GLP-1 Adverse Event Rates Explained: What the FDA Numbers Actually Mean
Published: · Last reviewed:
By The RX Index Editorial Team · Last verified: May 14, 2026 · Informational only — not personal medical advice. Talk to a licensed clinician about your situation.

If you searched “GLP-1 adverse event rates explained,” you probably opened the FDA label, saw “nausea 44%” sitting next to “pancreatitis warning,” and felt your stomach drop. Here's the short version: across the major FDA-approved GLP-1 and related incretin medicines for weight loss — Wegovy, Zepbound, Saxenda, and Foundayo — GI side effects are common, but permanent discontinuation specifically because of GI adverse reactions ran 1.9% to 6.2% across drugs and doses in the obesity-label trial programs. That number matters more than the raw nausea percentage, and it's not on the front of the label.
This guide shows you the math behind every major symptom rate, explains why the placebo column matters more than the drug column, and tells you exactly which symptoms are green (“track it”), yellow (“call before your next dose”), and red (“urgent care now”).
What we actually verified
- ✓We pulled adverse reaction tables from current FDA prescribing information posted on DailyMed (the NIH's official source for FDA-submitted labeling): Wegovy (label with major changes through March 2026), Zepbound (updated April 22, 2026), Saxenda (updated February 25, 2026), Foundayo (initial label April 1, 2026), Ozempic, and Mounjaro.
- ✓For head-to-head detail we pulled directly from the published Phase 3 papers (STEP 1, SURMOUNT 1--5, SCALE, and the ACHIEVE/ATTAIN orforglipron programs).
- ✓We separated clinical trial rates (which have a denominator and a placebo arm) from post-marketing reports like FAERS (which don't).
- ✓We used Reddit and patient forums only to identify what real people are searching -- never as evidence for a medical claim.
No fake “medically reviewed by” badge. No invented author credentials. The byline is what it says and our editorial policy explains who reviews our content and how corrections are handled.
What does “adverse event rate” actually mean?
Quick answer: An adverse event rate is the percentage of people in a clinical trial who reported a specific symptom during the study. It is not your personal risk. It includes events the drug probably caused and events that happened during treatment but may not be related. The useful number is usually the gap between the drug group and the placebo group, not the drug number alone.
Adverse event
Any unwanted thing that happens while a person is on the drug. It might be the drug. It might be the flu. It might be coincidence.
Adverse reaction
A subset — events the trial researchers thought were probably caused by the drug. A stricter standard than adverse event.

The most important thing this page can teach you: the placebo column matters more than the drug column.
In the Wegovy trials, 44% of people on the drug reported nausea. So did 16% of people on placebo (a fake injection). The piece that's actually about the drug is the difference: 44% minus 16% = about 28 percentage points of nausea that the drug seems to be adding. We show this math for every common symptom in the master table below.
One more critical point about what these percentages mean:
The percentages you'll see are not “your odds at any given moment.” They're cumulative over the whole trial — usually 40 to 72 weeks. A 44% nausea rate means 44% of people reported nausea at some point during 16+ months of treatment, not that 44% of people are nauseous on any given day.
GLP-1 adverse event rates explained: the master FDA label table
Quick answer: Below are the most common adverse event rates from each drug's current FDA label, with the placebo rate next to it, and the absolute difference (the “+X pts” column). Use this to compare what the drug seems to add over the baseline trial noise. Important caveat: the FDA explicitly warns that adverse reaction rates from one trial cannot be directly compared to another, because populations, doses, durations, and reporting methods differ. Treat this as a translation guide, not a leaderboard.

Table 1 — Common adverse events in adult weight-management trials
Format: Drug rate% vs Placebo rate% → +Difference pts. Source: current DailyMed labels accessed May 14, 2026.
| Drug & dose | Nausea | Vomiting | Diarrhea | Constipation | Source |
|---|---|---|---|---|---|
| Wegovy 2.4 mg (semaglutide injection, weekly) | 44% vs 16% → +28 pts | 24% vs 6% → +18 pts | 30% vs 16% → +14 pts | 24% vs 11% → +13 pts | Wegovy DailyMed label |
| Wegovy 7.2 mg (semaglutide injection, weekly) | 39% vs 13% → +26 pts | 22% vs 6% → +16 pts | Not separately listed in 7.2 mg table | 20% vs 8% → +12 pts | Wegovy DailyMed label |
| Zepbound 5 mg (tirzepatide injection, weekly) | 25% vs 8% → +17 pts | 8% vs 2% → +6 pts | 19% vs 8% → +11 pts | 17% vs 5% → +12 pts | Zepbound DailyMed label |
| Zepbound 10 mg | 29% vs 8% → +21 pts | 11% vs 2% → +9 pts | 21% vs 8% → +13 pts | 14% vs 5% → +9 pts | Zepbound DailyMed label |
| Zepbound 15 mg | 28% vs 8% → +20 pts | 13% vs 2% → +11 pts | 23% vs 8% → +15 pts | 11% vs 5% → +6 pts | Zepbound DailyMed label |
| Saxenda 3 mg (liraglutide injection, daily) | 39% vs 14% → +25 pts | 16% vs 4% → +12 pts | 21% vs 10% → +11 pts | 19% vs 9% → +10 pts | Saxenda DailyMed label |
| Foundayo 5.5 mg (orforglipron, oral, daily) | 26% vs 10% → +16 pts | 13% vs 4% → +9 pts | 21% vs 11% → +10 pts | 20% vs 9% → +11 pts | Foundayo DailyMed label |
| Foundayo 9 mg | 34% vs 10% → +24 pts | 21% vs 4% → +17 pts | 23% vs 11% → +12 pts | 27% vs 9% → +18 pts | Foundayo DailyMed label |
| Foundayo 17.2 mg | 35% vs 10% → +25 pts | 24% vs 4% → +20 pts | 25% vs 11% → +14 pts | 24% vs 9% → +15 pts | Foundayo DailyMed label |
Table 2 — Discontinuation rates: who actually stops the medicine?
