clinician conversation before starting if you have:
- Current pregnancy, plans for pregnancy, or breastfeeding
- History of pancreatitis
- Severe gastroparesis
- Active gallbladder disease
- History of severe hypoglycemia (especially if on insulin or sulfonylureas)
- Diabetic retinopathy (if you have T2D)
Real Patient Experiences (And Honest Limitations)
We don't fabricate testimonials and we don't use composite case studies. The community discussion themes below come from public Reddit, forum, and review content (r/Zepbound, r/Mounjaro, r/FattyLiver, r/PCOS). They are used here for voice and decision-friction insight, not as evidence for medical claims.
Common themes in real patient experiences
- Liver enzyme anxiety — readers consistently want to know if their ALT and AST will come down, and how fast
- FibroScan number improvement — anecdotally common, but variable, and patients want a clearer 'what's a good change' framework
- Insurance workarounds — when 'fatty liver' alone gets denied, patients consistently report re-coding under T2D, obesity, or OSA and getting approved
- Mounjaro vs Zepbound confusion — patients regularly mix up which brand goes with which indication
- 'Worth it through the nausea' — most common framing once past titration weeks 4–8
What tirzepatide will NOT do for your liver
- Will not work without lifestyle support. Weight loss is the engine. The medication is the accelerator.
- Will not reverse cirrhosis (F4). Current data is in F2–F3. Cirrhosis requires different management.
- Will not work if alcohol is a co-driver. Alcohol cessation is non-negotiable.
- Will not last after you stop. Weight tends to return, and liver fat can follow. Most patients on GLP-1s stay on them long-term.
- Will not produce overnight results. Liver enzymes improve over weeks to months. Histologic change at a year or more.
Frequently Asked Questions
Is tirzepatide FDA-approved for fatty liver disease?
No. As of May 2026, tirzepatide is FDA-approved for type 2 diabetes (Mounjaro, in adults and pediatric patients 10 years and older), chronic weight management (Zepbound), and moderate-to-severe obstructive sleep apnea in adults with obesity (also Zepbound). The Phase 3 SYNERGY-OUTCOMES trial for MASH started in October 2025; results are expected around 2027. The only GLP-1 with FDA approval for noncirrhotic MASH with F2-F3 fibrosis is Wegovy injection (semaglutide 2.4 mg), approved August 2025.
Is Zepbound the best tirzepatide for fatty liver?
Zepbound is the best tirzepatide for you if your prescribing reason is weight management or obstructive sleep apnea -- and it produced the highest MASH resolution rate of any GLP-1 in a randomized trial (73.3% at 15 mg in SYNERGY-NASH). But 'best for fatty liver' depends on your full picture. If you have confirmed F2-F3 MASH and insurance, Wegovy injection has the FDA-approved liver indication and is generally easier to get covered.
Is Mounjaro or Zepbound better for fatty liver?
Mounjaro and Zepbound contain the exact same molecule -- tirzepatide -- at the same adult dose strengths. The difference is the FDA-approved indication: Mounjaro for type 2 diabetes, Zepbound for chronic weight management and OSA. For fatty liver specifically, both work the same way because they're the same drug. Which one you take depends on which condition you also qualify for and which one your insurance covers.
Can I take tirzepatide if my ALT or AST is high?
This is a clinician decision. Elevated ALT or AST can come from MASLD/MASH, alcohol, viral hepatitis, medications, gallbladder disease, or other causes. Bring ALT, AST, platelets, A1C, medication list, alcohol pattern, and any ultrasound, FibroScan, or MRE results so the provider can decide whether to start, monitor, stage further, or refer to a specialist.
What's the best dose of tirzepatide for fatty liver?
In SYNERGY-NASH, the 15 mg dose produced the highest MASH resolution rate (73.3%), followed by 10 mg (62.8%) and 5 mg (51.8%). Higher doses produced larger liver benefits but also more GI side effects. Real-world dosing starts at 2.5 mg and titrates up every 4 weeks to the highest dose you tolerate. Most patients tolerate up to 10 mg comfortably; 15 mg has more nausea.