This is the number that tells you tolerability, not the symptom rates above.
| Drug & dose | Stopped (any adverse reaction) | Stopped (GI reactions specifically) | Placebo arm stopped (any / GI) |
|---|---|---|---|
| Wegovy 2.4 mg | 6.8% | 4.3% | 3.2% / 0.7% |
| Wegovy 7.2 mg | 5% | 3% | 2% / 0.3% |
| Wegovy 25 mg tablet | 6.9% | 3.4% | 5.9% / 2.0% |
| Zepbound 5 mg | 4.8% | 1.9% | 3.4% / 0.5% |
| Zepbound 10 mg | 6.3% | 3.3% | 3.4% / 0.5% |
| Zepbound 15 mg | 6.7% | 4.3% | 3.4% / 0.5% |
| Saxenda 3 mg | 9.8% | 6.2% | 4.3% / 0.8% |
| Foundayo 5.5 mg | 6% | 3% | 3% / 0.7% |
| Foundayo 9 mg | 9% | 6% | 3% / 0.7% |
| Foundayo 17.2 mg | 10% | 6% | 3% / 0.7% |
Table 3 — Serious or rare adverse events (all in the under-1%-to-low-single-digits range)
| Event | Wegovy 2.4 mg | Zepbound (all doses) | Saxenda 3 mg | Foundayo (highest dose) |
|---|---|---|---|---|
| Severe GI reaction | 4.1% vs 0.9% | 1.7%--3.1% vs 1.0% | 4.8% vs 1.4% | ~3% vs 1% |
| Adjudicated acute pancreatitis | 0.2 per 100 pt-yrs vs <0.1 | 0.2% vs 0.2% | 0.3% vs 0.1% | 0.14 vs 0.04 per 100 pt-yrs |
| Cholelithiasis (gallstones) | 1.6% vs 0.7% | 1.1% vs 1.0% | 2.2% vs 0.8% | 1% vs 0.7% |
| Cholecystitis | 0.6% vs 0.2% | 0.7% vs 0.2% | 0.8% vs 0.4% | 0.4% vs 0.3% |
| Acute kidney injury | Post-marketing reports | Post-marketing reports | Post-marketing reports | 0.2% vs 0.05% |
| Hypersensitivity reaction (immediate) | Reported | 2.1% vs 0.4% | Reported | 0.5% vs 0.3% |
| Resting heart rate increase | 1--4 bpm | 1--4 bpm | 2--3 bpm | Mean +4--5 bpm vs +0.5; tachycardia 3% vs 0.9% |
Boxed warning applies to all four drugs
The obesity-indicated medicines covered here carry a boxed warning about thyroid C-cell tumors based on rodent studies. None of these medicines should be used in people with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). The human relevance of the rodent findings is undetermined.
For a deeper dive into how FDA draws the line between contraindications, warnings, and adverse reactions, see our GLP-1 contraindication vs. warning vs. precaution guide.
Why “+X pts” matters: the simplest way to read any GLP-1 label
Quick answer: Take the drug's rate, subtract the placebo rate, and you have the absolute excess — the part the drug is probably adding. For Wegovy nausea, that's 28 points. For Zepbound 15 mg nausea, it's 20 points. For Saxenda nausea, it's 25 points. For Foundayo 17.2 mg, it's 25 points.
Nausea is the most reliably attributable symptom
Every GLP-1 here adds 16 to 28 percentage points to the baseline nausea rate. It's the symptom most clearly tied to the drug.
Diarrhea has a smaller drug effect than it looks
In Wegovy, the raw number is 30%, but placebo was 16%. Only about 14 points are likely the drug -- not the scary-sounding 30%.
Constipation rises modestly across the class
Usually 10 to 18 points above placebo. Higher Foundayo doses are on the upper end. It tends to persist longer than nausea.
Discontinuation gaps are small
Most GLP-1s push the "quit because of GI" rate up by only 1 to 5 percentage points over placebo. Most people ride it out.
How common is nausea on a GLP-1?
Quick answer: Nausea is reported by roughly 25% to 44% of trial participants on a GLP-1 in adult weight-management studies, compared to roughly 8% to 16% on placebo. Most cases are mild to moderate, cluster during the dose-escalation phase, and ease as the body adjusts. The drug-attributable portion is 16 to 28 percentage points.
In the STEP-1 trial of Wegovy, gastrointestinal symptoms peaked during the dose-escalation phase and then declined. The label-level takeaway: in the Wegovy 2.4 mg adult weight-reduction trials, 4.3% of Wegovy-treated patients discontinued because of GI reactions vs 0.7% on placebo. People get nauseous, and the great majority continue the medicine.
What helps — common clinician and pharmacist talking points (not personal medical advice):
- •Slow the dose escalation. Most labels allow staying at a tolerable dose longer.
- •Eat smaller meals. Don't eat past full.
- •Lower the fat content of meals. Fat slows gastric emptying further.
- •Stay hydrated. Sip water through the day.
- •Avoid alcohol during dose increases.
How common is vomiting on a GLP-1?
Quick answer: Vomiting was reported by 8% to 24% of trial participants on a GLP-1, depending on the drug and dose, versus 2% to 6% on placebo. It is more common with semaglutide and orforglipron than with tirzepatide at typical maintenance doses, and more common during dose escalation than maintenance. Repeated vomiting that prevents fluid intake is a red flag, not a normal symptom.
Wegovy 2.4 mg adds about 18 percentage points of vomiting reporting over placebo. Zepbound 10 mg adds about 9. Saxenda adds about 12. Foundayo at the highest dose adds 20.
How common is diarrhea or constipation?
Quick answer: Diarrhea was reported by 19% to 30% of trial participants on a GLP-1 versus 8% to 16% on placebo. Constipation was reported by 11% to 27% versus 5% to 11%. The drug-attributable portion is roughly 10 to 15 percentage points for diarrhea and 6 to 18 points for constipation. Both are usually mild to moderate and dose-related.
Fiber and fluid help constipation
In most cases. A bowel-motility supplement (talk to a clinician or pharmacist) can be added if needed.
Persistent diarrhea is a fluid-loss problem first
Replace electrolytes, not just water. GLP-1s slow stomach emptying and change bowel motility.
Severe diarrhea with cramping, blood, or fever
Not a GLP-1 side effect to ride out. That's a clinician call -- not a wait-and-see symptom.
Which GLP-1 has the fewest side effects?
Quick answer: There is no single “least toxic” GLP-1. Trial rates of common GI symptoms are broadly similar across the class once you adjust for dose, and the FDA explicitly warns against directly comparing rates between different trial programs. In the only head-to-head trial of Wegovy and Zepbound (SURMOUNT-5), tirzepatide came out modestly better on tolerability metrics. Individual tolerability varies far more than the trial averages suggest.

What we can say with reasonable confidence:
In SURMOUNT-5, treatment discontinuation because of adverse events was 6.1% with tirzepatide vs 8.0% with semaglutide 2.4 mg, GI-event discontinuation was 2.7% vs 5.6%, and serious adverse events were 4.8% vs 3.5%. That head-to-head result is more useful than comparing separate FDA label tables.
Weekly drugs (Wegovy, Zepbound) showed lower discontinuation rates than daily Saxenda in the data reviewed. Saxenda discontinuation came in at 9.8% in the label-level number.
Tirzepatide at 5 mg has lower side-effect rates than 15 mg. Some prescribers stop dose escalation when patients hit a tolerable level rather than pushing to the highest dose.
Foundayo (oral orforglipron) is not automatically "easier on the stomach" because it's a pill. In ACHIEVE-3, a type 2 diabetes trial, more participants in the orforglipron groups discontinued because of adverse events than in the oral semaglutide group.
The framework that's actually useful: what dose, what indication, what population, what would my prescriber recommend given my history? Someone with a history of severe GI sensitivity should bring that history to the prescriber and ask whether slower escalation, a lower tolerated maintenance dose, or a different product makes sense.
See our best GLP-1 with least side effects guide and GLP-1 side effects comparison chart for a deeper comparison.
Serious side effects: pancreatitis, gallbladder, kidney, NAION, and the boxed warning
Quick answer: Serious GLP-1 side effects are real but rare. Adjudicated pancreatitis occurred in about 0.2% to 0.4% of trial participants — similar to placebo in most trials. Gallbladder disease occurred in 1% to 4%. NAION was classified by EMA in June 2025 as a “very rare” side effect of semaglutide medicines. The boxed warning for thyroid tumors applies to the obesity-indicated medicines covered here and is based on rodent studies, not human data.
Pancreatitis
Adjudicated acute pancreatitis ran 0.2% to 0.4% across the obesity GLP-1 trials. In SURMOUNT trials, the tirzepatide rate was 0.2% versus 0.2% on placebo — essentially identical. In SAXENDA trials, liraglutide came in at 0.3% versus 0.1% on placebo. Wegovy reports 0.2 cases per 100 patient-years vs less than 0.1 on placebo.