Can tirzepatide reverse fatty liver?
SYNERGY-NASH showed that tirzepatide can resolve MASH (the inflammatory subset of fatty liver) without worsening fibrosis in up to 73.3% of patients at 15 mg over 52 weeks. Fibrosis improvement was 53-59% across doses vs 32.8% on placebo. 'Reversal' is the wrong word -- 'resolution' and 'improvement' are the trial endpoints. Sustained treatment plus lifestyle change is what produces sustained benefit.
How long does it take tirzepatide to improve fatty liver?
Trial and imaging studies show improvements in liver enzymes and liver-fat measures over weeks to months. Histologic MASH resolution and fibrosis improvement were measured at 52 weeks in SYNERGY-NASH -- that's the typical timeline for major structural change.
Will my insurance cover tirzepatide for fatty liver?
Generally not for the indication 'fatty liver' alone, because tirzepatide is not FDA-approved for MASH. Coverage is typically granted when tirzepatide is prescribed for a condition you also have that is on the label: Mounjaro for T2D, Zepbound for obesity (BMI 30+, or 27+ with weight-related comorbidity), or Zepbound for moderate-to-severe OSA. Wegovy injection is the GLP-1 with the MASH indication on its label and is the strongest insurance path when your liver is the primary reason.
Is compounded tirzepatide the same as Mounjaro or Zepbound?
No. Compounded tirzepatide is a non-FDA-approved formulation made by compounding pharmacies. It has not been evaluated by the FDA for safety, efficacy, or manufacturing quality. The SYNERGY-NASH trial used Eli Lilly's branded tirzepatide. Those results do not apply to compounded products. We do not describe compounded tirzepatide as clinically proven for any indication, including fatty liver.
Does tirzepatide damage the liver?
No, the opposite is the typical pattern -- liver enzymes improve on therapy in most patients. Tirzepatide has shown a favorable hepatic safety profile across its trials. Rare cases of drug-induced liver injury have been reported, which is why your clinician may want baseline and follow-up liver labs given your underlying fatty-liver context.
Should I switch from Wegovy injection or Ozempic to tirzepatide for my fatty liver?
Probably not, if you're responding well -- especially if you're on Wegovy injection with documented MASH, since it's the FDA-approved option for that indication. If you're on Ozempic for T2D and your liver labs and imaging are stable or improving, the semaglutide is likely helping. Switching should be a clinical decision driven by inadequate weight loss, blood sugar control, or tolerability -- not by chasing slightly higher numbers in a different trial.
I have cirrhosis (F4) -- can I take tirzepatide?
This is a hepatology decision, not a telehealth decision. Wegovy injection and tirzepatide trials excluded patients with cirrhosis, and Wegovy injection's MASH approval is specifically for noncirrhotic disease. If you have cirrhosis, please see a hepatologist before starting any GLP-1.
What's the cheapest way to get tirzepatide for fatty liver?
If you have insurance: prior authorization for Mounjaro (T2D), Zepbound (obesity or OSA), or Wegovy injection (MASH). Copays vary, but covered tirzepatide is usually $25-$200/month. If you don't have insurance: LillyDirect's Zepbound Self Pay Journey Program lists $299/month for 2.5 mg, $399/month for 5 mg, and $449/month for 7.5-15 mg when program terms are met. TrumpRx lists $299/month for Zepbound as of May 2026. Compounded tirzepatide is typically $179-$299/month cash but is not FDA-approved and was not studied in SYNERGY-NASH.
How We Built This Guide (Methodology)
The RX Index is an independent pricing intelligence and comparison resource for GLP-1 telehealth providers. We evaluate medications and providers by separating regulatory facts (FDA approval status), clinical trial evidence (with explicit phase and population caveats), commercial facts (verified pricing, insurance support, prior-authorization handling), and editorial judgments (which option may fit best for which reader — clearly framed as such).
What we did not do for this page:
- We did not use star ratings or aggregate 'scores' without showing how they were calculated.
- We did not fabricate testimonials, reviewers, or case studies.