See our GLP-1 contraindication vs. warning vs. precaution guide for how pancreatitis fits into the full label risk picture.
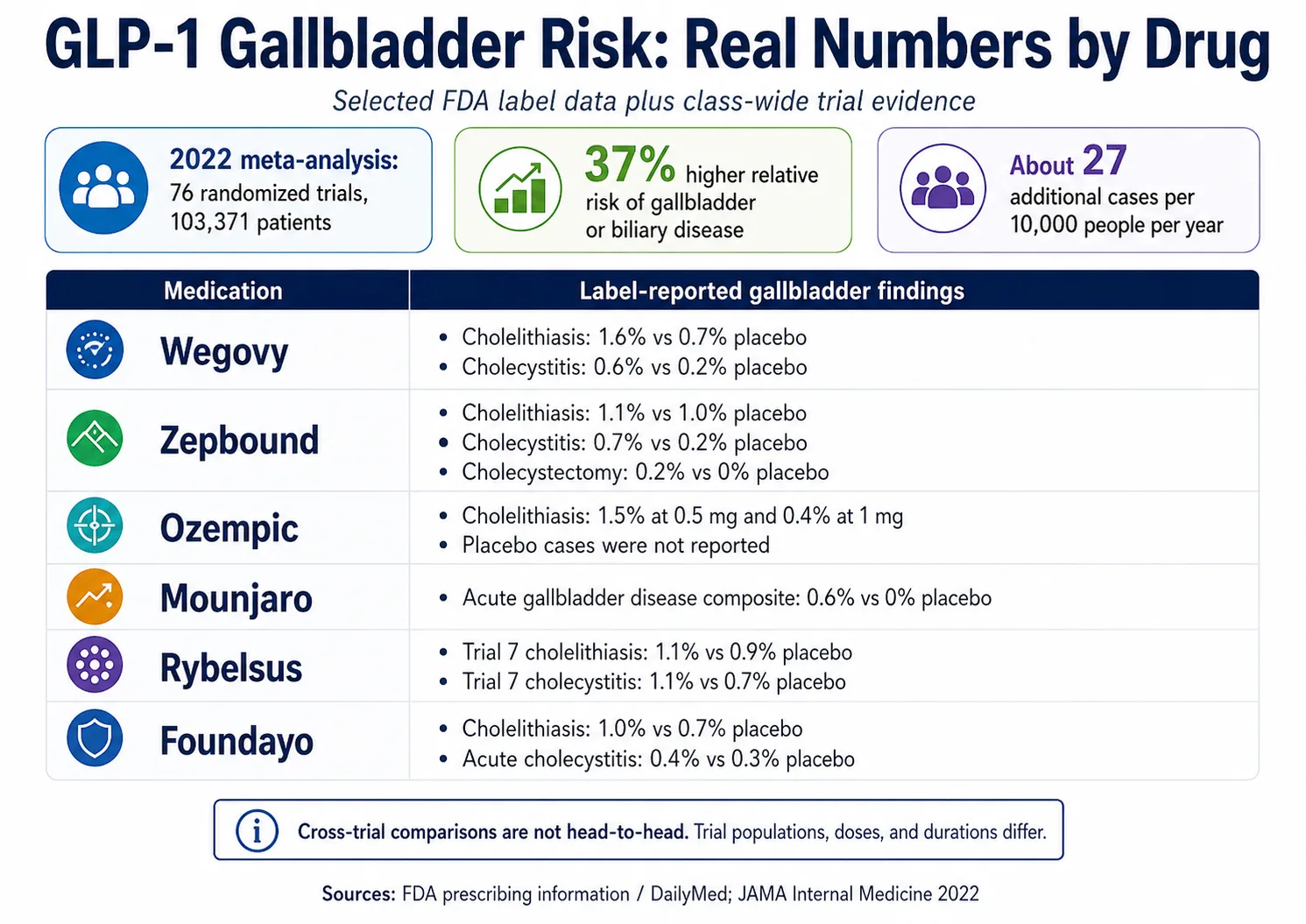
Gallbladder disease
Gallbladder disease is a label-supported concern, but the size of the signal differs by product. Wegovy and Saxenda labels state acute gallbladder disease occurred more often with treatment than placebo even after accounting for the rate of weight loss. The current Zepbound and Foundayo label numbers show smaller placebo differences in the pooled weight-reduction trials.
NAION (non-arteritic anterior ischemic optic neuropathy)
This is the eye-blindness story that made headlines in 2024 and was investigated by regulators throughout 2025. NAION causes sudden, usually painless, vision loss in one eye.
| Date | Development |
|---|---|
| 2024 | Small Harvard study found hazard ratio of 4.28 for NAION in semaglutide users vs non-GLP-1 diabetes drugs. |
| Jan 2025 | European Medicines Agency opened a formal NAION review. |
| Mid 2025 | Larger multi-database OHDSI study of 37 million adults with type 2 diabetes found a smaller increase than the Harvard study. |
| Jun 2025 | Study in American Journal of Ophthalmology found no NAION risk difference vs matched controls; cumulative incidence below 0.1% over five years. |
| Jun 2025 | EMA PRAC concluded NAION should be listed as a "very rare" side effect of semaglutide medicines. WHO issued a global medical product alert referencing the same update. |
As of this verification, the strongest label-update source for NAION is EMA/WHO semaglutide product-information guidance. The honest read: the absolute risk is small, and the label addition reflects regulatory caution.
Acute kidney injury
Current labels describe AKI as occurring in association with GI reactions or dehydration. Repeated vomiting or diarrhea with poor fluid intake should trigger clinician contact.
Hypoglycemia
GLP-1s alone rarely cause hypoglycemia in people without diabetes. Risk rises sharply with insulin or sulfonylureas. In Wegovy Study 3 (type 2 diabetes), hypoglycemia was 6.2% vs 2.5% placebo.
Severe allergic reactions
Zepbound trials reported immediate hypersensitivity in 2.1% vs 0.4% on placebo. Most were skin reactions. Anaphylaxis and angioedema have been reported post-marketing. Anyone with a prior serious allergic reaction should not retry.
Surgery and anesthesia
2024 multi-society guidance: most patients can continue GLP-1s before elective surgery, but higher-risk patients may need an individualized plan. Always tell your surgical and anesthesia team you're on a GLP-1.
Ileus and intestinal obstruction
Added to the Ozempic label in September 2023 after post-marketing reports. Rare in trials. Severe, persistent abdominal pain with vomiting and constipation that won't resolve is a clinical-evaluation symptom.
Suicidal behavior (monitoring warning removed)
The prior monitoring warning was removed from Wegovy, Zepbound, and Saxenda U.S. labels in February 2026 after FDA and EMA reviews found no causal link. New or worsening mood symptoms should still be discussed with your prescriber.
Red, Yellow, Green: how to triage a symptom
Quick answer: Most GLP-1 symptoms are green (track them, mention at the next visit) or yellow (call your prescriber before the next dose). A specific set of red flags — severe persistent pain, dehydration, allergic reaction, sudden vision loss, severe hypoglycemia symptoms — are not “wait and see” events.

GREEN — Track it, mention it next visit
- ✓Mild nausea, especially in the first few weeks of a new dose
- ✓Decreased appetite or feeling full faster than expected
- ✓Mild constipation that responds to fiber and fluids
- ✓Occasional burping or mild reflux
- ✓Mild fatigue or headache early on
- ✓One or two episodes of loose stool
What to do: write down what you ate, when you took the dose, when the symptom started, and how long it lasted. Bring it to your next appointment.
YELLOW — Call your prescriber before the next dose
- !Vomiting more than once a day for multiple days
- !Diarrhea keeping you from drinking or eating normally
- !Constipation that hasn't resolved in 5+ days despite fiber and fluids
- !New or worsening reflux affecting sleep
- !Dizziness when standing up
- !Feeling weak, lightheaded, or "off"
- !Worsening of any green-tier symptom across two consecutive doses
- !Hair shedding (common, usually temporary -- but worth flagging)
What to do: call the prescribing clinic. Do not skip or change doses on your own. Ask whether the next dose should be held, delayed, or escalated more slowly.
RED — Seek urgent medical care
- ✕Severe persistent abdominal pain, especially if it radiates to the back
- ✕Vomiting so frequent you can't keep down fluids
- ✕Signs of dehydration: little or no urination, very dark urine, racing heart, confusion, or inability to keep fluids down
- ✕Sudden vision loss in one eye (NAION can present this way)
- ✕Difficulty breathing, swelling of the face or throat (possible anaphylaxis)
- ✕Severe abdominal pain with fever and jaundice (possible gallbladder emergency)
- ✕Severe hypoglycemia symptoms (shaking, sweating, confusion, loss of consciousness) if you're on insulin or sulfonylureas
- ✕Persistent severe abdominal pain with vomiting and inability to pass stool (possible obstruction)
What to do: emergency department, urgent care, or call your local emergency line. Tell them you're on a GLP-1 medication and which one. This matters for diagnosis and for procedural decisions.
Will GLP-1 side effects go away?
Quick answer: Most common GI symptoms — nausea, vomiting, diarrhea — are more likely during dose escalation and often decline once the maintenance dose stabilizes. Constipation and reflux can persist longer. Hair shedding is usually temporary. Serious events like pancreatitis, gallbladder disease, and NAION are not transient — they require clinical evaluation.
| Symptom | Does it ease over time? | Notes |
|---|---|---|
| Nausea, vomiting, diarrhea | Yes -- usually after dose stabilizes | In every product reviewed, new GI event rates declined sharply after maintenance dose was reached. |
| Constipation | Partially -- can persist | Tends to be more persistent than nausea. May require ongoing fiber and fluid management. |
| Reflux | Sometimes | Can be persistent. Sometimes responsive to elevating head of bed, smaller meals, avoiding eating before lying down. |
| Hair loss | Yes -- usually temporary | Typically related to rapid weight loss itself, not the drug specifically. Usually improves within months of weight stabilizing. |
| Pancreatitis, gallbladder, AKI, NAION | No -- these are events | Not transient symptoms. They need clinical evaluation when they happen. |
For a deeper look at what happens after years on these medications, see our GLP-1 long-term side effects guide.
Why Reddit and FAERS sound scarier than the trials
Quick answer: Reddit and FAERS show what people are reporting, not how often events happen. Reddit captures the loud minority. FAERS captures voluntary reports without a denominator — the count tells you a signal exists, not how common the event is. The FDA explicitly states FAERS reports cannot be used to calculate incidence, do not establish causation, may be duplicative or incomplete, and are not by themselves an indicator of a drug's safety profile.
Clinical trial rates
What it is: Percentages from controlled studies with denominators, placebo arms, and adjudicated outcomes.
Tells you: Roughly how often a thing happens in the trial population.
Doesn't tell you: How often it happens in your population, or in the real world.
FAERS reports
What it is: Voluntary reports filed with the FDA by patients, doctors, manufacturers, lawyers, and pharmacists.
Tells you: A signal exists. If a thousand pancreatitis reports come in, the FDA wants to know.
Doesn't tell you: How often it happens. No denominator. No placebo arm. No causality check.
Reddit, TikTok, forums
What it is: What people are worried about, what symptoms feel like, what their care experience has been.
Tells you: What language people use and what their lived experience is.
Doesn't tell you: How common any of it is. 0.5% of a subreddit posting ≠ epidemic.
Do Ozempic and Mounjaro rates belong in this weight-loss table?
Quick answer: Not in the main weight-loss table. Ozempic and Mounjaro are FDA-approved for type 2 diabetes, so their label rates come from diabetes trial populations — different doses, different baseline health, often different background medicines. Many readers search Ozempic and Mounjaro because they're being used off-label for weight loss; the rates from diabetes trials are the only label data that exists for those specific products.
For diabetes-label context only (do not compare directly to the obesity-label table):
- Ozempic 0.5 mg / 1 mg: nausea 15.8% / 20.3% vs 6.1% placebo; vomiting 5% / 9.2% vs 2.3%; diarrhea 8.5% / 8.8% vs 1.9%; constipation 5% / 3.1% vs 1.5%.
- Mounjaro 5 mg / 10 mg / 15 mg: nausea 12% / 15% / 18% vs 4%; vomiting 5% / 5% / 9% vs 2%; diarrhea 12% / 13% / 17% vs 9%; constipation 6% / 6% / 7% vs 1%. GI-related discontinuation 3.0% / 5.4% / 6.6% vs 0.4% placebo.
See our GLP-1 FDA indication vs. off-label use guide for why these distinctions matter.
What changed in 2024–2026: the regulatory timeline
Quick answer: Between September 2023 and April 2026, FDA, EMA, and WHO added or updated several GLP-1 safety items. The most material updates: the surgery/anesthesia framework shifted from “hold the drug” to “individualized risk”; EMA classified NAION as a very rare side effect of semaglutide; the suicidal-behavior monitoring warning was removed from several U.S. obesity labels; and Foundayo became the first FDA-approved oral non-peptide GLP-1.
| Date | Drug or class | Update |
|---|---|---|
| September 2023 | Ozempic (semaglutide) | Label updated to add ileus/intestinal obstruction risk |
| October 2024 | GLP-1 class | New multi-society guidance replaces simplistic "hold GLP-1" framing before elective surgery |
| November 2024 | Wegovy (semaglutide) | Pulmonary aspiration during anesthesia warning added |
| January 2025 | Semaglutide products | EMA PRAC opened formal NAION review |
| June 2025 | Ozempic, Rybelsus, Wegovy | EMA PRAC concluded NAION is "very rare" side effect; WHO issued global alert June 27 referencing the same update |
| August 2025 | Wegovy | New MASH indication added (treatment of metabolic dysfunction-associated steatohepatitis) |
| February 2026 | Wegovy, Zepbound, Saxenda | Suicidal behavior and ideation warning removed from current U.S. labels |
| April 1, 2026 | Foundayo (orforglipron) | First new molecular entity approved under FDA's National Priority Voucher pilot -- first oral non-peptide GLP-1 for chronic weight management |
| April 22, 2026 | Zepbound | Most recent DailyMed label update |
Compounded GLP-1s are not the same as FDA-approved products
FDA clarification
- !Compounded GLP-1 medications are not FDA-approved and have not been studied in the same way as Wegovy, Zepbound, Saxenda, or Foundayo.
- !FDA reported 605 adverse-event reports associated with compounded semaglutide and 545 associated with compounded tirzepatide as of July 31, 2025.
- !Compounded versions may vary in concentration, formulation, and dosing instructions from pharmacy to pharmacy.
- !FDA has issued warnings about compounded GLP-1 dosing errors and adverse events.
- !The percentages on this page are from FDA-approved products only. Compounded-product reports cannot be combined with these rates.
When you read a story about someone hospitalized from “Ozempic,” check whether they were taking FDA-approved Ozempic or a compounded semaglutide from a pharmacy or telehealth source. The two are not the same product.
If you're researching all GLP-1 options including compounded paths, our GLP-1 path quiz routes you to the option that fits your situation and budget.
What to ask your prescriber
Use the adverse event rate data to prepare better questions, not to self-prescribe or switch drugs without input. The most useful questions cover the specific product you're taking, the escalation schedule, what's expected at your current dose, what should trigger a call, and what should trigger urgent care.
The 10-question prescriber checklist
Which exact product am I taking -- FDA-approved brand, generic, or compounded?
What dose am I starting on, and what's the escalation schedule?
What side effects are most common at this dose specifically?
Which symptoms should make me call your office before my next dose?
Which symptoms should send me to urgent care or the ER?
What should I do if I vomit repeatedly or can't keep down fluids?
Do my other medications (insulin, sulfonylureas, oral contraceptives, blood thinners) change the risk profile?
Do you want me to track anything? Blood pressure, glucose, weight, symptoms?
What's the plan if I can't tolerate the next dose increase?
What should I tell a surgeon or anesthesiologist if I need a procedure?
Ask this if…
If you have type 2 diabetes
- •Should my insulin or sulfonylurea dose change when I start the GLP-1?
- •How often should I check my glucose during the first 8 weeks?
- •What's my hypoglycemia action plan?
- •Should I adjust other diabetes medications if I have GI side effects affecting eating?
Frequently asked questions
How common is nausea on a GLP-1?
Nausea is reported by 25% to 44% of trial participants on a GLP-1 in adult weight-management studies, compared to 8% to 16% on placebo. Most cases are mild to moderate and improve as the maintenance dose stabilizes. The drug-attributable portion (subtracting the placebo rate) is roughly 16 to 28 percentage points depending on the drug.
How common is vomiting on a GLP-1?
Vomiting was reported by 8% to 24% of trial participants on a GLP-1, versus 2% to 6% on placebo. Foundayo at the highest dose has the highest reported rate; tirzepatide at lower doses has the lowest. Repeated vomiting that prevents fluid intake is a red flag -- not a normal side effect to ride out.
How common is pancreatitis with GLP-1 medications?
Adjudicated acute pancreatitis occurred in approximately 0.2% to 0.4% of GLP-1-treated trial participants, with similar rates in placebo arms in most trials. Post-marketing surveillance has logged thousands of pancreatitis reports, but those counts don't represent incidence and cannot establish causation. Persistent severe abdominal pain that radiates to the back is the symptom that requires urgent evaluation.
Does Ozempic or Wegovy cause blindness?
In June 2025, EMA's PRAC concluded that NAION (non-arteritic anterior ischemic optic neuropathy) should be listed as a "very rare" side effect of semaglutide medicines -- Ozempic, Rybelsus, and Wegovy -- and WHO issued a medical product alert referencing that classification. The cumulative incidence is estimated at less than 0.1% over five years in the largest cohort studies. Sudden vision loss in one eye is a red-flag symptom requiring urgent ophthalmology evaluation.
Which GLP-1 has the fewest side effects?
There is no single "least toxic" GLP-1 across the board. Trial rates of common GI symptoms are broadly similar across the class once you adjust for dose. The FDA explicitly warns against directly comparing rates between different trial programs. In the only head-to-head trial of Wegovy and Zepbound (SURMOUNT-5), tirzepatide came out modestly better on tolerability -- discontinuation 6.1% vs 8.0%, GI-event discontinuation 2.7% vs 5.6% -- but had a higher serious-adverse-event rate (4.8% vs 3.5%).
Are GLP-1 side effects permanent?
Most common symptoms -- nausea, vomiting, diarrhea -- fade after the dose-escalation phase. Exact timing depends on the drug, dose, and escalation schedule. Constipation and reflux can persist longer. Hair loss is usually temporary. Some serious events (pancreatitis, gallbladder disease, NAION) are not transient and require medical evaluation when they occur.
What percent of people stop taking a GLP-1 because of side effects?
Across current obesity-label datasets for Wegovy, Zepbound, Saxenda, and Foundayo, discontinuation specifically because of GI adverse reactions ran about 1.9% to 6.2%, and discontinuation for any adverse reaction ran about 4.8% to 10.0%. Saxenda has the highest reported discontinuation rate; Zepbound 5 mg the lowest.
Can FAERS or social media tell me how likely a GLP-1 side effect is?
No. FAERS can identify reports and possible safety signals, but the FDA states that FAERS data cannot be used to calculate the incidence of an event, does not establish causation, and is not by itself an indicator of a drug's safety profile. Social media has the same limitation plus stronger selection bias. Use either for signal context, not personal risk estimation.
Are compounded GLP-1 side effect rates the same as the FDA-approved versions?
No. Compounded GLP-1 medications are not FDA-approved and have not been studied in randomized clinical trials of comparable size or design. The rates on this page are from FDA-approved products only. FDA reported 605 adverse-event reports associated with compounded semaglutide and 545 associated with compounded tirzepatide as of July 31, 2025.
What's a "boxed warning" and why do these medicines have one?
A boxed warning (sometimes called a "black box warning") is the most serious type of FDA warning. The obesity-indicated medicines on this page carry one for thyroid C-cell tumors, based on rodent studies in which semaglutide and tirzepatide caused these tumors at clinically relevant doses. The relevance to humans is undetermined. These medicines are contraindicated in anyone with a personal or family history of medullary thyroid carcinoma or MEN 2 syndrome.
Should I worry about GLP-1s and surgery?
Tell every surgeon and anesthesiologist that you're on a GLP-1 well before any procedure. Current 2024 multi-society guidance says most patients can continue GLP-1 medicines before elective surgery, but higher-risk patients -- people in dose escalation, people with active GI symptoms, people on higher doses -- may need an individualized plan such as a 24-hour liquid diet, stomach ultrasound, full-stomach precautions, or delaying the procedure. This replaced the earlier 2023 "hold the drug" approach.
Was the suicidal behavior warning removed from GLP-1 labels?
Yes -- the prior monitoring warning was removed from current U.S. labels for Wegovy, Zepbound, and Saxenda in February 2026, after FDA and EMA reviews did not establish a causal link between GLP-1 use and suicidality. New or worsening mood symptoms should still be discussed with the prescriber, but suicidality monitoring is no longer a class-wide label requirement in the U.S. obesity products reviewed here.
What is the bottom line on GLP-1 adverse event rates?
Bottom line: the common symptoms are common, the serious events are specific and uncommon, and the safest move is to compare placebo-adjusted rates while knowing which symptoms require care.
The drug doesn't cause 44% of people to throw up. The drug pushes the trial-long nausea rate up by about 28 percentage points over placebo. Most of the people who get nauseous keep taking the drug. The serious events you've heard about are real and rare. The symptoms that need urgent care are specific and named. The math is doable, the signals are knowable, and “scared of the label” doesn't have to be your decision-making frame.
If you're starting a GLP-1 this week, save this page. If a symptom shows up, the Red/Yellow/Green table tells you which bucket it's in. If you're trying to decide between drugs, compare placebo-adjusted differences, not raw percentages — and talk to a clinician who can match the choice to your history.
We'll keep this page updated. Visible timestamp at the top reflects the last verification date.
Related guides on The RX Index
- GLP-1 Side Effects Comparison Chart (2026)
- Best GLP-1 with the Least Side Effects: 2026 Data
- GLP-1 Long-Term Side Effects: What We Know (2026)
- Foundayo (Orforglipron) Side Effects: Full FDA Label Review
- Orforglipron Side Effects: Trial Data and Red Flags
- GLP-1 Contraindication vs. Warning vs. Precaution Explained
- GLP-1 FDA Indication vs. Off-Label Use: The Real Difference
- GLP-1 Path Quiz -- Find the Right Medication for Your Situation
Sources and citations
- 1.Current DailyMed prescribing information accessed May 14, 2026: Wegovy label (major changes through March 2026), Zepbound label (updated April 22, 2026), Saxenda label (updated February 25, 2026), Foundayo label (April 1, 2026), Ozempic label, and Mounjaro label. DailyMed is the NIH's official source for FDA-submitted labeling.
- 2.SURMOUNT-5 head-to-head data: Aronne et al., NEJM 2025.
- 3.NAION classification: EMA PRAC June 2025 press release; WHO Medical Product Alert June 27, 2025.
- 4.FAERS limitations: FDA FAERS Public Dashboard FAQ.
- 5.Surgery and anesthesia guidance: 2024 multi-society GLP-1 guidance (ASA, ASMBS, AGA, ASGE, IASMC, SAGES).
- 6.Compounded GLP-1 reporting: FDA's "Concerns About Unapproved GLP-1 Drugs" page, current as of February 4, 2026.
- 7.Foundayo approval: FDA Press Release, April 1, 2026.
- 8.Phase 3 trials: STEP 1 (Wegovy), SURMOUNT 1--5 (Zepbound), SCALE (Saxenda), ACHIEVE/ATTAIN (orforglipron/Foundayo).
About The RX Index: The RX Index is a pricing intelligence and comparison resource for GLP-1 telehealth providers. We verify pricing, coverage, and policy directly from primary sources and update our guides quarterly or sooner when material changes occur.
Editorial standards: We do not pay for placement, do not accept paid testimonials, and do not include “medically reviewed by” claims unless a real, named clinician with verifiable credentials actually reviewed the content. This page was researched and written by The RX Index editorial team and is informational only — not personal medical advice. Talk to a licensed clinician about your specific situation.
Last verified: May 14, 2026. This page is updated when FDA labels change, EMA/WHO safety updates are issued, or new head-to-head trial data is published.