- We did not invent author or medical-reviewer credentials.
- We did not feature compounded medications as winners on FDA-approved-indication questions.
- We did not blur compounded with FDA-approved tirzepatide.
- We did not let affiliate relationships influence what we said about FDA status, trial evidence, or clinical recommendations.
What we did:
- Cited primary sources for every medical and regulatory claim (FDA prescribing labels, peer-reviewed publications, ClinicalTrials.gov, AASLD practice guidance, manufacturer pages, FDA enforcement announcements).
- Used Reddit and forum content only for voice-of-customer language, never as evidence for medical claims.
- Disclosed affiliate relationships transparently.
- Verified pricing directly from manufacturer and provider websites on May 12, 2026.
Full methodology: /methodology/ · Editorial standards: /editorial-standards/ · Affiliate disclosure: /affiliate-disclosure/
What We Actually Verified for This Page
- Verified May 12, 2026: SYNERGY-NASH Phase 2 efficacy-estimand outcomes from NEJM (Loomba R, Hartman ML, et al. 2024; updated September 4, 2025) and Eli Lilly's investor release
- Verified May 12, 2026: SYNERGY-NASH subgroup analysis from JHEP Reports (June 2025)
- Verified May 12, 2026: SYNERGY-OUTCOMES Phase 3 status from ClinicalTrials.gov NCT07189468
- Verified May 12, 2026: Wegovy injection FDA approval for noncirrhotic MASH with F2–F3 fibrosis (August 2025) from the FDA prescribing label
- Verified May 12, 2026: Rezdiffra FDA approval for noncirrhotic MASH with F2–F3 fibrosis (March 14, 2024) from the FDA prescribing label
- Verified May 12, 2026: AASLD November 2025 semaglutide-for-MASH practice guidance update from aasld.org
- Verified May 12, 2026: Zepbound FDA indications and prescribing information from the FDA prescribing label (NDA 217806)
- Verified May 12, 2026: Mounjaro FDA indications including adult and pediatric (10+) T2D from the FDA prescribing label (NDA 215866)
- Verified May 12, 2026: LillyDirect Zepbound Self Pay Journey Program pricing from lilly.com/lillydirect
- Verified May 12, 2026: Ro Body pricing ($39 first month; as low as $74/month annual; $149/month monthly) from ro.co
- Verified May 12, 2026: Sesame Care Success by Sesame pricing from sesamecare.com
- Verified May 12, 2026: TrumpRx live pricing ($299/mo Zepbound) from trumprx.gov
- Verified May 12, 2026: CMS Medicare GLP-1 Bridge program details (July 1, 2026 – December 31, 2027; $50 copay) from CMS
- Verified May 12, 2026: CVS Caremark Zepbound formulary removal (July 1, 2025) from Caremark business updates
- Verified May 12, 2026: FDA March 3, 2026 announcement warning 30 telehealth companies about illegal marketing of compounded GLP-1s from FDA.gov
- Verified May 12, 2026: FDA February 20, 2026 warning letter to MEDVi from FDA.gov
Still Not Sure Which Path Is Right for You?
You have seen the dose-by-dose evidence, the FDA-status reality, the brand-by-brand comparison, the insurance strategy, the cost ranges, and the honest tradeoffs. If you are ready to move, Ro evaluates you for Zepbound or Wegovy injection, runs insurance verification, and supports prior authorization. If you still want help deciding which medication fits your specific situation, our free quiz is built for that.
Sponsored affiliate link. Free quiz has no signup.
This page provides general information based on published clinical trial evidence, FDA-approved prescribing information, and society guidance. It is not a substitute for medical advice. Talk to your clinician before starting, changing, or stopping any medication.
Affiliate disclosure: The RX Index earns a commission when you sign up with some of the providers mentioned on this page. It does not affect what you pay, and it never determines our rankings or which providers we cover. Read the full disclosure.
The RX Index is an independent pricing intelligence and comparison resource for GLP-1 telehealth providers. Last evidence review: May 12, 2026.
Related Guides
Published: · Last reviewed: